Abstract
Background
Rheumatoid arthritis (RA) is a chronic, immune-inflammatory disease that could be managed by an attenuation in dietary habits. With the increase in research that links the effect of nutrition linked to RA.
Objectives
Our study aimed to determine the possible therapeutic effect of different types of milk in rat models experimentally induced with RA using 40 ml milk (camel (CM), goat (GM), sheep (SHM), cow (CWM), almond (AM), and soy milk (SM)).
Materials and Methods
Forty adult male Wistar Albino rats were divided into eight groups (five rats each) as follows: (Control group): nonarthritic, (RA): rheumatoid arthritis, (RA+CM): RA having CM, (RA+GM): RA having GM, (RA+SHM): RA having SHM, (RA+CWM): RA having CWM, (RA+AM): RA is having AM, and (RA+SM): RA has SM.
Results
It has been observed that in the RA group and milk-treated groups, but not in the RA+CM group, the arthritis score changed significantly (p ≤ 0.05) in comparison with the control group. CM and GM showed promising effects in downregulating various arthritis effector players including anticitrullinated peptide antibodies, C-reactive protein, prostaglandin E2, and other inflammatory mediators, which may be attributed to their lowest ω-6/ω-3 polyunsaturated fatty acid ratios.
Conclusion
A possible benefit of this research is the potential development of new functional foods that promote health, prevent disease, and potentially enable the milk to be used as a therapeutic agent for RA.
Introduction
Rheumatoid arthritis (RA) affects the adult population globally (McInnes & Schett, 2011). It is described by inflammation of the joint, chronic proliferative synovitis that ends in articular cartilage and bone destruction and joint deformity, and may also have prominent extra-articular features (Lee et al., 2007; Scott et al., 2010). Inflammation is a defensive response to injury or infection triggered due to physical trauma, harmful chemical substances, or microbial agents to eliminate or weaken the invading substance and to establish the conditions to heal and repair the tissues infected by these agents. It is generally characterized by pain, heat, and redness (Prakash et al., 2013). The inflammatory mediator’s production in RA has been linked to various genetic and environmental factors, including chronic stress, infectious agents, tobacco use, and dust exposure (Catrina et al., 2016; Silman & Pearson, 2002; Walker et al., 1999).
Despite its onset being unrestricted, studies suggest that it occurs mainly among 30–55 year old with females experiencing the disease 3–5 times as often as males. Older peoples tend to suffer more from this condition (Mendoza-Vázquez et al., 2013; Pandey, 2010). The diagnosis of RA is rather challenging as its symptoms can closely mimic other diseases, and it depends mostly on clinical information congregated from the history and physical examination of patients along with ultrasound imaging analysis and various blood tests. However, its prompt diagnosis works as a vital improvement factor for desirable outcomes such as lesser joint destruction and prevention of functional disability (Cho et al., 2019; Moura et al., 2015). Its pain affects life quality, impairs physical and social functioning, and raises healthcare costs (da Rocha Castelar Pinheiro et al., 2013).
The illness (RA) is systemic at all stages, rather than a prototypical autoimmune disorder in which sensitivity or inability to tolerance to a tissue-specific antigen leads to a selective, organ-destructive immune response (Goronzy & Weyand, 2009). The augmented manifestation of cytokines and transcription factors are the mechanisms intricate in joint destruction and the various interleukins (ILs) are the cytokines involved in the progress of arthritis (Zhu et al., 2016). Nonsteroidal anti-inflammatory medications (NSAIDs), disease-modifying antirheumatic drugs (DMARDs), historical (minocycline, auranofin, and cyclosporine), and biologic DMARDs. Antitumor necrosis factor-alpha (TNF-α) antibodies (Amjevita, Erelzi) and IL-6 receptor antibody (Kevzara) were used for its treatment (Guo et al., 2018; Scott et al., 2010; Smolen et al., 2014). These treatments have greatly improved and controlled the inflammation, but the pain persists even when the synovitis has been successfully suppressed (Altawil et al., 2016; McWilliams & Walsh, 2016; McWilliams et al., 2012). In addition, prolonged use of these medications has been linked to side effects, including gastric irritation that can lead to gastric ulcer (Gao et al., 2020) and the cost of these medications for long-term treatment poses another concern. Therefore, more studies on using alternative therapies are required.
Dietary habit plays a substantial role in human health and increasing reports from the past few years support the role of nutrition in RA.
