Abstract
Background
Lung cancer is a deadly type of cancer and is the primary cause of cancer-related deaths in both males and females worldwide. The number of lung cancer cases has risen worldwide due to a growing population of smokers and an increase in environmental pollutants.
Objectives
The current study was dedicated to studying the anti-cancer and immunomodulatory activities of nuciferine against B[a]P-induced lung cancer in mice.
Materials and Methods
The lung cancer was initiated in BALB/c mice by challenging them with B[a]P for 4 weeks. The post- and pre-treatment of 25 mg/kg of nuciferine was given to the B[a]P-challenged mice. The mice’s body weight, lung weight, tumor incidences, and tumor numbers were tabulated. The biochemical markers, immunoglobulins (Igs), phase-I and -II detoxification enzymes, and mitochondrial enzyme activities were assessed using standard techniques. The levels of inflammatory cytokines and the tumor biomarkers were evaluated using the assay kits. The histopathological analysis was conducted in the lung tissues of the experimental mice.
Results
Both post- and pre-treatment of nuciferine successfully increased body weight and reduced lung weight, tumor incidence, and tumor numbers. The nuciferine treatment effectively regulated the levels of biochemical parameters and Igs. Furthermore, the nuciferine treatment modulated the phase-I and -II enzymes, tumor biomarkers, mitochondrial enzymes, and inflammatory cytokine levels. The histopathological findings also proved the efficacy of nuciferine against lung cancer.
Conclusion
The present findings suggest that nuciferine exhibits both immunomodulatory and chemopreventive properties against lung cancer in mice. Therefore, it is evident that nuciferine possesses chemopreventive properties against lung cancer and can potentially enhance chemotherapy treatments.
Introduction
Lung cancer is a common form of malignant neoplasm that is widespread across the globe. It is a significant contributor to cancer-associated deaths, responsible for 18.4% of all cancer fatalities, amounting to almost 1.76 million mortalities in both males and females (Bade & Dela Cruz, 2020; Draškovič et al., 2023). Currently, lung cancer is the most often diagnosed cancer, representing 11.6% of all diagnosed cancer cases and contributing to 18.4% of overall cancer-associated deaths. The prevalence and mortality of lung cancer are increasing due to sociological and economic advancements, posing a significant threat to global health (Sung et al., 2021). Currently, the preferred therapy for lung cancer is surgical dissection of the tumor followed by further chemotherapy, which has proven to be beneficial in treating lung cancer in its early stages.
Chemotherapy is the conventional treatment method for the majority of lung cancer patients (Islam et al., 2022). Though it does not significantly enhance life expectancy because of the development of drug resistance, while anti-cancer medicines aim to induce apoptotic cell death in tumors, cancer cells might evolve resistance to apoptosis, hence reducing the drugs’ efficacy (Akash et al., 2023; Xu et al., 2021). Nevertheless, there remains a lack of efficacious therapy options for those who have advanced lung cancer (Rauf et al., 2021). Despite the substantial financial investments in cancer research, the mortality rates for cancer have remained markedly elevated (Emran et al., 2022). Hence, it is crucial to investigate innovative therapeutic approaches for lung cancer to enhance the survival rates and overall well-being of those diagnosed with this condition.
