Abstract
Background
The herbs Baidoukou (the fruit of Amomum compactum Sol. ex Maton), Baijusheng (the fruit of Lactuca sativa L.), and Biba (Piper longum L.) make up the traditional Mongolian medicine known as Sugemule-3 decoction.
Objectives
Heart failure (HF) is severely impacted; however, the pharmacological mechanism behind it is yet unclear. This investigation looked at the Sugemule-3 ethanol extract’s therapeutic mechanism for isoproterenol-induced HF in rats.
Materials and Methods
To create a HF model, isoproterenol was administered to Wistar rats. Following the model’s successful establishment, various medications were administered for 4 weeks. The electrical signals of cardiac fluctuations, cardiac morphology, and function were observed using electrocardiogram (ECG) and echocardiography. The rats were killed 4 weeks later, and the hearts were observed using an electron microscope and HE staining. Cardiomyocyte apoptosis was observed using terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) staining. The levels of markers in serum were determined using enzyme-linked immunosorbent assay (ELISA). Western blot and immunohistochemistry were used to detect the expression of proteins in rats.
Results
Our findings show that isoproterenol injection subcutaneously interfered with the structure and function of the left ventricle, particularly during systole. In the model group, the level of oxidative stress index malondialdehyde (p < 0.01) increased with the decrease in antioxidant enzymes such as superoxide dismutase (p < 0.01) and glutathione peroxidase (p < 0.01). Sugemule-3 ethanol extract considerably reduced the harmful effects of isoproterenol on the rat myocardium. Additionally, isoproterenol increased the level of peroxisome proliferator-activated receptor (PPAR), carnitine palmitoyl transferase-1 (CPT-1), phosphoenolpyruvate carboxylase (PEPCK), stearyl CoA desaturase-1 (SCD-1), and ubiquitin-conjugating (UCB) expression while decreasing the level of peroxisome proliferator-activated receptor γ coactivator-1 (PGC-1) expression, leading to cardiometabolic diseases. Sugemule-3 ethanol extract improved cardiac metabolism.
Conclusion
The findings demonstrate that Sugemule-3 ethanol extract may improve energy metabolism of cardiomyocytes and alleviate isoproterenol-induced HF through inhibiting the peroxisome proliferator-activated receptor gamma (PPARγ)/PGC-1 signaling pathway.
Keywords
Introduction
Heart failure (HF) is the advanced stage of cardiovascular conditions like coronary heart disease, hypertension, and heart valve disease. The prevalence and readmission rate of HF is increasing rapidly, with high mortality reported. About 50% of patients with advanced HF died within 1 year after diagnosis, with the remaining 50% reportedly dying within 5 years (Antonione et al., 2019). Additionally, the increasing economic and medical burden brought by HF is a major public health challenge in the cardiovascular field. Comprehensive studies of HF have revealed that substrate utilization, metabolic disorders, energy deficiency, and oxidative stress are the basis of systolic dysfunction and disease progression (Bertero & Maack, 2018). Goicoechea discovered that enhancing cardiomyocytes’ energy metabolism can lower HF mortality (Goicoechea et al., 2015). Therefore, there has been a lot of focus on enhancing the oxidative stress and energy metabolism of cardiomyocytes in HF patients. The primary medications for treating HF include angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and sodium corticosteroid receptor antagonists (Abdelnabi et al., 2022). These drugs primarily enhance myocardial contractility and reduce cardiac load, with none reported directly addressing cardiac metabolism. Therefore, it is essential to create medications that can enhance cardiomyocyte energy metabolism to treat HF more successfully, raise patient survival rates, and lower the hospitalization and fatality rates linked to the condition.
Cardiomyocytes are the cells with the highest mitochondrial content, accounting for 1/3 of their volume. For effective contraction, the heart must continuously produce substantial amounts of adenosine triphosphate (ATP) through mitochondrial oxidative phosphorylation, utilizing energy substrates like glucose, fatty acids, amino acids, and ketone bodies (Kolwicz et al., 2013). The metabolism of cardiomyocytes is flexible. When other substrates are abundant, however, the heart takes over the metabolism of those other substrates. Under normal conditions, mitochondria mostly employ fatty acids as substrates to obtain ATP (Young & Latimer, 2023). In HF, mitochondrial function is impaired, ATP production is insufficient, metabolic flexibility decreases, and mitochondrial circulation and oxidation capacity are weakened, resulting in impaired, causing insufficient generation of ATP that leads to a decrease in metabolic flexibility, weakening of mitochondrial circulation and oxidation capacity, which results in impaired energy metabolism, myocardial contractile disturbance (Karwi et al., 2018), and generation of a large number of reactive oxygen species (ROS), simultaneously. Additionally, excessive ROS typically leads to mitochondrial deoxyribonucleic acid (DNA) damage, lipid peroxidation, inhibition of antioxidant activity, and mitochondrial ATP production (Lopaschuk et al., 2021; Tsutsui et al., 2011). Abnormal cardiac energy metabolism significantly contributes to the progression of HF, despite limited understanding of its regulatory mechanisms.
