Abstract
Background
Coccidiosis is considered a protozoan disease affecting wild and domestic animal species which leads to economic losses. Regarding drug resistance, scientists are focused on alternative sources rather than coccidiostats with fewer side effects. Recently, nanotechnology has been a new trend against parasitic infections by manipulating atoms and molecules at the nanoscale.
Objectives
This study assessed the hepatoprotective effects of biosynthesized nano selenium (BSN) derived from Azadirachta indica leaf extracts on biological, biochemical, and oxidative stress markers in mice infected with Eimeria papillata.
Materials and Methods
The formation of nano selenium was achieved using leaf extract from A. indica, and their morphology and size were characterized using scanning electron microscopy (SEM). Twenty-five male C57BL/6 mice were divided into five groups, with five mice in each group. The first group received distilled water, while the second group was administered 0.5 mg/kg of BSN. The third, fourth, and fifth groups were infected with the parasite’s oocysts. For 5 days, the fourth group received BSN (0.5 mg/kg), and the fifth group was treated with amprolium (120 mg/kg). Oocyst shedding was assessed on day 5 post-infection (p.i.). After sacrificing the mice, the liver and blood from each mouse were collected and processed for histological, biochemical, and oxidative damage analyses.
Results
Oocyst shedding in the feces of mice decreased following oral treatment with BSN. Eimeria infection reduced feed consumption to 31.44%, but this increased to 45.57% after BSN treatment. The infection caused significant liver damage, as evidenced by pathological changes and a notable increase in liver function parameters, including glutamic-pyruvic transaminase (ALT), glutamic-oxaloacetic transaminase (AST), alkaline phosphatase (ALP), and direct bilirubin. These parameters improved after treatment with BSN. Additionally, Eimeria infection disrupted nutrient homeostasis. Levels of soluble proteins and carbohydrates in the liver tissue decreased, while blood glucose and total protein levels increased. Treatment with BSN in mice infected with E. papillata significantly improved nutrient homeostasis. Our research also demonstrated that BSN significantly reduced levels of nitric oxide (NO) and hydrogen peroxide (H2O2), while increasing levels of catalase (CAT) and superoxide dismutase (SOD) to help mitigate oxidative damage during Eimeria infection.
Conclusion
Our findings demonstrate that BSN has a significant in vivo hepatic response against murine coccidiosis and could be valuable as a coccidiostat in treating the disease.
Introduction
Protozoan parasites are widely distributed and could affect wild and domestic animals. Coccidiosis is an infectious disease caused by obligate protistan parasites within the genus Eimeria (Pakandle, 2009). Infection occurs when the infectious stage, sporulated oocyst, hatches to release eight sporozoites and then a new host ingests them. This disease causes animals to lose weight gain and feed efficiency because of symptoms including diarrhea and appetite loss (Williams, 2005). It can be treated with a range of anti-coccidial drugs (such as sulfaquinoxaline, nitrofurazone, nicarbazin, and amprolium) (Mehlhorn, 2014), but the usage of these therapeutic agents contributed to multidrug resistance and an overabundance of parasite infections in tissues (Onyiche et al., 2021). Scientists are concerned about moving away from the chemotherapeutic control of coccidiosis and moving toward non-medicated methods, such as natural and safe resources, to circumvent undesirable health consequences.
Nanotechnology is a promising field for the treatment of various infectious diseases (Sun et al., 2019). Selenium nanoparticles (SN) serve as effective anti-oxidant and anti-inflammatory agents (Alkhudhayri et al., 2018; Rayman, 2012). Additionally, SNs exhibit strong anti-parasitic activity against several protozoans, including Leishmania major (Beheshti et al., 2013), Giardia duodenalis (Malekifard et al., 2020), and Eimeria papillata (Abdel-Gaber et al., 2023; Alkhudhayri et al., 2018, 2020; Dkhil et al., 2012). Moreover, potential applications of SN have also been documented in cases of murine schistosomiasis (Dkhil et al., 2016, 2019) and murine trichinosis (Sarhan et al., 2022). While SN can be synthesized through various chemical processes, these methods tend to be costly, complex, and sometimes hazardous. To address these challenges, several eco-friendly strategies for the biosynthesis of SN have been developed, which include utilizing different plant sources as alternatives to traditional chemical methods.
