Abstract
Background
Animal-derived remedies are prominent in folk medicine worldwide, and discovering potent anti-inflammatory agents is attractive for rheumatoid arthritis (RA) treatment. Himalayan marmot oil (HMO), a traditional Tibetan medicine topically or orally used for burns and arthritis, has been common in Tibetan areas for centuries. However, the composition and systematic pharmacological evaluation of HMO are still unclear.
Objectives
This study focused on the chemical composition and anti-inflammatory effects of HMO in vitro and in vivo to expand the topical value of the application of HMO in RA.
Materials and Methods
First, the chemical composition of HMO was explored by gas chromatography–mass spectrometry (GC–MS). Then, we established a bioassay system for L929 cells and RAW 264.7 cells to evaluate the effect of HMO on cytotoxicity and inflammatory responses. The anti-arthritic evaluation involved testing for paw swelling, swelling inhibition, and histological analysis in collagen-induced arthritis (CIA) rats. Enzyme-linked immunosorbent assay, Western blot analysis, and quantitative real-time polymerase chain reaction were performed to determine the effect of HMO on the production and secretion of inflammatory cytokines.
Results
GC–MS results showed that seven categories of chemical substances, including 261 compounds in HMO, and the main components of HMO were saturated fatty acids and unsaturated fatty acids. HMO showed low toxicity in cells according to the CCK-8 tests. In CIA model rats, HMO obviously inhibited hind paw swelling and reduced the arthritis index. Pathological staining results and micro-CT imaging indicated that HMO attenuated synovial hyperplasia and inflammatory cell infiltration in joint tissues. Meanwhile, in vivo and in vitro, HMO significantly inhibited interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α) levels, while the expression of interleukin-10 (IL-10) was increased.
Conclusion
The current work identified an excellent anti-inflammatory animal-derived medicinal agent, HMO, in alleviating synovial inflammation and joint destruction by regulating IL-6, IL-10, and TNF-α expression with low toxicity, which provides a protective effect against RA. Our study expands the topical value of the application of HMO to inhibit the progression of inflammation, and it could be a safe, novel RA complementary therapy.
Introduction
Rheumatoid arthritis (RA) is a chronic systemic autoimmune disease. It is characterized by inflammation of the synovial tissue and autoantibody production (Weyand & Goronzy, 2021). As a chronic inflammatory disease, the prevalence of RA is approximately 1% in the general population. The disease can affect anyone at any age and is more prevalent in women (Quan et al., 2021). The disease is characterized by progressive joint destruction, with bone erosion and synovial proliferation leading to the destruction of bones and joints, accompanied by severe joint pain and, eventually, deformation of the joints (Singh et al., 2020). Advanced bone and joint deformities can lead to irreversible loss of function and physical disability, dramatically deteriorating the patient’s quality of life and potentially further exacerbating associated complications, even to the point of being life-threatening. More than 30% of RA patients lose their ability to work after 10 years, and in more severe cases, the life expectancy of RA patients is reduced by 5–10 years (Emery et al., 2016; Takeuchi et al., 2017).
Currently, the primary treatment for RA is to protect the patient’s functional activity and prevent further disease progression and joint destruction by controlling the pain and inflammatory response of the joints. It is possible to relieve symptoms of this disease and slow its progression with early detection and appropriate treatment. Clinically, a variety of anti-rheumatic drugs are commonly used to treat RA, including non-steroidal anti-inflammatory drugs (NSAIDs), biosynthetic anti-rheumatic drugs (bDMARDs), glucocorticoids (GCs), targeted synthetic disease-modifying anti-rheumatic drugs (tsDMARDs), and traditional synthetic anti-rheumatic drugs (csDMARDs) (Qindeel et al., 2020). However, these drugs have significant side effects, and patients’ gastrointestinal reactions and cardiovascular function need to be closely monitored while using them (Quan et al., 2021).
As a traditional Tibetan medicine, Himalayan marmot oil (HMO) was recorded in Chinese Materia Medica (Zhonghua Bencao). It is mainly obtained from the adipose tissue of Marmota himalayana Hodgson, which is distributed in the Qinghai-Tibetan Plateau. HMO is widely used for the treatment of burns and rheumatic joint pain and as an adjunct in the treatment of lumbar disc herniation (Ai, 2008). HMO exhibits significant anti-bacterial and bactericidal properties, which facilitate wound protection and skin tissue repair. Consequently, it is also utilized in the treatment of burns, prurigo, and psoriasis (Ma et al., 2003; Wang & Zhou, 1997). Studies have shown that HMO is mainly composed of different fatty acids, which contain a large amount of oleic acid and n-9 and n-3 polyunsaturated fatty acids (PUFAs). HMO contains approximately 70% C12–C18 unsaturated fatty acids, of which n-3 PUFAs can promote the release of large amounts of cell anti-inflammatory factors, and these cytokines can promote tissue and cellular uptake and clearance of inflammatory response products (Zhang et al., 2022). The highly unsaturated fatty acid composition of HMO has been associated with anti-inflammatory properties. Marmot oil is often made into ointment in Europe for the treatment of alleviation of joint and muscle pain (Yu et al., 2024). However, Tibetan medicine often administers HMO directly to the skin as a treatment for burns or arthritis. Therefore, HMO has effective anti-inflammatory and bacteriostatic properties and can alleviate some chronic inflammatory diseases. However, no systematic studies have been reported on the therapeutic effects of HMO on arthritis.
