Abstract
Background
Disease prevention prevailed in the post-COVID world. The global trends are trailing toward prevention and immunity enhancement with alternative therapies and herbal drugs.
Objectives
The current research was performed to prepare and standardize a herbal formulation reported to have multiple health benefits including immunomodulation.
Materials and Methods
Ethanolic extracts of Asparagus racemosus, Tinospora cordifolia, and Trigonella foenum-graecum were prepared and used in the proportion of 1:1:1. The extracts were analyzed for phytochemicals and further standardized by high-performance thin layer chromatography (HPTLC) with analytical standards for all three drugs, that is, shatavarin IV, trigonelline, and berberine. The A. racemosus, T. cordifolia, and T. foenum-graecum extract blend (ATTEB) was tested for neutrophil adhesion and carbon clearance to check for overall immunomodulatory activity. The effervescent tablets were formulated and evaluated for various pharmacopoeial parameters. Accelerated stability studies of the formulations were done for 6 months duration as per the International Council for Harmonisation of Technical Requirements of Pharmaceuticals for Human Use (ICH) guidelines.
Results
The phytochemical and HPTLC results confirmed the presence of bioactive constituents in extracts and formulation. The blend of extract ATTEB (neutrophil adhesion: 52.17 ± 0.79, phagocytic index: 0.0292 ± 0.002) enhanced neutrophil adhesion by approximately 35% from the control group (16.91 ± 1.94), with almost five-fold higher phagocytic index compared to the control group (0.0056 ± 0.004) and even higher compared to the standard drug group (0.022 ± 0.001). A ready-to-use formulation of effervescent tablets was prepared and evaluated. The B7 batch showed a suitable formulation based on the various evaluation parameters. The formulation was found to be stable under 6-month accelerated stability studies.
Conclusion
The research study successfully developed a novel formulation of the blend with immunomodulatory activity.
Introduction
A widespread dependence on complementary and alternative medicine to treat both acute and chronic illnesses led to extensive research and the identification of some plants that may be able to prevent disease. Numerous herbs from Ayurvedic formulations have been used, singly or combined to modulate the immune system. Immune activity alterations brought on by dietary choices and environmental contaminants are known to occur in diets high in micronutrients and antioxidants (Bafna & Mishra, 2010).
Herbs are used in the indigenous medicine system as immunomodulators to alter the body’s defense mechanism. Numerous experimental models have established the influence of plant derivative active components, namely polysaccharides, flavonoids, peptides, lectins, and tannins on the immune system (Bafna et al., 2021; Shivaprasad et al., 2006).
The Asparagus racemosus, Tinospora cordifolia, and Trigonella foenum-graecum extract blend (ATTEB) is widely used by rural people in the Indian subcontinent as traditional medicine for its multiple activities like antimicrobial, anti-inflammatory, immunomodulator, adaptogen, female tonic, galactagogue, and so on (Deshpande & Ranade, 2021; Khare, 2011; Nadkarni & Nadkarni, 1954; Sharma & Dash, 1999). Thus, the blend of these three extracts was selected for the study.
A. racemosus is a member of the plant family, Liliaceae. It contains a variety of compounds, including vitamins A, B1, B2, C, and E, minerals (primarily magnesium, phosphorus, calcium, and iron), folic acid, essential oils, asparagine, arginine, tyrosine, flavonoids (kaempferol, quercetin, and rutin), mucilage, resin, tannin, and oligospirostanoside (immunoside) (Alok et al., 2013; Kanwar & Bhutani, 2010; Tiwari et al., 2017). It is used as a potent antioxidant, antibacterial, antimicrobial, immune stimulant, adaptogenic, anti-ulcerogenic, analgesic, anti-hepatotoxic, anti-dyspepsia, galactagogue, antitussive, antitumor, anti-inflammation, anti-neuropathic, treatment of cough, bronchitis, treatment of neurodegenerative disorders, cardioprotective, and antibacterial (Alok et al., 2013).