Cigarette consumption significantly influences the prevalence of lung cancer. Long-term smoking relates to a tenfold rise in the likelihood of acquiring lung cancer in comparison to individuals who do not smoke (Faselis et al., 2022). B[a]P, a polycyclic aromatic hydrocarbon (PAH), is a highly carcinogenic substance. PAHs are the primary ingredient in tobacco smoke and have a significant role in causing lung cancer (Bauer et al., 2022). B[a]P, a highly potent PAH, was the first carcinogen discovered in cigarette smoke. It is frequently employed to provoke lung cancer in animal models. It has been established that the harmful effects of B[a]P are caused by its metabolites and oxidative stress, which have a crucial role in lung cancer growth (Letelier et al., 2023). Recent data have confirmed that chronic exposure to tobacco smoke can cause premalignant lesions and disrupt deoxyribonucleic acid (DNA) repair mechanisms. This can ultimately lead to the development of invasive cancer by activating cancer-associated inflammatory mediators like nuclear factor kappa B (NF-κB) (Doukas et al., 2022). Carcinogenesis involves a complex interaction between tumor suppressor genes, oncogenes, and pathways that regulate cell death. The deregulation of apoptotic pathways can result in cancer development by allowing for cell dedifferentiation and metastasis. Therefore, many consider initiating apoptosis a beneficial treatment strategy in chemoprevention (Hänggi & Ruffell, 2023).
Nuciferine, a well-known alkaloid compound extracted from Nelumbo nucifera leaves, has been reported to have several biological activities. Numerous recent studies have highlighted that nuciferine attenuated myocardial infarction (Harish Kumar et al., 2022), protected against acute kidney injury (Li et al., 2021), protected against liver injury (Zhou et al., 2023), protected against hepatic steatosis and insulin resistance (Du et al., 2022), and regulated immunity and gut microbiota in colitis (Zhu et al., 2022) in experimental animal models. The anti-cancer effects of nuciferine against several malignancies, such as oral cancer (Xie et al., 2023), melanoma (Xu et al., 2020), and so forth, have been well reported. However, the anti-cancer activities of nuciferine against lung cancer models have not been reported yet. Therefore, the current study was dedicated to studying nuciferine’s anti-cancer and immunomodulatory activities against B[a]P-induced lung cancer in mice.
Materials and methods
Chemicals
The primary chemicals and reagents, including nuciferine, B[a]P, and so forth, were obtained from Sigma–Aldrich, USA. The biochemical markers were determined using kits obtained from Elabscience and Abcam, USA.
Experimental Mice
The study used healthy BALB/c mice weighing 25–30 g. The mice were procured and housed in polypropylene cabins under regular laboratory conditions, including a temperature of 25 ± 3°C, a dark/light sequence of 12 h, and an air moisture level of 55 ± 10%. The mice were provided with regular food and access to drinking water. The institutional animal ethics committee verified and approved all the experimental protocols conducted on the mice.
Experimental Design
The mice acclimating to the laboratory environments were distributed into four experimental groups. Group I: Control mice received only vehicle corn oil. Group II: The mice were given a dose of 50 mg/kg bw. of B[a]P twice a week for 4 weeks (2nd to 7th weeks; remaining weeks given only vehicle corn oil) to initiate lung cancer. Group III: The mice were post-treated with 25 mg/kg of nuciferine 1 day after the B[a]P challenge for 16 weeks. Group IV: The mice were given a pretreatment of 25 mg/kg of nuciferine before the B[a]P challenge for the 16th week. The body weight of each mouse was carefully noted throughout the study. After the completion of treatments, the mice from all groups were sacrificed under anesthesia by cervical dislocation, and samples were obtained for additional investigations.
Analysis of Lung Weight, Tumor Incidence, and Tumor Numbers
After sacrificing the mice under anesthesia, the lung tissues were excised from the mice, rinsed with buffered saline, and subsequently weighed to assess alterations in lung weight. The occurrences and total numbers of tumors were evaluated, and the results were documented.
Analysis of Biochemical Markers
The lung tissue homogenate was prepared and utilized for the following biochemical parameters: The previous technique (Mildred et al., 1981) was employed to assess the aryl hydrocarbon hydroxylase (AHH) activity. The Orlowski and Meister (1965) technique was employed to measure the gamma-glutamyltransferase (GGT) activity. The activity of 5′-nucleotidase (5′-NT) was assessed using the Hardonk and De Boer (1968) technique, and King’s (1965) technique was employed to measure the lactate dehydrogenase (LDH) activity.
