Abstract
Background
Eimeria papillata is a parasitic protozoan that causes intestinal coccidiosis in mice. Due to the rise of drug-resistant strains, medicinal plants are now employed to treat parasite diseases.
Objectives
This study investigated the beneficial and therapeutic effects of Persea americana extract (PAE) on biological, hematology, biochemical, and oxidative stress markers, as well as trace elements changes in the hepatic tissue of E. papillata-infected mice.
Materials and Methods
The animals were divided (five mice/group), as follows Group 1: Control group for non-infected-nontreated mice, Group 2: Treated group for mice orally treated with PAE (500 mg/kg b.w.), Group 3: Infected group for mice orally infected with Eimeria oocysts, Group 4: Infected-treated group for mice orally infected with Eimeria oocysts and then treated for 5 days with PAE (500 mg/kg b.w.), and Group 5: Infected-treated group for mice orally infected with Eimeria oocysts and then treated for 5 days with amprolium (120 mg/kg b.w.). On the 5th day postinfection, animals were euthanized, and samples (blood and livers) were collected from them.
Results
The coccidial infection caused weight loss of –2.54%, which improved to –1.36% with PAE. PAE therapy reduced the number of oocysts in mice’s feces. Furthermore, the Eimeria infection increased lymphocytes and neutrophils. The infection resulted in severe liver impairment, as evidenced by pathological changes as well as an elevation in liver function parameters such as aspartate aminotransferase, alanine aminotransferase, and alkaline phosphatase. Infection also resulted in a considerable reduction in glutathione reduced and an elevation in nitric oxide, and malonaldehyde. PAE demonstrated significant hepatoprotective activity by increasing these parameters. Our data revealed that E. papillata infection resulted in considerable increases in Fe, Cu, Zn, Ni, and Co concentrations, which were then reduced following PAE treatment.
Conclusion
Our results showed that PAE treatment significantly reduced hepatic damage and oxidative stress caused by E. papillata infections.
Introduction
Coccidiosis is an infectious protozoan disease caused by parasites of the genus Eimeria (Phylum Apicomplexa) (Pakandl, 2009). Eimeria species showed unique features for the site and host specificity, having a monoxenous life cycle (López-Osorio et al., 2020). This infection develops after consuming feed or water contaminated with sporulated oocysts. Oocysts reside and multiply in the intestinal tract, causing significant pathological alterations in infected animals (Abudabos et al., 2017). Clinical indications include decreased feed intake resulting in body weight loss, dehydration, and increased ability to other infections (Grès et al., 2003). Furthermore, the host may alter its micronutrient habitat, depriving Eimeria species of the nutrient requirements needed for replication (Doherty et al., 2002). Few investigations have found trace element alterations in organs involved in the process of disease (Abdel-Maksoud et al., 2012; Ilbäck et al., 2004, 2007; Metwaly et al., 2013). Danforth et al. (1992) mentioned that Eimeria papillata’s intracellular development in the mouse jejunum makes it an excellent model for studying animals (Danforth et al., 1992).
Coccidiosis can be treated with a variety of synthetic coccidiostats, such as amprolium, which limit coccidia’s normal growth, metabolism, and reproduction. However, the use of these drugs has been linked to immune system interference (Yunus et al., 2004), toxic effects on animal health (Nogueira et al., 2009), multidrug resistance (Abbas et al., 2011), and an excess of parasite infections in tissues (Onyiche et al., 2021). Scientists all over the world are turning to alternative methods for controlling parasite issues (Hamad et al., 2014). Several studies have shown that several compounds extracted from botanicals have great anticoccidial and other therapeutic properties (Abdel-Tawab et al., 2020; Al-Quraishy et al., 2014; Hussain et al., 2021).
Numerous studies have shown that avocado (Persea americana) is effective in treating and preventing a variety of ailments, as well as in confirming its traditional uses. Avocado, for example, has been shown to have antihypertensive (Adeboye et al., 1999), anti-inflammatory (Adeyemi et al., 2002), hypoglycemic (Antia et al., 2005), larvicidal (Leite et al., 2009), antiparasitic (Jiménez-Arellanes et al., 2013), antimicrobial (Cardoso et al., 2016), and antidiabetic properties (Umoh et al., 2019). As a result, avocado has achieved global awareness, and consumption has expanded significantly in recent years. This study established the hepatoprotective effects of P. americana extract (PAE) and its impact on changes in trace elements in the livers of infected mice.
