Abstract
Background
Atherosclerosis (AS) is the pathological basis of acute myocardial infarction, acute stroke, and other acute critical illnesses. Vascular smooth muscle cell (VSMC) phenotypic transformation plays a remarkable role in the occurrence and development of AS. Previous studies have found that GSK3β-TCF21 is a key signaling pathway regulating VSMC phenotypic transformation. Baicalin is an important herbal monomer for cardiovascular protection, but whether it can regulate the GSK3β-TCF21 signaling pathway and VSMC phenotypic transformation remains unclear.
Purpose
The study aimed to explore the role of Baicalin in AS and VSMC phenotypic transformation and whether the GSK3β-TCF21 signaling pathway participates in this process.
Materials and Methods
In this study, through the AS mouse model and ox-LDL-induced VSMC phenotypic transformation model in vitro, the effect of Baicalin on the GSK3β-TCF21 signaling pathway and VSMC phenotypic transformation was investigated.
Results
Pathological staining showed that the AS plaque area of the Baicalin group decreased significantly (p < 0.001). The expression of the marker gene (TAGLN, ACTA2, CNN1, and MYH11) of VSMC phenotypic transformation was significantly increased (p < 0.001) after Baicalin treatment. Baicalin could activate GSK3β and increase the expression of TCF21 significantly (p < 0.001) to inhibit VSMC phenotypic transition. Inhibiting the GSK3β-TCF21 signaling pathway was able to reduce the effects of Baicalin on reducing VSMC phenotypic transition and protecting AS.
Conclusion
Our study indicates that Baicalin is protective in inhibiting VSMC phenotypic transformation and ameliorating AS by regulating GSK3β-TCF21.
Introduction
Atherosclerosis (AS) is the main cause of myocardial infarction, stroke, and other acute cardiovascular and cerebrovascular events (Libby, 2021). It is a complex pathological process with many kinds of cells involved (Wolf & Ley, 2019). Recently, genetic lineage–tracing studies have shown that most of the cells present in AS plaques are derived from vascular smooth muscle cells (VSMCs) differentiated from the vascular media (Miano et al., 2021). As the main cells in AS plaques, converting VSMCs from systolic phenotypes to other phenotypes plays an important role in the formation and progression of AS (Zhang et al., 2021). Normally, VSMCs, located in the media of blood vessels, control the vessel’s diameter and tension by constriction, and mature VSMCs hardly proliferate and maintain low synthetic activity (Grootaert & Bennett, 2021). They show a contractile phenotype and stable expression of smooth muscle contraction marker proteins, such as TAGLN, ACTA2, CNN1, MYH11, etc (Basatemur et al., 2019). When vascular injury or local environmental changes occur, the expression of cell contraction markers decreases and the ability of proliferation, migration, and matrix synthesis is enhanced (Aherrahrou et al., 2020). Studies have shown that phenotypic transformation of VSMCs is a key step in the occurrence and development of AS (Liu et al., 2021).
Scutellaria is clinically used alone or in combination with other Chinese medicines to treat respiratory tract infections, acute dysentery, viral hepatitis, allergic diseases, and gynecological diseases (Maleki et al., 2022). Scutellarin is a flavonoid extracted from the dried root of Scutellaria, which is one of the flavonoids with the highest content of Scutellaria, and it is an effective component in the body. It is rapidly converted into Baicalin and other metabolites in the blood. Baicalin is the most important active ingredient. Previous studies have found that Baicalin has antibacterial, antiviral, antioxidant, oxygen-free radical scavenging, antipyretic, analgesic, anti-inflammatory, anti-tumor, and significant cardiovascular and cerebrovascular protective effects (Shah et al., 2022; Xu et al., 2020). However, there have been no relevant studies on whether Baicalin as a monomer can inhibit the phenotypic transformation of VSMCs and their effect on AS progress.