T. cordifolia, which belongs to the family Menispermaceae, strengthens the body’s defenses against illness. The Ayurvedic Pharmacopoeia of India recognizes the medicinal use of the entire plant, specifically the stem because it contains more alkaloids than leaves. Alkaloids, namely, berberine, palmetine, and tinosporine, are found to be present in sufficient amounts in the stem. The glycosides found are tinocordifolioside, cordifolioside A and B, diterpenoid lactone furanolactone, and clerodane derivatives (amritoside A, B, C, and D), (Agrawal et al., 2012; Reddy et al., 2009; Sharma et al., 2012). It has been used as a general tonic, anti-inflammatory, anti-arthritic, anti-allergic, antimalarial, antidiabetic, and aphrodisiac properties (Upadhyay et al., 2010).
The Ayurvedic Pharmacopoeia of India lists the stem as the official medication. The T. cordifolia stem is one of the ingredients in several Ayurvedic remedies used for fever, dyspepsia, general debility, and urinary disorders.
Due to its immunomodulatory properties, T. cordifolia has been reported to be used successfully to treat various infections, including infections of the mammary gland (Abiramasundari et al., 2012; Mukherjee et al., 2010).
T. foenum-graecum plant contains high levels of soluble mucilage, fiber, and galactomannan, which inhibit the digestion and absorption of starch and bile salts. Seeds of this plant have been shown to contain 0.2%–0.9% diosgenin, 4.8% saponins, 35% alkaloids, and 100 mg of flavonoids per gram of fenugreek seeds (Jani et al., 2009; Meghwal & Goswami, 2012). In addition, the steroidal sapogenin and diosgenin found in fenugreek have been shown to boost milk production in nursing mothers (Bin-Hafeez et al., 2003; Forinash et al., 2012; Mandegary et al., 2012). The body weight (BW) gain was reduced, and an increase in water intake, a decrease in blood sugar, and an increase in alanine transaminase (ALT) levels were observed. A decrease in BW in male mice was observed (Kandhare et al., 2015). It has been proven to act as an immunomodulator (Bin-Hafeez et al., 2003).
However, to date, no scientific evaluations have been conducted to confirm the immunomodulatory role of the combination of all three plants. Thus, this study was designed to study the immunomodulatory activity of the blend (1:1:1) of extracts of these three plants. A formulation of the same was also prepared and evaluated.
Extraction and Phytochemical Characterization
Plant Material and Extraction
The dried roots of A. racemosus, dried stems of T. cordifolia, and dried seeds of T. foenum-graecum L. were purchased from D. G. Ayurvedic Sangrah, Andheri, Mumbai. Its authenticity was certified by the Department of Botany, Government Holkar Science College, Indore, Madhya Pradesh, India, and voucher specimens were deposited in the department for future reference.
For the extraction, the collected roots, stems, and seeds (100 g each) of A. racemosus, T. cordifolia, and T. foenum-graecum, respectively, were grounded into powder in a grinder and extracted by using a soxhlet apparatus with 95% ethanol. The extracts were concentrated using a rotary evaporator at a temperature of 45°C and dried in a vacuum oven at 40°C (Jha & Sit, 2022). The dried extracts were collected and stored in a well-closed container at a cool and dry place for future use. All three extracts were named and labeled as A. racemosus extract (ARE), T. cordifolia extract (TCE), and T. foenum-graecum extract (TFGE), respectively.
The blend of all three extracts in 1:1:1 W/W/W (ATTEB) was prepared and used for further studies.
Phytochemical Analysis of Extracts
The individual extracts and ATTEB were subjected to a preliminary phytochemical investigation (Bhavsar et al., 2023; Gadamsetty et al., 2013; Khandelwal, 2008).