Materials and Methods
Methanolic Extract Preparation
Fruits of the P. americana were bought at the Riyadh local market (Saudi Arabia). According to the procedure of Al-Otaibi et al. (2023), the 70% P. americana methanolic extract was prepared.
Animals and Infection
Twenty-five male C57BL/6 mice weighing between 20 and 23 g with 10–12 weeks of age were procured from the animal facility available in the College of Pharmacy at King Saud University. The experiment was conducted using mice following the institution’s policies regarding the care and use of animals in research (approval no. KSU-SE-22-40). Mice were allowed unlimited access to water and normal laboratory animal nutrition.
Mice were divided into five groups (five mice/group), as follows: Group 1: Control group for non-infected-non-treated mice, Group 2: Treated group for mice orally treated with PAE (500 mg/kg b.w.), Group 3: Infected group for mice orally infected with Eimeria oocysts, Group 4: Infected-treated group for mice orally infected with Eimeria oocysts and then treated with PAE (500 mg/kg b.w.), and Group 5: Infected-treated group for mice orally infected with Eimeria oocysts and then treated with amprolium (120 mg/kg b.w.).
Prof Heinz Mehlhorn of the Heinrich-Heine-Universitat in Germany provided the oocysts of E. papillata, which were then sporulated and propagated following Dkhil et al. (2011). Groups of 3–5 underwent oral gavage with 1,000 sporulated E. papillata oocysts suspended in 100 µl sterile saline. Following 60 min of infection, groups 4 and 5 received the recommended dosages of PAE and amprolium for 5 days, following the earlier study conducted by Al-Otaibi et al. (2023).
Using a McMaster chamber, oocyst shedding was quantified on day 5 and expressed as the number of oocysts per gram (OPG) of feces (Schito et al., 1996). The average weight gain was calculated as the difference between the weight on day 0 before treatment and day 5 after treatment. After that, on day 5, ether was used to euthanize every mouse to collect samples. Moreover, the mortality rate of dead mice was recorded throughout the experiment according to El Dakroury et al. (2016).
Sample Collection
Blood from the heart was collected into heparinized tubes, and plasma was separated and stored at –20°C for biochemical analysis. The livers of mice were taken and then divided into small pieces. For histology, parts of the liver were fixed in neutrally buffered formalin (10%), while others were kept at –80°C for trace element and oxidative stress analysis.
Neutrophils and Lymphocytes Count
After collecting blood, 3 µl was used to prepare a thin smear, stained with Giemsa, and examined under a Leica DM 2500 microscope with a software of NIS ELEMENTS (ver 3.8).
Biochemical Analysis
Liver functions were assessed in blood plasma using commercial kits (Biodiagnostic Co., Egypt). Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were determined using the Reitman and Frankel (1957) method. Alkaline phosphatase (ALP) was quantified by applying Belfield and Goldberg’s (1971) method. A molecular device (Spectra MAX 190) equipped with the software SoftMax® Pro (ver 6.3.1) was used to measure the absorbance.
Histological Study
Before being embedded in paraffin wax, fixed liver samples in 10% formalin were repeatedly diluted with ethyl alcohol to dehydrate them. As described by Drury and Wallington (1980), serial sections of 5 µm were cut, deparaffinized, and stained with hematoxylin and eosin (H&E). A Leica DM 2500 microscope (NIS ELEMENTS software, version 3.8) was used to examine and photograph the liver sections. The methodology for liver histology scoring followed Ishak et al. (1995).
Oxidative Status
Liver samples were processed according to Tsakiris et al. (2004) and then homogenized. Using commercial kits (Biodiagnostic Co., Egypt), the supernatant was colorimetrically analyzed. The conventional protocols of Beutler et al. (1963), Montgomery and Dymock (1961), and Ohkawa et al. (1979) were utilized to assess oxidative stress markers, including glutathione reduced (GSH), nitric oxide (NO), and malonaldehyde (MDA), respectively. Absorbance was measured using a molecular device (Spectra MAX 190) supplied with SoftMax® Pro software (ver 6.3.1).