Analysis of Immunoglobulins (Igs)
The serum of experimental mice was analyzed to determine the levels of Igs, including IgG, IgM, and IgA, using the method developed by Tennant et al. (1979). The levels are quantified, and the final results are as mg/dL.
Analysis of Phase‑I and -II Enzymes
The enzymatic activities of cyt-b5, cyt-P450, and NADPH-cyt-c-reductase were assessed using previously developed techniques (Omura & Sato, 1964; Wharton & Tzagoloff, 1967). The uridine 5′-diphospho-glucuronosyltransferase (UDP-GT), GST, and QR were determined using the previous methods (Benson et al., 1980; Habig et al., 1974; Luquita et al., 1994).
Analysis of Tumor Biomarker Levels
The serum samples of experimental mice were analyzed to assess the CYFRA 21‑1 and carcinoembryonic antigen (CEA) levels using ELISA kits bought from Elabscience, USA. The studies were conducted per the manufacturer’s guidelines, and the findings were illustrated as ng/mL.
Analysis of Inflammatory Cytokines
The experimental mice’s interleukin (IL)-6, tumor necrosis factor (TNF)-α, and IL-1β levels were examined using Abcam, USA assay kits. The assays were performed per the manufacturer’s guidelines, and the findings are reported in pg/mL.
Analysis of Mitochondrial Enzymes
The lung tissues from the experimental mice were homogenated with a solution of 0.01 M saline and then centrifuged at 10,000 rpm for 15 min. The supernatant was employed for the mitochondrial enzyme assays. The activities of succinate dehydrogenase (SDH) (Slater & Borner, 1952), malate dehydrogenase (MDH) (Mehler & Kornberg, 1948), ketoglutarate dehydrogenase (KDH) (Reed & Mukherjee, 1969), and isocitrate dehydrogenase (ICDH) (King, 1965) in the lung tissue homogenates were assayed using the previously described methods.
Histopathological Analysis
The lung tissues were processed with formalin (10%) and then subjected to paraffin block preparation. Hematoxylin and eosin (H&E) staining was conducted on paraffinized tissue sections of 5 µm in diameter. The slides were examined and captured using a microscope at a magnification ×40.
Statistical Analysis
The values are analyzed statistically using the SPSS software. The values are analyzed using one-way analysis of variance (ANOVA) and Tukey’s post hoc test. The results are presented as the Mean ± Standard Deviation (SD) of three replicate assays.
Results
Nuciferine Treatment Increases Body Weight and Reduces Lung Weight, Tumor Incidence, and Tumor Numbers
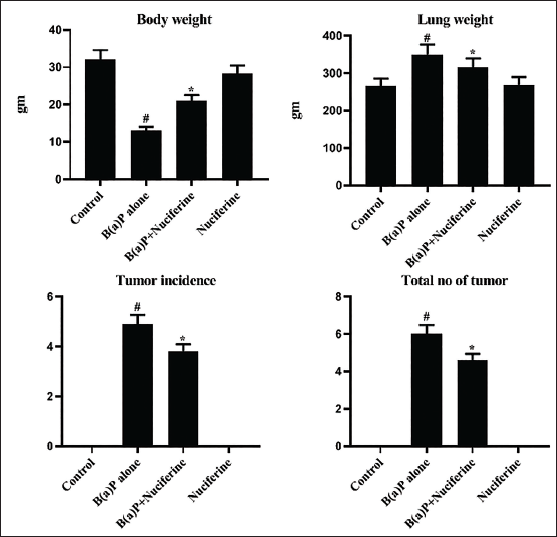
The levels of body weight, lung weight, and tumor incidence and numbers in the experimental mice are illustrated in Figure 1. The B[a]P-induced mice exhibited a reduction in body weight and an elevation in lung weight. Furthermore, it also demonstrated an elevated incidence of tumors and an overall increase in the number of tumors. Notably, the 25 mg/kg of nuciferine-treated mice showed a remarkable upsurge in body weight and a reduction in lung weight. The nuciferine treatment also decreased both tumor numbers and incidences in the B[a]P-exposed mice.