Materials and Methods
Materials
Dulbecco’s Modified Eagle’s Medium (DMEM), 0.25% trypsin-ethylenediaminetetraacetic acid (EDTA), penicillin-streptomycin, and fetal bovine serum (FBS) were purchased from Thermo Fisher Scientific, Inc. The CCK-8 kit and Calcein-AM/PI kit were purchased from Dojindo Laboratories, Inc. RIPA lysis buffers, BCA protein quantity kits, RNA extraction kits, cDNA synthesis kits, and quantitative real-time polymerase chain reaction (qRT-PCR) kits were purchased from ServiceBio Inc. Dimethyl sulfoxide (DMSO) and 1 × Tris-buffered saline with Tween-20 (TBST) buffer were purchased from Aladdin, Inc. HMO is from the dried adipose tissue of M. himalayana Hodgson. The sample was authenticated by Professor Hao Zhang (Sichuan University, China). Herbarium vouchers (No. MH2007) were prepared and deposited at the Chinese Materia Medica Herbarium of Sichuan University.
Animals
Sprague-Dawley (SD) rats (male, 180–200 g) were provided by Beijing WeiTong LiHua Experimental Animal Technology Co., Ltd. All animals were housed in cages with free access to food and water and then fed adaptive diets for 7 days. The rooms were maintained at a controlled temperature of 23 ± 1°C and a relative humidity of 65 ± 5%. Additionally, the rooms were subjected to a 12-h light/dark cycle. The animal experiments conducted in this study adhered to the guidelines set forth by the European Union for experimental animals. Animal experiments were conducted in full compliance with ethical standards.
Extraction of HMO
The pretreated (thawed, washed, drained) Himalayan marmot adipose tissue was minced with cryogenic grinding and mixed evenly. Five hundred grams of the mixed marmot adipose tissue was weighed and mixed with 2,000 mL of water. The mixture was heated in a water bath at 60–70°C for 2 h and then centrifuged at 7,200 g for 30 min. The upper oily sample was taken to obtain HMO.
Analysis of HMO with Gas Chromatography–Mass Spectrometry (GC–MS)
The following procedures were used to prepare the HMO samples. Briefly, 1 g of accurately weighed sample was transferred to a 20 mL headspace flask. A separate sampling of n-alkanes (C7-C40) mixed standard (20 µg/mL) was used for retention time correction. The flask was immersed in a thermostatic water bath at 60°C, stirring at 450 rpm for 15 min until temperature equilibrium. Afterwards, the 50/30 µm DVB/CAR/PDMS solid phase microextraction head was inserted into the headspace above the sample. Separation and identification were performed by GC–MS. A DB-wax fused-silica capillary column (30 m × 0.25 mm × 0.25 µm, Agilent J&W Scientific, Folsom, CA, USA) was used. The flow rate was constant at 1.0 mL/min, the injection port temperature was 260°C, and high-purity helium was used as the carrier gas. Analyzing the derived samples was performed using an Agilent 7890B gas chromatography system coupled to an Agilent 5977B quadrupole mass spectrometer.
Cell Culture
L929 and RAW264.7 cells were cultured in DMEM with 10% FBS, 100 units/mL penicillin, and 100 µg/mL streptomycin, and then incubated in 5% CO2 at 37°C in a humidified atmosphere. During the passage, the medium was changed every 2 days, and cells were routinely passaged when 80–85% confluent. All experiments included a vehicle control group containing 0.1% DMSO.
Cell Viability Assay
L929 cells were seeded at 1 × 104 per well in 96‑well plates and treated with HMO at various concentrations of 0 µg/mL, 1 µg/mL, 10 µg/mL, 100 µg/mL, and 1,000 µg/mL when the cells attached to the bottom. CCK-8 reagent was added to each well after treatment with HMO for 24 h, 72 h, and 120 h. The plates were placed in the incubator for 2 h, and then the absorbance was measured at 450 nm with a microplate reader.
Live/Dead Staining
Calcein AM/PI assays were performed after HMO treatment for 24 h. Fifty microliters of calcein AM (2 µmol/L) and PI (4.5 µmol/L) in a phosphate-buffered saline (PBS) mixture were added to each well and incubated at 37°C in a humidified atmosphere of 5% CO2 in the dark for 30 min. Live cells (yellow-green fluorescence) were detected using a fluorescence microscope with excitation filters at 490 ± 10 nm.
Establishment of the Inflammation Model and Drug Administration
RAW264.7 cells were seeded at 1 × 106 per well in six‑well plates for 24 h. For treatments, the medium was replaced by HMO at different concentrations of 0 µg/mL, 1 µg/mL, 10 µg/mL, 100 µg/mL, and 1,000 µg/mL at 37°C. After 24 h, cells were treated with 1 µg/mL lipopolysaccharide (LPS) for 6 h.