High-performance Thin Layer Chromatography (HPTLC) Analysis
Phytochemical fingerprinting of the obtained extract and ATTEB was performed using HPTLC. The ethanolic solution of each sample, that is, all extracts and ATTEB (1 mg/mL) was prepared and utilized for HPTLC. The HPTLC system comprises a CAMAG Linomat-V—an automated sample applicator with 100 µL syringe and a CAMAG TLC scanner-4. The analysis was performed on a 20 × 10 cm aluminum plate coated and pre-activated with a 0.2 mm silica gel 60 F254 layer. The samples were applied using the Linomat-V—an automated sample applicator with a bandwidth of 6 mm and space between two bands ranging from 8 to 17 mm. Linear ascending development of the plate was carried out in twin—a trough glass chamber previously saturated with mobile phase for 20 minutes at room temperature (250°C ± 2°C) and relative humidity 60% ± 5%. The solvent front (chromatogram length) run was approximately 70 mm. After development, the plate was removed and dried. The scanning was performed using the CAMAG TLC scanner-4; peak height, peak area, and spectra were measured and recorded. The mobile phase for A. racemosus extract with shatavarin IV—ethyl acetate:methanol:water (8:2:1 V/V/V); T. foenum-graecum L. with trigonelline—propanol-2-ol:methanol:water (4:1:4 V/V/V); T. cordifolia with berberine—n-butanol:acetic acid:water (14:3:4 V/V/V) and for ATTEB—ethyl acetate:methanol:water (8:2:1 V/V/V) were used. The phytochemical fingerprinting of all extracts was performed using the toluene:ethyl acetate:methanol (4:4:1 V/V/V) as mobile phase (Mahata et al., 2012; Shinde et al., 2011).
Experimental Animals
The animal studies designed for the immunomodulatory activity of ATTEB were approved by the Institutional Animal Ethics Committee-Committee for the Purpose of Control and Supervision of Experiments on Animals (IAEC-CPCSEA) (approval number: IAEC/2019/01/01). The study used Swiss albino mice (25–30 g) of both sexes. During the week before and during the tests, they were housed in cages with six animals per cage, with standard conditions being 26.2°C, 44–56% relative humidity, and daily cycles of 12 hours of light and 12 hours of darkness. All the animals received water and a regular diet of rat pellets.
Experimental Design
The drug solutions were prepared in distilled water for oral administration. The immunomodulatory activity of ATTEB was checked at the cellular level. Cellular immunity was evaluated using a neutrophil adhesion test and carbon clearance assay. Both the experimental models had four common groups consisting of six animals each. Group I—Control (received only vehicle 10 mL/kg p.o.), Group II—Standard (levamisole 25 mg/kg), Group III—ATTEB (150 mg/kg p.o.), Group IV—ATTEB (300 mg/kg p.o.), respectively.
Neutrophil Adhesion Test
For 14 days, the animals received the oral treatment described above. Blood samples were taken on the 14th day of medication administration by puncturing the retro-orbital plexus into a vial containing heparin. The samples were then examined for differential leukocyte count (DLC) and total leukocyte count (TLC). After initial counts, blood samples were incubated with 80 mg/mL of nylon fibers for 15 minutes at 37°C. The incubated blood samples were again analyzed for TLC and DLC (Bhavsar et al., 2023; Mungantiwar et al., 1999).
The product of TLC and the percentage of neutrophils give the neutrophil index (NI) of the blood sample. Percent neutrophil adhesion was calculated as shown below:
Neutrophil adhesion (%) = [(NIu – NIt)/NIu] × 100
where, NIu = Neutrophil index of the untreated blood sample; Nit = Neutrophil index of the treated blood sample.
Carbon Clearance Test
Animals received the above-described oral treatment for 5 days. After 5 days, 48 hours after the previous treatment, 0.1 mL of carbon ink suspension was injected into the tail vein. Blood samples were extracted from the retro-orbital sinus (in 5 mL of ethylenediaminetetraacetic acid (EDTA) solution) at 0 and 15 minutes.
A 25 mL sample was combined with 2 mL of a 0.1% sodium carbonate solution, and its absorbance at 660 nm was measured (Bhavsar et al., 2023; Gokhale et al., 2003; Jayathirtha & Mishra, 2004).
The phagocytic index was calculated using the following equation:
Phagocytic index = k (sample)/k (control)
where, k = (Log OD1 – Log OD2)/15
OD1 = optical densities at 0 minutes and OD2 = optical densities at 15 minutes, respectively.
Formulation and Evaluation of ATTEB Effervescent Tablet
For the preparation of tablets, the following method was followed:
Step I: Add extract (ARE, TCE, and TFGE) with lactose and mix well. Add tartaric acid, sodium bicarbonate, and sucrose to this mixture and mix.