TCF21 is a transcription factor closely related to the occurrence and progression of AS and myocardial infarction; overexpression of TCF21 can significantly inhibit the progression of AS (Deng et al., 2022). Recent studies have shown that TCF21 is a key molecule in the regulation of VSMCs and that overexpression of TCF21 could significantly inhibit the associated markers of secreted VSMCs and the transcription factors critical to the phenotypic transition of VSMCs (Xie & Martin, 2020). A mechanical study found that TCF21 could inhibit the occurrence of the phenotypic transition of VSMCs by blocking the association of MYOCD and SRF (Nagao et al., 2020). Therefore, it has been suggested that TCF21 is a vital protective factor of VSMC phenotypic transition and AS.
Previous studies have shown that the GSK3β signaling pathway can be activated by targeting the PH domain of AKT, and GSK3β activation can promote the expression of TCF21 (Yang et al., 2019). However, whether the GSK3β-TCF21 signaling pathway can inhibit the phenotype conversion of VSMCs and protect the progression of AS has not been reported. Therefore, we aim to explore the molecular mechanism of the regulation of VSMC phenotypic transition and protection of AS by Baicalin, providing new ideas and targets for future prevention and treatment of AS.
Materials and Methods
Establishment of an Animal Model
Male Apoe-/- mice were obtained from Shanghai Model Organisms Center (Shanghai, China). All the mice were fed a high-fat diet (HFD), which contains 15% fat and 0.25% cholesterol, for 8 weeks to establish the AS model. 80 male Apoe-/- mice were randomly assigned to each group, and there were 10 mice in each group. For Baicalin treatment, Baicalin (purity > 98%) (MedChem Express, USA) was dissolved in saline, and the ultimate doses of Baicalin were 50 mg/kg. Baicalin was injected into mice intragastrically once a day.
Cell Culture and Cell Treatment
Human VSMCs (HVSMCs) were provided by ATCC (Manassas, VA, USA) and grown in a DMEM medium (Shanghai BasalMedia Technologies) with 10% FBS (Thermo Fisher Scientific) at 37° with 5% CO2. For ox-LDL (Sigma, USA) exposure, cells were cultured in a medium that contained 100 µg/mL ox-LDL for 24 h. For treatment with Baicalin (Guanghan Bio-Tech, China), VSMCs were treated with 10 µmol Baicalin (MedChem Express) for 24 h.
HE Staining
An HE staining kit (Nanjing Jiancheng Bioengineering Institute) was used for tissue staining and was operated according to the protocol. After HE staining was completed, photographs were taken under light microscope observation, the images were used to calculate the lumen area and plaque area using software such as Image-Pro Plus, and the total area of the plaque was divided by the lumen area to determine the relative size of the plaque. Plaque size was quantified in 5– 10 sections per mouse using ImagePro Plus software by a trained observer under blinded conditions.
Quantitative Real-time Polymerase Chain Reaction
TRIzol was used to extract total RNA from VSMCs and vascular tissues. Total RNA was then transcribed into cDNA strands reversely. Subsequently, PCR amplification was conducted, and the expression levels of phenotypic transformation–related genes were detected by quantitative real-time polymerase chain reaction (qRT-PCR). The expression of messenger RNA (mRNA) in the cells and tissues was calculated using the 2-∇∇Ct method. The primers used in the study included the following:
Western Blotting Assay
Vascular tissues of the mice were added with a lysis buffer for tissue homogenization after being cut into pieces. VSMC protein samples were extracted directly. Total protein concentration was detected by the BCA method. Proteins were subjected to electrophoresis followed by membrane transfer and closure. The primary antibody was then added, and the secondary antibody was added and incubated for 1 h. The primary antibody was then added and incubated overnight. 5% skim milk powder was slowly used to block protein at 37°C for 1 h. Protein expression levels were detected using anti-GS3Kβ (1:1,000, abcam, ab185141), anti-p-GS3Kβ (1:1,000, abcam, ab68476), anti-TCF21 (1:1,000, abcam, ab182134), and β-actin (1:2,000, abcam, ab8226). The luminescent solution (A and B solutions mixed 1:1, Thermo Fisher Scientific, A38555) was dropped onto the PVDF membrane and was performed into blot imaging. Protein levels to be measured were corrected with β-actin. The Western botting (WB) bands were quantified using Image Lab software, and expression levels of the corresponding proteins were calculated.
