Abstract
Background
The potential of cyanidin 3-O-β-D-glucoside (C3G), known for its anti-obesity and anti-diabetic properties, in suppressing arteriosclerosis progression remains unexplored.
Objectives
This study aimed to elucidate whether C3G prevents or ameliorates arteriosclerosis by investigating the effects of C3G on the transformation, proliferation, and migration of vascular smooth muscle cells (SMCs). Herein, we evaluated the effects of C3G on the migratory ability of SMCs in the presence of adipocytes.
Materials and Methods
SMCs and 3T3-L1 adipocytes were cocultured in a Boyden chamber, with SMCs positioned in the upper chamber and pre-adipocytes in the lower chamber. Pre-adipocytes were initially seeded in the lower chamber and prompted to undergo differentiation over days 0, 2, 4, 6, and 8. SMCs were seeded in the upper chamber. The quantification of cells that migrated through the membrane was carried out. The migratory potential of SMCs within the SMC/adipocyte coculture (SACC) setup was assessed by placing the upper chamber into wells containing adipocytes, with or without 0.1% C3G.
Results
The findings indicated that the accumulation of lipid droplets in adipocytes increased as the differentiation stage progressed. The amount of lipid droplets that accumulated upon the addition of C3G was significantly lower than that in the control. C3G was found to inhibit the active form of plasminogen activator inhibitor-1 (PAI-1) while enhancing the levels of the anti-inflammatory adipocytokine adiponectin. In the SACC, SMC migration increased alongside lipid droplet accumulation in adipocytes. We revealed that C3G decreased the active form of PAI-1 in the SACC and suppressed vascular SMC migration.
Conclusion
C3G inhibited the migration of SMCs in the SACC, highlighting its potential as a compound that can prevent arteriosclerosis and thrombus formation progression by suppressing hyperplasia of the vascular intima.
Introduction
The increase in the number of patients with arteriosclerosis is one of the most critical challenges. Extensive research on obesity and diabetes has yielded several human intervention trials and meta-analyses demonstrating that anthocyanin intake contributes to weight management and reduces the risk of type 2 diabetes (Guo et al., 2016; Stull et al., 2010; Wedick et al., 2012). In obesity, adipocytes enlarge and increase in number, enhancing the response to inflammation and oxidative stress, resulting in the suppression of glucose and lipid metabolism. Adipocytes secrete physiologically active substances known as adipocytokines. The adiponectin levels, which have anti-inflammatory and anti-diabetic effects, decrease as obesity progresses. In contrast, tumor necrosis factor-α and interleukin-6 (IL-6), which induce insulin resistance, increase. Furthermore, anthocyanins increase the expression of adiponectin and decrease the expression of IL-6 and plasminogen activator inhibitor-1 (PAI-1) in adipocytes (Tsuda et al., 2004, 2006). PAI-1 inhibits fibrin degradation by inhibiting plasminogen activators (PAs) and is involved in thrombus formation, thereby contributing to post-atherogenic vascular lesions.
In arteriosclerosis, vascular smooth muscle cells (SMCs) are induced to accumulate abnormally and form vascular neointima. The vascular neointima is composed of SMCs and an extracellularly deposited matrix. Although cell motility consists of several important steps, the mechanism by which cell migration leads to SMC accumulation is not completely understood (Mitchison & Cramer, 1996). In our previous studies, we focused on identifying components that modulate adipocytokine production during SMC migration in the presence of adipocytes in vitro by applying the Boyden chamber method to assess cell migration (Yuui et al., 2020; Zhang et al., 2013). Cyanidin 3-O-β-
To date, the role of C3G in controlling the progression of arteriosclerosis has not yet been clarified. Therefore, we hypothesized that C3G may control the risk of developing arteriosclerosis and lifestyle-related diseases by regulating adipocyte differentiation and adipocytokine secretion. In the present study, we investigated the effect of C3G on SMC migration in the presence of adipocytes. Because the mortality rate of patients with arteriosclerosis-related vascular disease is extremely high, there is a need for novel strategies to limit the negative vascular implications of related disorders. We believe that our study will be useful because C3G has the capacity to mitigate the thickening of the vascular intimal membrane linked to the worsening of atherosclerosis and potentially inhibit the progression of atherosclerosis during the early stages of the disease.