Effect of Nuciferine on the Body Weight, Lung Weight, Tumor Incidence, and Tumor Numbers in the B[a]P-induced Mice. All the Values Were Analyzed Using SPSS Software and Illustrated as Mean ± Standard Deviation (SD) Triplicates. The Data Were Evaluated by One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Assay. #p < 0.01 Compared with Control (Group I) and *p < 0.05 Compared with Cancer-bearing Mice (Group II).
Nuciferine Treatment Modulates Biochemical Markers and IGS Levels
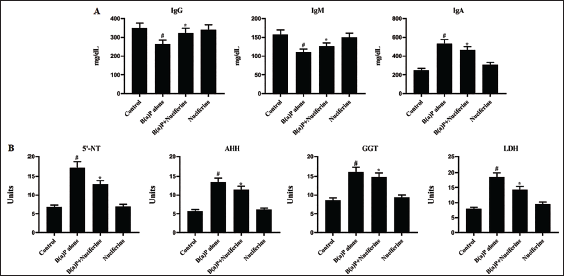
The biochemical markers and Igs in the experimental mice were evaluated, and the outcomes are depicted in Figure 2A and B. The B[a]P-exposed mice revealed decreased IgM and IgG levels and increased IgA in their serum. It also demonstrated decreased biochemical marker levels, including AHH, LDH, GGT, and 5′-NT, in their serum. Remarkably, the 25 mg/kg nuciferine treatment efficiently increased the IgM and IgG levels while reducing the IgA levels. Both post- and pre-treatments with nuciferine demonstrated a considerable increase in the GGT, 5′-NT, AHH, and LDH levels in B[a]P-induced mice.
Effect of Nuciferine on the Biochemical Markers in Control and Experimental Mice. (A) Immunoglobulin (Ig), IgM, and IgA Levels. (B) 5′-Nucleotidase (5-NT), Aryl Hydrocarbon Hydroxylase (AHH), Gamma-glutamyltransferase (GGT), and Lactate dehydrogenase (LDH) Status. All the Values Were Analyzed Using SPSS Software and Illustrated as Mean ± Standard Deviation (SD) Triplicates. The Data Were Evaluated by One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Assay. #p < 0.01 Compared with Control (Group I) and *p < 0.05 Compared with Cancer-bearing Mice (Group II).
Nuciferine Treatment Regulates Phase-I and -II Enzymes in the B[a]P-induced Mice
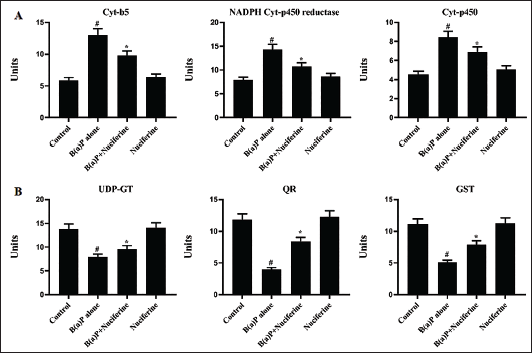
The serum of experimental mice was analyzed to determine the phase-I and -II enzyme levels. The outcomes of this assay are illustrated in Figure 3A and B. Elevated activities of cyt-P450, cyt-b5, and NADPH-cyt-P450 reductase enzymes (phase-I) were detected in the serum of B[a]P-induced mice. In contrast, it exhibited reduced UDP-GT, GST, and QR (phase-II) enzyme activities. Remarkably, the nuciferine treatment efficiently reduced the phase-I enzymes while augmenting the phase-II enzyme activities. Both post- and pre-treatment of nuciferine significantly regulated the phase-I and -II enzymes.