CCK8 Assay
Take cells in good growth condition, prepare a certain concentration of cell suspension, and add 2 × 10³ cells/100 µl per well into a 96-well cell culture plate. Pre-cultivate the cells at 37℃ for 2–6 h in the incubator before carrying out the experiments. Take 10 µl CCK-8 solution and add it to a 96-well cell culture plate and continue incubation for 0.5–4 h in a 37°C incubator. Determine the absorbance of the cell; the detection wavelength is 430–490 nm, and the reference wavelength is 600–650 nm. The calculation formula is as follows:
Cell viability = [(As − Ab)/(Ac − Ab)] × 100%.
RNA Interference
VSMCs were transfected with siRNA (Origene, China) for TCF21 (si-TCF21). The expression of TCF21 was examined by qRT-PCR after 72 h. The manufacturer’s instructions were used to guide the use of Lipofectamine 2000 (Thermo Fisher Scientific). Solution A: siRNA was diluted with Opti-MEM; Solution B: lipo2000 was diluted with Opti-MEM. Solution B was absorbed and added to solution A, gently mixed, and left for 20 min at room temperature. The transfection reagent was added to the medium in each well, and after 6 h, the culture was replaced with a complete medium and continued until 72 h. After transfection was operated, cells were incubated with ox-LDL and Baicalin for 24 h after being starved with 0.1% FBS for 24 h.
Lentivirus Transfection Injection
TCF21 lentiviral vectors shRNA and control shRNA were obtained from Origene. Lentiviruses were prepared by co-transfection of the lentiviral vector with lenti-vpak packaging plasmid (Origene, China). Concentrated lentivirus (2×108 Tfu) was injected intravenously into mice via the tail vein. Mice were executed 8 weeks after the first injection for analysis.
Statistical Analysis
SPSS 22.0 software was used to perform the statistical analysis. All data were expressed as mean ± SD. Each experiment was conducted at least three times. Two-tailed Student’s t-test was applied to analyze differences in the two groups, and one-way ANOVA was applied to analyze differences in three or more groups.
Results
Baicalin Inhibited the AS Progression and VSMC Phenotypic Transformation
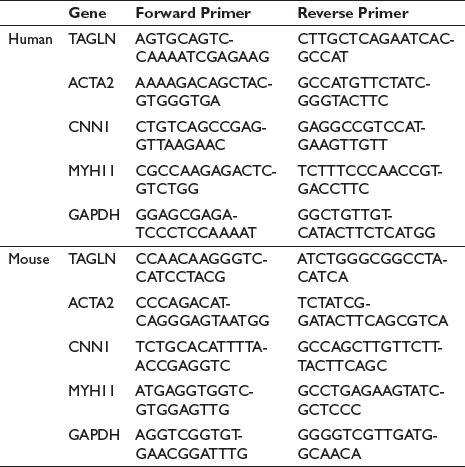
To investigate the roles of Baicalin on the AS progression and the phenotypes of VSMCs, Apoe-/- mice were divided into two groups randomly. One group was given Baicalin (50 mg/kg) intragastrically, and the other group was given normal saline. Mice in both groups were fed an HFD for 8 weeks. HE pathological staining showed that the AS plaque area of the Baicalin group was significantly decreased (Figure 1A,B), and the percentage of plaque size of reduction was about 24.70%. This result suggested that Baicalin played a protective role in AS. Previous studies have found that ox-LDL could lead to VSMC phenotypic transformation. To study the effects of Baicalin on VSMC phenotypic transformation, first, VSMCs were treated with Baicalin in different concentrations and CCK8 assay was followed to detect cell viability (Figure 1D). It was found that 10 µM Baicalin could not affect its viability. So, 10 µM was chosen to conduct the following experiments. After Baicalin treatment, qRT-PCR detection showed that the expression of the marker gene of VSMC phenotypic transformation was significantly increased (Figure 1E). Besides, Baicalin could also significantly up-regulate the expression of the marker gene of VSMC phenotypic transformation in arteries from mice (Figure 1C). These results suggested that Baicalin could inhibit ox-LDL-mediated phenotypic transformation of VSMCs.
