Abstract
Tripterygium wilfordii Hook F. (TwHF), a member of the Celastraceae family and belonging to the genus Tripterygium, is a vine-like shrub. It contains numerous active components, such as Tripterygium glycosides (GTW) and triptolide (TP), widely known for their significant reproductive toxicity. This toxicity is characterized by its detrimental effects on the testes and ovaries, leading to reduced libido, apoptosis of germ and follicle cells, and abnormal secretion of sex hormones. Thus, it is necessary to explore the mechanisms behind TwHF’s reproductive toxicity, which will lead to advancements in detoxification and efficacy enhancement strategies. This article offers a comprehensive overview and analysis of the research conducted on reproductive toxicity and its reversibility induced by TwHF, aiming to provide a reference for its rational use in clinical settings.
Introduction
Recent years have witnessed extensive research on Tripterygium wilfordii Hook F. (TwHF), uncovering a wide range of therapeutic properties, including immunosuppressive, anti-inflammatory, anti-atherosclerosis, anti-tumor, anti-rejection, and neuroprotective effects (Yang & Liu, 2018). Hence, it is extensively utilized (Song & Gao, 2023) in the management of conditions such as purpuric nephritis, rheumatoid arthritis, diabetic nephropathy, and psoriasis, among others (Clinical indications for the use of TwHF are listed in Table 1). However, a critical concern arises with its pronounced reproductive toxicity. The effective dose for anti-fertility (20 mg/d) is significantly lower than that required for other treatments, being just 1/3 of the immunosuppressive dose (1–1.5 mg/kg) (Ye et al., 1992). Presently, in clinical practice, the reproductive toxicity of TwHF is often managed by coadministration with detoxifying agents like Cistanche deserticola, resveratrol, and dodder seed (Liu, Qi, et al., 2018). Nonetheless, its efficacy for individuals necessitating prolonged TwHF use remains limited. This article is committed to providing an exhaustive summary and analysis of the reproductive toxicity mechanisms attributed to TwHF and illuminating strategies to reduce toxicity while enhancing its therapeutic efficacy.
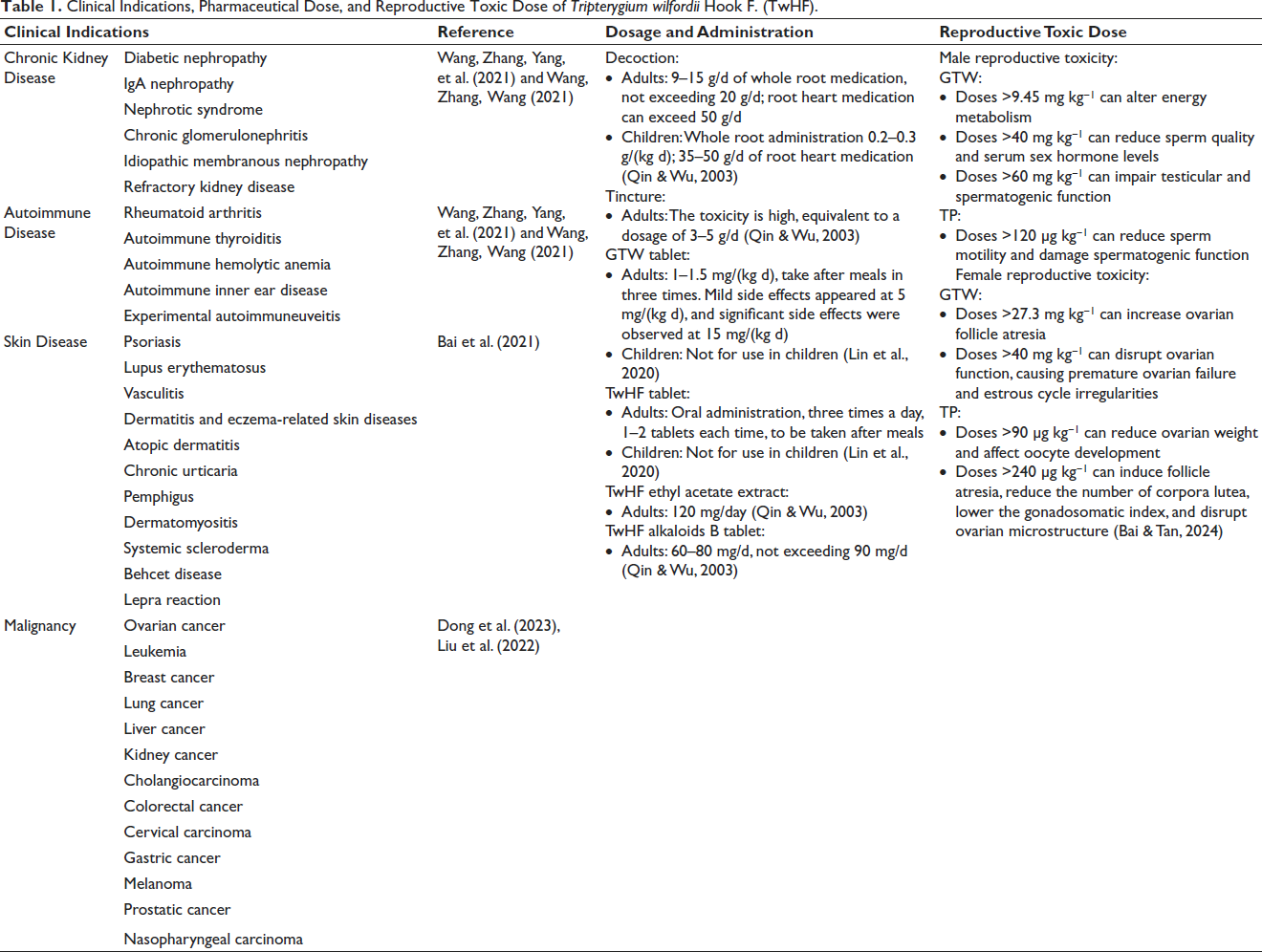
Clinical Indications, Pharmaceutical Dose, and Reproductive Toxic Dose of Tripterygium wilfordii Hook F. (TwHF).