RNA Isolation and qRT-PCR
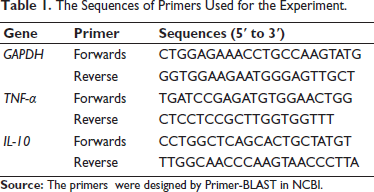
LPS- and HMO-treated RAW264.7 cells were harvested for RNA extraction. Total RNA was isolated with an RNA extraction reagent (ServiceBio Inc.) following the manufacturer’s protocol. A total of 1 µg of RNA was processed for reverse transcription using a FastQuant RT kit (ServiceBio Inc.) to acquire cDNA. The resulting cDNA was combined with an SYBR‑Green mixture (ServiceBio Inc.) in accordance with the manufacturer’s instructions for qRT‑PCR. The 2–∇∇Cq method was used for data analysis. The sequences of primers used for the experiment are shown in Table 1.
The Sequences of Primers Used for the Experiment.
Animals and Treatment
The SD male rats were allocated into four groups using a randomization process. Group A was used as a blank control, and groups B, C, and D were injected with type II chicken collagen to induce collagen-induced arthritis (CIA). Type II chicken collagen was phase-dissolved in 0.1 M glacial acetic acid on the previous day to make a concentration of 2 mg/mL and stocked at 4°C. On the second day, before modeling, the chicken type II collagen and the glacial acetic acid mixture were given an equal volume of complete Freund’s adjuvant to completely emulsify it. Animals were anesthetized with isoflurane using an anesthesia machine. Anesthesia was induced with 3% isoflurane, and the isoflurane level was maintained at 1.5% during the surgery. The previously configured chicken type II collagen was injected into the periarticular area from the medial plantar skin of the rat approximately 2 cm from the joint in the amount of 100 µL per toe to achieve initial immunization. Fifty microliters of chicken type II collagen were reinjected 15 days later in the same way. Beginning on day 18 of modeling, the animals received saline or indicated drugs at the ankle joints for 32 consecutive days. Group A, as the control group, and group B, as the model group, were given 0.5 mL saline. Group C was given 0.5 g of diclofenac sodium cream (DSC), and group D was given 0.5 mL HMO. On day 32, after modeling, the rats were sacrificed for experimental measurements.
Evaluation of RA
Body weight, paw thickness, and arthritis index were measured every 3–4 days after modeling. The degree of swelling of the ankle was recorded by Vernier calipers. By observing the appearance of the ankle joint, a five-point scale was used to score the arthritis of rat paws: Grade 0: there is no swelling in the ankle joint or toes; Grade 1: there is no swelling of the ankle joint or toes, minor redness of the skin, and no restrictions on activities; Grade 2: there is redness and swelling present in the toes and ankles, and activities are slightly affected; Grade 3: there is evident swelling and redness observed in the toes and ankles, and activity is affected clearly; Grade 4: there is severe swelling and redness of the ankle and toes, as well as deformity and stiffness, and activity is obstructed. The maximum arthritis score of each rat was set at 8 (4 points × 2 paws).
Imaging Examination
On day 32, after modeling, the rats were sacrificed with an intraperitoneal injection of 2% sodium pentobarbital. Then, the ankle joints of rats were removed from the tibia by dissection with scissors and fixed by immersion in a neutral fixative. The fixative was washed off and placed in the micro-CT machine, and the rat ankle tissue was scanned by micro-CT with three-dimensional imaging.
Histological Assessment
The ankle joints of the rats were collected, dehydrated, and fixed with 4% paraformaldehyde, embedded in paraffin, and sectioned into 4 µm thick sections. Then, the sections were subjected to hematoxylin–eosin (HE), toluidine blue (TB), and safranin-O/fast green staining.
Immunohistochemistry
The dewaxed and hydrated ankle tissue sections were incubated in 3% H2O2 for 10 min at room temperature, rinsed with water, soaked in PBS for 5 min, serum blocked and incubated with primary antibody for 2 h at 37°C, washed 3 times in PBS for 5 min each, dried around the tissue in PBS and added secondary antibody, then incubated in 37°C for 30 min; rinsed and dried again and added streptavidin–biotin complex (SABC) for 30 min; rinsed and developed color with chromogenic agent, then dehydrated and sealed the film.
Statistical Analysis
All in vitro experiments were performed in triplicate. Two-tailed Student t-tests and one-way analysis of variance (ANOVA) were performed using GraphPad Prism software v.5.01 (San Diego, CA, USA). All values are expressed as the means ± standard deviations (SDs). Dunnett’s or Tukey’s tests were applied if significant differences were observed between the treated groups. Statistical significance was attributed to differences observed at the *p < 0.05 and **p < 0.01 levels.