PPAR, or peroxisome proliferator-activated receptor, is significant in managing the metabolism of fats and carbohydrates, including α, δ, and γ subtypes. Despite its low expression in the heart, peroxisome proliferator-activated receptor gamma (PPARγ) is crucial for normal cardiac function (Duan et al., 2005). Research has shown that mice with elevated PPARγ expression develop cardiac insufficiency, and PPARγ transgenic mice develop dilated cardiomyopathy. It was also reported that after treatment with PPARγ agonist, the mice developed cardiac hypertrophy, peripheral edema, and aggravation of HF. PPARγ messenger ribonucleic acid (mRNA) levels are significantly higher in the human heart compared to the mouse heart, by a factor of 8.1–14.5, indicating a potentially greater role of PPARγ in humans, despite the mechanism being unclear (Duan et al., 2005; Son et al., 2007). In addition, mitochondrial energy metabolism is significantly regulated by peroxisome proliferator-activated receptor γ coactivator-1 (PGC-1), which regulates the generation and utilization of myocardial ATP and induces the expression of key downstream genes involved in cardiac mitochondrial biogenesis and metabolic regulation (Di et al., 2018; Ranasinghe et al., 2023). PGC-1 expression was downregulated in HF animal models, and PGC-1-deficient mice had ventricular arrhythmias and worsened myocardial fibrosis (Di et al., 2018; Sano et al., 2004).
One of the most well-known ethnic medical systems in China is Mongolian medicine, which has a well-developed theoretical framework, distinctive diagnostic and therapeutic approaches, and high levels of clinical efficacy. Sugemule-3 decoction (SD-3) was first referenced as a traditional Mongolian remedy in The Four Medical Scriptures. SD-3 includes Baidoukou, derived from the fruit of Amomum compactum Sol. Maton, Baiju, and Biba (Piper longum L.) are mixed in a ratio of 3:2:1. The inhibitory effects of SD-3 on Heyi, Heyi with the role of mitochondria in HF. Mitochondria are important sites for energy metabolism. Clinical studies indicate that the Mongolian medicine SD-3 decoction is effective in treating acute myocardial infarction and provides significant myocardial protection (Bai, 2017). The mechanism by which SD-3 exerts its protective effect on isoproterenol (ISO)-induced HF, specifically through enhancing cardiomyocyte energy metabolism, remains uncertain. This research explored the potential of SD-3 ethanol extract to enhance cardiomyocyte energy metabolism and mitigate oxidative stress via the PPARγ/PGC-1 pathway. This research establishes a theoretical foundation for employing SD-3 in HF treatment.
Materials and Methods
Materials
ISO (1351005) and metoprolol (MP, 1441301) were obtained from Sigma–Aldrich, USA. Pentobarbital sodium was obtained from Merck, Germany. Terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay kit, bicinchoninic acid (BCA) protein quantitative kit, and Tissue Mitochondria Isolation kit was purchased from Beyotime Biotechnology in Shanghai, China. The mitochondrial respiratory chain enzyme complex activity assay kit was sourced from Abcam. The enzyme-linked immunosorbent assay (ELISA) kit was obtained from the Sizhengbai company (Beijing, China). Anti-bodies to PGC-1 [cell signaling technology (CST), 2178S], PPARγ (Abcam, ab310323), carnitine palmitoyl transferase-1 (CPT-1) (Abcam, ab234111), phosphoenolpyruvate carboxylase (PEPCK) (Abcam, ab115693), stearyl CoA desaturase-1 (SCD-1) (Abcam, ab236868), ubiquitin conjugating (UBC) (Covalab, pab74564) were used to detect the expression of protein. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and horseradish peroxidase-labeled secondary anti-bodies were sourced from CST, USA, while enhanced chemiluminescence (ECL) was acquired from Merck Millipore, USA.
Ethanol Extracts Preparation
SD-3 powder weighed 100 g and was soaked in 1 L of 70% ethanol solution (diluted with water). Over a period of 3 days, the samples were stirred repeatedly at room temperature, and the extracts were filtered twice through Whatman filter paper with an 8 µm pore size. The extract was dried using a rotary evaporator and stored for future experiments.
Animals
Specific pathogen-free (SPF) Wistar rats (male, 200 ± 20 g) were purchased from the Faculty of Laboratory Animal Sciences, Harbin Medical University (SCXK2022-004). In this study, experiments were approved by the Institutional Animal Care and Use Committee of Heilongjiang University of Chinese Medicine and complied with National Institutes of Health (NIH) laboratory animal care and use guidelines (ISBN: 0-309-4).