Consuming fish oil in mice delayed the onset and decreased the severity of type II collagen-induced arthritis, compared to animals given vegetable (Leslie et al., 1985). Goldberg and Katz (2007) suggested that any person having RA should consume 3–6 g of ω-3 fatty acids every day for 12 weeks. After the consumption of ω-3 supplements for this time frame, the intensity of joint pain and the need for NSAIDs dose decreased in patients (Goldberg & Katz, 2007). Milk and dairy products are considered as balanced nutritious foods in the human diet. Researchers have reported that feeding CM for 3 weeks to arthritic mice at 10 ml/kg decreased the level of TNF-α and improved the anti-inflammatory IL-10 levels in serum (Arab et al., 2017). So far, no studies have been conducted to analyze and compare the therapeutic potential of various types of milk in rat models artificially produced with RA. Complete Freund’s adjuvant (CFA) stimulates acute local inflammatory reactions, including arthritis, and produces results identical to human RA. In this work, we investigated the possible therapeutic advantages of different types of milk in RA-induced rat models. In this study, we therefore explored the potential therapeutic benefits of various types of milk in RA-induced rat models. In this study, we therefore explored the potential therapeutic benefits of various types of milk in RA-induced rat models. A possible benefit of this research is the potential development of new functional foods that promote health, prevent disease, and potentially enable the milk to be used as therapeutic agent for RA.
Materials and Methods
Materials
All types of milk (camel (CM), goat (GM), sheep (SHM), cow (CWM), almond (AM), and soy milk (SM)) were purchased from a local market, Riyadh region, Saudi Arabia in 2019.
Milk Sample Preparation
First, 20 ml of each sample of fresh milk was placed in a beaker and frozen for the whole night at −30°C. Then the frozen samples were freeze-dried in an Alpha 1–2 LD plus freeze dryer (Martin Christ, Osterode am Harz, Germany).
The freeze-dryer was set for 20 minutes of initial freezing at –51°C and 0.035 bar, followed by 12 hours of primary drying at 30°C and 0.10 mbar pressure to achieve a fine powder. The milk powder was stored in the freezer at –30°C for future investigations.
Fatty Acids Analysis
To obtain fatty acid methyl esters (FAMEs), milk was methylated (brochures number MAK224) and then purified with a FAME purification kit (brochures number MAK225) from Sigma-Aldrich Company using the manufacturer’s instructions.
The resultant FAMEs were investigated with gas chromatography. The fatty acids were separated using an Omegawax™ 320 capillary column (30 m × 0.32 mm i.d. × 0.25 µm film thickness, Supelco, Inc., Bellefonte, PA, USA) at an oven temperature of 200 °C; carrier gas helium 25 cm/s at 200°C; detector, flame ionization (FID) 260°C; injection, 1 µl divided 100:1 at 250°C. FAMEs in samples were identified by comparing the retention periods of standard fatty acids (Supelco, Inc., Bellefonte, USA), polyunsaturated fatty acid (PUFA) 2, animal source, and FAME Mix RM-1, oil reference (Sigma-Aldrich, St. Louis, USA), respectively (Lepage & Roy, 1986).
Animal Model
Forty adult male Wistar Albino rats (200 ± 20 g) were provided and maintained in the animal house of experimental Animal Care Center, College of Education, King Saud University, Saudi Arabia. Animals were housed individually in plastic cages at 22 ± 1°C, a 12/12-hour cycle (dark/light), and a humidity of 55–60%. Free access to normal food and drinking water was guaranteed throughout the entire experimental period. Furthermore, the rats were habituated to the lab setting for a duration of seven days prior to the commencement of the study. The rats were divided equally into eight (five rats each) different groups based on their weight.
Group 1 (Control group): Non-arthritic rats (control negative) normal rats + 30 g standard diet (nonarthritic).
Group 2 (RA): RA-induced rats (control positive) + 30 g standard diet.
Group 3 (RA+CM): RA-induced rats + 30 g standard diet + 40 ml CM.
Group 4 (RA+GM): RA-induced rats + 30 g standard diet+ 40 ml GM.
Group 5 (RA+SHM): RA-induced rats + 30 g standard diet+ 40 ml SHM.
Group 6 (RA+CWM): RA-induced rats + 30 g standard diet+ 40 ml CWM.
Group 7 (RA+AM): RA-induced rats + 30 g standard diet+ 40 ml AM.