Results
GC–MS Analysis of HMO
The GC–MS raw data were transferred using a software analysis base file converter to facilitate efficient data retrieval. All components of HMO are represented by the total ion chromatogram in Figure 1. Based on standards and related databases, 20 main compounds of HMO were initially identified. The classes of compounds included organooxygen compounds, fatty acids, hydroxyl compounds, carboxylic acids, and their derivatives (Table 2). After analyzing the raw data, a total of 7 classes and 261 compounds were further confirmed. Among them were 67 organooxygen compounds, 29 fatty acyls, 14 carboxylic acids and derivatives, 8 saturated hydrocarbons, 8 phenols, 7 lactones, 6 prenol lipids and other types (*Table S1 in the supplemental material).
The 20 Chemical Components Present at the Highest Concentration in Himalayan Marmot Oil (HMO).
Gas Chromatography–Mass Spectrometry (GC–MS) Chromatogram of Himalayan Marmot Oil (HMO).
Cytotoxicity Test of HMO on L929 Cells
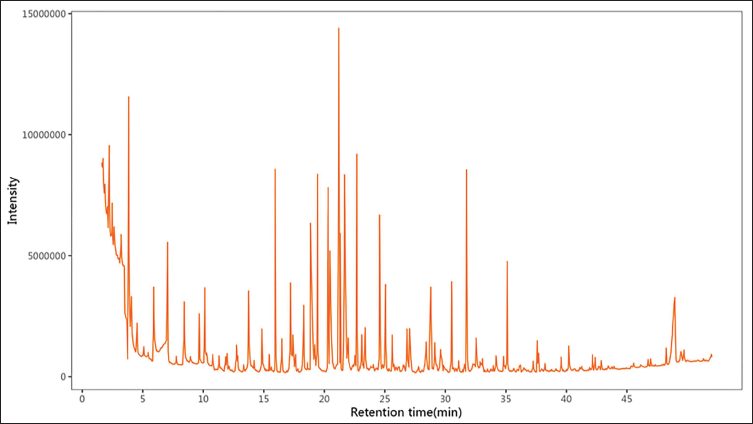
The CCK-8 assay was used to determine the cytotoxicity of HMO (1 µg/mL, 10 µg/mL, 100 µg/mL, 1,000 µg/mL) in L929 cells treated for 24 h, 72 h, and 120 h. As shown in Figure 2, HMO at 1 µg/mL, 10 µg/mL, 100 µg/mL, and 1,000 µg/mL had no toxicity to cells. In comparison to the control group (0 µg/mL), the cell viability of the group exposed to concentrations ranging from 1 to 1,000 µg/mL had no significant change. These results showed that the range between 1 µg/mL and 1,000 µg/mL HMO had no apparent effects on cell viability.
Cytotoxic Effect of Himalayan Marmot Oil (HMO) on L929 Cells. Cell Viability was Analyzed by CCK-8 Assay. Preincubation with Increasing Concentrations (from 0 to 1,000 µg/mL) of HMO in L929 Cells.
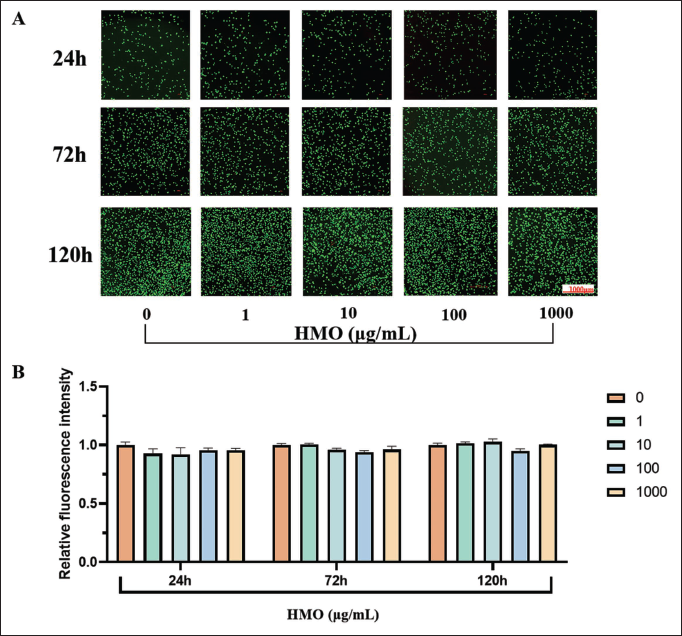
Furthermore, the evaluation of live/dead cell immunofluorescence staining (calcein-AM/PI) was performed to ascertain the cytotoxicity of HMO in L929 cells. The fluorescence image of cells is shown in Figure 3A. Adequate proliferation of live cells was found at 24 h, 72 h, and 120 h. These results were consistent with those obtained by the CCK-8 assay. The fluorescence intensity showed that HMO did not exhibit significant cytotoxicity in L929 cells at concentrations ranging from 0 to 1,000 µg/mL (Figure 3B). The abovementioned results validated that HMO at 0–1,000 µg/mL concentrations exhibits minimal cytotoxicity and favorable cell compatibility.
The Viability of L929 Cells Treated with Himalayan Marmot Oil (HMO) at Different Concentrations (0 µg/mL, 1 µg/mL, 10 µg/mL, 100 µg/mL, and 1,000 µg/mL) for 24 h, 72 h, and 120 h as Determined by Live/Dead Staining. (A) Images of Live/Dead Staining were Obtained Using Fluorescence Microscopy. (B) Relative Fluorescence Intensity in Each Group.