Trace Elements Detection
Liver samples were digested after drying according to UNEP/FAO/IOC/IAEA (1984). The inductivity-coupled plasma iCAP-6500 Duo (Thermo Scientific, UK) was used to quantify the levels of iron (Fe), zinc (Zn), copper (Cu), cobalt (Co), and nickel (Ni). Values were expressed in mg/g dry weight.
Statistical Analysis
SigmaPlot® version 11.0 (Systat Software, Inc., Chicago, IL, USA) was used for statistical data analysis. Values were presented as mean ± SD. The p value ≤ 0.05 was considered a statistically significant difference.
Results
At day 5 postinfection (p.i.), oocyst shedding was extremely varied between groups, with the highest levels reported in the feces of mice experimentally infected with E. papillata 4,075 ± 129.7 × 106 oocyst/g of feces (Table 1). PAE inhibited oocyst shedding in the infected-PAE group by 594.3 ± 89.4 × 106 oocyst/g of feces. PAE, similar to amprolium, was highly effective against E. papillata (Table 1). Moreover, the addition of PAE induced a significant reduction (20%) in the mortality rate than those observed in the infected group (40%). The control and non-infected-PAE groups showed no inhibitory effect since they had not been infected with Eimeria species.
Oocyst Output and Weight Change in Mice at Day 5 Postinfection (p.i.) with E. papillata.
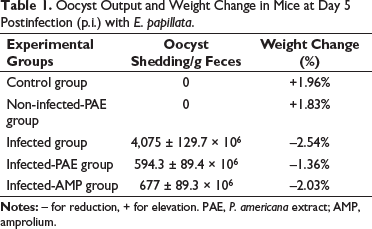
At day 5 p.i., mice infected with E. papillata had about –2.54% weight loss compared to mice in the control and non-infected-PAE groups who gained 1.96% and 1.83%, respectively (Table 1). After treatment, mice treated with PAE lost 1.36% body weight and amprolium lost 2.03% less than the infected group (Table 1).
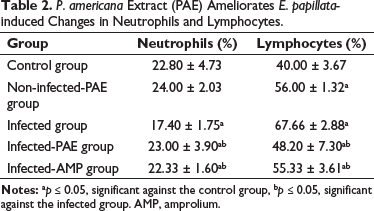
The effect of PAE on white blood cells (WBCs) in mice infected with Eimeria oocysts is displayed in Table 2. The neutrophil count (17.40 ± 1.75), when compared to the control and non-infected-PAE groups, was significantly lower in the infected group. Table 2 shows the lymphocyte levels in the infected group were significantly higher (67.66 ± 2.88) compared to the control and non-infected-PAE groups. When compared to the infected group, treatment with PAE led to a significant reduction in neutrophils and lymphocytes (Table 2).
P. americana Extract (PAE) Ameliorates E. papillata-induced Changes in Neutrophils and Lymphocytes.
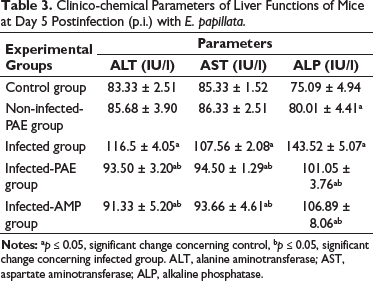
The clinico-chemical parameters that indicate liver functions in mice treated with PAE and amprolium after experimental infection with E. papillata are displayed in Table 3. Following Eimeria infection, the infected group’s ALT (116.5 ± 4.05 IU/l), AST (107.56 ± 2.08 IU/l), and ALP (143.52 ± 5.07 IU/l) were all considerably higher than those of the control group. Between treatments, there were significant differences in the levels of ALT, AST, and ALP. In comparison to the drug group, PAE reduced the increase to 93.50 ± 3.20 IU/l (ALT), 94.50 ± 1.29 IU/l (AST), and 101.05 ± 3.76 IU/l (ALP) (Table 3).
Clinico-chemical Parameters of Liver Functions of Mice at Day 5 Postinfection (p.i.) with E. papillata.