Group 8 (RA+SM): RA-induced rats +30 g standard diet+ 40 ml SM.
Ethics
All the procedure of this study was approved by King Saud University ethical committee (Reference #: KSU-SE-19-21).
Animal Diet
The rats were fed on a basal diet prepared based on the American Institute of Nutrition-93 diet formulated for maintenance (AIN-93M) diet (Reeves, 1997). Throughout the experimental period, they were fed with 30 g/day diet and had free access to drinking water
Induction Arthritis in Animals
In this experiment, arthritis was induced in rats according to the methods of Ahmed et al. (2019) and Zhang et al. (1999) with little modification.
Thirty-five animals (groups 2–8) were sedated with CO2 inhalation before being injected once intradermally in the right hind paw with CFA (0.1 ml, Cat. No. 7024, Chondrex Inc., Washington, USA) including 10 mg/ml of heat-killed Mycobacterium tuberculosis. On day 0, the CFA injection was delivered. Between days 14 and 21, rats with arthritis showed increased paw thickness, redness, and edema.
Measurement of Growth Indicators
The difference between each rat’s weight at the start of the study (Wi) and at the end (Wf) was used to calculate its weight gain (Wg): Wg (g) = Wf – Wi (Paiz et al., 2010)
Food efficiency (FE): The ratio of weight gain (Wg) to total food intake (TFI) was used to estimate FE: FE = (Wg (g))/TFI (Gidenne & Maertens, 2016)
Milk efficiency (ME): The ratio of weight gain (Wg) in grams to total milk consumption (TMC) was used to calculate ME: ME = (Wg (g))/TMC
Evaluation of Arthritis Score
The severity of RA in each paw of rats was assessed as previously described by Cui, Wang et al. (2019), based on a scale from 0 to 4 as follows:
Grade 0.0: Absence of swelling.
Grade 1: Presence of mild swelling or redness in one of the fingers in the paw.
Grade 2: Presence of swelling/redness in one or more than one finger of the paw.
Grade 3: Presence of swelling of foot or ankle.
Grade 4: Presence of swelling in the whole paw including all fingers, foot, and ankle.
The clinical arthritis scores (0–4 scale) were evaluated for each limb, and the maximum score was 16 (for 4 limbs). The maximum score based on this method was 16.
Serum Collection and Biochemical Measurements
All rats were anesthetized with CO2 at the end of the experiment (day 28). The blood samples (3 ml) were then collected directly from the heart into simple tubes by cardiac puncture. This blood was collected in a tube and centrifuged for 10 minutes at room temperature at 3,000 rpm. The serum obtained was stored at −80°C for further analysis and measurement of inflammatory indicators in the blood.
Pro-inflammatory and Anti-inflammatory Cytokines
Serum levels of TNF-α, IL-1β, IL-6, and IL-10 in serum were determined using rat cytokine/chemokine magnetic bead panel, Merck KGaA Kit (Cat. No. RECYTMAG-65K Darmstadt, Germany) according to the manufacturer’s instructions.
Prostaglandin E2 (PGE2), C-reactive Protein (CRP), Anticitrullinated Peptide Antibodies (ACPA), and Rheumatoid Factor (RF) Levels
Serum levels of RF (Cat. No. MBS9399647), CRP (Cat. No. MBS564107), ACPA (Cat. No. MBS762429), and PGE2 (Cat. No. MBS7606497) were determined using My BioSource assay kit (My BioSource, CA, USA) following the manufacturer’s instructions.
Statistics
Results are presented as mean ± standard deviation. We used one-way analysis of variance (ANOVA) to determine the statistical significance of our results. The p value was assumed to be significant at p ≤ 0.05. Study groups were further compared using Duncan’s multiple range tests when the overall p value was statistically significant (p ≤ 0.05). Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS, version 22. Inc., Chicago, IL, USA).