Anti-inflammatory Effects of HMO in an LPS-induced Cell Model
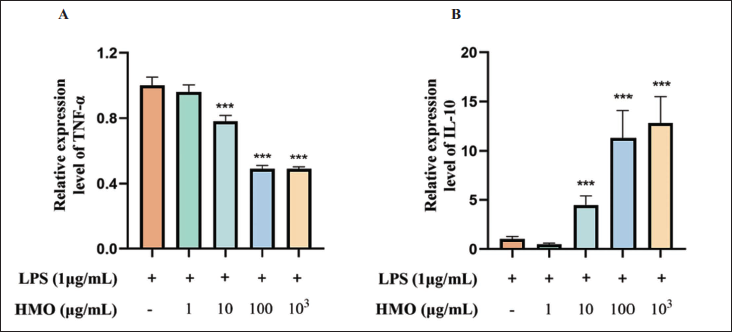
Previous studies have shown that inflammatory cytokines play a critical role in the progression of RA, and tumor necrosis factor-α (TNF-α) and interleukin-10 (IL-10) are essential cytokines involved in regulating the inflammatory response. Therefore, we performed qRT-PCR to identify the effects of HMO on the expression of TNF-α and IL-10. Compared with the LPS-induced group, the RNA expression of the intracellular pro-inflammatory factor TNF-α was decreased (Figure 4A), while that of the anti-inflammatory factor IL-10 was upregulated after treatment with HMO in RAW264.7 cells (Figure 4B). The regulatory effects of HMO on TNF-α and IL-10 were significantly dose-dependent. These results demonstrated that HMO could significantly inhibit LPS-induced cellular inflammation. The anti-inflammatory mechanism of HMO may be related to the inhibition of TNF-α expression and the promotion of IL-10 production
Effects of Himalayan Marmot Oil (HMO) on the Expression of Tumor Necrosis Factor-α (TNF-α) and Interleukin-10 (IL-10) by Quantitative Real-time Polymerase Chain Reaction (qRT-PCR). (A) Relative Expression Levels of TNF-α (n = 3). (B) Relative Expression Levels of IL-10 (n = 3). *p < 0.05, **p < 0.01, ***p < 0.001 versus the Lipopolysaccharide (LPS) Group.
Effect of HMO on the Body Weight of CIA Rats
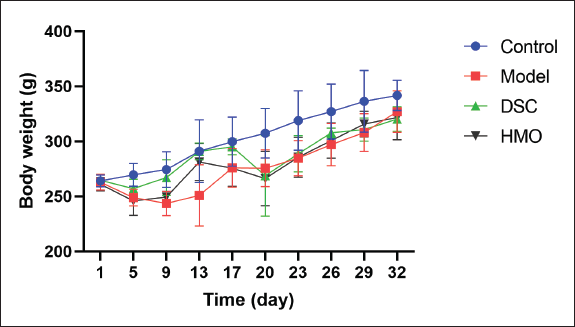
The body weight of the rats in this experiment was evaluated at 3- to 4-day intervals from day 1 to day 32. During the 32-day treatment period, the rats in the normal control group were in good health, with glossy fur, free movement, and a noticeable reaction to stimulation. In the model group, rats showed a weak state, rough and dull hair, poor mental health, and reduced diet and activity. After treatment, the HMO and DSC rats were in good spirits. The rats had a good diet and activity. As shown in Figure 5, the body weight of the control group rats increased steadily. However, the body weight slowly increased in CIA model rats, which lasted until the end of the drug administration. Compared with the model group, there was no significant difference in body weight change in the HMO group.
Effect of Himalayan Marmot Oil (HMO) on the Body Weight of Collagen-induced Arthritis (CIA) Rats (n = 6).
Effect of HMO on the Severity and Arthritis Score of CIA Rats
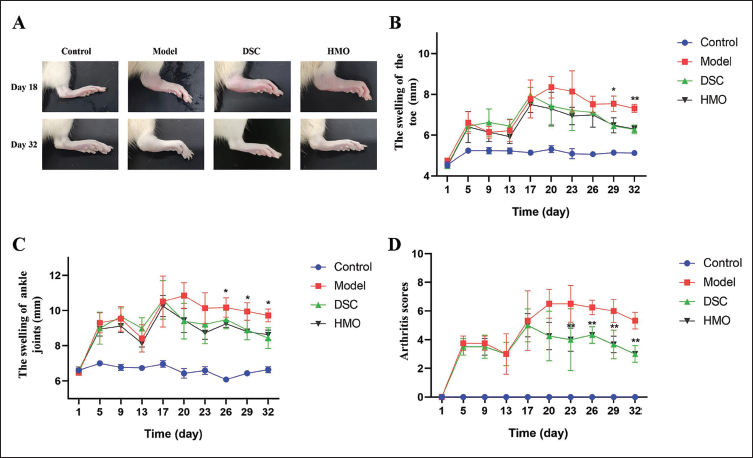
By observing the toe and ankle joints of rats (Figure 6A), it was found that CIA rats after modeling showed significant redness and swelling of the toe and ankle joints. The swelling condition and inflammation of the toe and ankle joints in the HMO-treated rats were significantly suppressed compared with those in the model group. As shown in Figure 6B and C, after the initial immunization, the rats showed an early local inflammatory response with a significant increase in toe and ankle joint swelling, which then gradually tapered off. After the second immunization, the swelling of the toe and ankle joints of the rats increased significantly again. The thickness of the toe and ankle joints of the rats gradually decreased from the beginning of treatment with HMO intervention. Eventually, compared with the model group, the toe and ankle joint swelling of the rats in the HMO treatment group was significantly improved. As shown in Figure 6D, the arthritis score of the model group rats was significantly higher than that of the normal control group. After drug treatment, arthritis scores were significantly lower in the HMO and DSC groups from day 18 than in the model group. These results suggest that HMO exhibits a significant anti-RA effect in rats with CIA.