Step II: Mix 2% polyvinylpyrrolidone (PVP) in isopropyl alcohol (IPA) 2% and sodium benzoate.
Add the mixture obtained from Step II to the mixture obtained by Step I drop-wise to obtain lumps. Pass these lumps through sieve number 20. Tray dry the product for 1 hour at 60°C–70°C. Pass the dried blend through sieve number 100 to get granules. Mix 1.5% W/W talc and magnesium stearate for 15 minutes. The dried granules were compressed to get the tablets through a tablet compression machine (Maru & Belemkar, 2021; Shirsand et al., 2010).
Evaluation
The following evaluation parameters were assessed as per Indian Pharmacopoeia.
Weight variation: Twenty tablets were weighed altogether. The average weight was calculated using the individual weight of the tablet. No more than two tablets should exceed 5% of the average weight.
Thickness and diameter: The thickness and diameter of the tablets were measured using Vernier calipers.
Hardness: Hardness or crushing strength determinations are made during tablet production and are used to determine the need for pressure adjustment on tablet machines. A Monsanto hardness tester measured the hardness of the tablet. The hardness of the tablet should be between 3 and 5 kg/cm2.
Friability: The friability of the tablet was determined by the friability tester. The weight of the six tablets was done before and after rotating in a friability tester at 25 rpm for 4 minutes. The % friability should be less than 1% in all the formulations, ensuring that the tablets are mechanically stable.
Effervescent time: The disintegration test is carried out using the disintegration tester, consisting of a basket rack holding six plastic tubes, open at the top and bottom, the bottom of the tube is covered by a 10-mesh screen. The basket is immersed in a bath of suitable liquid held at 37°C, preferably in a 1 L beaker.
A single tablet is placed in a beaker containing 200 mL of purified water at 20°C ± 1°C whenever a clear solution without particles is obtained, it is considered effervescent time.
Stability Studies of Formulation
Stability studies were performed as per the International Council for Harmonisation of Technical Requirements of Pharmaceuticals for Human Use (ICH) guidelines (European Medicines Agency, 2003). Specifically, three time points were selected for accelerated stability studies. Tablets were kept at a temperature of 40°C ± 2°C, with relative humidity of 75% ± 5% for 0, 3, and 6 months. Following keeping tablets for a selected duration, tablets were evaluated for various parameters, including weight variation, thickness, diameter, hardness, friability, and disintegration time (Patel & Siddaiah, 2018; Suksaeree et al., 2023).
Results
The total amount of ethanolic extracts obtained (in percentage) from all three plant materials are A. racemosus (ARE): 10%, T. cordifolia (TCE): 10%, and T. foenum-graecum (TFGE): 12.5%.
Phytochemical Screening of Plant Extracts
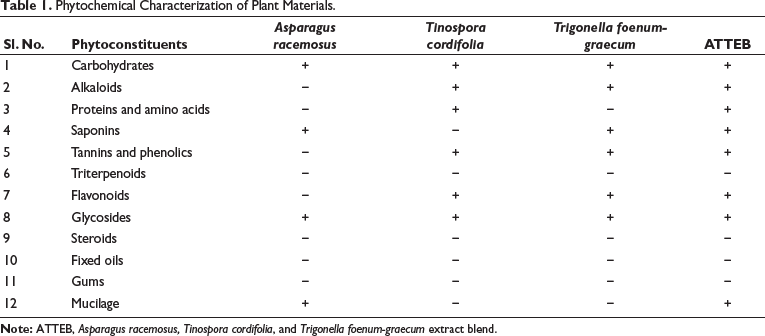
A preliminary phytochemical analysis was performed to identify the phytochemicals in the obtained extracts and ATTEB. The obtained results confirmed the presence of bioactive constituents in respective extracts and a combination of all three in the ATTEB. The detailed observations are given below in Table 1.
Phytochemical Characterization of Plant Materials.