The Damage and Reversibility of Tripterygium wilfordii to the Male Reproductive System
Clinical Observation
Documented evidence since the 1980s has consistently established a link between the clinical use of TwHF and male reproductive dysfunction. Research by Bao (2021) highlighted that Tripterygium glycosides (GTW) tablets used in treating male patients with proteinuria could induce asthenozoospermia. In a related study, Su et al. (1987) found a reduction in libido in 2 out of 18 male patients treated with a TwHF decoction, averaging 27.5 years of age. In an attempt to treat 26 male patients with mild psoriasis, a clinical dosage ranging from 1/3 to 1/4 of the GTW standard dose was employed. This treatment led to a significant decline in sperm density and vitality, with the most severe cases experiencing a total loss of fertilization capability (Qian et al., 1989).
Experimental Study
Testicular Reproductive Toxicity
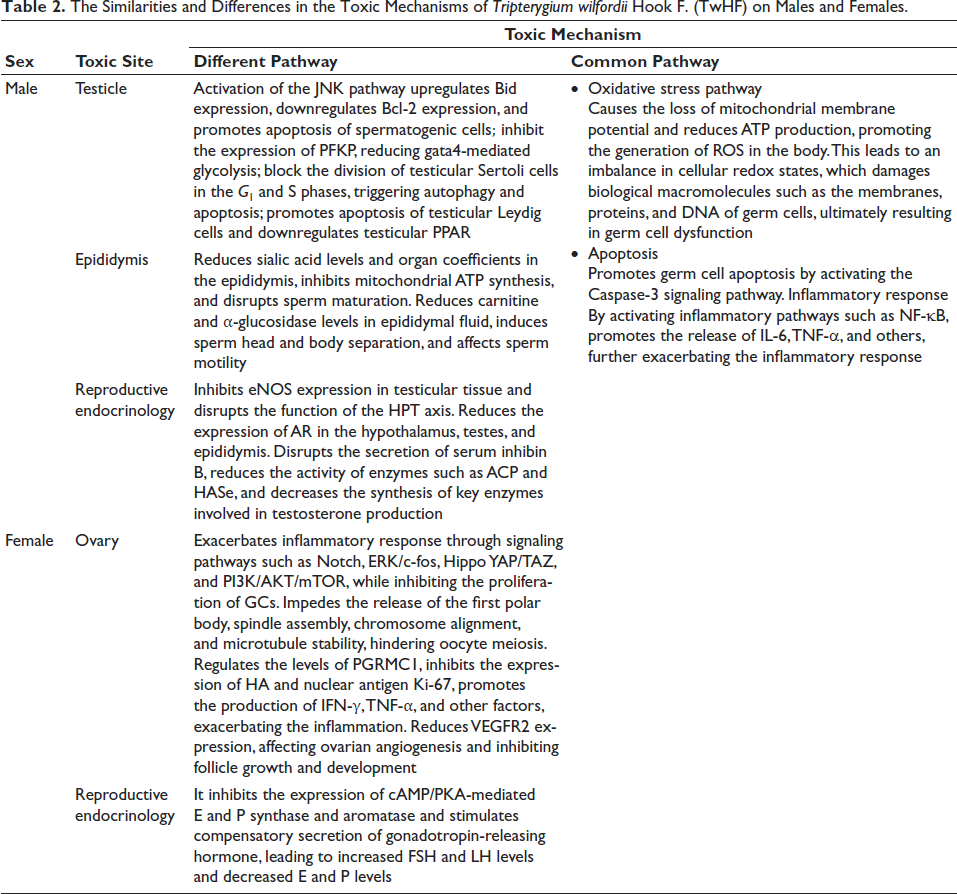
The testicle plays a critical role in facilitating spermatogenesis and synthesizing male hormones (Ren et al., 2021). Research conducted by Li et al. (2015) revealed that TwHF primarily exhibits reproductive toxicity by targeting the testicle, leading to its accumulation in it. In a study conducted by Huang et al. (2009), it was observed that exposure to GTW led to the abnormal expression of 354 genes associated with reproductive function in the testicles of mice, with 112 genes being upregulated and 242 genes downregulated. Researchers (Xiu et al., 2020) have identified that triptolide (TP) significantly affects primary spermatocytes during the pachytene stage in C57BL/6 rats. Numerous studies (Huang et al., 2019; Qiu et al., 2019; Wang et al., 2018; Ye & Chen, 2019) have shown that TwHF mainly affects rats’ germ cells through three mechanisms: oxidative stress, apoptosis, and inflammatory response. GTW can increase reactive oxygen species (ROS) levels, disrupting redox balance and triggering oxidative stress reactions. At the same time, GTW can reduce the messenger ribonucleic acid (mRNA) expression of anti-apoptotic genes (Bcl-2, PIWIL2, STAT3, CREM) in rats’ testicular tissue and increase the mRNA expression of apoptotic genes (BAX, FAS, FASL, CASPASE3) in spermatocytes, thereby promoting germ cell apoptosis. Additionally, the release of tumor necrosis factor-α (TNF-α) and other factors activates pathways such as nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and c-Jun N-terminal kinases (JNK), exacerbating the inflammatory response. Furthermore, TwHF reduces the content of sperm protein and total alkaline protein, thereby promoting apoptosis in spermatogenic cells. Additionally, it has been observed (Zhang, Fang, et al., 2021; Zhang, Tang, et al., 2021) in GATA4-treated TM4 cells BALB/c mice that TwHF inhibits the expression of phosphofructokinase, platelet (PFKP) and decreases GATA4-mediated glycolysis, leading to testicular toxicity.
Liu, Qi, et al. (2019), Liu, Tian, et al. (2019) and Liu, Yin, et al. (2019) discovered that administering GTW tablets can have harmful effects on the seminiferous tubules of male rats afflicted with collagen-induced arthritis. Earlier studies have shown that TP can hinder the division of testicular supporting cells during the G1 and S phases. This interference leads to a cascade of detrimental outcomes in SD rats, including reduced contraction, decreased activity, compromised adhesion capabilities, and ultimately, the initiation of autophagy and apoptosis (Zhang et al., 2020). Additionally, research by Guan et al. (2019) has unveiled that TP can adversely affect the secretion of sex hormones in SD rats by promoting apoptosis in testicular interstitial cells. In a related study by Ma et al. (2015), rats were subjected to intraperitoneal injections of TP (60 µg kg−1) over a 2-week period. This research highlighted a significant decrease in the levels of a marker enzyme indicative of testosterone levels and a downregulation of the testicular peroxisome proliferator-activated receptor (PPAR), underscoring the profound impacts of TP on male reproductive health.
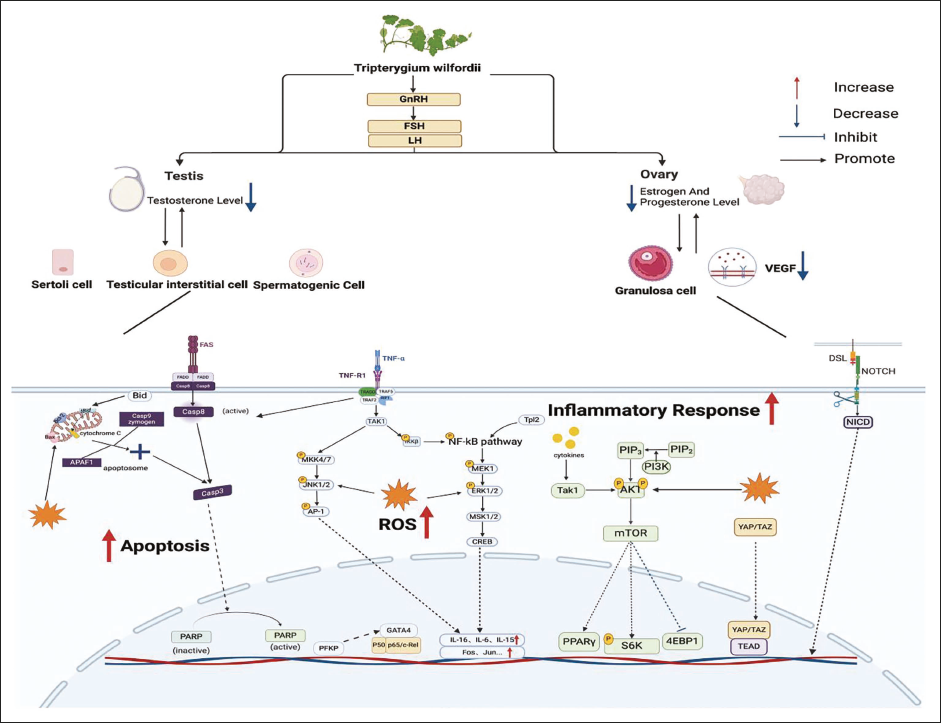
One mechanism by which TwHF induces reproductive toxicity is through the suppression of endothelial nitric oxide synthase (eNOS) expression in testicular tissues and the disturbance of the hypothalamus–pituitary–gonadal (HPG) axis’s functionality. Guo et al. (2019) observed in their studies that in SD rats, the administration of high doses of TwHF significantly reduces androgen receptor expression within the hypothalamus, testicles, and epididymis. Moreover, GTW can interfere with the secretion of serum inhibin B, and diminish the synthesis of key enzymes involved in testosterone production. These disruptions collectively compromise rats’ spermatogenesis and fertilization potential (Jing et al., 2017; Yang et al., 2002, 2010). The testicular reproductive toxicity mechanism of TwHF is shown in Figure 1 and Table 2.