Materials and Methods
Reagents
Reagents utilized in this study included C3G (lot number 10011233) and Oil Red O (lot number 154-02072), both procured from Wako Pure Chemical Industries located in Osaka, Japan. Antibodies targeting extracellular signal-regulated kinase (ERK)1/2 (K-23), phosphorylated ERK 1/2 (p-ERK) (sc-16982), and β-actin were obtained from Santa Cruz Biotechnology in Santa Cruz, CA, USA. The horseradish peroxidase-conjugated anti-mouse IgG antibody was sourced from Southern Biotech in Birmingham, AL, USA. Bovine fibrinogen, which contains trace amounts of plasminogen, was acquired from Organon Teknika in Dublin, Ireland, while bovine thrombin was purchased from Mochida in Tokyo, Japan. The murine PAI-1 activity assay kit was obtained from Molecular Innovation in Novi, MI, USA (lot number MPAIKT-910) and was utilized in accordance with the manufacturer’s guidelines. Additionally, a mouse adiponectin/Acrp30 assay kit was acquired from R&D Systems in Minneapolis, MN, USA (lot number 278969) and was employed following the manufacturer’s instructions. All other reagents and chemicals employed in this study were of the highest commercial quality.
Cell Culture
The 3T3-L1 pre-adipocyte cell line was obtained from the Health Science Research Resources Bank (JCRB9014, Osaka, Japan). In accordance with our previous report (Yuui et al., 2020), adipocytes were properly differentiated. The medium was changed every 2 days from day 0 to 8 to maintain differentiation. The medium containing C3G, which constitutes the black bean seed coat, was replaced every 2 days. Cells were pretreated with C3G (final concentration: 20 µM). Methanol (final concentration: 0.1%) was used as the vehicle, which was subsequently added to the control culture. Previous research has indicated that the concentration of C3G affects 3T3-L1 cells within a range of 10–100 µM (Matsukawa et al., 2015). In the present study, we performed a preliminary cytotoxicity assessment to investigate the effects of low concentrations of components derived from natural products. Our findings revealed that a concentration of 20 µM does not exhibit cytotoxic effects, leading us to classify it as a physiological concentration. All controls utilized in this study were cultured in a medium supplemented with 0.1% methanol. Comprehensive supplementary methodologies were described as additional materials provided alongside the article (Supplemental material 1). SMCs obtained from the thoracic aorta of mice were prepared as previously described (Hou et al., 2004; Zhang et al., 2013). The SMCs utilized in this investigation were derived from cultured cells isolated from smooth muscle tissue obtained from the thoracic aorta of mice, in accordance with established protocols (Hou et al., 2004; Zhang et al., 2013). The established cultured cells were subsequently passaged for use in our previous report (Hou et al., 2007) and cultured at 37°C in a humidified atmosphere containing 5% CO₂.
Oil Red O Staining Analyses
The 3T3-L1 adipocytes were subjected to fixation with a 2.5% glutaraldehyde solution (lot number KWQ0074, Wako, Osaka, Japan) for a duration of 10 min. Following fixation, the cells were stained with Oil Red O for 15 min and subsequently rinsed with distilled water. Intracellular lipids were then extracted using isopropanol. The quantification of extracted lipids was then conducted utilizing a microplate reader (Model 357-00045T, Thermo Fisher Scientific, Waltham, MA, USA) at a wavelength of 492 nm.
Adipocytokine Assay
The adipocytokine assay utilized the methodology described by Yuui et al. (2020) to evaluate the effects of C3G on the production of PAI-1 and adiponectin in adipocytes. The culture fluid on differentiation day 8 was also obtained and used as a specimen for quantifying the levels of active PAI-1 and adiponectin. Specific assay kits were used to measure levels of active PAI-1 and adiponectin in the adipocyte monoculture fluid. The PAI-1 active assay kit uses an active PAI-1 antibody that reacts with coated and dried urokinase. Therefore, this kit did not detect latent or complex PAI-1.
Boyden Chamber Assay for Assessing the Migratory Ability of SMCs
To evaluate migration ability, migrated SMC cells were fixed and stained according to the SMC/adipocyte coculture (SACC) method reported by Yuui et al. (2020). Coculture was performed using the Boyden chamber. Pre-adipocytes were initially seeded at a concentration of 4.5 × 103 cells per well in the lower chamber and prompted to undergo differentiation over a period of days 0, 2, 4, 6, and 8. SMCs were seeded in the upper chamber at a density of 3.0 × 104 cells per well. The quantification of cells that migrated through the insert wall membrane was carried out. The migratory potential of SMCs within the SACC setup was assessed following a 4-h cocultivation period by placing the upper chamber into wells containing adipocytes, with or without 0.1% C3G. On the indicated days, the migrated SMCs were fixed and stained on the migrating side of the upper chamber using the Diff Quick Kit 2 (Sysmex, Hyogo, Japan). Subsequently, the number of cells on the detached membrane was measured under a microscope. The culture fluid was also used as a specimen quantifying the levels of active PAI-1 and adiponectin under coculture. After coculture on differentiation day 8, the culture fluid was obtained and used to measure fibrin degradation using electrophoretic zymography (Ueshima et al., 2004). Comprehensive supplementary methodologies were described as additional materials provided alongside the article (Supplemental material 2).