Baicalin Inhibited the AS Progression and VSMC Phenotypic Transformation. Representative Images of Quantification of the AS Plaque Size and HE Staining in Aortic Root from AS and AS + Baicalin Mice. Scale Bars, 50 mm (n = 10) (A and B). Quantification of the qRT-PCR Detection of mRNA Expression of TAGLN, ACTA2, CNN1, and MYH11 in Arteries from AS and AS + Baicalin Mice After 8 Weeks of HFD (n = 10) (C). Quantification of CCK8 Assay Detecting Cell Viability in VSMC Incubated with ox-LDL After Baicalin Pretreatment (D). Quantification of the qRT-PCR Detection of mRNA Expression of TAGLN, ACTA2, CNN1, and MYH11 in VSMCs Incubated with ox-LDL After Baicalin Pretreatment (E). **p < 0.01, Two-tailed Student’s t-test.
Baicalin Activated the GSK3β Signaling Pathway and Promoted the Expression of TCF21
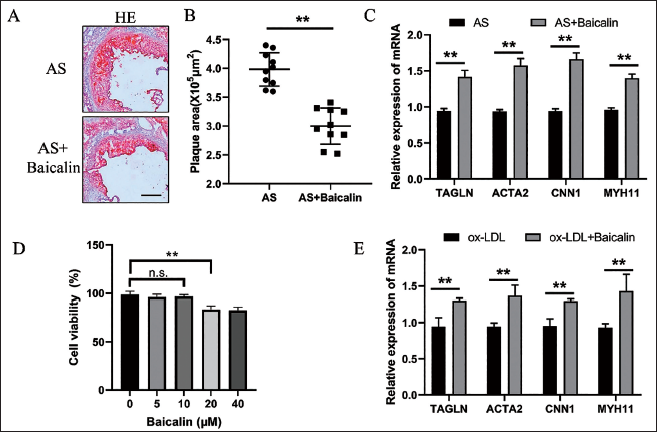
Previous studies have reported that TCF21 is a vital protective molecule in VSMC phenotypic transformation, and overexpression of TCF21 can inhibit the VSMC phenotypic transformation and play a protective role in AS progression (Xie & Martin, 2020). To further explore the mechanism of the effects of Baicalin on inhibiting AS progression and VSMC phenotypic transformation, the expression level of TCF21 was measured by WB and qRT-PCR, and it was found that the expression level of TCF21 was significantly increased in the Baicalin group (Figure 2A,B). A previous study found that GSK3β activation is an important mechanism to regulate TCF21 (Gao et al., 2019). Therefore, the expression of GSK3β and p-GSK3β by WB was detected, and it was found that Baicalin could be able to increase the expression of p-GSK3β markedly (Figure 2A). These results suggest that Baicalin may regulate the phenotypic transformation of VSMCs by activating the GSK3β-TCF21 signaling pathway.
Baicalin Activated the GSK3β Signaling Pathway and Promoted the Expression of TCF21. Representative WB Images and Quantification of p-GSK3β, GSK3β, and TCF21 Expressions in VSMCs After Incubating with ox-LDL After Baicalin Pretreatment (A). Quantification of the qRT-PCR Detection of mRNA Expression of TCF21 in VSMCs Incubated with ox-LDL After Baicalin Pretreatment (B). **p < 0.01, Two-tailed Student’s t-test.