The Similarities and Differences in the Toxic Mechanisms of Tripterygium wilfordii Hook F. (TwHF) on Males and Females.
Epididymal Reproductive Toxicity
The epididymis acts as a crucial intermediary structure, bridging the testicle and the vas deferens, where it plays essential roles in sperm storage, transport, and maturation. Li et al. (2009) found in their research that TP can lead to a decrease of Sprague–Dawley male rats in sialic acid levels and organ coefficients in the epididymis, inhibit the synthesis of mitochondrial adenosine triphosphate (ATP) in sperm, and interfere with sperm maturation processes. In another study by Wang et al. (1999), a group of SD rats received TP orally for 5 weeks. The findings showed a marked decrease in carnitine and α-glycosidase levels in the epididymal fluid, alongside the separation of the sperm head from the acrosome and the presence of detached sperm cells within the epididymal cavity. Moreover, further research has indicated that in SD rats TP leads to negative changes in sperm motility, including their speed and linear trajectory (Chen et al., 2022). The reproductive toxicity mechanisms of TwHF in the epididymis are shown in Figure 1 and Table 2.
Reversibility Research
Research conducted in the 1980s concluded the male infertility caused by TwHF could indeed be reversed (Su et al., 1987). Subsequent research demonstrated that the reductions in sperm density and viability in male mice, rats, and macaques caused by TwHF could recover within 4–10 weeks after discontinuation of the treatment (Jing et al., 2016; Lin et al., 2000). Su et al. (1987) investigated the condition of 18 male patients who had stopped taking TwHF decoction and found that patients taking a daily decoction of TwHF (10–20 g) experienced a significant reduction in sperm count within 2 weeks. However, for those who discontinued the medication for more than 3 months, sperm count increased significantly or returned to normal, suggesting that the toxicity of TwHF on the male reproductive system is reversible. However, other studies have noted variability in the fertility-related effects of different TwHF sources, suggesting that the degree of recovery after discontinuing the medication might vary (Wu et al., 2015). Through gas chromatography–mass spectrometry (GC–MS) analysis, Zhang, Fang, et al. (2021) and Zhang, Tang, et al. (2021) found that the composition and relative content of volatile components in TwHF from different regions vary significantly, possibly due to factors such as different ecological environments and germplasm resources.
The Damage and Reversibility of Tripterygium wilfordii to the Female Reproductive System
Clinical Observation
Female patients treated with TwHF face an increased risk of menstrual cycle disorders, amenorrhea, decreased sexual function, and premature ovarian failure. Studies show a 65.1% likelihood of experiencing menstrual disorders or amenorrhea among these patients (Shu et al., 1987). Research involving 81 female patients revealed that 34% suffered from reproductive system damage after GTW treatment (Qiu et al., 2004). Ding et al. (2013) reported that 26.92% of adolescent female patients with purpuric nephritis encountered temporary menstrual abnormalities following medication treatment. Furthermore, Yang et al. (2006) demonstrated that administering GTW at dosages ranging from 20 to 60 mg/d can lead to amenorrhea in women.
Experimental Study
The ovary is essential for egg production, ovulation, and sex hormone secretion. Research aimed at developing a rat model to investigate ovarian dysfunction induced by TwHF found that while the uterine index remained unaffected, the ovarian index was significantly reduced (Ni et al., 2015). This indicates that the ovary is the primary organ affected by TwHF’s reproductive toxicity.