The leaves of Azadirachta indica, commonly known as neem, are widely utilized in medicine (Latif et al., 2020). One benefit of using natural extracts like neem is their ability to minimize the risk of developing resistance. Additionally, the residues from such natural products in meat pose no health risks for human consumption. Neem has been identified to possess several pharmacological properties, including hepatoprotective effects (Chattopadhyay et al., 1992), hypoglycemic (Bopanna et al., 1997), hypolipidemic (Bopanna et al., 1997), cardioprotective (Peer et al., 2008), anti-ulcer (Pillai & Santhakumari, 1984), anti-inflammatory (Lee et al., 2017), anti-bacterial (Herrera-Calderon et al., 2019), chemotherapeutic (Paliwal et al., 2005), and anti-fungal (Sudan et al., 2020) properties. Furthermore, A. indica has shown effectiveness against various parasites, including Plasmodium falciparum (Khalid et al., 1986), Plasmodium berghei (Habluetzel et al., 2019), Leishmania amazonensis (Carneiro et al., 2012), Trypanosoma brucei (Mbaya et al., 2010), and E. papillata (Dkhil et al., 2013).
In mice infected with E. papillata, this study demonstrated the hepatoprotective potential of biosynthesized SN derived from A. indica leaf extracts and its effects on oxidative stress marker alterations.
Materials and Methods
Plant Collection and Extract Preparation
A. indica (Meliaceae) leaves were harvested from the botanical gardens located in Riyadh, Saudi Arabia. The botanical identity of the plant was confirmed by a taxonomist at King Saud University’s herbarium (voucher no. KSU-10804). The procedure of Abdel-Gaber et al. (2023) was followed to prepare the 70% A. indica methanolic. After being lyophilized, the resulting crude extract was kept at −20°C.
Biosynthesized Nano Selenium (BSN) Preparation
The synthesis of nano selenium was achieved through a reduction reaction of sodium selenite (Na2Se2O3·5H2O) using ascorbic acid, followed by stabilization with polysorbate, as described by Vahdati and Moghadam (2020). In summary, 30 mg of sodium selenite was dissolved in 90 mL of Milli-Q water. Ascorbic acid (10 mL at a concentration of 56.7 mM) was added dropwise while stirring vigorously. After every 2 mL of ascorbic acid, 10 µL of polysorbate was incorporated to form nano selenium. To further develop the colloidal solution, it was mixed in a 9:1 ratio with 1 mL of A. indica methanolic extract. This mixture was stirred for 15 min at 45°C in a water bath. Following the mixing process, lyophilization was performed to enhance both the long-term stability of the nano selenium and the shelf life of the final product. A color change in the aqueous solution served as an indicator for the formation of nano selenium (BSN). The shape and size of the BSN were characterized using scanning electron microscopy (SEM) with a JEOL JSM-6060L (JEOL Ltd., Tokyo, Japan) at an accelerating voltage of 15 kV (Abdel-Gaber et al., 2023).
Parasite Passage
E. papillata was utilized as a model coccidial parasite in mice. The Eimeria oocysts were passage through five laboratory mice, Mus musculus. Oocysts were collected and processed from the feces of the infected mice (refer to Abdel-Gaber et al., 2023. The oocysts were sporulated using a 2.5% potassium dichromate solution and then washed three times in phosphate buffer solution before being used in the experiment.