Effect of Nuciferine on the (A) Phase-I (Cyt b5, NADPH Cyt P450 & Cyt P450) B) Phase-II (UDP-GT, GST & QR) Enzymes Levels in experimental Mice. All the Values Were Analysed Using SPSS Software and Illustrated as Mean ± Standard Deviation (SD) Triplicates. The Data Were Evaluated by One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Assay. #p < 0.01 Compared with Control (Group I) and *p < 0.05 Compared with Cancer-bearing Mice (Group II).
Nuciferine Treatment Regulates Tumor Biomarker Levels
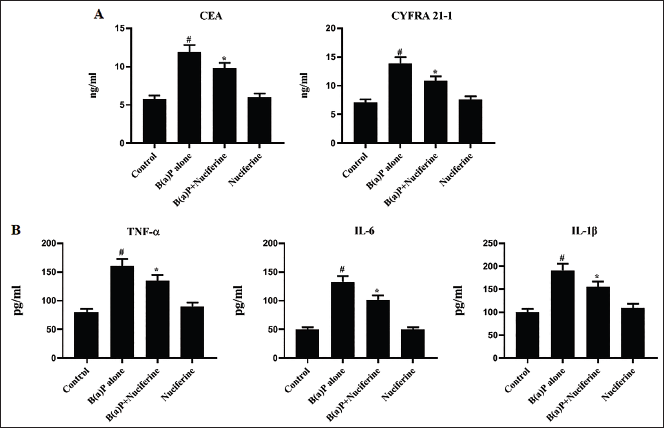
CYFRA 21-1 and CEA concentrations in the control and treated mice were evaluated, and the results are presented in Figure 4A. The B[a]P-induced mice exhibited increased CYFRA 21-1 and CEA levels in the serum. Notably, both post- and pre-treatment of 25 mg/kg of nuciferine significantly reduced these marker levels in the B[a]P-challenged mice.
Effect of Nuciferine on the (A) Carcinoembryonic Antigen (CEA), CYFRA 21-1 and (B) Inflammatory Cytokine Levels in the B[a]P-induced Mice. All the Values Were Analyzed Using SPSS Software and Illustrated as Mean ± Standard Deviation (SD) Triplicates. The Data Were Evaluated by One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Assay. #p < 0.01 Compared with Control (Group I) and *p < 0.05 Compared with Cancer-bearing Mice (Group II).
Nuciferine Treatment Reduces the Inflammatory Cytokine Levels
Figure 4B displays the concentrations of IL-6, TNF-α, and IL-1β in the serum of the mice used in the experiment. A drastic upsurge in the IL-6, TNF-α, and IL-1β levels was observed in the B[a]P-treated mice. Considerably, the treatment of 25 mg/kg of nuciferine (both post- and pre-treatments) successfully decreased these cytokine levels in the B[a]P-challenged mice.
Nuciferine Treatment Increases Mitochondrial Enzyme Activities
The mitochondrial marker enzyme activities, including SDH, MDH, KDH, and ICDH, in the lung tissues of both control and treated mice (Figure 5A). The mice exposed to B[a]P exhibited decreased SDH, MDH, KDH, and ICDH activity. Remarkably, 25 mg/kg of nuciferine treatment resulted in a significant increase in SDH, MDH, KDH, and ICDH activities in the lung tissues, as depicted in Figure 5A.
(A) Effect of Nuciferine on the Mitochondrial Enzyme Activities in the B[a]P-induced Mice. All the Values Were Analyzed Using SPSS Software and Illustrated as Mean ± Standard Deviation (SD) Triplicates. The Data Were Evaluated by One-way Analysis of Variance (ANOVA) and Tukey’s post hoc Assay. #p < 0.01 Compared With Control (Group I) and *p < 0.05 Compared With Cancer-bearing Mice (Group II). (B) Effect of Nuciferine on the Lung Histopathology of the B[a]P-induced Mice. Scale Bar: 50 µm. Group I: Control Mice Treated with Vehicle Corn Oil; Group II: Mice with 50 mg/kg B[a]P-induced Lung Cancer; Group III: B[a]P-induced Lung Cancer + 25 mg/kg of Nuciferine Treated Mice; Group IV: B[a]P-induced Lung Cancer + 25 mg/kg of Nuciferine Pretreated Mice.