HPTLC Analysis of Extracts
The HPTLC studies of all the extracts and ATTEB provided valuable insights into the phytochemical profiles of the extracts and ATTEB. The optimized mobile phase system for each extract and respective analytical standard is as follows: for A. racemosus with shatavarin IV—ethyl acetate:methanol:water (8:2:1 V/V/V); for T. foenum-graecum L. with trigonelline—propanol-2-ol:methanol:water (4:1:4 V/V/V); for T. cordifolia with berberine—n-butanol:acetic acid:water (14:3:4 V/V/V), and ATTEB—ethyl acetate:methanol:water (8:2:1 V/V/V). The obtained results indicated the presence of shatavarin IV, trigonelline, and berberine in A. racemosus, T. foenum-graecum L., and T. cordifolia extract, respectively.
HPTLC performed phytochemical fingerprinting of individual extracts and ATTEB. For the phytochemical fingerprinting of all extracts, another mobile phase system was optimized, that is, toluene:ethyl acetate:methanol (4:4:1 V/V/V), which ensured efficient separation of the compound. The HPTLC analysis confirmed the presence and appropriately separated bioactive principal components of the respective extracts and ATTEB in the chromatographic system. The Rf values of analytical standards, that is, shatavarin IV, trigonelline, and berberine were found to be at Rf 0.42, Rf 0.32, and Rf 0.408, respectively (Figure 1). The Rf value calculations from the observations obtained from the HPTLC plates of all standards, extracts, and ATTEB confirm the presence of shatavarin IV, trigonelline, and berberine in extracts as well as in ATTEB. These phytoconstituents are the active/major principles of the respective plants for the pharmacotherapeutic activity/response.
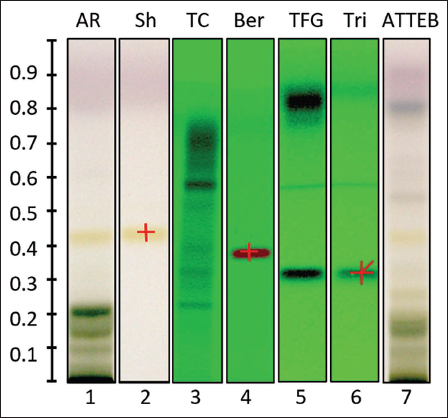
The ATTEB and each component of ATTEB, that is, extracts of A. racemosus, T. cordifolia, and T. foenum-graecum were analyzed. All confirmed the presence of bioactive compounds, that is, shatavarin IV, trigonelline, and berberine.
Neutrophil Adhesion Test
Neutrophil counts decrease as a result of their adhesion to nylon fibers post-incubation with blood. The ATTEB-treated groups showed higher neutrophil adhesion as compared to the control group, that is, 16.9% ± 1.94%. Groups administered with a dose of 150 mg/kg ATTEB and 300 mg/kg ATTEB exhibited a significant increase in neutrophil adhesion, that is, 47.42% ± 1.87% and 52.17% ± 0.79%, respectively, confirming the immune stimulant effect in animals after drug treatment. The results obtained on treatment with the standard drug levamisole 25 mg/kg BW—52.9% ± 1.25% and those treated with 300 mg/kg BW ATTEB—52.17% ± 0.79% were comparable (Table 2 and Figure 2).
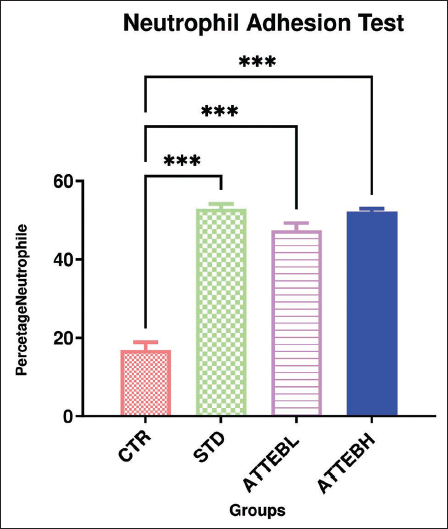
Neutrophil Adhesion Test.
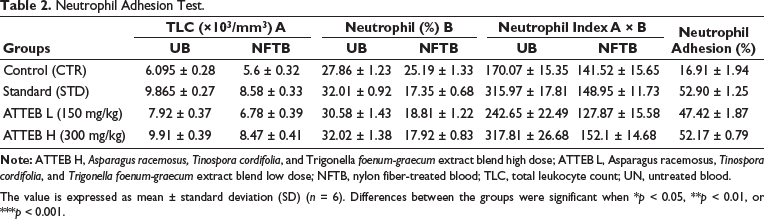
The value is expressed as mean ± standard deviation (SD) (n = 6). Differences between the groups were significant when *p < 0.05, **p < 0.01, or ***p < 0.001.