The random number table method was used to group the experimental animals according to the needs of the study. Wistar rats were randomly separated into four groups: control, ISO, SD-3 treatment, and MP treatment, with each group comprising 10 rats. Rats in all groups, except the blank control, received subcutaneous injections of ISO at 10 mg/kg/day for 7 days to induce HF rats. Echocardiography confirmed the cardiac function in HF model rats. Following successful modeling, the SD-3 treatment group was administered 0.4 g/kg of SD-3 ethanol extract intragastrically once daily. The MP treatment group was administered a daily dose of 10 mg/kg. The blank control and model groups received intragastric solvent administration daily. After 28 days, echocardiography was conducted to assess the cardiac function of rats in each group. The heart, kidney, and spleen were excised, rinsed with saline, weighed, and all measurements were recorded. The heart of rats was stained by HE staining.
Echocardiography
Four weeks after intragastric administration, mice underwent echocardiography using an Acuson Sequoia C256 (Siemens) ultrasound machine, following intraperitoneal anesthesia with 2% pentobarbital sodium (2.25 mL/kg).
TUNEL Assay
Sections embedded in paraffin were dewaxed using xylene and then rehydrated with a series of alcohol concentrations. Apoptosis in cardiomyocytes was identified following the TUNEL staining kit instructions (G002-2-2, Nanjing Jiancheng Bioengineering Institute), and the observation was conducted using a microscope. In the experiment, horseradish peroxidase reacts with DAB and hydrogen peroxide to form brown phenylethylhydrazine polymers. Cells that turn brown are marked as positive, and apoptosis is assessed by calculating the percentage of TUNEL-positive cells in various sections.
Determination of Mitochondrial Respiratory Chain Complex Activity
The reagent required for the experiment was prepared according to kit instructions. Kits for ATP synthase (A016-1-1) and mitochondrial respiratory chain complexes I (A089-1-1), II (A089-2-1), and III (A089-3-1) were employed. Reagents and samples were added to the designated wells, thoroughly mixed, and promptly placed in the enzyme plate detector for analysis. Absorbance values were measured at specific wavelengths: 340 nm for MitoCheck complexes I and V Activity, 600 nm for complex II, and 550 nm for complex III. Vitality was assessed according to the provided guidelines.
Measurement of Malondialdehyde (MDA), Superoxide Dismutase (SOD), and Glutathione Peroxidase (GSH-Px) Content
The heart tissue was washed with physiological saline to remove residual blood. The tissue homogenate was obtained by the Automatic Sample Rapid Grinder (JXFSTPRP, Shanghai). Samples were processed according to the ELISA kit instructions (MDA, SOD, GSH-Px kits from Nanjing Jiancheng Technology Co.), involving the application to the coated orifice plate and incubation with an enzyme.
Western Blotting
The rat heart tissue was lysed in radioimmunoprecipitation assay (RIPA) lysis buffer containing phenylmethylsulfonyl fluoride (PMSF) and then centrifuged to obtain the supernatant. Use BCA protein detection kit to detect the protein levels in the hearts of rats in each group. The protein (10 µg) was separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and then the protein was transferred to polyvinylidene fluoride (PVDF) membrane. Seal the membrane with a blocking solution under shaking conditions at room temperature for 2 h. The membrane was incubated overnight with the first anti-body at 4°C and then treated with the second anti-body on a shaker at room temperature for 90 min. Visualize the protein using ECL luminescent chromogenic reagent and analyze it using ImageJ 1.8.0 software.
Immunohistochemistry
After dewaxing the paraffin-embedded sections of each group’s heart, they were dehydrated with alcohol, soaked in citrate buffer, and treated with 0.3% hydrogen peroxide in the dark for 10 min. The slices were first incubated with goat serum for 30 min and then incubated with primary and secondary anti-bodies, respectively. DAB staining was performed for 2–10 min, and the color of the slices changed to brown or brownish under a microscope before stopping the experiment. Stain again with hematoxylin, differentiate with 1% ethanol for 30 s, treat with 1% ammonia solution for 15 s, dehydrate with ethanol gradient, clarify with xylene, and seal with neutral gel.
Statistical Analysis
The results in this experiment are shown as mean ± standard deviation (SD). Single factor analysis of variance (ANOVA) was performed using GraphPad Prism (Version 9.0), followed by Fisher’s least significant difference post hoc test for multi-group comparison. Conduct the Student’s t-test to compare two groups. Use a two-tailed test to determine the p value. Use Spearman correlation coefficient to evaluate the strength of the correlation, with statistical significance determined by a p value less than 0.05.
Results
Effect of SD-3 on the Functions of Left Ventricular of Rats with ISO-induced HF
Following intragastric administration for 4 weeks, echocardiography was performed. The study investigated the impact of ISO, SD-3, and MP on echocardiographic structural parameters in rats (Figure 1). The ISO group showed significant increases in left ventricular anterior wall thickness at systolic (LVAWs), left ventricular end-diastolic diameter at systolic (LVDs), and interventricular septum thickness (IVST) compared to the control group. In the SD-3 group, LVAWs, LVDs, and IVST showed significant reductions compared to the ISO group, while in the MP group, significant decreases were observed in LVAWs, LVDs, left ventricular posterior wall thickness at systolic (LVPWs), and IVST. Although the echocardiographic parameters left ventricular anterior wall thickness at diastolic (LVAWd), left ventricular end-diastolic diameter at diastolic (LVDd), and left ventricular posterior wall thickness at diastolic (LVPWd) were not significantly different from control, they exhibited an upward trend in HF rats.