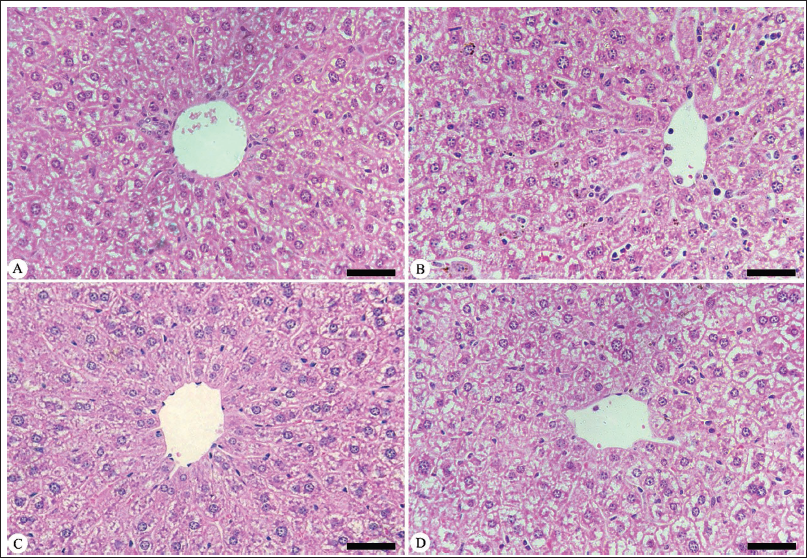
The Eimeria infection caused a moderate pathological change in the hepatic tissue, as evidenced by inflammatory cellular infiltrations, cytoplasmic vacuolation, and sinusoid dilatations, with more Kupffer cells than the control group (Figure 1). According to Ishak’s score, all visible hepatic alterations in the infected group are classified as 9–10 at day 5 p.i., compared to the control group (Table 4). PAE treatment significantly reduced the E. papillata-induced pathological alterations in the hepatic tissue compared to the infected group (Figure 1).
Protective Role of PAE Against Infection with E. papillata in the Hepatic Tissue of Mice. (A) Non-infected Mouse Liver. (B) Infected Mouse Liver on Day 5 Postinfection (p.i.). (C) Liver Treated with P. americana Extract (PAE). (D) Liver Treated with Amprolium (AMP). Sections Stained with Hematoxylin and Eosin (400×).
Total Histological Scores in the Hepatic Tissue of Mice Infected with E. papillata on Day 5 Postinfection (p.i.).

Figure 2 depicts the GSH, NO, and MDA levels used to explore PAE’s antioxidant properties. The infected group had a significantly lower GSH level (15.40 ± 0.76 mg/g) than the control group (Figure 2). However, treating mice with PAE was able to significantly elevate GSH levels to those of the infected group. The infected group with E. papillata had significantly higher NO levels (36.72 ± 2.51 µmol/g) and MDA concentrations (25.07 ± 0.93 nmol/g) compared to the control group (Figure 2). The change in the levels of NO and MDA caused by the Eimeria infection was significantly ameliorated, and the oxidative stress in hepatic tissue was reduced following the PAE treatment (Figure 2).
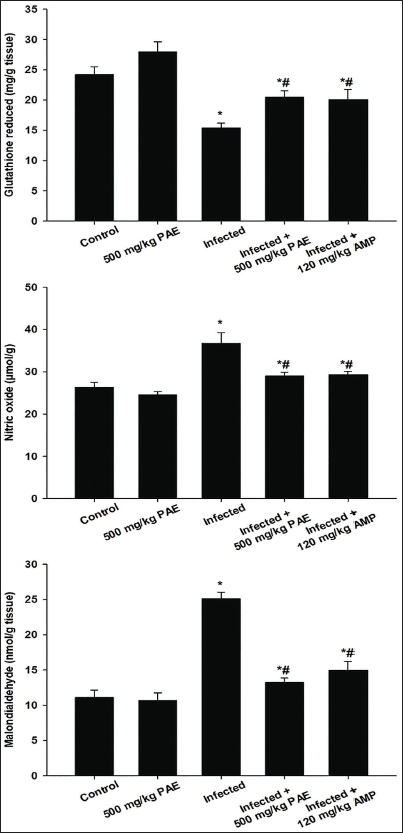
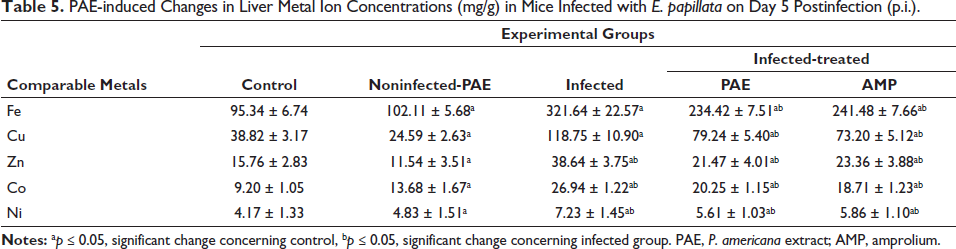
The levels of five trace elements were evaluated in the hepatic tissue of all experimental groups on day 5 p.i. (Table 5). The Eimeria infection significantly altered the level of trace elements. Infected mice had a significantly higher Fe concentration (321.64 ± 22.57 mg/g) compared to the control group (95.34 ± 6.74 mg/g) (Table 5). Infected mice had significantly higher levels of Zn and Ni (38.64 ± 3.75 mg/g and 7.23 ± 1.45 mg/g, respectively) compared to the control group (Table 5). Infected mice had a threefold higher quantity of Cu in hepatic tissue (118.75 ± 10.90 mg/g) compared to the control group (38.82 ± 3.17 mg/g) (Table 5). Infection with E. papillata increased Co levels in mice by around 50% (26.94 ± 1.22 mg/g) compared to the control group (13.68 ± 1.67 mg/g) (Table 5). Table 5 shows that treatment with PAE and amprolium significantly reduced the rise in Fe, Cu, Zn, Ni, and Co metal levels caused by E. papillata.