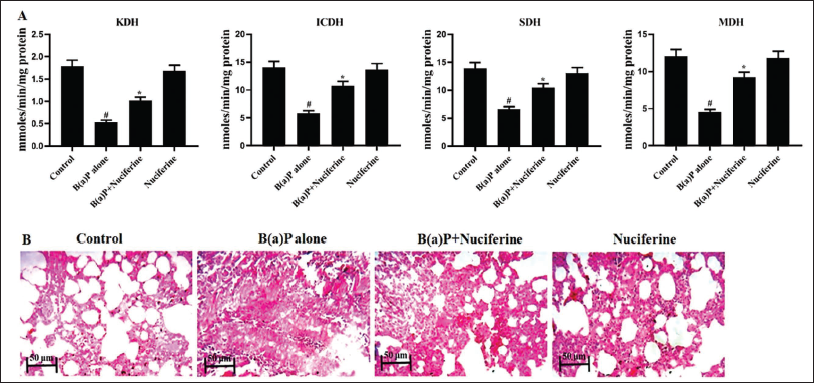
Nuciferine Treatment Improves Lung Histopathology
Histopathological analysis of lung tissues from experimental mice was conducted, and the results are presented in Figure 5B. The mice subjected to B[a]P exhibited an altered alveolar structure, elevated hyperchromatic nuclei, increased growth of abnormal cells, localized bronchial damage, and excessive growth of cells in the alveolar epithelium (hyperplasia) in the lung tissues. The mice that were given a dose of 25 mg/kg of nuciferine (pretreatment) showed significant improvement in proliferative lesions and reduced hyperplasia in the lung tissues.
Discussion
Lung cancer is a deadly type of cancer and is the primary cause of cancer-associated mortalities in both sexes. The prevalence of lung cancer has risen in recent years due to a growing population of smokers and the presence of environmental toxins (Nooreldeen & Bach, 2021). The increased rate of deaths associated with lung cancer has urged the exploration of novel therapeutic interventions to overcome this disease. The present work has evaluated nuciferine’s anti-cancer and immunomodulatory potentials against B[a]P-exposed lung cancer in mice. The association between inflammatory responses and cancer formation is particularly significant when considering oncogenic signaling pathways. Inflammation plays a pivotal role in cancer growth by causing genetic changes in stem cell DNA and the subsequent development of cancer, which is heavily influenced by the surrounding tumor microenvironment. Exposure or continuous attack of stem cells’ DNA by inflammatory stresses without repair can result in genetic changes. These mutations can gradually transform a somatic stem cell into a tumor stem cell (Zhao et al., 2021). Hence, targeting inflammation in therapy could significantly augment the efficacy of the treatment.
Cytokines are widely recognized for their crucial involvement in host defense and pathogenesis in inflammatory diseases. Cytokines are the pivotal players in cancer advancement (Hosseinzadeh et al., 2019). TNF-α, IL-6, and IL-1β are the critical players in inflammation-related tumor growth. IL-6 is an essential cytokine that plays an active role in various diseases, including the development of tumors. Some tumor cells can enhance tumor growth through the self-activation of IL-6 (Essogmo et al., 2023). Elevated levels of TNF-α can exacerbate the activation of lymphocytes and eosinophils at the site of inflammation, leading to a rise in the release of many other cytokines. Previous studies have linked IL-1β, a cytokine that promotes inflammation, to tumor progression in individuals with lung cancer (Bezel et al., 2021). The outcomes of our work revealed an upsurge in the TNF-α, IL-6, and IL-1β levels in the B[a]P-exposed mice. Furthermore, the administration of nuciferine successfully reduced these cytokine levels.