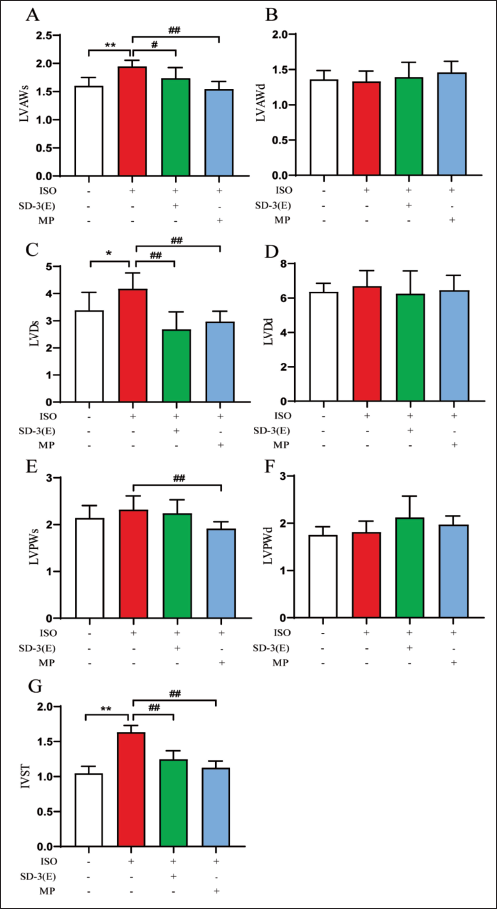
The effects of ISO, SD-3, and MP on echocardiographic functional parameters were examined in rats (Figure 2). Compared with control, ejection fraction (EF) and fraction shortening (FS) in ISO group decreased significantly, while those of SD-3 and MP groups increased significantly (p < 0.01).
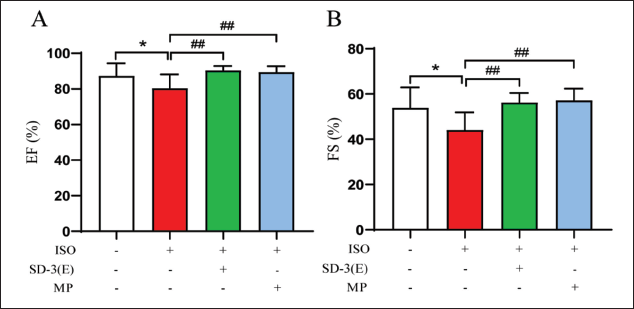
The study indicates that ISO significantly damages cardiac structure and function in HF rats, while SD-3 effectively inhibits these structural changes and mitigates ISO-induced cardiac function impairment.
Effect of SD-3 on Body Weight and Organ Weight of Rats with ISO-induced HF
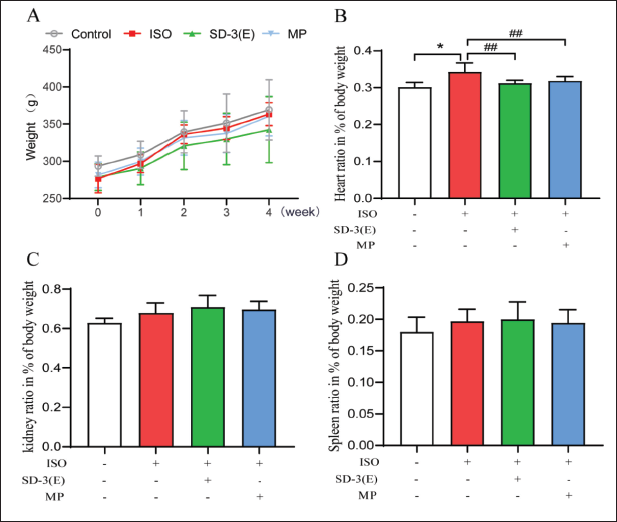
Rats’ weight changes were recorded weekly at the same time during intragastric administration (Figure 3A). The findings reveal that the percentage of heart weight increased compared to the control and that, when compared with ISO group, the percentage of heart weight decreased significantly in SD-3 and MP groups (p < 0.01) (Figure 3B). The percentage weights of the kidney and spleen showed no significant difference (Figure 3C–D). These results demonstrate that ISO treatment did not change the weight of rats and other organs but only increased the weight of the heart. The results also reveal the protective effect of SD-3 ethanol extract on HF rat hearts, as indicated by the reduced heart weight.