PAE-induced Changes in Liver Metal Ion Concentrations (mg/g) in Mice Infected with E. papillata on Day 5 Postinfection (p.i.).
Discussion
Coccidiosis is caused by protozoan Eimeria parasites following fecal-oral uptake of sporulated oocysts, which affects a variety of animal species and results in significant economic losses (Al-Quraishy et al., 2011). This disease has been treated with around 30 sulfanilamide drugs, including amprolium, which have a wide range of toxic effects on animal host tissues (Noack et al., 2019). Nowadays, a key area of research is determining substitutes for anticoccidial agents in the treatment and management of coccidiosis. Natural plants currently provide new approaches for innovative therapies against parasitic infections (Newman & Cragg, 2007). This study investigated the ameliorative role of PAE as a hepatoprotective agent in addition to its anticoccidial properties.
In this study, PAE was found to be an efficient agent in treating Eimeria infection because it reduced oocyst shedding on the 5th-day p.i., as well as decreasing the mortality of experimental mice, in comparison to the infected group. This suggests that the extract has anticoccidial effects. This agreed with Kosińska et al. (2012), Di Stefano et al. (2017), Melgar et al. (2018), Castro-López et al. (2019), Rosero et al. (2019), and Abd Elkader et al. (2022), who found bioactive compounds in P. americana that have a strong antimicrobial activity by disrupting microbial cell walls and inhibition of the electron transport system within Eimeria mitochondria. In our earlier studies of Al-Otaibi et al. (2023), we verified that phytochemicals in PAE, which change the integrity of the parasite membrane and cause loss of homeostasis and ultimately the death of the parasite before the oocysts are formed in the intestinal mucosa and released with feces, are the cause of the reduced oocyst output.
According to our findings, the E. papillata infection caused weight loss. Previous studies clarified that this loss results from the Eimeria stages consuming the nutrients inside the intestinal epithelium, which leads to notable alterations (Al-Quraishy et al., 2020; Anwar et al., 2008; Dkhil et al., 2014; Ghanem & Abdel-Raof 2005; Gilbert et al., 2011; Hamid et al., 2019; Metwaly et al., 2013; Ogbe et al., 2009; Qaid et al., 2021). Following treatment with PAE, a significant improvement in body weight was observed due to the phytochemicals in the plant extract, which improved the nutritional status of mice compared to the control group, which is similar to Al-Otaibi et al. (2023). Furthermore, the percentage of neutrophils and lymphocytes changed significantly as a result of the Eimeria infection. This may relate to the crucial role of these cells during E. papillata invasion infected mice’s intestinal epithelium (Amer et al., 2015).