Effect of Himalayan Marmot Oil (HMO) on Foot Swelling and Arthritis Score of Collagen-induced Arthritis (CIA) Rats (n = 6). (A) Representative Images of Hind Paws from Rats in Each Group. (B) Swelling of the Toes of CIA Rats. (C) Swelling of Ankle Joints of CIA Rats. (D) The Arthritis Score of CIA Rats. *p < 0.05, **p < 0.01, ***p < 0.001 versus Model Group.
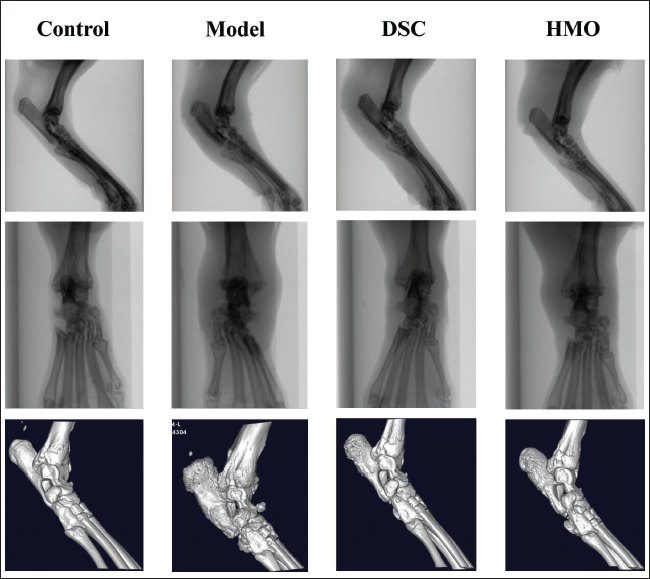
Effects of HMO on Ankle Joint Imaging in CIA Rats
The computed tomography (CT) images of the hind paws of the rats were obtained by scanning and 3D reconstruction of the micro-CT system (Figure 7). The results showed that the bone and joint gaps observed in the control group of rats exhibited distinct characteristics, including regular shapes, organized arrangements, and firm and intact bone structures. The rats in the model group showed obvious tissue destruction, blurring and narrowing of the joint space, formation of many oval or round bone fragments, bone erosion, and destruction of joint integrity. Compared to the model group rats, the HMO group showed significantly better joint destruction, clearer joint gaps, fewer bone fragments, and better bone erosion. The imaging results demonstrated that HMO could improve the degree of bone destruction in the CIA animal model.
Effects of Himalayan Marmot Oil (HMO) on Ankle Joint Imaging in Collagen-induced Arthritis (CIA) Rats.
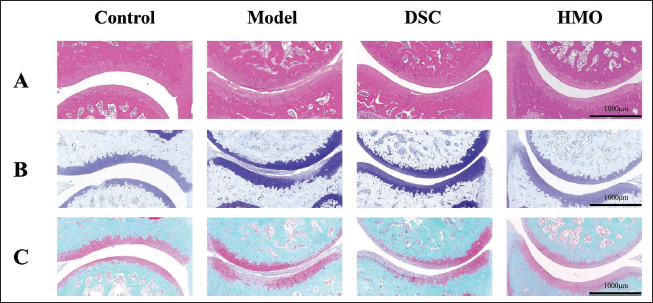
Effects of HMO on Pathological Features in CIA Rats
The HE staining results (Figure 8A) showed that the joint structure of the blank control group was normal, the joint cavity was clear, the surface of articular cartilage was smooth and intact, and the synovial cells were neatly arranged without inflammatory cell infiltration. In the model group, the joint structure was severely damaged, the joint cavity was narrowed, and the cartilage was damaged. In the HMO group, the joint cavity was widened, synovial hyperplasia was reduced, the synovial surface was smoother, inflammatory cell infiltration was reduced, and fewer vessel opacities appeared. The results of HE staining showed that HMO effectively reduced proliferation and inflammatory cell infiltration in rat joints and inhibited cartilage erosion and vascular opacification formation.