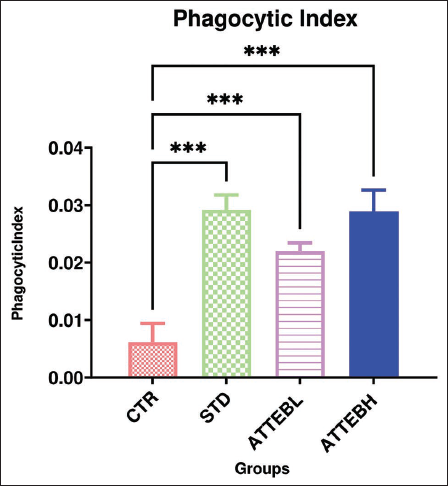
Carbon Clearance Test (Phagocytic Index)
Eliminating diseased cells, such as debris, microorganisms, inorganic particles, or tissue derbies by ingesting and digesting is a constant process that goes on in the body called phagocytosis. A number of cells belonging to the reticuloendothelial system (RES) perform this and are collectively called phagocytes. Several diseases are linked to the abnormalities in or malfunctioning of phagocytosis. The same activity is utilized to assess carbon clearance from the body (blood). A dose-related increase in phagocytic index, that is, 0.022 ± 0.0015 and 0.029 ± 0.004, was observed on treatment with 150 and 300 mg/kg BW ATTEB, respectively, for 5 days. The results are represented in Table 3 and Figure 3.
Carbon Clearance Test (Phagocytic Index).

Formulation Development and Evaluation of Formulation of Effervescent Tablet
Formulation
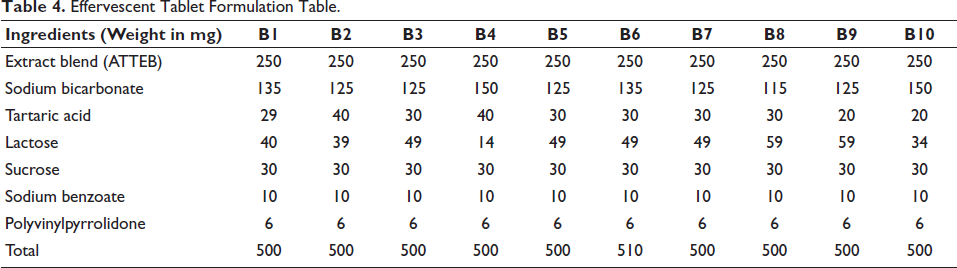
With the standard ingredients, a formula was developed. A total of 10 formulations were prepared and evaluated for the study. B7 formulation was found to be optimum in all parameters (Maru & Belemkar, 2021) (Table 4 and Figure 4).
Effervescent Tablets.

Effervescent Tablet Formulation Table.
Evaluation of Tablet Formulation
Based on the pharmacopoeial guidelines, various parameters were evaluated to observe the appropriateness of the developed formulation.
Tablet Evaluation Parameters were Assessed
Weight variation, thickness, diameter, hardness, friability, and disintegration time. The parameters complied with the requirements of the Indian Pharmacopoeia.
Among the 10 formulations prepared, the B7 formulation complied with all the pharmacopeial standards and was the most suitable formulation. The parameters recorded for B7 formulation are as shown here: average tablet weight (500.4 mg), table diameter (7.76 mm), tablet thickness (3.34 mm), friability (0.47%), hardness (3.08 kg/cm2), and disintegration time (86 minutes). Specifically, the formulation B7 was found to have optimum tablet weight and effervescent time, which led to the selection of the B7 formulation.