Inhibiting the GSK3β-TCF21 Signaling Pathway Significantly Reduced the Inhibitory Effects of Baicalin on VSMC Phenotypic Transformation
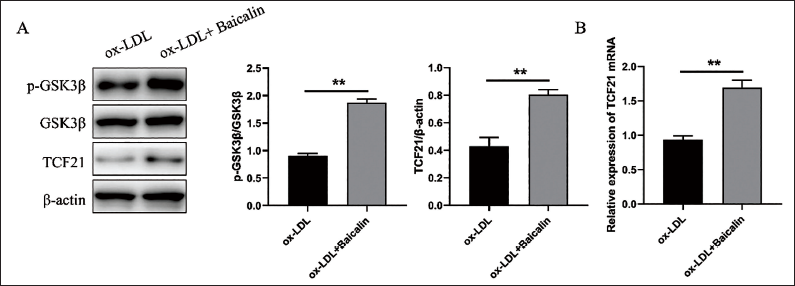
To further investigate the role of GSK3β-TCF21 activation in the regulation of VSMC phenotypic transformation, VSMCs were pretreated with SB216763, a specific inhibitor of GSK3β activation (Chen et al., 2023). It was found that after pretreatment with SB216763, the expression levels of marker gene (TAGLN, ACTA2, CNN1, and MYH11) related to the phenotypic transformation of VSMCs were significantly reduced by qRT-PCR detection (Figure 3A). In addition, si-TCF21 was used to knock down the expression of VSMCs (Figure 3B). After the treatment of VSMCs with Baicalin, the expression levels of marker gene decreased in the si-TCF21 group markedly (Figure 3C). The above results indicated that the inhibition of GSK3β could weaken the inhibition of Baicalin on VSMC phenotypic transformation.
Inhibiting the GSK3β-TCF21 Signaling Pathway Significantly Reduced the Inhibitory Effects of Baicalin on VSMC Phenotypic Transformation. Quantification of the qRT-PCR Detection of mRNA Expression of TAGLN, ACTA2, CNN1, and MYH11 in VSMCs Incubated with ox-LDL After Baicalin Pretreatment With or Without SB216763 (A). Quantification of the qRT-PCR Detection of mRNA Expression of TCF21 in VSMCs After RNA Interference (B). Quantification of the qRT-PCR Detection of mRNA Expression of TAGLN, ACTA2, CNN1, and MYH11 in VSMCs Incubated with ox-LDL After Baicalin Pretreatment with si-NC or si-TCF21 (C). **p < 0.01, One-way ANOVA.
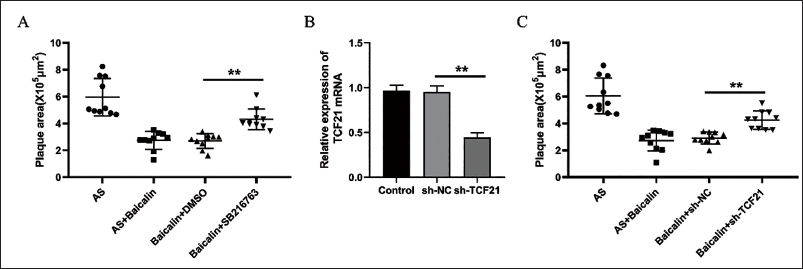
Inhibiting the GSK3β-TCF21 Signaling Pathway Mitigated the Protective Effects of Baicalin on AS Progression
To further clarify the role of GSK3β-TCF21 signaling pathway activation in AS progression, Apoe-/- mice were injected intraperitoneally with SB216763. It was found that the AS plaque area of Apoe-/- mice was significantly increased after treatment with SB216763 (Figure 4A). In addition, sh-TCF21 lentivirus was used to inject Apoe-/- mice through the tail vein. qRT-PCR detection showed that sh-TCF21 could significantly reduce the expression of TCF21 of the blood vessels and lead to a significant increase in the area of AS plaques in mice (Figure 4A, B). The above results indicated that inhibiting the activation of the GSK3β-TCF21 signaling pathway could inhibit the protective effects of Baicalin on AS progression.
Inhibiting the GSK3β-TCF21 Signaling Pathway Mitigated the Protective Effects of Baicalin on AS Progression. Quantification of the AS Plaque Size in Aortic Root from AS, AS + Baicalin, Baicalin + DMSO, and Baicalin + SB216763 Mice After 8 weeks of HFD (A). Quantification of the qRT-PCR Detection of mRNA Expression of TCF21 in Arteries After RNA Lentivirus Transfection Injection (B). Quantification of AS Plaque Size in Aortic Root from AS, AS + Baicalin, Baicalin + sh-NC, and Baicalin + sh-TCF21 Mice After 8 Weeks of HFD (C). **p < 0.01, One-way ANOVA.