Results
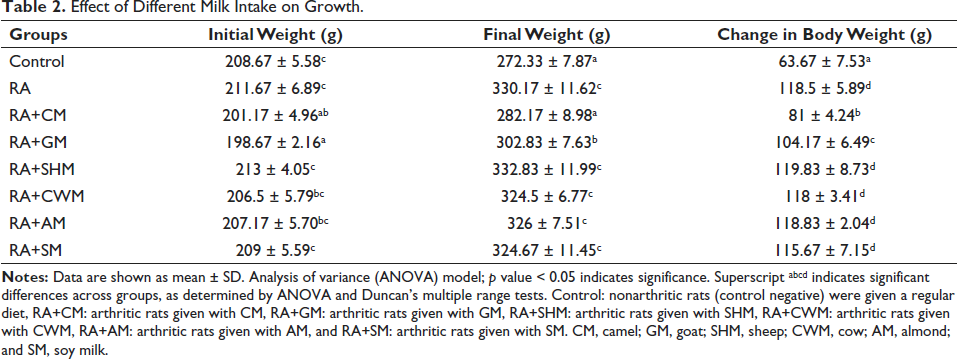
Total ω-3, ω-6, and ω-6/ω-3 Ratios Obtained from Different Milks
Table 1 represents the total ω-3, total ω-6, and ω-6/ω-3 ratios of milk obtained from different animals. The highest amount of ω-3 was observed in GM followed by CM > CWM > AM > SHM. Similarly, the highest amount of ω-6 was noted in CWM followed by GM > CM > SM > AM > SHM. A high PUFA ratio (ω-6/ω-3) is believed to have adverse health effects and from Table 1 it can be predicted that the lowest PUFA ratio (ω-6/ω-3) was found in CM and the highest PUFA ratio (ω-6/ω-3) was found in cows’ milk.
ω-3, ω-6, and ω-6/ω-3 Ratios Obtained from Different Milks (g/100 g).
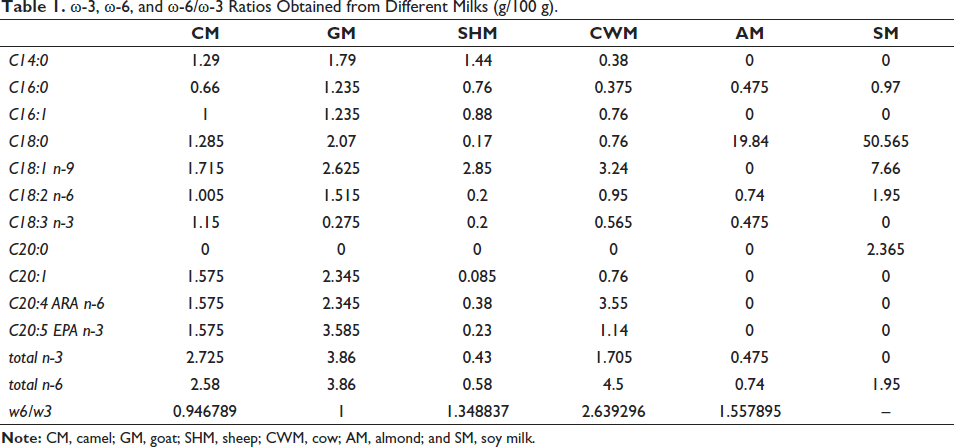
Effect of Different Milk on External Morphological Features of the Rat’s Paws and Arthritis Score
Figure 1 presents the outcome of different types of milk consumption on external morphological features of the rat’s paws and arthritis score. RA-induced rats developed severe swelling, redness, pain, and limping joint deformities in almost all paws. In the RA group and milk-treated groups, but not in the RA+CM group, the arthritis score changed significantly (p ≤ 0.05) in comparison with the control group. The arthritis score observed in the RA group of rats was 11.62 ± 1.42 and a significant reduction in the arthritis score with a concomitant reduction in the degree of swelling, redness, and joint deformities was observed in the RA-induced rats, which received CM or GM (0.92 ± 0.66 and 8.08 ± 0.86, respectively). On the other hand, insignificant differences were observed in the other groups (RA+SHM, RA+CWM, RA+AM, RA+SM) when compared with the RA group, and the same degree of paw swelling, redness, limpness, and joint deformities were observed in these groups.