Mice and Coccidial Infection
Male C57BL/6 mice, weighing between 20 and 22 g, were divided into five groups, each consisting of five mice. The first group served as a negative control and was administered distilled water. The second group received daily oral gavage inoculations of 0.5 mg/kg of BSN. The third, fourth, and fifth groups were orally administered 103 sporulated E. papillata oocysts to induce infection. The fourth group was treated with 0.5 mg/kg of BSN 1 h after infection for 5 days, following the protocol established by Abdel-Gaber et al. (2023). The fifth group was treated with 120 mg/kg of amprolium, also administered 1 h after infection for 5 days, according to the same reference. On day 5 post-infection (p.i.), fresh fecal pellets were collected from each mouse in the E. papillata-infected untreated and treated groups. The number of oocysts per gram of wet feces was quantified using a McMaster chamber, based on the method outlined by Schito et al. (1996). Feed consumption was calculated by subtracting the weight of the residual food collected on day 5 p.i. from the initial weight of the food offered at the start of the experiment (day 0 p.i.).
Sample Collection
On the 5th day p.i., all mice were euthanized by CO2 asphyxiation. Blood was collected from the heart into heparinized tubes, and then plasma was separated and kept at −20°C for biochemical analysis. The livers of mice were removed and then divided into small pieces. Parts were fixed in neutral buffered formalin (10%) for histology, while others were preserved at −80°C for oxidative stress analysis.
Biochemical Analysis
Plasma biochemical parameters indicative of liver function were measured using commercial kits from Biodiagnostic Co., Egypt. The methods established by Reitman and Frankel (1957) were employed to determine the levels of glutamic-oxaloacetic transaminase (AST) and glutamic-pyruvic transaminase (ALT). Alkaline phosphatase (ALP) levels were assessed using the method described by Belfield and Goldberg (1971). Direct bilirubin was measured with commercial kits from Human Gesellschaft für Biochemica und Diagnostica mbH, Germany, following the procedure outlined by Thomas (1998). Additionally, the levels of glucose and total proteins in the blood plasma of mice were determined using commercial kits from Bio-Diagnostic Co., Egypt, following the protocols of Trinder (1969) and Gornall et al. (1949), respectively. Absorbance readings were taken using the Ultrospec 2000 ultraviolet (UV) spectrophotometer from Amersham Pharmacia Biotech, Cambridge, UK.
Histological and Histochemical Preparation
The fixed liver samples were dehydrated using a series of ethyl alcohol dilutions, then embedded in paraffin wax and sectioned to a thickness of 5 µm. For histological examinations, the sections were stained with hematoxylin and eosin (H&E) following the method outlined by Drury and Wallington (1980). Histological scoring of the liver was performed according to the criteria established by Ishak et al. (1995). For the histochemical analysis, sections were stained using the periodic acid-Schiff’s method to assess total carbohydrates (Hotchkiss, 1948) and the mercuric bromophenol blue method for total proteins (Mazia et al., 1953). The sections were examined and photographed using a Leica DM 2500 microscope with NIS ELEMENTS software (version 3.8).
Tissue Preparation and Homogenization
Freshly expelled hepatic tissue was homogenized on ice using a phosphate buffer (0.1 M, pH 7.4) in a 1:10 weight-to-volume (w/v) ratio, following the method described by Tsakiris et al. (2004). The homogenate was then centrifuged at 5,000 rpm for 15 min at 4°C. The supernatant was stored at −20°C until needed.
Oxidative Stress Assays
The hepatic homogenate was utilized to assess oxidative stress biomarkers, specifically catalase (CAT), hydrogen peroxide (H2O2), nitric oxide (NO), and superoxide dismutase (SOD). This was carried out following the standard techniques established by Aebi (1984), Montgomery and Dymock (1961), and Nishikimi et al. (1972), using the appropriate kits provided by Bio-Diagnostic Co. in Egypt. The absorbance measurements were quantified using the Ultrospec 2000 UV spectrophotometer from Amersham Pharmacia Biotech, based in Cambridge, UK.
Statistical Analysis
Data are presented as mean ± standard deviation (SD). Differences among groups were considered significant at a p value ≤ 0.05, determined using one-way analysis of variance (ANOVA) with SigmaPlot® version 11.0 (Systat Software, Inc., Chicago, IL, USA).