In addition, we conducted TB staining (Figure 8B) and safranin-O/fast green staining (Figure 8C) to evaluate the degree of articular cartilage damage. The staining results indicated a significant reduction in thickness and severe erosion of the articular cartilage in the experimental rats. In contrast, joint erosion was significantly suppressed in the HMO group. These results suggest that HMO can effectively improve the severity of arthritis after CIA.
Effects of Himalayan Marmot Oil (HMO) on Pathological Features in Collagen-induced Arthritis (CIA) Rats. (A) Hematoxylin–Eosin (HE) Staining. (B) Toluidine Blue (TB) Staining. (C) Safranin-O/Fast Green Staining.
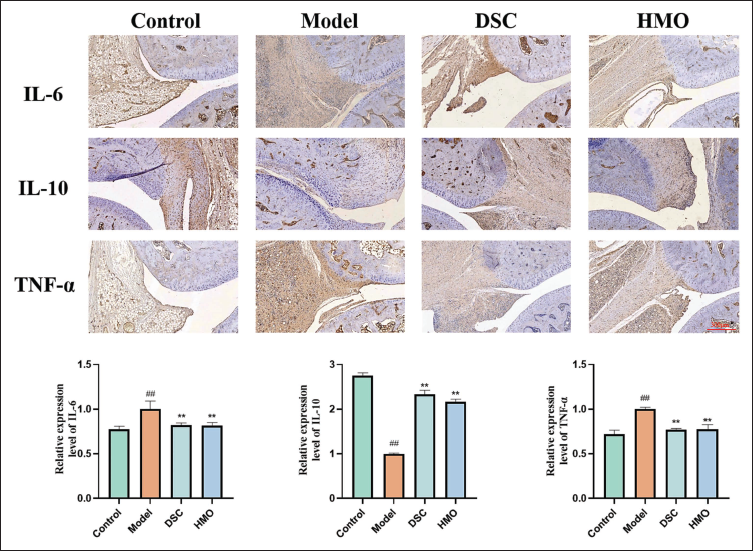
Anti-inflammatory Effects of HMO in CIA Rats
Inflammatory cytokines in ankle joints were further investigated by immunohistochemistry. As Figure 9A shows, the expression levels of interleukin-6 (IL-6), IL-10, and TNF-α in the HMO group were more similar to those in the control group. The quantitative analysis of immunohistochemical staining (Figure 9B) showed that IL-10 expression was significantly increased in rats after HMO administration compared with the model group, while the expression of IL-6 and TNF-α was inhibited. Therefore, HMO can reduce the inflammatory response in CIA rats. These effects could potentially be modulated through the inhibition of IL-6 and TNF-α, which are pro-inflammatory factors, and increasing the augmentation of IL-10, an anti-inflammatory factor.
The Results of Immunohistochemical Analysis in Collagen-induced Arthritis (CIA) Model Rats. (A) Image of Immunohistochemical Staining in CIA Model Rats. (B) Relative Expression Levels of Interleukin-6 (IL-6). (C) Relative Expression Levels of Interleukin-10 (IL-10). (D) Relative Expression Levels of Tumor Necrosis Factor-α (TNF-α). ##p < 0.05 versus the Control Group; **p < 0.01, ***p < 0.001 versus the Model Group.
Discussion
RA is an autoimmune inflammatory disease with persistent synovitis, progressive cartilage destruction, and osteoporosis as the main manifestations (Liu et al., 2021). As the disease progresses, RA can also be associated with mild or severe disability, which can affect the patient’s quality of life (Machin et al., 2020). Studies have shown that inflammatory cells secrete more pro-inflammatory factors, including IL-1β and IL-6, to aggravate inflammation when TNF-α is activated in synovial tissues. Therefore, inhibition of TNF-α expression is a crucial factor in the treatment of RA (Kondo et al., 2021; Zamri & de Vries, 2020). The absence of IL-10 may lead to the exacerbation of RA inflammation and expedite the advancement of the condition (Ye et al., 2014). Therefore, as an immunosuppressive cytokine, IL-10 can effectively inhibit the activation of inflammatory factors in macrophages and has a potential therapeutic effect on the treatment of RA (Chen et al., 2017). In this study, the expression of IL-6, TNF-α, and IL-10 in the synovium of rats with CIA was assessed using immunohistochemistry. Our findings revealed that HMO effectively suppressed the secretion of the pro-inflammatory cytokines IL-6 and TNF-α while enhancing the expression of the anti-inflammatory cytokine IL-10, thereby mitigating the inflammatory response. These results also preliminarily explain why the therapeutic effect of HMO on RA may occur through its immunomodulatory effect, inhibiting the synthesis and release of pro-inflammatory factors such as TNF-α and IL-6 and promoting the synthesis of anti-inflammatory factors such as IL-10.