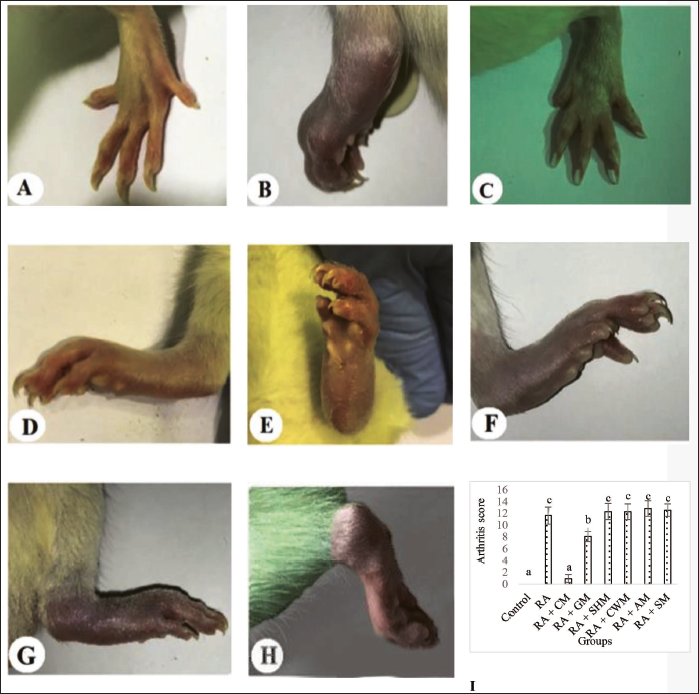
Different Milk on Growth Indicators and FE and ME
Table 2 shows the outcome of different treatments on growth indicators in the control and experimented groups. It has been noted that in comparison with those in the control group, final weight and body weight change in the RA and RA+milk treatment groups significantly (p ≤ 0.05) increased. A significant (p ≤ 0.05) reduction in rats’ final body weights was observed in RA+CM and RA+GM when compared to the RA group. However, the insignificant (p ≥ 0.05) difference in final body weights of groups that consumed sheep (RA+SHM), cow (RA+CWM), almond (RA+AM), and soy (RA+SM) milk was observed in comparison with the RA group. An almost similar pattern was seen in weight gain. FE significantly (p ≤ 0.05) reduced in the RA group when compared with control, but insignificant change was observed in RA+CM and RA+GM group when compared with control. Similarly, while comparing with RA group, insignificant (p ≥ 0.05) difference in FE was observed in arthritic groups that consumed SHM, CWM, AM, or SM. ME of the RA+CM group was highest followed by RA+GM and the least efficiency was observed in the RA+SM group (Table 2 and Figure 2).
Effect of Different Milk Intake on Growth.
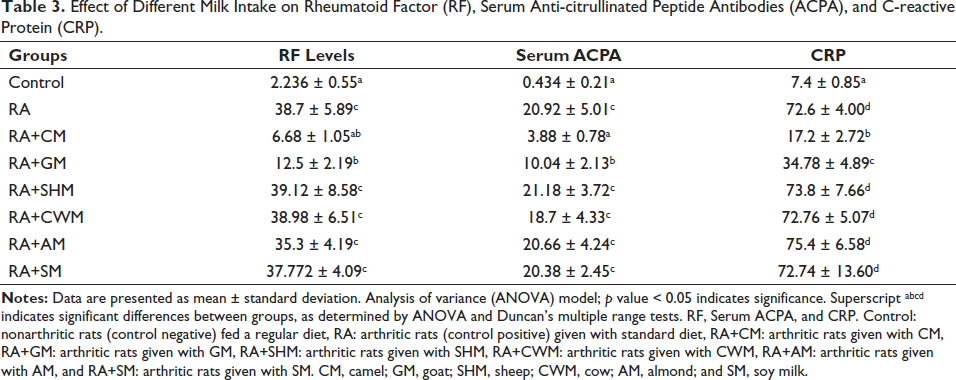
Effect of Different Milk on RF, Serum ACPA, and CRP
Table 3 summarizes the effect of different types of milk on serum levels of RF, ACCPA, and CRP. The serum RF levels, serum ACPA levels, and serum CRP levels increased significantly (p ≤ 0.05) in the RA group in comparison with the control group. Treatment with CM (RA+CM) and GM (RA+GM) has a significant (p ≤ 0.05) effect on serum RF levels, serum ACPA, and serum CRP, and a significant decrease has been observed in these two groups when compared with the RA group. Contrary to this, the difference was insignificant (p ≥ 0.05) between other milk-treated groups (RA+SHM; RA+CWM; RA+AM; RA+SM) and RA group.
Effect of Different Milk Intake on Rheumatoid Factor (RF), Serum Anti-citrullinated Peptide Antibodies (ACPA), and C-reactive Protein (CRP).