Results
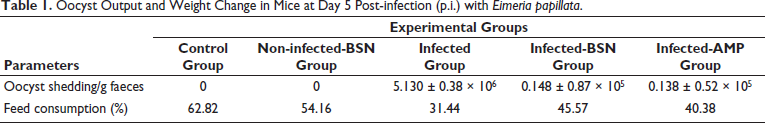
On the 5th day, fecal oocyst output was highly variable between infected and treated groups and reached its peak of approximately 5.130 ± 0.38 × 106 oocyst/gram of feces in the infected group (Table 1). Using SEM examination, BSN has a spherical morphology with a size of 62.8–71.5 nm with an average diameter of ⁓67.7 nm (Supplementary Figure 1). After treatment with BSN, the rate of Eimeria oocyst shedding on day 5 p.i. was lowered to 0.148 ± 0.87 × 105 oocyst/gram of feces. Similar to amprolium, BSN exhibited excellent efficacy against E. papillata (Table 1). There was no evidence of Eimeria infection among the control and BSN groups as there were no oocysts present in their fecal matter, confirming the absence of contamination between infected and control mice. Moreover, there was a significant interaction between Eimeria infection and diet uptake, as shown in Table 1. The feed consumption of the infected mice was reduced in the 5th-day p.i. to 31.44%, compared to the control and non-infected-BSN groups of 62.82% and 54.16%, respectively (Table 1). This rate was affected during treatment with BSN (45.57%) and amprolium (40.38%) than those of the infected group (Table 1).
Oocyst Output and Weight Change in Mice at Day 5 Post-infection (p.i.) with Eimeria papillata.
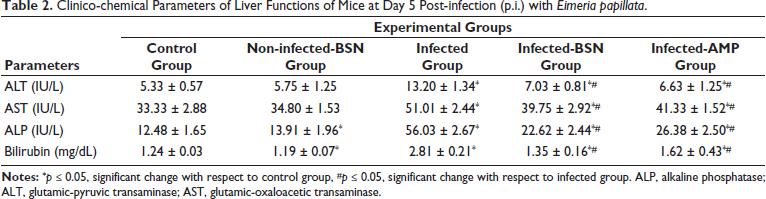
The clinical-chemical parameters were indicative of the liver functions in mice experimentally infected with E. papillata and those treated with BSN and amprolium are presented in Table 2. There was a significant increase in the infected group for the levels of ALT (13.20 ± 1.34 IU/L), AST (51.01 ± 2.44 IU/L), ALP (56.03 ± 2.81 IU/L), and bilirubin (2.81 ± 0.21 mg/dL) than that of the control group. Liver parameters including ALT, AST, ALP, and bilirubin levels showed substantial differences owing to treatments. BSN inhibited their increase to be 7.03 ± 0.81 IU/L (ALT), 39.75 ± 2.92 IU/L (AST), 22.62 ± 2.44 IU/L (ALP), and 1.35 ± 0.16 mg/dL (bilirubin) in comparison to the drug group (Table 2).
Clinico-chemical Parameters of Liver Functions of Mice at Day 5 Post-infection (p.i.) with Eimeria papillata.