Tumor biomarker levels provide prognostic information on lung cancer. People with elevated biomarker levels have a greater possibility of developing tumors compared to people with normal levels. Tumor markers are commonly used to indicate the presence of cancer. AHH, LDH, GGT, and 5′-NTs are important and sensitive prognostic markers. Their levels increase significantly due to cellular injury, leading to the leaking of cytoplasmic contents into the circulation (Chen et al., 2021). AHH serves as a significant biomarker for prompt lung cancer diagnosis (Kasala et al., 2015). LDH activity is elevated in numerous malignant tumors, making it a prognostic biomarker for various cancers. LDH is crucial in controlling glycolysis, the sole energy-generating pathway responsible for promoting rapid tumor growth. Cancer cells’ survival and proliferation depend on LDH (Xie et al., 2014). The lung cancer cell lines exhibited higher LDH activity than non-malignant cells. The elevation in lung LDH activity caused by heightened glycolysis during tumor development may be attributed to this phenomenon. Proliferating tumor cells have been discovered to exhibit heightened activity of 5′-NT. The lung cancers exhibited an elevated concentration of 5′-NT (Stabile et al., 2012). Our findings demonstrated the increased AHH, LDH, GGT, and 5′-NT levels in the B[a]P-challenged mice. Remarkably, the treatment with nuciferine significantly reduced these biomarkers in the B[a]P-exposed mice.
Mitochondria serve as the primary source of cellular energy, and any damage to mitochondria results in reduced energy output, ultimately promoting cell death. Carcinogens disrupt cell functioning via binding to thiol-bearing enzymes and subcellular membranes, which serve as sensitive binding sites. The elevated ROS production and tumorigenic conditions might lead to a reduction in mitochondrial enzyme activities (Patel et al., 2022). The decrease in these enzyme activities may result from alterations in tumor cell structure and metabolic changes in mitochondria (Musicco et al., 2023). The ICDH, SDH, MDH, and KDH activities in the B[a]P-induced mice were substantially decreased, as found in the present investigation. Nevertheless, the mice treated with nuciferine exhibited a substantial upsurge in the mitochondrial enzyme activities in their lung tissues.
The correlation between low blood Ig levels and severe lung conditions has already been elucidated. Several Igs, including IgA, IgM, IgG, and IgE, are widely present in the tumor cells. Recently, there has been significant study interest in the IgA isotype since it is found on mucosal surfaces. However, studies have also been conducted on IgA antibodies and their participation in tumor growth. Elevated serum concentrations of IgA antibodies have been detected in individuals with lung cancer. Earlier research has emphasized that immunomodulation is a potent and viable strategy for cancer treatment (Shapiro et al., 2021). The findings of this work exhibited that the IgG and IgM levels were diminished in the lung cancer mice, indicating a decrease in the humoral immune response. These Igs are pivotal players in neutralizing chemical toxicants, opsonization, and activating the complement system. The results demonstrate that the B[a]P challenge caused immunosuppression, leading to compromised immunological responses in mice. The IgM and IgG levels in the B[a]P-exposed mice were remarkably augmented by nuciferine, whereas the level of IgA was decreased. This provides evidence of the immunomodulatory effects of nuciferine.
Tumor markers are well recognized to have a crucial impact on tumor growth, and researchers focus on these indicators to hinder tumor growth. CYFRA 21-1 and CEA are the most valuable and sensitive tumor biomarkers. CEA is widely recognized as a significant biomarker for cancer, particularly as a prognostic predictor for lung cancer. CEA frequently induces surrounding cells outside the malignant epithelial tumor cell, enhancing the viability of non-malignant cells and promoting metastasis (Yang et al., 2023). The CEA is the most extensively researched tumor biomarker. Moreover, the elevated concentration of CEA is associated with diverse malignant tumors. CYFRA 21-1 is essential for maintaining the stability of epithelial cells (Baj et al., 2022). Elevated CYFRA21-1 levels have been documented in cases with lung neoplasms. Patients exhibiting higher CYFRA 21-1 levels demonstrate a reduced overall survival rate. In this work, we found that the CEA and CYFRA21-1 levels in the serum of B[a]P-challenged mice were increased. However, the administration of nuciferine significantly decreased the CEA and CYFRA21-1 levels.