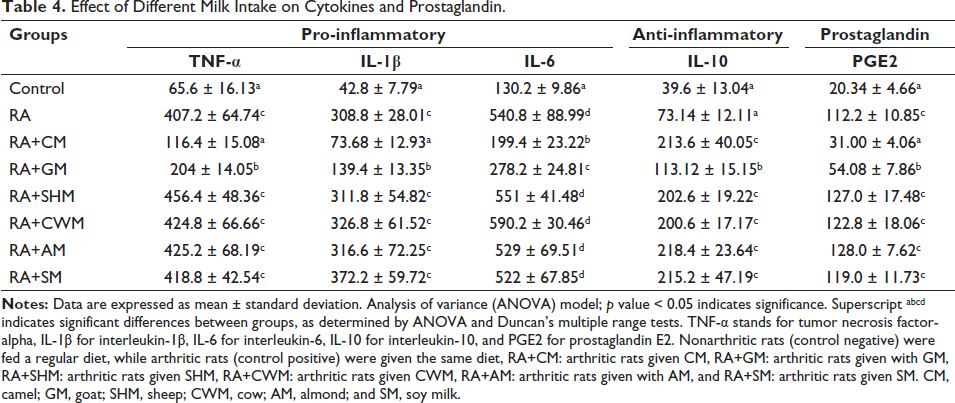
Effect of Different Milk on Cytokines (Pro-inflammatory and Anti-inflammatory) and Prostaglandin
Table 4 lists the effect of different treatments on cytokines (TNF-α, IL-1β, IL-6, and IL-10) and prostaglandin (PGE2). As established by the CFA-induced arthritis model, the pro-inflammatory cytokines significantly (p ≤ 0.05) increased in RA group in comparison with those of control group. When treatment with milk was considered, a significant reduction in the pro-inflammatory cytokines was observed between the CM and GM groups, whereas there was no significant difference between the RA group and other milk-treated groups (RA+SHM, RA+CWM, RA+AM, RA+SM). Contrary with this, an upsurge in anti-inflammatory cytokine (IL-10) was observed in all milk-treated groups when compared with RA group. Levels of PGE2 increased significantly (p ≤ 0.05) in the serum of RA-induced rats in comparison with the control rats. In CM and GM significantly reduced PGE2 level was observed when compared with the RA group, while an insignificant effect of other milk (RA+SHM, RA+CWM, RA+AM, and RA+SM) was observed on PGE2 level.
Effect of Different Milk Intake on Cytokines and Prostaglandin.
Discussion
In this study, we assessed the therapeutic effects of several animal milk and other milk types against RA in an animal model and also examined the essential fatty acid content and their proportions in these milk types. Essential fatty acids of milk such as linoleic acid (ω-6) and α-linolenic acid (ω-3) are major components of PUFAs and are essential for human health as these cannot be synthesized by humans (Hu et al., 2001). However, the ratio of ε6 and ω-3 determines the actual health benefits of milk as a high ω-6/ω-3 PUFA ratio is believed to have adverse health effects while a low ratio seems beneficial (Jensen, 2002). In our study, we found that the highest amount of ω-3 was found in GM followed by CM and the highest amount of ω-6 was found in CWM followed by GM and CM. However, the lowest ω-6/ω-3 PUFA ratio was found in CM and highest in cows’ milk. To study the beneficial health effects of different milk types against RA, we used an arthritic rat model (Ahmed et al., 2019; Zhang & Davidson, 1999). Arthritis was induced in the rats by injecting heat-killed M. tuberculosis in CFA. Morphological changes, RA factor levels, and arthritis scores all confirmed the development of arthritis in these rats (Cui, Cui et al., 2019). These arthritic rats were subsequently used to test the therapeutic effects of different kinds of animal milk and also AM and SM. In the present study, we found that RA rats fed on different milk types demonstrated varying morphological features and arthritis scores. As compared to arthritic rats, a significant reduction in the arthritis score with a concomitant decrease in the degree of swelling, redness, and joint deformities was observed in arthritic rats that received CM or GM. On the other hand, insignificant differences in arthritis scores and morphological changes were observed between arthritic rats and those that received other milk types, demonstrating the superiority of CM and GM in downgrading arthritis effects. Similar to our findings, CM and GM have been shown previously to possess antiarthritic properties (Arab et al., 2017; Rohmah et al., 2015). Arthritic rats fed camel and GM also showed lower body weight and weight gain, as well as increased efficiency of food and milk intake, compared to arthritic rats. However, arthritic rats fed other types of milk resulted in lower food and milk intake efficiency. Importantly, despite the reduction in food and milk intake efficiencies these rats gained significant body weight. This could partly explain the reduction in arthritis symptoms in rats that consumed camel and GM compared to those that received other milk types. Generation of ACPA and inflammatory molecule, CRP is the hallmark of RA. ACPAs are autoantibodies that act on citrullinated peptides and play a major role in arthritis pathology (Behl et al., 2021). Similarly, elevated CRP levels are recorded in inflammatory diseases including RA (Qin et al., 2022). Our current data showed that arthritis rats exhibited elevated ACPA and CRP levels and these levels were significantly downregulated in those fed on camel or GM. No significant effect on ACPA and CRP levels was noted in arthritis rats that consumed other milk types tested in the study. To the best of our knowledge, this study is the first to observe the capacity of CM and GM to lower ACPA levels in arthritic rats. Consistent with our finding, CM was previously reported to alleviate CRP levels in mice intoxicated with carbon tetrachloride (Hamed et al., 2018). Inline, goat cheese, naturally enriched in PUFAs (ω-3 PUFA) and conjugated linolenic acid (CLA), significantly decreased CRP levels in overweight and obese subjects (Santurino et al., 2020). As the overactivated immune system of the body plays a critical role in RA etiology (Mueller et al., 2021), we sought to understand the capacity of studied milk types to lower inflammatory parameters. Monocytes and macrophages infiltrate synovial membranes and are activated to produce inflammatory mediators by activating transcription factor nuclear factor kappa B (NF-κB) (Guo et al., 2018). As expected, our data showed increased TNF-α, IL-1β, IL-6, and IL-10.