Effect of SD-3 on the Tissue Structure of Rats with ISO-induced HF
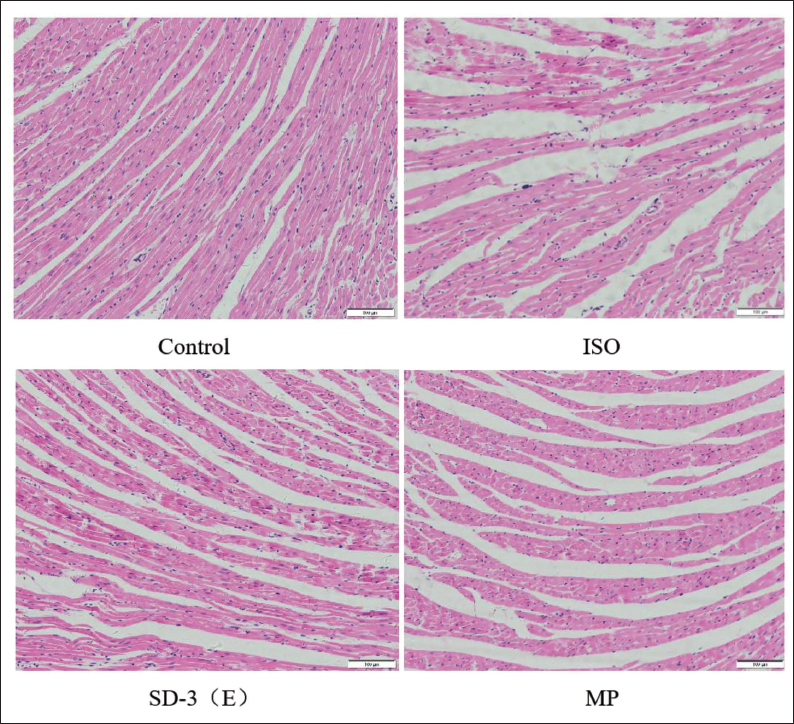
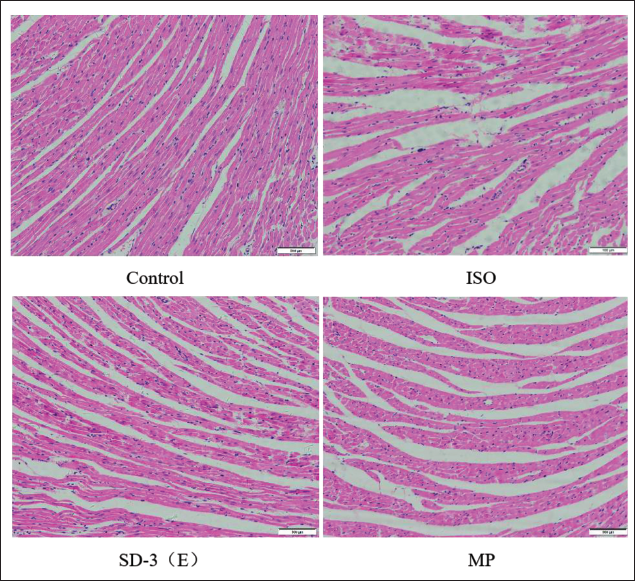
Histopathological changes in rat myocardium were observed using HE staining. Compared with control, the ISO group exhibited local myocardial necrosis, interstitial edema, irregular arrangement of muscle fibers with fiber breakage, inflammatory cell infiltration, and other pathological changes. Treatment with SD-3 and MP improved the injury in the myocardial structure of rats subjected to ISO (Figure 4). The findings indicate that SD-3 ethanol extract might attenuate the effects of ISO on rat cardiac tissue structure and myocardial histological alterations.
Effect of Sugemule-3 Decoction (SD-3) on the Myocardial Tissue Structure of Rats with Isoproterenol (ISO)-induced Heart Failure (HF) by HE Staining. Scale Bar 100 µm.
Effect of SD-3 on Cardiomyocyte Apoptosis of Rats with ISO-induced HF
TUNEL assay was used to study the protective role of SD-3 on cardiomyocyte apoptosis in rats with HF. Compared with the control group, apoptosis of cardiomyocytes in rats with HF was significantly increased, while SD-3 and MP treatment significantly reduced the number of apoptotic cardiomyocytes compared with the HF group, indicating that SD-3 ethanol extract has an improvement effect on cardiomyocyte apoptosis in rats with HF (Figure 5).
Effect of Sugemule-3 Decoction (SD-3) on Cardiomyocyte Apoptosis of Rats with Isoproterenol (ISO)-induced Heart Failure (HF) by Terminal Deoxynucleotidyl Transferase dUTP Nick-end Labeling (TUNEL) Assay. Scale Bar 100 µm.