ERK Signaling in SMCs
The effect of p-ERK signaling on protein expression was also examined according to the method reported by Yuui et al. (2020). ERK signaling in SMCs was studied by seeding SMCs with the culture fluid and lysing them after stimulation. Comprehensive supplementary methodologies were described as additional materials provided alongside the article (Supplemental material 3).
Statistical Analyses
All the experiments were performed in triplicate (n = number of independent experiments). The data are presented as mean ± SEM and were analyzed using the Student’s t-test. Statistical significance was set at p < 0.05.
Results
C3G Extracted from Black Beans Suppressed Adipocyte Differentiation
To explore the influence of C3G on adipocyte differentiation, 3T3-L1 cells were exposed to differentiation conditions for 8 days with or without the extract. Treatment with C3G led to a notable decrease in lipid droplet content in adipocytes differentiated for 6 and 8 days, in comparison to the untreated control cells (Figure 1).
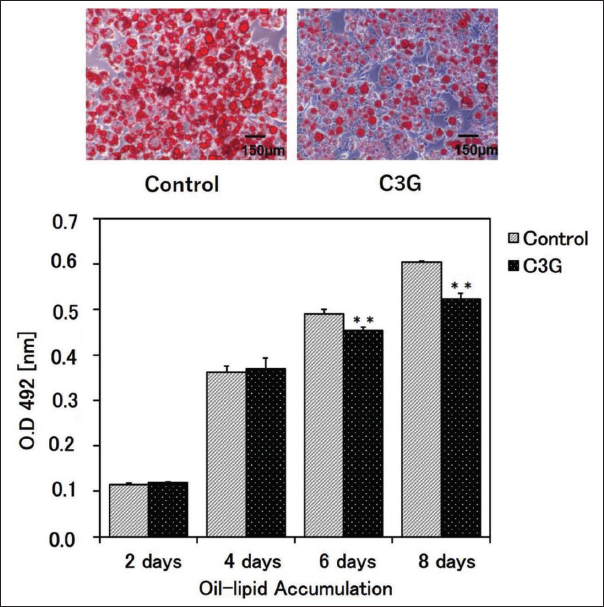
C3G Inhibited Active PAI-1 and Promoted the Secretion of Adiponectin
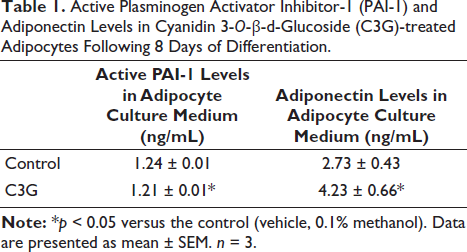
We investigated the levels of adipocytokines generated by adipocytes in a single-culture setting and observed that C3G significantly inhibited the production of active PAI-1 while enhancing the secretion of adiponectin compared to cells that were not treated (Table 1).
Active Plasminogen Activator Inhibitor-1 (PAI-1) and Adiponectin Levels in Cyanidin 3-O-β-d -Glucoside (C3G)-treated Adipocytes Following 8 Days of Differentiation.
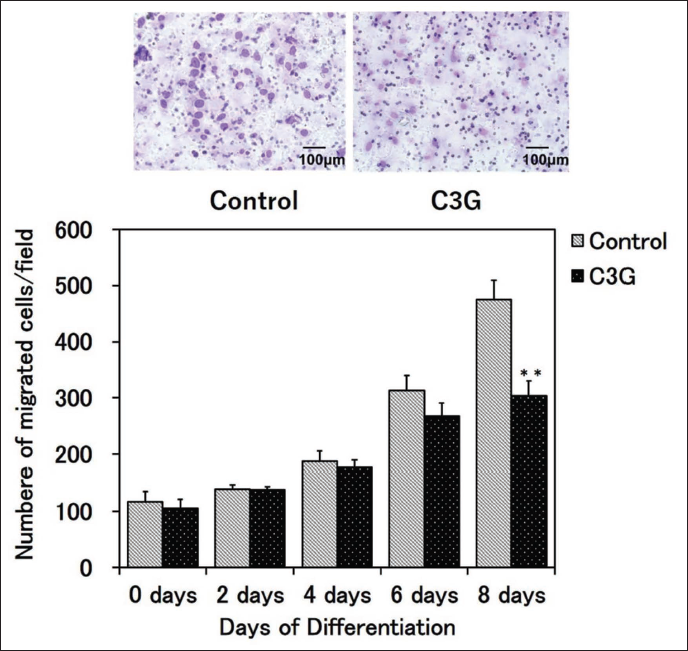
C3G Inhibited SMC Migration in SACC
Analysis of the migratory ability of SMCs in SACC revealed that C3G treatment inhibited SMC migration in a differentiation-dependent manner, with a significant reduction following 8 days of differentiation (Figure 2).