Levels in arthritic rats. Milk and milk-derived products have been shown previously to lower the symptoms of arthritis through downmodulating inflammatory effectors (Aarts et al., 2021). For example, CM has been shown to prevent anti-inflammatory actions in adjuvant-induced arthritis through attenuating TNF-α, NF-κBp65, cyclooxygenase-2 (COX-2), and inducible nitric oxide synthase (iNOS) by the inhibition of the mitogen-activated protein kinase (MAPK) pathway signaling molecules including p38 MAPK, extracellular signal-regulated kinase 1/2 (ERK1/2), and c-Jun N-terminal kinases 1/2 (JNK1/2[23]). Consumption of Lactobacillus casei fermented milk blunted the expression of IL-17, TNF-α, IL-23, IL-6, and IL-1β in lymph nodes (Noto Llana et al., 2013). T helper 17 cells (Th17) and gamma delta T cells (γδ T) cells, which play a role in reactive arthritis, require these cytokines to differentiate. The movement of these inflammatory cells from the stomach to the joints has been hypothesized as a mechanism for the development of reactive arthritis. Likewise, oral administration of bovine milk-derived extracellular vesicles attenuates arthritis in mouse models by lowering monocyte chemoattractant protein-1 (MCP-1) and IL-6 levels (Arntz et al., 2015). Milk fat globule-epidermal growth factor 8 was found to be a novel anti-inflammatory factor and able to mitigate RA in mice and humans by decreasing inflammatory cytokine levels (Albus et al., 2016). In an arthritic model, a low-molecular-weight component obtained from hyperimmunized cow milk inhibits neutrophil emigration, suppressing inflammation (Ormrod & Miller, 1992). In vivo and in vitro studies showed that bovine milkfat globule (MFGM)-derived complex lipid fractions inhibited inflammatory mediators IL-1β, nitric oxide, superoxide anion, COX-2, and neutrophil elastase in an arthritis rat model (Palmano et al., 2020). The above data underscore the anti-inflammatory effects of milk and milk components in alleviating arthritis. In agreement with the above studies, we found that camel and GM significantly decreased TNF-α, IL-1β, and IL-6, levels in arthritic rats, while other milk types failed to have any anti-inflammatory effect due to their lack of effects on studied inflammatory parameters. Prostaglandins are a group of lipids derived from arachidonic acid catalyzed by COX isoenzymes (Zhu et al., 2022). Production of prostaglandin is generally very low in uninflamed tissues but increases immediately in acute inflammation before the recruitment of leukocytes and the infiltration of immune cells (Ricciotti & FitzGerald, 2011). PGE2 is ubiquitously produced by all cells but predominantly by macrophages and therefore is a critical player in generating inflammatory reactions (Tilley et al., 2001). In this study, we found that PGE2 is significantly increased in arthritic rats. We further observed that these elevated PGE2 levels were markedly downmodulated by camel and GM, substantiating the anti-inflammatory properties of these two milk types. Corroborating with our result, alpha-lactalbumin (alphaLA) and beta-lactoglobulin (betaLG), which are major components of milk whey protein, inhibited the increased formation of IL-6 and PGE(2) via suppressing COX enzyme expression in an adjuvant-induced arthritis rat model (Yamaguchi et al., 2009). Likewise, bovine milk-derived α-lactalbumin inhibited colon inflammation and carcinogenesis in azoxymethane and dextran sodium sulfate-treated mice by blocking PGE2 production (Yamaguchi et al., 2014)
Conclusion
In this study, we assessed the therapeutic efficacy of different milk types against adjuvant-induced RA in a rat model. Among the studied milk types, only camel and GM showed promising effects in downmodulating various arthritis effector players including ACPAs, CRP, PGE2, and other inflammatory mediators. These favorable effects of camel and GM may be attributed to their lowest ω-6/ω-3 PUFA ratios. Thus, this study substantiates the therapeutic of camel and GM against RA. The creation of novel functional foods that support health, fend off illness and might make it possible to employ milk as a treatment for RA is one potential advantage of this research.