The treatment goal of RA is to achieve clinical, structural, and functional remission (Maeda et al., 2022). Clinical remission is defined as the alleviation of inflammatory symptoms and signs, such as joint swelling and tenderness, which can be assessed through the clinical disease activity index (CDAI), disease activity index (DAS-28), and simplified disease activity index (SDAI). Structural remission means that the destruction of bones and joints is almost stopped and can be evaluated by the Sharp score (van der Heijde, 1996). Functional remission refers to a state in which body function no longer declines, which can be evaluated by a health assessment questionnaire. Through drug intervention, pain and swelling may be quickly eliminated, and clinical relief can be achieved. However, from the analysis of joint structure, it may be found that the degree of joint damage is still occurring and will continue to worsen and lead to dysfunction. Therefore, it is very important to achieve mitigation of these three states.
CIA could best reflect the clinical and pathological characteristics of RA, and it is currently the gold standard model for RA study. Therefore, in this study, we followed traditional use and applied HMO directly to the ankle joints of CIA rats in a single dose. Our data showed that the body weight of rats treated with HMO showed a dynamic upward trend, indicating that the state of rats began to recover. Comparing the swelling degree of the ankle joint and toe, it can be seen that HMO can significantly eliminate the symptoms of local joint swelling and redness caused by RA, indicating that HMO can effectively relieve the inflammatory reaction. The rats treated with HMO exhibited noteworthy enhancements in joint destruction, synovial hyperplasia, and pannus formation in micro-CT imaging compared to the CIA model group. These outcomes demonstrated significant differences, suggesting that HMO treatment possesses a certain efficacy in alleviating the structural alterations associated with RA.
HMO is extracted from the fatty oil of the Himalayan marmot of the squirrel family. Analysis by GC-MS revealed that HMO and its active components have several pharmacological effects. It contains many oleic acid and PUFAs, which means it may have anti-inflammatory and bacteriostatic properties (Vila et al., 2022). In addition, HMO is rich in carboxylic acids, methoxyphenols, and dihydrofurans. These active ingredients have good anti-inflammatory and antioxidant properties (Huang et al., 2015; Kim et al., 2019; Ren et al., 2020). Therefore, HMO is suitable for anti-rheumatism swelling and pain treatments. Based on the traditional application of Tibetan medicine, HMO is an oily substance derived from the adipose tissue of marmots after heating and extraction to remove water (Ma et al., 2003; Wang et al., 2000). The cytotoxicity of HMO has been investigated and determined to be significantly minimal. In this study, we explored the extraction process of HMO and selected the optimal extraction scheme for follow-up research. The extraction rates of HMO extracted by this method are higher, and it does not contain extra moisture. In this way, the HMO is less prone to deterioration and is convenient for later preservation. The results of CCK-8 and cell staining showed that HMO had good biocompatibility and low toxicity. To verify the efficacy and safety of HMO in the treatment of RA, we established an inflammatory model with RAW cells and treated them with HMO. The results of qRT-PCR analysis showed that compared with the control group, HMO significantly inhibited the secretion of the pro-inflammatory factor TNF-α and promoted the expression of the anti-inflammatory factor IL-10 in a dose-dependent manner. Although the specific mechanism of HMO and the regulation of related molecular channels were not profoundly investigated in this study, the available results demonstrate that HMO exhibits significant anti-inflammatory properties in vitro and in vivo with less toxicity and thus holds great promise for the treatment of RA inflammation.
Conclusion
Taken together, the current work identified an excellent anti-inflammatory animal-derived medicinal agent HMO in alleviating synovial inflammation and joint destruction by regulating IL-6, IL-10, and TNF-α expression. It also showed low toxicity both in vitro and in vivo, which provides a protective effect against RA. As a traditional medicine, our study expands the topical value in the application of HMO to inhibit the progression of inflammation. It could be a safe, novel RA complementary therapy.
Footnotes
Abbreviations
CIA: Collagen-induced arthritis; DMEM: Dulbecco’s Modified Eagle’s Medium; GCs: Glucocorticoids; GC-MS: Gas chromatography–mass spectrometry; HE staining: Hematoxylin–eosin staining; HMO: Himalayan marmot oil; IL-6: Interleukin-6; IL-10: Interleukin-10; LPS: Lipopolysaccharide; NSAIDs: Non-steroidal anti-inflammatory drugs; PUFAs: Polyunsaturated fatty acids; qRT-PCR: Quantitative real-time polymerase chain reaction; RA: Rheumatoid arthritis; TB staining: Toluidine blue staining; TNF-α: Tumor necrosis factor-α.
Acknowledgments
The authors would like to thank Qingquan Kong and Zhifeng Zhang for their valuable contributions to this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animal experiments conducted in this study adhered to the guidelines set forth by the European Union for experimental animals. This study has been approved by the Division of Animal Control and Inspection, Department of Food and Animal Inspection and Control, Instituto para os Assuntos Cívicos e Municipais (IACM), Macao (Approval number: AL072/DICV/DIS/2021).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sichuan Science and Technology Program (2020YFQ0007 to Professor Q. Q. Kong), Science and Technology Major Project of Tibetan Autonomous Region of China (XZ202201Z0001G to Professor Q, Q. Kong), Science and Technology Project of Tibet Autonomous Region (XZ202303ZY0007G to Professor Q. Q. Kong), and the Macao Science and Technology Development Fund (0043/2021/AGJ to Dr. Z. F. Zhang).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