Effervescent Tablets were Prepared as Ready-to-use Formulations
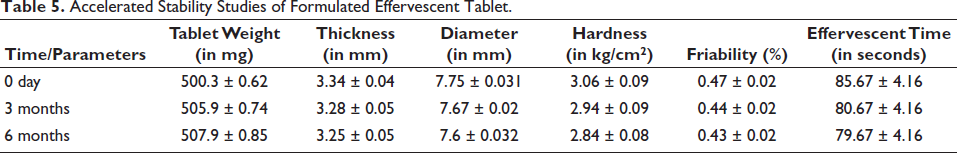
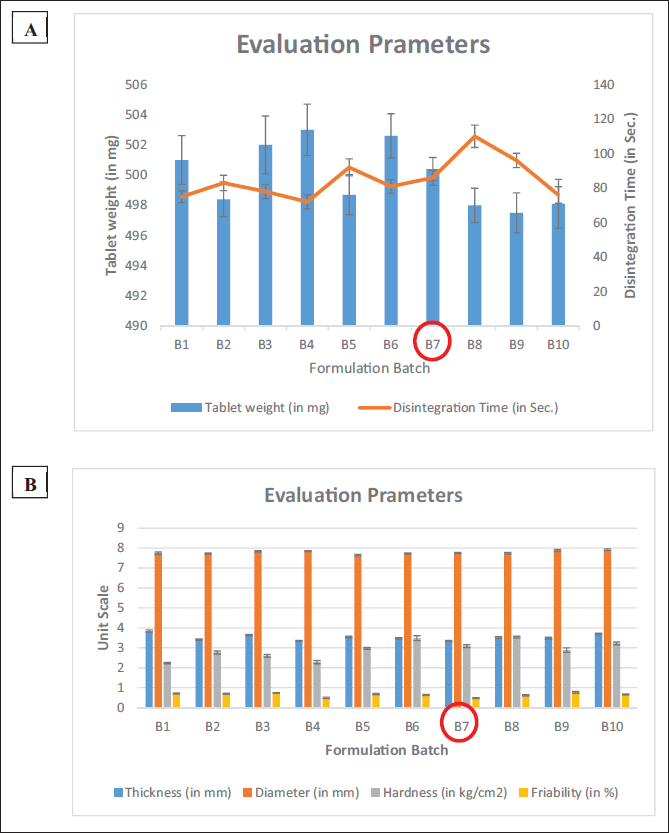
Stability Studies of Tablets
As per the standards of the ICH guidelines, evaluation of the formulation was carried out in triplicate. The study was carried out for 3 and 6 months (Table 5). Based on various evaluation parameters, the formulation was stable for all the time points (Figures 5 and 6). Though there was a slight increase in the weight of the formulation, it was insignificant as per the guidelines. The formulation complied with the stability requirements of pharmaceuticals for 3 and 6 months.
The stability was established based on the percentage of the evaluation parameters recorded during the evaluation. None of the parameters should be less than 90% of the freshly prepared formulation. The formulation qualified for the stability studies as per prescribed limits in official books.
Accelerated Stability Studies of Formulated Effervescent Tablet.
Stability Studies of Effervescent Tablets.
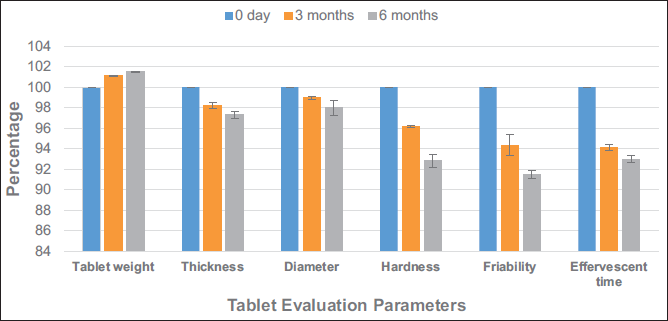
The percentage calculation (Figure 6) for stability studies data (Table 5) at 3 and 6 months is calculated with reference to the data obtained at 0 days of the study.