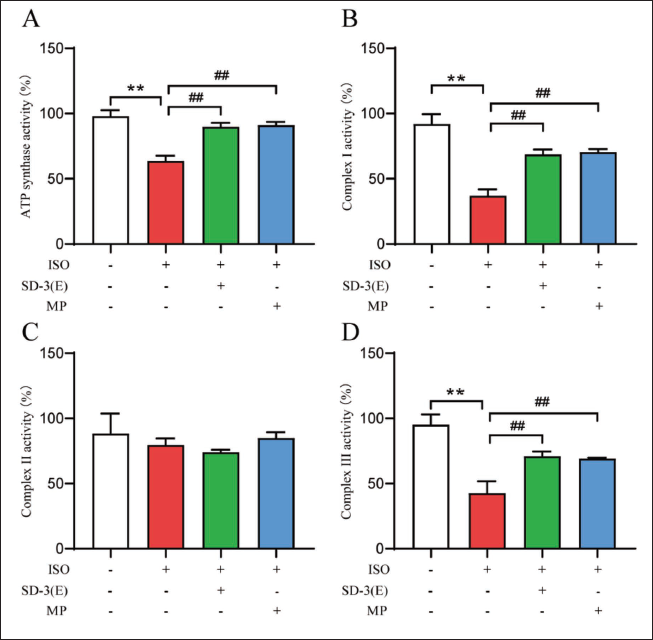
Effect of SD-3 on Mitochondrial Function of Rat with ISO-induced HF
The study examined SD-3’s effects on rat mitochondrial function by isolating mitochondria from tissues and evaluating ATP synthase activity along with mitochondrial respiratory chain complexes I, II, and III. The ISO group exhibited a notable decrease in ATP synthase activity and mitochondrial respiratory chain complexes I and III relative to the control. The study found that SD-3 and MP treatments significantly increased the activities of mitochondrial respiratory chain complexes I and III, along with ATP synthase, as shown in Figure 6. This indicates that SD-3 ethanol extract enhances mitochondrial function in rats with HF.
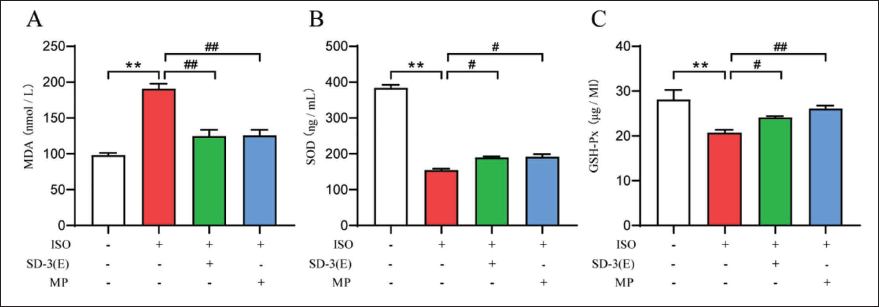
Effect of SD-3 on Oxidative Stress in Rats with ISO-induced HF
The study investigated the relationship between SD-3 ethanol extract’s impact on rat cardiac structure and function and oxidative stress by measuring MDA levels and the antioxidant enzymes GSH-Px and SOD with an ELISA kit. The ISO group showed a significant enhancement in MDA content and a marked decrease in the antioxidant enzymes SOD and GSH-Px relative to the control group. After administering SD-3 and MP, MDA levels significantly decreased, while antioxidant enzymes SOD and GSH-Px levels significantly increased compared to HF (Figure 7). The study indicates that SD-3 ethanol may affect cardiac structure and function via oxidative stress in HF rats.
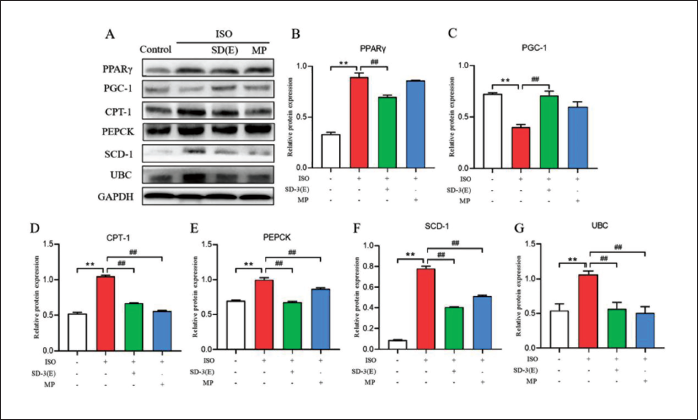
Effect of SD-3 on the Expression of PPARγ/PGC-1 Pathway-related Proteins of ISO-induced HF in Rats
To assess if SD-3 ethanol extract modulates cardiac metabolism via the PPARγ/PGC-1 pathway to mitigate ISO-induced HF, protein expression associated with this pathway was examined using Western blot and immunohistochemistry. Western blot analysis revealed that the ISO group showed significantly increased expressions of PPARγ, CPT-1, PEPCK, SCD-1, and UBC compared to the control, while PGC-1 expression was significantly decreased. Following SD-3 treatment, the expressions of PPARγ, CPT-1, PEPCK, SCD-1, and UCB were notably reduced, whereas the expression of PGC-1 was significantly elevated compared to HF. In addition, the expressions of PPARγ, CPT-1, PEPCK, SCD-1, and UCB decreased following treatment with MP, while the expressions of PGC-1 increased significantly compared with those of ISO group (Figure 8). Immunohistochemical analysis revealed that the protein was mainly expressed in cardiomyocytes, as indicated by the brown DAB stain. In the ISO group, PPARγ, CPT-1, PEPCK, SCD-1, and UCB expression levels increased, while PGC-1 expression decreased, relative to the control and drug-treatment groups (Figure 9).