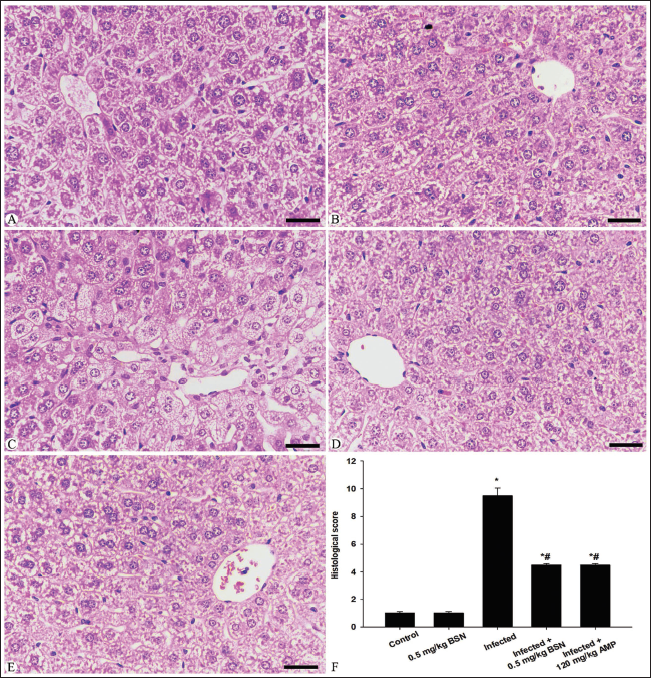
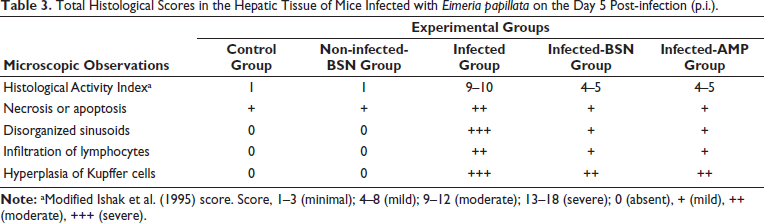
Microscopic examination of liver sections in the control group, as shown in Figure 1, revealed a normal histological structure of hepatic tissue. In contrast, the infected group that contained E. papillata oocysts exhibited moderate pathological changes in the liver. These changes included necrotic hepatocytes, inflammatory cellular infiltrations, cytoplasmic vacuolation, and degeneration of hepatocytes (Figure 1). Additionally, the hepatic sinusoids in the infected group showed a higher presence of Kupffer cells compared to the control group (Figure 1). According to Ishak’s score, all observable hepatic changes in the infected group were categorized as a score of 10–11 on day 5 p.i. when compared to the control group (Figure 1, Table 3). Furthermore, the histopathological changes in the liver tissue were significantly reduced in the BSN-treated mice, which exhibited fewer Kupffer cells than those in the infected group (Figure 1).
Total Histological Scores in the Hepatic Tissue of Mice Infected with Eimeria papillata on the Day 5 Post-infection (p.i.).
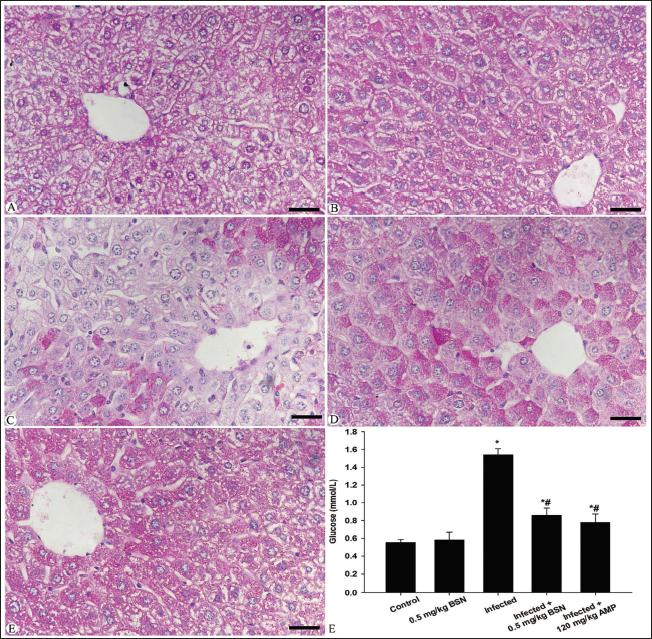
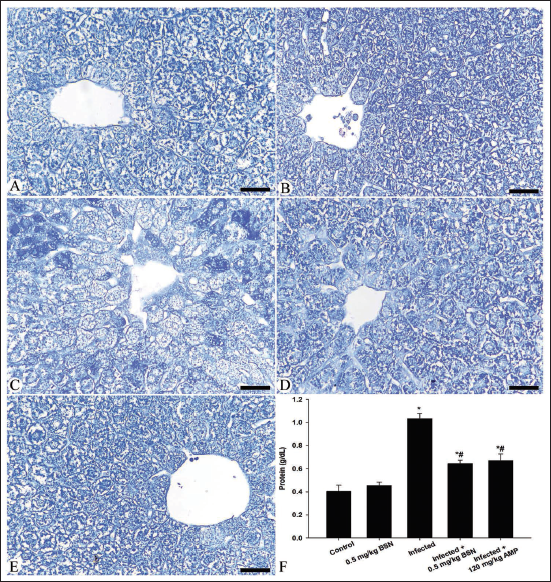
As shown in Figures 2 and 3, the nutrient constituents were detected in the blood plasma and hepatic tissue of all experimental mice groups. Microscopic examination revealed a depletion in the carbohydrate content within the hepatic tissue of mice infected with E. papillata (Figure 2), associated with an elevation of the glucose level in the blood plasma from 0.55 ± 0.03 mmol/L in the control group to 1.54 ± 0.07 mmol/L in the infected group (Figure 2). Oral administration of BSN caused a