Ovarian Cell Growth and Development Disorders and Apoptosis
In their study, Ye et al. (2016) found that rats with premature ovarian failure induced by GTW exhibited disturbances in the estrous cycle, developmental abnormalities in follicles, and a reduction in both ovarian function and the ovarian index. Further research has shown that apoptosis in mammalian ovarian granulosa cells (GCs) can trigger follicular atresia, leading to decreased reproductive capacity in mature gilts (Liu et al., 2002). Numerous studies (Bai et al., 2019; Chen et al., 2014; Jiang et al., 2021; Liu et al., 2015; Liu, Qi, et al., 2018; Liu, Qi, et al., 2019; Liu, Tian, et al., 2019; Liu, Yin, et al., 2019; Liu, Zhang, et al., 2018; Ruan et al., 2017; Su et al., 2014; Xu et al., 2016; Zeng et al., 2014; Zhang et al., 2014; Zhao et al., 2015) have shown that in rats, the effects of TwHF can be achieved through the promotion of ROS, apoptosis, and inflammatory responses. GTW can disrupt the redox balance, increase the levels of ROS, and exacerbate oxidative stress. Additionally, GTW can affect signaling pathways such as Caspase, Notch, and Hippo YAP/TAZ, increasing the mRNA expression of apoptotic genes (BAX, FAS, FASL, CASPASE3) in GCs, thereby promoting apoptosis. Furthermore, the release of TNF-α and other factors activates pathways such as ERK/c-fos, and PI3K/Akt/mTOR, intensifying the inflammatory response. Additionally, TwHF disrupts the balance of ovarian apoptotic genes BCL2/BAX and affects the regulation of progesterone receptor membrane component 1, decreases the expression of hyaluronic acid, inhibits the expression of nuclear antigen Ki-67, and enhances the production of interferon-gamma (IFN-γ) and TNF-α. These changes lead to the apoptosis of GCs and consequent impairment of ovarian function.
Research by Hao et al. (2023) has shown that TwHF can obstruct the release of the initial polar body and disrupt spindle assembly, chromosome alignment, and the stability of microtubule proteins in rats, thereby impeding the meiotic progression of oocytes. Moreover, studies by Zhao et al. (2021) suggest that TwHF may reduce the expression of vascular endothelial growth factor, resulting in decreased ovarian angiogenesis in scenarios of insufficient blood supply. (The reproductive toxicity mechanisms of TwHF on GCs are shown in Figure 1 and Table 2).
Disrupting Reproductive Endocrine System
Estrogen (E) and progesterone (P) are essential for maintaining female reproductive function. Recent studies have revealed that in rat models, TwHF can stimulate the compensatory secretion of gonadotropin-releasing hormone, leading to a marked increase in serum levels of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) while concurrently reducing the levels of E and P. These effects are likely due to the inhibition of cyclic adenosine monophosphate (cAMP)/protein kinase A (PKA)-mediated expression of E and P synthase, along with the modulation of aromatase activity (Wang, Zhang, Wang, 2021; Wang, Zhang, Yang, et al., 2021). The reproductive toxicity mechanisms of TwHF on the reproductive endocrine system are shown in Figure 1 and Table 2.
Reversibility Research
There is a divergence of opinions on the potential reversibility of female reproductive toxicity caused by TwHF. In research by Meng and Ma (2019), the administration of GTW for 4 weeks led to a decrease in ovarian volume, follicles, and corpus luteum in all female patients. However, a notable recovery of these pathological changes was observed after an 8-week cessation period, with a full return to normal observed after 12 weeks of discontinuation. This indicates that the ovarian pathological changes induced by TwHF might be a delayed and reversible process. Contrastingly, Lin et al. (2019) found that hormone levels and the rate of follicle cell apoptosis in female rats showed no signs of recovery even after a 35-day period following discontinuation, suggesting that the reproductive toxicity induced by TwHF could be irreversible. In a clinical setting, Lin et al. (2008) analyzed 25 patients suffering from amenorrhea due to TwHF and found that promptly stopping the medication and starting hormone therapy could effectively restore reproductive function. However, Yang et al. (2006) observed that female patients experienced varied durations of menstrual recovery after discontinuing TwHF, with dosages ranging from 20 to 60 mg/d, highlighting the complex and variable nature of TwHF’s effects on female reproductive health.
Numerous clinical cases have established a clear correlation between the degree of female reproductive toxicity caused by TwHF and both the dosage and duration of its administration, with age identified as a significant determinant. Remarkably, Zhou (2011) found that the total dosage in patients who developed amenorrhea often exceeded 8,000 mg, and more than half of these patients were able to restore their menstrual cycles after treatment. Additionally, younger patients tend to have a higher chance of recovery after ceasing the medication compared to older individuals, especially those aged. In particular, adolescent female patients diagnosed with adolescent purpura nephritis who halt the medication for 2 months can achieve recovery, with normalcy generally observed within 6 months. Additionally, the amenorrhea seen in premenopausal female patients post-TwHF administration tends to resolve within 26–40 days after stopping the medication. Within a group of 46 perimenopausal female patients treated with GTW for uterine bleeding, only nine cases (19.6%) succeeded in reinstating regular menstrual cycles after a 60-day medication discontinuation period (Zheng, 2011).