Discussion
The morbidity and mortality of acute myocardial infarction and acute stroke are increasing significantly, and AS is an important pathological basis. The pathogenesis of AS involves many mechanisms and has not been fully elucidated (Williams et al., 2020). Most of the previous studies on AS focused on the intima and the outer membrane of blood vessels. In recent years, more and more researchers have emphasized the important role of VSMCs in the occurrence and progression of AS (Cao et al., 2022). VSMC phenotypic transformation is a key change involved in the pathophysiological processes of AS (Sorokin et al., 2020). VSMCs exhibit low proliferative and synthetic capacity physiologically and enable contraction and relaxation of blood vessel walls by expressing a unique set of contractile proteins. The mutual transformation of VSMC phenotypes plays a physiological role in the maintenance of vascular homeostasis and vascular response to inflammation and injury (Aherrahrou et al., 2020). However, in the disease state, this equilibrium state of mutual transformation may be disequilibrated, resulting in the emergence of a pathologic change (Swiatlowska et al., 2022). Therefore, a correct understanding of VSMC phenotypic transformation will provide a new strategy for the future remedies of angiogenesis and atherosclerotic diseases.
Scutellaria is a commonly used Chinese herb for clearing heat and drying dampness in clinics. It mainly contains flavonoids, glycosides, terpenoids, volatile oils, and other components. It has many protective effects for the body, especially for the digestive system, cardiovascular system, and nervous system, among others (Ahmadi et al., 2022). Baicalin is the main and important active component of scutellarin. It has been reported that Baicalin has a remarkable inhibitory effect on AS progression. A previous study found that Baicalin could suppress ox-LDL-induced proliferation and migration of VSMCs in AS by up-regulating miR-126-5p (Chen et al., 2019). Another study found Baicalin could inhibit the development of AS by increasing the expression of Wnt1 and inhibiting the expression of dickkopf-related protein 1 (Wang et al., 2016). Moreover, Baicalin can improve as by inhibiting NLRP3-associated inflammation in Apoe-/- mice (Zhao et al., 2020). However, it has not been reported whether Baicalin can regulate VSMC phenotypic transformation.
Previous studies have screened out key genes closely related to the occurrence and development of AS and coronary heart disease through bioinformatics methods, among which TCF21 has been identified as a key gene in the local immune infiltration and AS progression (Iyer et al., 2018). Through vascular single cell transcriptome analysis, Rober et al. (Wirka et al., 2019) found that the expression of TCF21 was closely related to the regulation of VSMC phenotype, and the specific knockout of TCF21 gene in VSMCs significantly inhibited the regulation of VSMC phenotype in mice. Moreover, knockout of TCF21 could result in vascular injury and significant decrease in the number of protective VSMCs in the fibrous cap, which would lead to the progression of AS (Nagao et al. 2020). Higher TCF21 expression levels were found to be associated with reduced CAD risk through clinical population studies, suggesting that TCF21 is an important regulator of VSMC phenotypic transformation and AS progression (López-Mejías et al., 2017). Further study found that TCF21 could directly interact with MYOCD, blocking the association between MYOCD and SRF and inhibiting the VSMC phenotypic transition (Nagao et al., 2020). Our study confirmed for the first time that Baicalin could up-regulate the expression of TCF21 by activating the GSK3β signaling pathway to inhibit VSMC phenotype transition and play a significant role in inhibiting AS progression. Inhibiting the GSK3β-TCF21 signaling pathway significantly reduced the inhibitory effects of Baicalin on VSMC phenotypic transformation and AS progression. This suggested that the GSK3β-TCF21 signaling pathway played a key role in the protective effects of Baicalin on AS. Our study elaborated on the mechanism of Baicalin inhibiting the progression of AS, providing a theoretical basis for further expanding the role and mechanism of TCM in improving AS. We also provided a research basis for the next step of applying Baicalin-related drugs in the clinic for the prevention and treatment of AS. We suggest that clinical studies of Baicalin in the treatment of AS should be conducted in the future to provide an initial exploration of the clinical application of Baicalin in the prevention and treatment of AS.
Authors’ Contribution
MH and QZY wrote the manuscript; QZY, JJL, and QQX collected the data; MH, JJL, and QQX analyzed the data. All authors approved the manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was supported by the Ethics Commitment of The First Affiliated Hospital of Wenzhou Medical University.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