Abbreviations
ACPA: Anti-citrullinated peptide antibodies; AIN-93M: American Institute of Nutrition-93 diet formulated for maintenance; alphaLA: Alpha-lactalbumin; AM: Almond milk; ANOVA: Analysis of variance; betaLG: Beta-lactoglobulin; CA: California; CFA: Complete Freund’s adjuvant; CLA: Conjugated linolenic acid; CM: Camel milk; CO2: Carbon dioxide; COX-2: Cyclooxygenase-2; CRP: C-reactive protein; CWM: Cow milk; DMARDs: Disease-modifying antirheumatic drugs; ERK1/2: Extracellular signal-regulated kinase 1/2; FAMEs: Fatty acid methyl esters; FE: Food Efficiency; FID: Flame ionization detector; γδ T: Gamma delta T cells; GM: Goat milk; IL: Illinois; IL-1β: Interleukin-1beta; IL-6: Interleukin-6; IL-10: Interleukin-10; IL-17: Interleukin-17; IL-23: Interleukin-23; iNOS: Inducible nitric oxide synthase; JNK1/2: c-Jun N-terminal kinases 1/2; MAPK: Mitogen-activated protein kinase; MCP-1: Monocyte chemoattractant protein-1; ME: Milk efficiency; MFGM: Milk fat globule membrane; NF-κB: Nuclear factor-kappa B; NSAIDs: Nonsteroidal anti-inflammatory drugs; ω-3 PUFA: Omega-3 polyunsaturated fatty acids; ω-6 PUFA: Omega-6 polyunsaturated fatty acids; PA: Philadelphia; PGE2: Prostaglandin E2; RA: Rheumatoid arthritis; RF: Rheumatoid factor; rpm: Round per minute; SHM: Sheep milk; SM: Soy milk; SPSS: Statistical Package for the Social Sciences; TFI: Total food intake; Th17 cells: T helper 17 cells; TMC: Total milk consumption; TNF-α: Tumor necrosis factor-alpha; USA: United States of America; Wf: Final weight; Wg: Weight gain; Wi: Initial weight.
Footnotes
Acknowledgments
The authors extend their sincere appreciation to the Researchers Supporting Project number (RSPD2024R805), King Saud University, Riyadh, Saudi Arabia.
Authors Contributions
Conceptualization, DA, GS, LA, WA, and SA; methodology, LA, SA, and DA; software, GS and GA; validation, DA, GS, IE, and SA; formal analysis, DA and GS; investigation, LA, WA, GA, and IE; resources, DA, GS, and LA; data curation, SA; writing—original draft preparation, SA and GS; writing—review and editing, DA and GS; visualization, SA and LA; supervision, DA, GS, and WA; project administration, DA, GS, GA, and IE; funding acquisition, DA and GS. All authors discussed the results and commented on the manuscript. DA and GS contributed equally to the manuscript in the role ascribed to first authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was supported by the Researchers Supporting Project Number (RSPD2024R805), King Saud University, Riyadh, Saudi Arabia.
Institutional Review Board Statement
The Research Ethics Committee (REC) at King Saud University, Riyadh, Saudi Arabia (IRB Number: KSU-SE-19-21) approved all experimental procedures used in the present study.