Our findings suggested that the Eimeria infection caused certain pathological alterations, including a significant increase in AST, ALT, and ALP activities as compared to the control group as a result of the trace element imbalance. This is consistent with McCall et al. (1971), who reported that trace elements are regarded as critical factors for the maintenance of the structural and functional integrity of tissues inside organisms. Our findings are consistent with those published by Çam et al. (2008), Patra et al. (2010), Mondal et al. (2011), and Dkhil and Al-Quraishy (2012), who found that the Eimeria infection caused cell membrane permeability and damage in the hepatocytes. As a result, the destructed hepatocytes release an overabundance of hepatic enzymes into the bloodstream, increasing their levels in serum. Indeed, the Eimeria infection induced an inflammatory response in mouse hepatic tissue of mice due to the abundance of liver resident macrophages (particularly Kupffer cells) within sinusoidal lumen and leukocytes (particularly lymphocytes), which is a common response of body tissues facing injury impact. This was consistent with Dkhil and Al-Quraishy (2012), who found liver damage in mice after infection with E. papillata. PAE treatment decreases pathological injury and inflammation in the hepatic tissue by lowering enzyme levels compared to the infected group, implying a potential hepatoprotective effect in mice. This finding supported our previous studies in Al-Otaibi et al. (2023) that phytochemicals in PAE have therapeutic properties.
According to Georgieva et al. (2006), Eimeria infection results in an imbalance in the body’s natural antioxidant defense and production of free radicals (Georgieva et al., 2006). Our findings showed that E. papillata infection causes oxidative damage to the hepatic tissue of mice, resulting in the reduction of GSH levels and the depletion of antioxidant enzymes. During Eimeria infection, this oxidative parameter is crucial for shielding the animal’s body from damage caused by free radicals. The current study confirms the findings of Hürkul et al. (2021) and Al-Otaibi et al. (2023), who reported that the main cause of PAE’s significant antioxidant activity is the high concentration of phenolic compounds with antioxidant properties in the PAE. PAE also prevents the Eimeria infection-induced loss of GSH in the hepatic tissue.
The hepatic tissue of the infected mice exhibited higher levels of NO and MDA due to their imbalanced status. This suggested the presence of oxidative stress resulting from the Eimeria infection and the disruption of the equilibrium between pro-oxidants and antioxidants. Our results supported the findings of Koinarski et al. (2005) and Georgieva et al. (2006), who found that elevated MDA and NO levels were an immunological reaction to the presence of infectious sporozoite stages, which infiltrate intestinal cells and induce inflammation. The levels of MDA and NO decreased following PAE treatment. This is consistent with the study of Al-Otaibi et al. (2023), which showed that phytochemicals in PAE bind to important macromolecules on parasite membranes, disrupting them and subsequent death of the parasite.
Hepatic tissue is considered both a master player in the detoxification process and a victim of trace elements toxicity. Previous studies have shown that trace element levels alter in different organs involved in the disease process (Ilbäck et al., 2004, 2007). This was consistent with our findings, which showed that Eimeria infection can modify trace element levels (Fe, Cu, Zn, Co, and Ni) in mice’s hepatic tissues. Furthermore, Ilbäck et al. (2008) found that most viral disorders cause changes in the amounts of many trace elements in the blood and tissue. Nimbalkar et al. (2018) reported that trace elements function as cofactors for many enzymes in multiple metabolic pathways, therefore, variations in distribution have toxicological effects, which is consistent with our findings. The level of these elements increased following PAE administration, and their effect was combined with the balance of hepatic enzymes and oxidative stress indicator levels due to phytochemicals that play an effector function in this homeostasis.
Conclusion
Overall, our findings show that treatment with the PAE can successfully alleviate the disturbances in biological and biochemical indicators, histopathological damage, oxidative stress as well as trace elements imbalance in infected mice’s hepatic tissues. Further studies are required to include the investigation of the inflammatory markers in the hepatic tissue regarding the Eimeria infection and the role of PAE in ameliorating its impacts.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This research was approved by the Research Ethics Committee (REC) at King Saud University (approval number KSU-SE-22-40).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Researchers Supporting Project (RSP2024R25), King Saud University, Riyadh, Saudi Arabia; and also supported by Princess Nourah Bint Abdulrahman University Researchers Supporting Project (Number PNURSP2024R437), Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