Effect of Sugemule-3 Decoction (SD-3) on the Expression of Proteins Related to Peroxisome Proliferator-activated Receptor Gamma (PPARγ)/Peroxisome Proliferator-activated Receptor γ Coactivator-1 (PGC-1) Pathway in Rats with Isoproterenol (ISO)-induced Heart Failure (HF) by Immunohistochemistry. Scale Bar 100 µm.
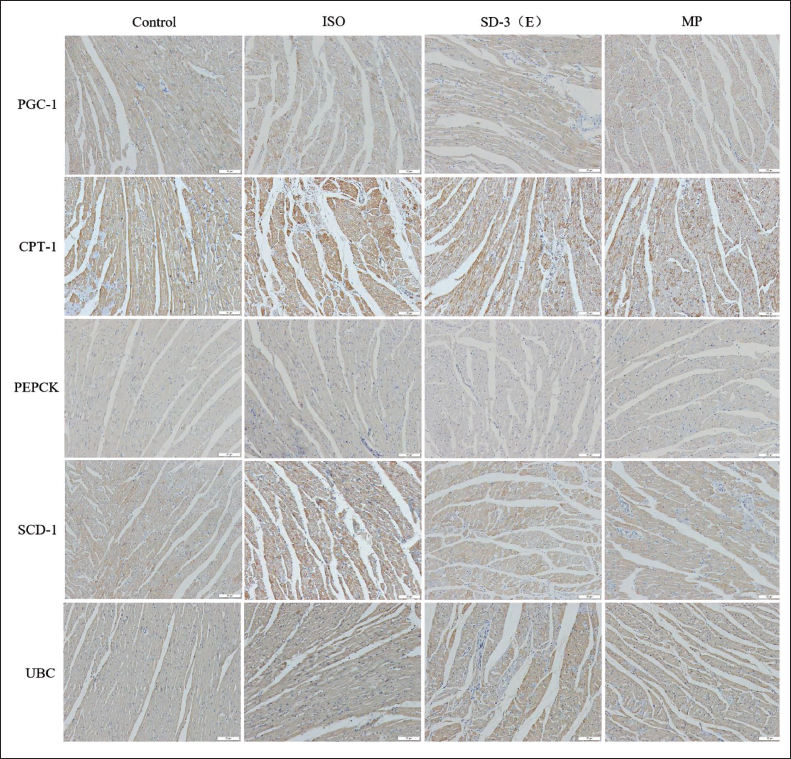
The findings from Western blot and immunohistochemistry supported our hypothesis that SD-3 ethanol extract moderates ISO-induced HF by regulating cardiac metabolism through PPARγ/PGC-1 pathway.
Discussion
HF is a complex syndrome characterized by diminished cardiac output and the heart’s failure to adequately deliver essential nutrients, including blood and oxygen to tissues and organs. Cardiovascular problems can cause feelings like weariness and dyspnea, in addition to physical manifestations like tachycardia and edema (Tanai & Frantz, 2016). HF exhibits high morbidity and mortality rates, with an increasing prevalence among younger populations. A study found that cardiac energy metabolism is inactivated during HF, and ATP production is reduced, further aggravating HF (Lopaschuk et al., 2021). Therefore, improving the energy metabolism of cardiomyocytes in patients with HF may be the key to discovering the treatment strategy for HF. A non-selective β-adrenergic agonist, ISO can raise heart rate, myocardial contractility, and oxygen consumption while also causing myocardial damage by releasing free radicals. The ISO-induced HF model is extensively utilized in experimental research (Krenek et al., 2009; Lopaschuk et al., 2021; Yang et al., 2020). This study utilized ISO to develop the HF model, investigating the impact of SD-3 ethanol extract on cardiac structure and function, alongside its effects on the PPARγ/PGC-1 pathway. The study demonstrated that ISO notably decreased EF% and FS% in HF rats while significantly increasing LVAWs, LVDs, and IVST. HE and TUNEL labeling revealed that ISO-induced cardiac tissue exhibited local necrosis, interstitial edema, disrupted muscle fiber organization with breakage, inflammatory cell infiltration, and other pathological changes, along with an increased number of apoptotic cells. The study successfully established the ISO-induced rat HF model.
Mongolian medicine SD-3 is a traditional prescription of Mongolian medicine, which has the effect of inhibiting heart damage (Wang et al., 2017; Zhen et al., 2022). The study found that EF% and FS% increased significantly, while LVAWs, LVDs, and IVST decreased significantly in rats treated with SD-3 ethanol extract. HE and TUNEL staining indicated that the SD-3 ethanol extract ameliorates ISO-induced HF, potentially through activating the PPARγ/PGC-1 pathway and enhancing cardiomyocyte energy metabolism.