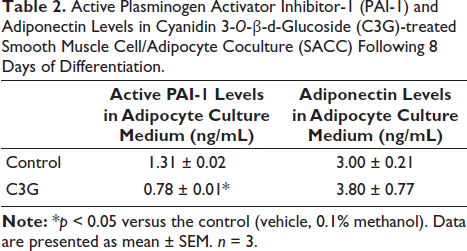
Effect of C3G Treatment on the Levels of Active PAI-1 and Adiponectin in SACC
During the SACC differentiation process over 8 days, treatment with C3G extract significantly decreased active PAI-1 levels and increased adiponectin levels compared to those observed in the control medium (Table 2).
Active Plasminogen Activator Inhibitor-1 (PAI-1) and Adiponectin Levels in Cyanidin 3-O-β-d -Glucoside (C3G)-treated Smooth Muscle Cell/Adipocyte Coculture (SACC) Following 8 Days of Differentiation.
Effect of C3G on ERK Signaling in SMCs
To clarify the molecular mechanism responsible for C3G’s inhibitory impact on SMC migration, an examination was conducted to assess the influence of the extract on ERK phosphorylation. SMCs were stimulated for 30 min using the medium exposed to C3G-pretreated adipocytes during an 8-day differentiation process. The findings revealed a reduction in ERK phosphorylation in SMCs following treatment with C3G compared to controls (Figure 3).
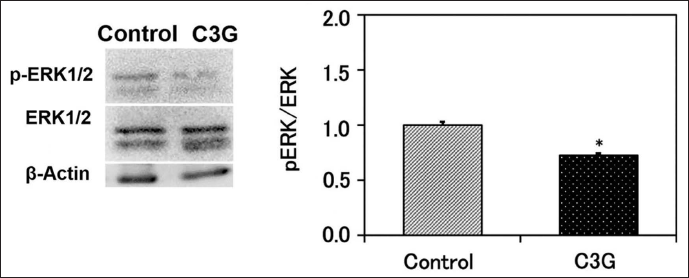
Effect of C3G on Fibrin Degradation
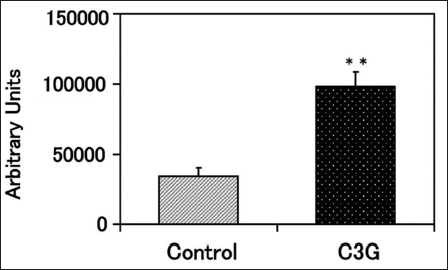
The effect of C3G on fibrin degradation in SACC was investigated after 8 days of differentiation. PA released by SACC showed significantly increased activity levels upon introduction of C3G compared to the activity observed in control cells (Figure 4).
Discussion
This study demonstrated that C3G regulates the levels of adipocytokines produced by adipocytes and suppresses SMC migration, thereby inhibiting the progression of atherosclerosis during the early stages of the disease.
Normal blood vessels have three layers on the lumen side: the intima, tunica media, and adventitia. The intima’s primary structure is comprised of endothelial cells, and the tunica media consists of SMCs, collagen fibers, elastin, and extracellular matrix. Furthermore, an internal elastic plate separates the inner membrane from the tunica media. SMCs in the tunica media regulate vascular contraction and relaxation; however, when vascular homeostasis is disrupted, as in the case of obesity, SMCs migrate to the inner membrane and subsequently proliferate. This causes intimal thickening, leading to atherosclerosis. Thrombosis is the final complication of atherosclerosis, and the breakdown of atherosclerotic plaques leads to an increase in blood-clotting factors that come into contact with tissue factors above the plaque, causing the clots to obstruct vascular flow. Furthermore, atherosclerosis progresses into the initial lesion of endothelial injury, which is followed by the formation of macrophage foam, migration, and proliferation of smooth muscle from the tunica media to the intima, and an increase in the extracellular matrix, resulting in the formation of atheroma. Tunica media SMCs are predominant in the intima (Grootaert & Bennett, 2021; Shi et al., 2023; Wan et al., 2012).