significant improvement in the hepatic tissue structure and lowered the plasma glucose level (0.86 ± 0.08 mmol/L) than that of the infected group (Figure 2). Moreover, the E. papillata infection caused quantitative elevation in the total protein content within the blood plasma of mice (1.03 ± 0.04 g/dL) in comparison to the control group (0.40 ± 0.05 g/dL) (Figure 3), associated with a qualitative observation for a depletion along the histological sections of the infected group (Figure 3). Treatment with BSN restored the plasma soluble proteins (0.64 ± 0.03 g/dL) with a significant improvement along the histological structure in the hepatic tissue when compared to the infected group (Figure 3).
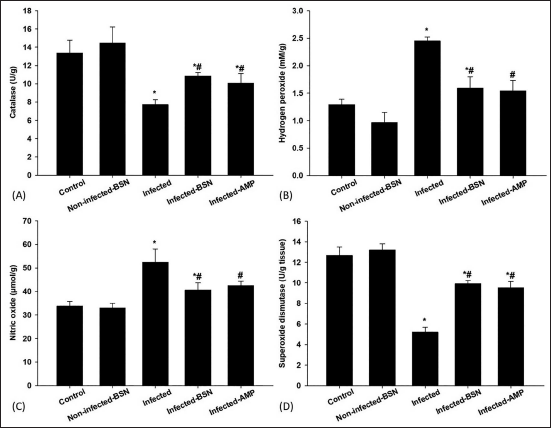
To investigate the effect of BSN on oxidative stress markers in the hepatic tissue, the CAT, NO, H2O2, and SOD levels were examined in Figure 4. There was a significant decrease in CAT level (7.71 ± 0.56 U/g) and SOD level (5.21 ± 0.46 U/g) in the infected group when compared to the control group (Figure 4). The anti-oxidant system within the infected hepatic tissue improved when BSN was given orally to the infected mice. In this case, treated mice showed a significant increase in the anti-oxidant activity of SOD and CAT, reaching 10.87 ± 0.35 U/g and 9.91 ± 0.33 U/g, respectively. Compared to the control group, infection with E. papillata induced a highly significant increase in the NO level (52.40 ± 5.64 µmol/g) and H2O2 concentration (2.45 ± 0.07 mM/g) (Figure 4). Treatment of mice with BSN significantly alleviated these E. papillata-induced increases with a significant decrease in NO (40.63 ± 3.03 µmol/g) and H2O2 (1.59 ± 0.21 mM/g) levels in the hepatic tissue (Figure 4).