Oxidative phosphorylation of mitochondria is the center of cell metabolism. The heart obtains ATP by oxidative phosphorylation of mitochondria to maintain normal contraction (Kolwicz et al., 2013). Mitochondrial oxidative phosphorylation requires the involvement of respiratory chain complexes. ATP generation is insufficient when mitochondrial activity is compromised, which results in HF. The study found decreased activities of mitochondrial ATP synthase and respiratory chain complexes I and III in the cardiac tissues of ISO rats. This study shows that the SD-3 ethanol extract alleviated ISO-induced inhibition of ATP synthase and mitochondrial respiratory chain complexes I and III activities.
The oxidative stress associated with ISO-induced HF is caused by an imbalance between the production of ROS and the antioxidant defense system, leading to an increase in lipid peroxidation (Kumari et al., 2020; Li et al., 2021; Wu et al., 2021). MDA, a mutagenic byproduct of lipid peroxidation, serves as a marker for oxidative stress and damage (Ayala et al., 2014). The study indicated elevated MDA levels in the ISO group, alongside reduced SOD and GSH-Px, key antioxidant enzymes that mitigate ROS. Treatment with SD-3 ethanol extract countered the ISO-induced rise in MDA and alleviated the suppression of SOD and GSH-Px in cardiac tissue.
PPARs are a subfamily of nuclear receptor superfamily composed of α, δ, and γ subtypes. PPARs are crucial for mitochondrial production, lipid metabolism, and antioxidant defense. Studies have disclosed that PPARγ is closely linked to cardiovascular diseases, and PPARγ protein expression is significantly upregulated under certain pathophysiological conditions, such as the increased expression of PPARγ in macrophages with atherosclerotic lesions (Kim & Yang, 2013). In arrhythmogenic right ventricular cardiomyopathy, the PPARγ pathway is activated, leading to increased expression at both mRNA and protein levels (Djouadi et al., 2009). Moreover, PGC-1 plays a crucial part in cardiac protection, and its expression is decreased in HF animal models, which results in impaired cardiac function (Di et al., 2018). PGC-1 also induces the expression of downstream genes that regulate cardiac metabolism. Therefore, to further investigate whether SD-3 ethanol extract can improve the energy metabolism of cardiomyocytes and attenuate ISO-induced HF through the PPARγ/PGC-1 signaling pathway, we determined the expression level of PPARγ, PGC-1, CPT1, PEPCK, SCD-1, and UCB. Research indicates that UBC upregulation plays an anti-stress role under stress conditions (Han et al., 2019). Suppression of CPT-1 and SCD-1 expression can alleviate heart disease, improve HF symptoms, and provide cardiac protection (George et al., 2016; Miklankova et al., 2021). Additionally, PEPCK upregulation accelerates cardiac hypertrophy (Ma et al., 2019). Western blot and immunohistochemistry results show that compared to the control group, the ISO group had significantly increased protein expression of PPARγ, CPT-1, PEPCK, SCD-1, and UBC, while PGC-1 protein expression was significantly reduced. Treatment with SD-3 ethanol extract resulted in significantly increased expression of PPARγ, CPT-1, PEPCK, SCD-1, and UCB and significantly decreased expression of PGC-1.
Conclusion
In summary, SD-3 ethanol extract may improve cardiomyocyte energy metabolism and alleviate ISO-induced HF through the PPARγ/PGC-1 signaling pathway. This research offers a novel theoretical basis for the application of SD-3 ethanol extract in HF treatment.
Footnotes
Abbreviations
CPT-1: Carnitine palmitoyl transferase-1; EF: Ejection fraction; FS: Fraction shortening; GSH-Px: Glutathione peroxidase; HF: Heart failure; ISO: Isoproterenol; IVST: Interventricular septum thickness; LVAWd: Left ventricular anterior wall thickness at diastolic; LVAWs: Left ventricular anterior wall thickness at systolic; LVDd: Left ventricular end-diastolic diameter at diastolic; LVDs: Left ventricular end-diastolic diameter at systolic; LVPWd: Left ventricular posterior wall thickness at diastolic; LVPWs: Left ventricular posterior wall thickness at systolic; MDA: Malondialdehyde; MP: Metoprolol; PEPCK: Phosphoenolpyruvate carboxylase; PGC-1: Peroxisome proliferator-activated receptor γ coactivator-1; PPAR: Peroxisome proliferator-activated receptor; ROS: Reactive oxygen species; SCD-1: Stearyl CoA desaturase-1; SD-3: Sugemule-3 decoction; SOD: Superoxide dismutase; UBC: Ubiquitin-conjugating.
Authors Contribution
XDP has carried out research, gathered data, and analyzed it. HYW has carried out research, performed experiments gathered data, and analyzed it. QL performed experiments, conducted formal analyses, edited the manuscript, and provided data Availability. FLZ designed, coordinated, and coordinated the entire research project and edited the manuscript. All authors contributed to the article, and the final version was approved by all authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animal experiments were approved by the Animal Ethics and Welfare Committee of Heilongjiang University of Chinese Medicine. The study was conducted in accordance with the local legislation and institutional requirements.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Foundation for Heilongjiang Province Traditional Chinese Medicine Research Project (ZHY2023-218).