Adipocytes play a vital role in atherosclerosis, which is the basis of lifestyle-related diseases. Accordingly, various adipocytokines, including inflammatory substances secreted by adipocytes, are involved in the development of atherosclerosis. The types and quantities of adipocytokines secreted by adipocytes vary depending on the differentiation and maturation of adipocytes and the quantity of accumulated lipid droplets in adipocytes.
In this study, we demonstrated that C3G reduced the number of lipid droplets accumulated in adipocytes and suppressed the active form of PAI-1 in adipocyte monocultures and SACC. A cytotoxicity assessment was performed on adipocytes utilizing a control comprising 0.1% methanol alongside a concentration of 20 µM C3G. The results indicated no significant reduction in cell viability at either concentration (data not shown). Additionally, given that the findings of this investigation revealed no decrease in cell viability attributable to C3G, the impact of cytotoxicity was determined to be negligible. These findings suggest that the suppressive effect of C3G on the migratory ability of SMCs may not be due to the increased secretion of adiponectin induced by C3G but rather attributed to the suppression of PAI-1 secretion. Similar to our previous reports on cell migration, we hypothesized that PAI-1 exhibits anti-adhesive properties and plays a role in cell migration by demonstrating its ability to compete with integrin-binding vitronectin and the PA receptor (Yuui et al., 2020). This is consistent with previous reports suggesting that PAI-1 is an important factor in the migration mechanism owing to its anti-adhesive properties (Durand et al., 2004).
The amount of lipid droplets that accumulated upon the addition of C3G was significantly lower than that in the control, indicating its potential to delay the progression to mature adipocytes (Lee et al., 2014). Anthocyanins suppress lipid accumulation without cytotoxicity by extensively inhibiting a gene and protein of transcription factor liver X receptor, sterol regulatory element-binding protein, peroxisome proliferator-activated receptor-γ, and CCAAT enhancer-binding protein-α controlling lipogenesis. Anthocyanins activate AMP-activated protein kinase, inhibiting adipocyte differentiation (Han et al., 2018). Furthermore, anthocyanins and C3G suppress PAI-1 and IL-6, which are increased by hypertrophic adipocytes. Anthocyanins and C3G increase the expression of uncoupling protein 2 and acyl-CoA oxidase 1, which are involved in adipogenesis and lipolysis in adipocytes (Tsuda et al., 2004). Furthermore, C3G improves insulin resistance in adipocytes by suppressing the activity of c-Jun NH2-terminal kinase induced by inflammatory cytokines (Guo et al., 2008). Considering the reduced lipid droplet accumulation that accompanied the addition of C3G in the present study, we speculate that C3G may be used to regulate lipogenesis and inhibit adipocyte differentiation. In the present study, pretreatment of adipocytes with C3G in SACC significantly decreased the migratory ability of SMCs in the presence of mature adipocytes compared to controls. C3G is known to decrease lipid droplet accumulation and PAI-1 secretion in adipocytes (Tsuda et al., 2004, 2006). In this study, C3G decreased PAI-1 secretion in the presence of SMCs and adipocytes. These results suggest that adipocyte-derived adipocytokines are essential for SMC migration and that C3G reduces the active form of PAI-1 in SACC and inhibits SMC migration.
There was no difference in the concentration of adiponectin between the SACC and adipocyte monocultures. Previous reports have shown that adiponectin interacts with T-cadherin present in SMCs, resulting in a notable intracellular uptake and possibly facilitating anti-atherosclerotic effects (Balatskaya et al., 2019). Consequently, in the present study, given that adiponectin was internalized by SMCs in SACC, it can be inferred that the concentration of adiponectin in the culture medium increased slightly.
Adiponectin, an anti-atherogenic agent that decreases as obesity progresses, in contrast to PAI-1 (which increases), reportedly suppresses ERK phosphorylation induced by insulin-like growth factor-1 and inhibits the migratory ability of SMCs (Arita et al., 1999; Kubota et al., 2002; Matsuda et al., 2002; Moon et al., 2004; Motobayashi et al., 2009). Our results agree with previously published reports that C3G increased adiponectin levels in SACCs and decreased ERK phosphorylation in SMCs. This suggests that C3G contributes to the decreased migratory ability of SMCs mediated by ERK phosphorylation, a signaling pathway that contributes to cell motility.