Damage to the Reproductive System of Children/Adolescents and Its Reversibility
There is a limited body of literature pertaining to the clinical observation and experimental investigation of the reproductive toxicity of TwHF on children, with a predominant emphasis on the enduring impacts of TwHF on the gonads of children. Through an examination of clinical cases, it has been determined that the occurrence of TwHF toxicity in children is more prevalent in males compared to females (Li et al., 2023). In a study, Zhang et al. (1994) studied a group of pediatric patients who had an average administration of GTW at 1.29 mg/kg d for an average duration of 102.8 days, with a cumulative average dosage of 120.9 mg/kg, and found that 8 years after discontinuation, male children were more likely to experience spermatogenic dysfunction, while the impact on female reproductive function was comparatively less. The subsequent findings of Ding et al. (2013) demonstrated a correlation between the adverse effects of TwHF on the gonads of children and factors such as the manufacturer’s production quality standards, duration of medication, dosage, and concurrent therapy. In the realm of experimental investigation, certain scholars have observed a heightened susceptibility of young male mice’s germ cells to GTW, resulting in genetic toxicity (Yang et al., 2008). A study conducted by Leng et al. (2011) has demonstrated that the administration of 6 mg/kg d GTW for 4 weeks can induce transient and reversible harm to the testicular tissue of adolescent rats, whereas 12 mg/kg d GTW for 4 weeks can lead to enduring damage.
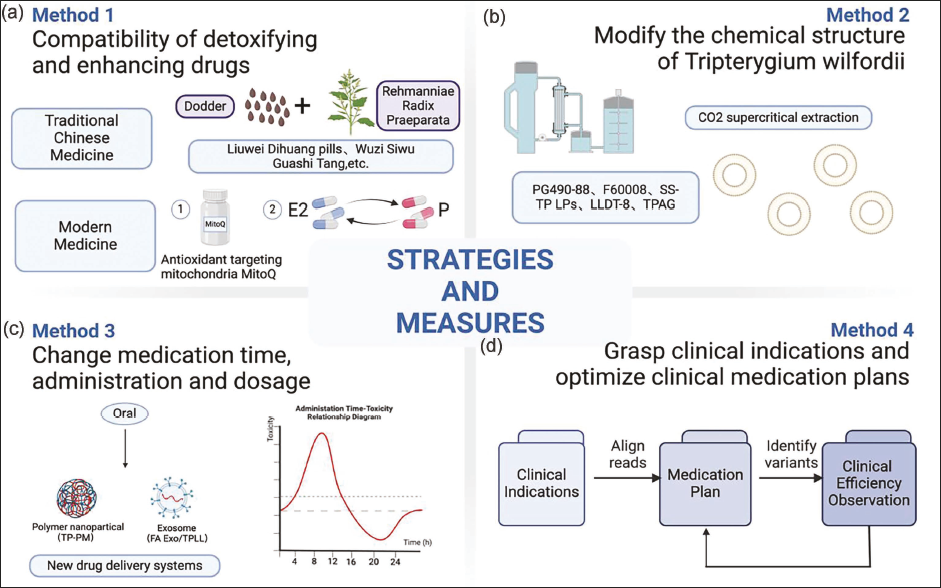
Strategies and Measures
Compatibility of Detoxifying and Enhancing Drugs
Traditional Chinese Medicine
In Traditional Chinese Medicine, the kidney is considered the cornerstone of vitality, primarily tasked with storing essence, which is pivotal for reproductive and developmental processes, closely linked to male ejaculation and the female menstrual cycle. The reproductive toxicity of TwHF mainly stems from the damage to essence and blood. Treatment, therefore, focuses on nourishing the kidney, regulating the Chong meridian, and enhancing blood circulation to restore the balance between kidney yin and yang, thus alleviating reproductive dysfunction (Liu, Qi, et al., 2018; Liu, Zhang, et al., 2018). It is generally believed that herbs such as Cistanche deserticola, resveratrol, and Cuscuta used in combination with TwHF can reduce toxicity. Research by Sun et al. (2023) showed that the main chemical components of Cuscuta exhibit E-like activity, can promote the function of the HPG axis, and increase the expression of SMAD 4 mRNA, which is inhibited by TwHF in rats. They also regulate the endocrine system and provide antioxidant and anti-aging effects, offering protection against ovarian dysfunction induced by TwHF in rats. Meanwhile, adding resveratrol to the culture medium increases mitochondrial and ATP content promotes ovarian cell proliferation and the expression of the anti-apoptotic peptide BCL2, and enhances the expression of genes related to oocyte and embryo development in ovarian GCs and oocytes, thereby promoting oocyte maturation and improving embryo production and quality of rats (Sirotkin, 2021). Furthermore, Liu, Qi, et al. (2019), Liu, Tian, et al. (2019), and Liu, Yin, et al. (2019) found that Cistanche can reduce oocyte apoptosis by regulating sex hormone levels in rats, inhibiting IFN-γ and TNF-α, and upregulating the expression of the anti-apoptotic proteins BCL2/BAX. When paired with sweet herbs such as licorice, Rehmannia glutinosa, and Angelica sinensis, it effectively addresses conditions of rigidity and dryness. Clinical prescription trends show that women often use formulas that nourish yin and blood, while men prefer formulas that warm yang and replenish essence to combat reproductive toxicity from TwHF. Kidney-tonifying formulas also play a role in detoxification and enhancing efficacy. For instance, Sun (2013) found that a kidney-tonifying and blood circulation-promoting formula helped in endometrial angiogenesis and growth in rats affected by GTW-induced ovarian reserve function decline. Various formulations, such as Yujing Granules, Bushenyichongkangshuai Decoction, Xin Jia Cong Rong Tu Si Zi Wan, and others, have been shown to effectively mitigate the reproductive toxicity of TwHF (Deng et al., 2022; Hou et al., 2003; Xu et al., 2015; Zhang et al., 2014). Additionally, research by Song et al. (2021) on different processing techniques for TwHF, such as vinegar roasting and frying with Lysimachia, mung beans, white peony, and licorice, revealed reducing apoptosis of testicular cells and correcting their lipid peroxidation/antioxidant ratio in male SPF mice.