Discussion
Previous studies discussed the ability of natural resources to counter murine coccidiosis rather than sulfanilamide drugs, with special reference to the intestinal site of infection. However, few research studies are concerned with the feedback of this disease on vital body organs (Dkhil & Al-Quraishy, 2012; Dkhil et al., 2011, 2012; Metwaly et al., 2012; Thagfan et al., 2023). This study assessed the potential role of SN using A. indica leaf extracts as a hepatoprotective effector against the impact of murine coccidiosis.
In this study, the BSN exhibits an efficient agent in ameliorating Eimeria infection as it could minimize the oocyst output on 5th-day p.i. than that of the infected group. The anti-coccidial activity of SN capped with molecules of A. indica was confirmed previously based on: (a) the presence of phytochemicals (such as saponins and alkaloids) in A. indica leaf extracts, which has a significant effect against parasites such as Eimeria species (Biu et al., 2006; Dkhil et al., 2012, 2013; Eze et al., 2020; Gotep et al., 2016; Ishaq et al., 2022; Toulah et al., 2010), (b) the therapeutic activity of SN may be attributed to an elevated immune response in infected mice against Eimeria species (Abdel-Gaber et al., 2023; Alkhudhayri et al., 2020; Frölich et al., 2012).
Our finding demonstrated the depletion in feed mice consumption following the Eimeria infection. Previous studies by Al-Quraishy et al. (2020), Abdel-Gaber et al. (2023) explained a possible hypothesis for this issue as Eimeria and its life stages causing severe damage in the site of infection (intestine) leads to poor absorption of nutrients. In addition, Ruff and Fuller (1975) reported that depletion in this rate may be attributed to malabsorption and disturbed metabolic status. Upon oral administration of BSN, improvement in food consumption rate was recorded due to the inhibition of Eimeria stages which is suggestive of the elevation of nutrient absorption across the intestinal wall and enhanced feed conservation ratio compared to the control group. This is consistent with Dkhil et al. (2012), Gotep et al. (2016), Alkhudhayri et al. (2020), Abdel-Gaber et al. (2023) reported the most beneficial health effects following treatment with SN and/or A. indica leaf extracts.
Our findings indicate alterations in liver function parameters due to Eimeria infection, which were associated with a significant increase in the activities of the enzymes ALT, AST, ALP, and bilirubin compared to the control group. These results are consistent with the studies by Çam et al. (2008), Patra et al. (2010), Mondal et al. (2011), Dkhil et al. (2011, 2012), and Esch and Petersen (2013), which reported that Eimeria infection leads to the destruction of hepatocyte cell membranes, impairing their permeability and resulting in the release of hepatic biomarkers into the bloodstream. Furthermore, Najafian et al. (2014) demonstrated that elevated levels of hepatic enzymes indicate damage to hepatocytes. Consequently, damaged hepatocytes release an excessive amount of hepatic enzymes into the bloodstream, resulting in increased serum levels. The Eimeria infection triggered an inflammatory response in the hepatic tissue of mice, primarily due to the presence of liver-resident macrophages, particularly Kupffer cells, within the sinusoids, as well as lymphocytes, which are a key component of the body’s response to injury. This observation aligns with the findings of Dkhil et al. (2012), who reported histopathological damage in the hepatic tissue of mice following Eimeria infection. Following treatment with BSN, the improvement in liver enzymes can be attributed to the recovery of hepatocytes and the epithelial lining, along with the inhibition of the parasite’s sexual stage development, especially when compared to the infected group. This suggests a potential hepatoprotective effect in mice. Dkhil et al. (2012) also noted that A. indica leaf extracts contain substances such as tannins, flavonoids, and glycosides that exhibit hepatoprotective properties. Furthermore, Alkhudhayri et al. (2018) reported a positive enhancement in the affected tissues of mice treated with SN after Eimeria infection.