Recent studies have demonstrated a correlation between the activity of ERK, mediated by PAI-1, and the metastasis and proliferation of cancer cells (Humphries et al., 2019; Li et al., 2024; Zeitlmayr et al., 2023). Additionally, PAI-1 activity has been implicated in the ERK signaling pathway during the progression of atherosclerosis (Takeda et al., 2001).
Previous investigations into the migration of SMCs in the context of atherosclerosis have frequently utilized ERK phosphorylation as a key marker. Furthermore, it has been established that factors such as platelet-derived growth factor and transforming growth factor-β, along with its type II receptor, play a significant role in the proliferation and migration of SMCs associated with atherosclerosis. Various studies have employed a combination of methodologies to explore the mechanisms underlying SMC migration; however, a comprehensive investigation into these mechanisms has not yet been undertaken (Tamura et al., 1998; Waltenberger et al., 1997). The findings of this study indicate that C3G exerts an inhibitory effect on cell migration related to atherosclerosis through the modulation of adiponectin activation. This process subsequently leads to a reduction in ERK phosphorylation. Additionally, C3G inhibits the activity of PAI-1, which further contributes to the decrease in ERK phosphorylation. The regulation of ERK phosphorylation by these two mechanisms suggests that C3G may significantly mitigate cell migration associated with atherosclerosis.
However, prior research has indicated that the expression of PAI-1 in atherosclerosis progression is modulated by the RAS homolog family, member A (Rho A)/Rho-kinase, and angiotensin II (Takeda et al., 2001). It is important to note that our study did not investigate whether C3G influences PAI-1 expression through these pathways. Future research should focus on assessing these factors to elucidate the underlying mechanisms in greater detail.
Thrombus formation following atherosclerosis occurs when platelets collect at the site of endothelial injury and form a thrombus composed mainly of fibrin. The clot-dissolving fibrinolytic system involves a mechanism in which PA converts plasminogen to plasmin, which in turn degrades fibrin. Plasmin activity is associated with tissue remodeling, and PA on the surface of migrating cells is thought to degrade the extracellular matrix and promote migration (Stefansson et al., 2001). Our results showed that C3G increased fibrin degradation activity in SACC and suppressed secretion of the active form of PAI-1 in SACC, suggesting that C3G inhibits the progression of atherosclerosis as well as thrombus formation following atherosclerosis.
A limitation of the present study is that the relationship between PAI-1 and vitronectin, a matrix that promotes cell adhesion and migration, is unknown. However, vitronectin expression is temporally and spatially regulated during development and is upregulated in various tumors. The αvβ3 and αvβ5 integrin receptors for vitronectin have been shown to function during angiogenesis and tumor invasion (Delannet et al., 1994; Drake et al., 1995; Jones et al., 1996). The role of C3G in SACC suggests that the migratory ability of SMCs is reduced by suppressing the binding between PAI-1 and vitronectin. Although this was an in vitro experiment using cultured cells, in vivo experiments using mice and rats are required to confirm the effects of the food components identified in this study.
Notably, the effect of C3G on the migratory ability of adipocytokines, such as adiponectin and PAI-1, was reported for the first time in this study. We believe that the risk of atherosclerosis and lifestyle-related diseases may be controlled by regulating adipocyte differentiation and adipocytokine secretion. Furthermore, we suggest that C3G has the potential to suppress the thickening of the vascular intimal membrane associated with the exacerbation of atherosclerosis. Thus, as a dietary supplement, consuming purified C3G from a natural source, such as C3G-rich beans, may have health benefits, especially for patients with metabolic syndromes.
Conclusion
The findings indicated that C3G suppressed the migratory ability of SMCs in the presence of adipocytes. We propose that C3G may function as a compound that influences the migration and proliferation of SMCs by modulating the adipocytokines secreted by adipocytes, thereby potentially inhibiting the progression of atherosclerosis.
Footnotes
Abbreviations
C3G: Cyanidin 3-O-β-
Acknowledgments
None.
Authors’ Contribution
Katsuya Yuui performed the laboratory studies, data analysis, and interpretation and drafted the manuscript. Risa Kudo designed the study, interpreted the data, and drafted the manuscript. Shogo Kasuda assisted in performing the laboratory studies and edited the manuscript. Shigeru Ueshima conceptually designed, directed, and interpreted the experiments and wrote the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