Modern Medicine
A clear link between the dosage, duration of use, and the reproductive toxicity of TwHF has been established (Cheng et al., 2021). Zhang (2023) found that combining TwHF with methotrexate enhances its water solubility, reduces the necessary dosage, and lessens side effects. Furthermore, Zhang et al. (2019) discovered that pairing TP with MitoQ, an antioxidant that targets mitochondria specifically, helps rats maintain the testicular microstructure, sperm morphology, and the integrity of the blood–testis barrier. Moreover, a study involving a 4-week intervention with E and P on Sprague–Dawley rats, following 12 weeks of GTW administration via gastric gavage, showed no significant changes in the estrus cycle, reproductive organ quality, tissue morphology, or sex hormone levels. These findings indicate that E and P replacement therapy may effectively prevent and treat the reproductive toxicity associated with TwHF (Wang et al., 2008). Regrettably, the new methods mentioned are still in the animal testing phase and have not progressed to clinical trials.
Modify the Chemical Structure of Tripterygium wilfordii
Altering the chemical composition of TwHF is a promising strategy for reducing its reproductive toxicity. Research has shown that the active ingredients of TwHF can be made more effective and less toxic through CO2 supercritical extraction techniques (Zhang et al., 2018). What’s more, compounds such as PG490-88 and F60008, developed by Liu (2011), have shown notable success in increasing the water solubility of TwHF.
Off-target effects refer to unintended biological effects that occur in areas outside the intended target of a drug (Mohammadi et al., 2020). The off-target effects of TwHF may cause damage to non-target tissues, such as hepatotoxicity, nephrotoxicity, cardiovascular toxicity, and hematologic toxicity (Lin et al., 2020). To mitigate these off-target effects and improve biosafety, researchers have developed various strategies, such as using TP derivatives like triptolide aminoglycoside (TPAG) and liposome-targeted drug delivery systems. Qi et al. (2015) conducted a histopathological examination of rats’ testicular tissue to assess the treatment effects of TPAG. They found that, compared to the TP group, which exhibited severe degeneration, exfoliation, and tubular atrophy of germ cells, both the control and TPAG groups showed normal morphology of testicular interstitial tissue and spermatocytes, with almost no pathological changes. In addition, the liposome-targeted drug delivery system designed by Zhao et al. (2023) showed good biocompatibility and low toxicity in both in vivo and in vitro experiments in tumor-bearing nude mice. However, it is unfortunate that these innovative approaches remain limited to animal experiments and have yet to reach the clinical trial stage.
Change Medication Time and Administration
The reproductive toxicity of TwHF follows a time-dependent pattern, with studies indicating that its toxicity peaks at 10
Oral administration of TwHF preparations has been linked with significant toxic side effects, largely due to their “first pass effect” and limited water solubility (Xu et al., 2019). However, recent advancements in drug delivery systems have shown promising results in addressing these issues. Xue et al. (2011) developed solid lipid nanoparticles, and Chen et al. (2015) reported on TP liposome hydrogel patches. Both technologies have been effective in significantly reducing toxicity and enhancing the drug’s efficacy. Xu et al. (2013) introduced an innovative carrier, TP polymer micelles (TP-PM), in the form of polymer nanoparticles. This development has been shown to increase testicular acid phosphatase (ACP) activity and decrease serum malondialdehyde levels, effectively reducing the reproductive toxicity associated with TwHF. In a similar vein, Gu et al. (2021) developed an exosome-targeted drug delivery system (FA Exo/TPLL), which demonstrated a significant reduction in drug toxicity through in vivo experiments. Additionally, research by Wang et al. (2012) has highlighted the effectiveness of the TwHF transdermal microemulsion delivery system in mitigating the reproductive toxicity induced by TwHF in male rats. Unfortunately, these innovative approaches remain limited to animal experiments and have yet to reach the clinical trial stage.
Grasp Clinical Indications
Currently, TwHF is mainly used in clinical practice for treating various conditions such as rheumatoid arthritis, chronic kidney diseases, autoimmune disorders, skin diseases, and malignant tumors, among others. Its clinical use should be judicious, guided by precise indications, and with particular caution when prescribing to children and individuals concerned with fertility issues. This careful approach ensures the therapeutic benefits of TwHF are harnessed effectively while minimizing potential risks, especially in sensitive populations. Table 1 details out the specific clinical indications for TwHF.