In this study, Eimeria parasite-disturbed energy homeostatic status leads to the depletion of the hepatic carbohydrates and protein contents. In that case, the infected mice are in bodily demand for glucose, in which, hepatocytes synthesize glucose through gluconeogenesis leading to an increased level of plasma glucose, which is consistent with Mondal et al. (2011) and Metwaly et al. (2013). In addition, Dkhil et al. (2012) reported the necessity to keep the glucose energy source in its normal range in blood after infection, which occurs via lysis of the stored carbohydrate content within tissues. Moreover, the increase in the blood total protein levels as compared to the control group is suggestive of the proper maintenance of the integrity of the liver, which is responsible for the synthesis of plasma proteins. Our results are in agreement with Dkhil et al. (2012) and Onyiche et al. (2021). Moreover, Trenam et al. (1992) and Koinarski et al. (2006) reported that the excessive production of free radicals leads to the destruction of cellular bio-molecules such as proteins and carbohydrates, which is considered as another possible hypothesis for the depletion of these contents. Upon treatment, BSN restored hepatic content due to the active ingredients in the A. indica leaf extracts as well as the therapeutic activity of SN, which both combined in action to inhibit the activity of the Eimeria parasite.
In the present study, Eimeria infection was found to induce oxidative stress and disrupt the balance between anti-oxidants and pro-oxidants. This was evidenced by an increase in NO and H2O2 levels, alongside a decrease in CAT and SOD levels. These findings confirm that oxidative stress triggers a series of biochemical changes that can hinder the maintenance of homeostasis, consistent with the research of Dkhil and Al-Quraishy (2012), Dkhil et al. (2011, 2012), Metwaly et al. (2012), and Thagfan et al. (2023). Furthermore, the administration of BSN suspension was shown to lower oxidative enzyme levels and enhance the anti-oxidant capacity of liver tissue in mice by normalizing the values of CAT, SOD, NO, and H2O2. This aligns with previous studies highlighting the significant anti-oxidant effects of the biologically active compounds found in A. indica leaf extracts (i.e., nimbin, azadirachtin, nimbidiol, quercetin, and nimbidin) (Abdel-Gaber et al., 2023; Abdel Moneim et al., 2014; Dkhil et al., 2012, 2013; Manikandan et al., 2008; Xiang et al., 2018) as well as those of SN (Abdel-Gaber et al., 2023; Alkhudhayri et al., 2020). Furthermore, the increased activity of CAT following BSN treatment suggests a heightened capacity to scavenge H2O2 produced in response to oxidative stress caused by Eimeria infection, which is supported by the findings of Onyiche et al. (2021). The depletion of SOD can lead to irreversible cell damage due to the accumulation of reactive oxygen species (ROS), resulting in enhanced protein oxidation and destabilization of intracellular conditions (Georgieva et al., 2006). After oral administration of BSN, SOD activity was also found to increase, likely due to its activation through interaction with free radicals released during the infection. Additionally, the SOD enzyme neutralizes superoxide radicals by converting them to H2O2, thereby reducing the toxic effects associated with these radicals, as noted by Alkhudhayri et al. (2020) and Onyiche et al. (2021). Treatment with BSN also resulted in decreased levels of NO, indicating a downregulation of inducible nitric oxide synthase (iNOS) enzymes and a reduction in the inflammatory response following the recovery of hepatocytes after Eimeria infection. These findings are consistent with the work of Dkhil et al. (2011, 2012) and Thagfan et al. (2023).
Conclusion
Our data collectively suggest that biosynthesized nano selenium has the potential to reduce liver damage by improving pathological alterations, enhancing anti-oxidant status, and positively influencing biological markers such as oocyst output and feed consumption. Additionally, it affects other biochemical markers related to enzymatic activity in mice experimentally infected with E. papillata oocysts. Thus, biosynthesized nano selenium may serve as a potential alternative to the anti-coccidial drugs currently in use.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This research was approved by the Research Ethics Committee (REC) at King Saud University (approval number KSU-SE-22-66). All authors agreed to participate in this study. All authors agreed to publish in this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Researchers Supporting Project (RSP2025R25) from King Saud University, Riyadh, Saudi Arabia; and Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2025R437), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