The liver is a vital organ that plays a significant role in the metabolism of carcinogens. Phase-I detoxifying enzymes facilitate the metabolic activation of carcinogens, resulting in the production of tumorigenic metabolites. Recurrent carcinogen exposure has been associated with phase-I and -II enzyme levels. Suppressing the phase-I enzyme activities and promoting the phase-II enzyme activities is believed to decrease the chances of developing cancer (Bagdasaryan et al., 2022). Cyt-b5 is a hemoprotein and a pivotal player in numerous metabolic activities, including the desaturation of fatty acids, the generation of steroid hormones, and the decrease of methemoglobin. The higher cyt-P450 and cyt-b5 levels were already noted in lung cancer. The cyt-P450 and cyt-b5 inhibitors are beneficial in decreasing the intensity of malignancies (Szaefer et al., 2008). Our findings showed that the cyt-P450 and cyt-b5 levels were increased in the B[a]P-exposed mice. Significantly, these increases were inhibited after treatment with nuciferine, demonstrating its chemopreventive potential through its stabilizing effects on these enzymes.
Cells protect themselves against damage caused by free radicals through various anti-oxidant mechanisms. The GST is a crucial diagnostic and monitoring tool for lung cancer. In addition, GST, in conjunction with glutathione, is a powerful scavenger of free radicals, thereby safeguarding cells from carcinogens and mutagens (Sorokina et al., 2010). Furthermore, QR and UDP-GT enzymes protect cells against the cancer-causing and poisonous effects of chemical carcinogens. UDP-GT and QR are renowned for their involvement in eliminating harmful metabolites through detoxification processes. A decrease in these enzyme activities results in an upsurge in oxidative stress, which in turn promotes the development of tumors (Grishanova & Perepechaeva, 2022). The findings of our study indicate that the mice exposed to B[a]P displayed reduced activity of UDP-GT, QR, and GST enzymes. In contrast, the nuciferine treatment increased these enzymes’ levels in the serum of B[a]P-challenged mice.
Conclusion
The present findings suggest that nuciferine exhibits immunomodulatory and chemopreventive properties in lung cancer-induced mice. The chemopreventive activity of nuciferine is linked to its immunomodulatory activities and its capacity to regulate biochemical markers and phase-I and -II enzymes. In addition, the nuciferine treatment reduced the incidence of tumors, inflammatory cytokines, and tumor markers. The nuciferine treatment leads to increased activity of mitochondrial enzymes. Therefore, it is evident that nuciferine possesses chemopreventive properties against lung cancer and can potentially enhance chemotherapy treatments.
Footnotes
Abbreviations
AHH: Aryl hydrocarbon hydroxylase; GGT: Gamma-glutamyltransferase; ICDH: Isocitrate dehydrogenase; Igs: Immunoglobulins; KDH: Ketoglutarate dehydrogenase; LDH: Lactate dehydrogenase; MDH: Malate dehydrogenase; PAH: Polycyclic aromatic hydrocarbon; B[a]P: Benzo(a)pyrene; SDH: Succinate dehydrogenase; UDP-GT: Uridine 5′-diphospho-glucuronosyltransferase; 5′-NT: 5′-Nucleotidase.
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animals were subjected to humane care as per the regulations, and necessary authorization was secured from the Institutional Animal Ethics Committee of Xi’an Zhongkai (Approval No. 4902).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