Optimize Clinical Medication Plans
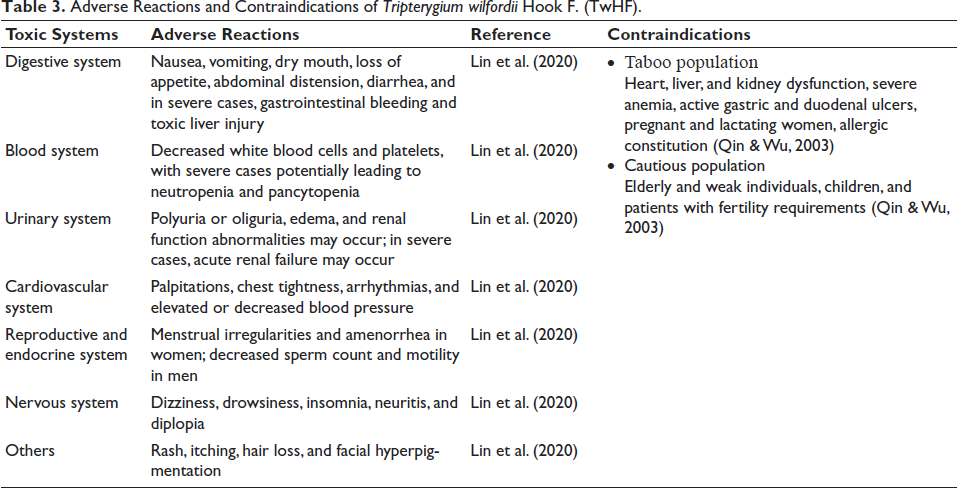
To optimize the clinical use of TwHF, a comprehensive evaluation of factors such as the patient’s medical history, medication efficacy, safety, and tolerance is essential. In addition, TwHF also has significant hepatotoxicity and nephrotoxicity. Therefore, conducting liver and kidney function tests is critical before starting treatment. Common toxic side effects and contraindications associated with TwHF are listed in Table 3. Due to TwHF’s dose-dependent toxicity, it is advisable to begin treatment with a low dose and gradually adjust to an optimal level (Yang & Liu, 2018). The therapeutic and toxic doses of TwHF for different populations are listed in Table 1. Luo et al. (2022) suggest a decoction dosage of 10–15 g and, for external use, a range of 1.85–11.25 g. Research by Ru et al. (2019) and Wang (2018) indicates that a dosage of 1–1.5 mg/kg over 3 months minimizes adverse reactions. Moreover, educating patients about their treatment is crucial to ensure they understand and adhere to medical advice, take medications as prescribed, and maintain regular follow-up appointments (Relevant strategies are shown in Figure 2).
Adverse Reactions and Contraindications of Tripterygium wilfordii Hook F. (TwHF).
Conclusion
Recent research has extensively examined the reproductive toxicity caused by TwHF, with a significant focus on GTW and TP. In females, the primary reproductive issues include menstrual disorders and amenorrhea, with concerns about the reversibility of these effects. Males often experience reduced sperm count and testicular atrophy, although these symptoms may recover 4–10 weeks post-medication cessation. In children, the main concern is the long-term impact on gonadal function. TwHF has been shown to cause germ cell apoptosis, hinder germ cell growth, and affect sex hormone secretion in the body. In clinical practice, efforts to reduce toxicity and increase efficacy involve using appropriate drug combinations, altering the chemical structure, understanding clinical indications, and fine-tuning medication plans. Despite progress in understanding TwHF’s reproductive toxicity, its clinical use is still limited by several factors: (a) a lack of studies on the relationship between the reversibility of TwHF-induced female reproductive toxicity and age; (b) the absence of standardized criteria for producing TwHF products and determining the optimal dosage range; (c) the current drug delivery systems are in the animal testing phase, with their clinical application yet to be confirmed. Future research will aim to clarify the mechanism of TwHF’s reproductive toxicity and develop strategies to minimize toxicity while improving therapeutic efficacy.
Footnotes
Abbreviations
E: Estrogen; FSH: Follicle-stimulating hormone; GCs: Granulosa cells; GTW: Tripterygium glycosides; HPG: Hypothalamic–pituitary–gonadal; LH: Luteinizing hormone; P: Progesterone; ROS: Reactive oxygen species; TP: Triptolide; TwHF: Tripterygium wilfordii Hook F.
Author Contributions
Wang, J J, Jin De, and Zhu Q proposed and designed the research. Wang J J conducted an investigation process as well as wrote the draft of the manuscript. Jin De and Zhu Q revised the manuscript and supervised the process of the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Natural Science Foundation of China for Young Scholars (Grant No. 82205008); Medical Scientific Research Foundation of Zhejiang Province, China (Grant No. 2023RC242); Hangzhou Municipal Health Commission Project (Grant No. A20210083); Zhejiang Traditional Medicine and Technology Program, China (Grant No. 2023ZF137); Key Research Project of the research project of Zhejiang University of Traditional Chinese Medicine Affiliated Hospital (Grant No. 2022FSYYZZ14); Chinese Association of Traditional Chinese Medicine Youth Talent Nurturing Program (Grant No. QNRC2-B21).