Discussion
In the current study, the extract blend of A. racemosus, T. cordifolia, and T. foenum-graecum was evaluated for its immune-modulating activity. The ethanolic extracts of all three drugs and their blend were subjected to phytochemical analysis. They were found to contain all the primary active constituents as reported earlier in the literature. The presence of these active constituents was further confirmed by HPTLC. During the development of the mobile phase system for the above-mentioned phytochemicals, various solvents as well as the combination of the various solvent systems were tailored to each sample’s unique chemical composition and achieve efficient separation. Various problems were encountered during the development of the mobile phase system; such as dragging of the peaks and others. All the problems were resolved with the optimized mobile phases, that is, toluene:ethyl acetate:methanol (4:4:1 V/V/V).
Neutrophil adhesion and carbon clearance tests were performed to analyze the immune-modulatory activity. The blend of extract ATTEB enhanced neutrophil adhesion by approximately 35% compared to the control group. Polymorphonuclear leukocytes (neutrophils, PMNs) get activated and play a predominant role in the clearance of microorganisms and foreign particles, initiating the host response to infection and foreign particle invasion. On activation, neutrophils reach the site of damage/infection by the process of margination in blood vessels, adhesion to endothelial cells, penetration through endothelial cells, diapedesis, and migration to damaged or infected tissue. In the whole process, adhesion is the key step to stop neutrophils from flowing along with the blood. Neutrophils recognize foreign particles through their surface pattern recognition receptors (PRRs) such as selectins and integrins. The activation of surface receptors is mimicked through the nylon fiber as the foreign particle (Liu et al., 2019). The ATTEB showed a considerable impact on the process by quick outreach to the site of damage.
To eliminate foreign particulate matter from circulation, RES, wandering, and stationary-type phagocytic cells are used in various organs. Especially, the stationary phagocytic cells like liver Kupffer cells, splenic macrophages, and others play a vital role in the elimination of such particles. Clearance of intravenously administered carbon particles (carbon ink suspension acts as foreign particles) in laboratory animals can mimic the phagocytic activity and thus, the overall capacity of the immune system to eliminate foreign particles from the bloodstream (Liu et al., 2019).
The results indicated a five-fold increase in the phagocytic clearance of carbon particles with a high dose of ATTEB compared to the control group. This indicates the possible enhancement of phagocytic activity by the ATTEB. Altogether, enhanced adhesion to endothelial cells and enhanced phagocytosis activity may contribute to exerting an efficient immune response in trauma or infection. Thus, it can be concluded that ATTEB demonstrated to have immunomodulatory activity in the condition of immune system activation.
The drug that induces adhesion may help in reaching neutrophils at the site of infection or damage quickly and clearing it rapidly from the vicinity. Thus, overall quicker control of infection and recovery from the trauma.
In order to get a ready-to-use formulation of ATTEB, which has been studied for its immunomodulatory activity, an effervescent tablet formulation has been prepared and evaluated, out of 10 formulations, formulation seventh was found to be the most suitable. The effervescent time was found to be 86 seconds, which is optimum for developing a drink from the effervescent tablet. The hardness and friability were found to be optimum for preventing the breaking of tablets or shedding a considerable number of small particles from the tablets during transportation and bearing other shears. The formula was also found to be suitable for uniform hoper sliding and so on to endure uniformity in the size, diameter, and weight of the tablet.
The accelerated stability study is designed to observe the chemical degradation and physical alteration in the formulation at exaggerated (temperature and humidity) conditions of storage. The formulation passed all the pharmacopeial standards and was found to be stable for 6 months as observed in accelerated stability studies as per ICH guidelines (European Medicines Agency, 2003). The crushing strength, that is, hardness of the tablet, can be altered by moisture content. It can also affect other parameters like weight, thickness, diameter, and friability of the tablet. On storing the tablet for 3 and 6 months at the temperature of 40°C ± 2°C, relative humidity of 75% ± 5% could not induce a significant change in various pharmaceutical parameters (Maru et al., 2023; Tesfa et al., 2007). Thus, the tablet can be considered stable as per the regulatory requirements. The research develops a herbal product with an immunomodulatory potential that can be administered easily to the patient population of any age.
Conclusion
In the current study, the blend of extracts of ATTEB was prepared and standardized. The extract blend was found to comply with pharmacopoeial standards. The ATTEB was studied for immunomodulatory activity in mice and was found to enhance phagocytic activity and stimulate neutrophil activation that contributes to the immune system activity. Effervescent tablets as ready-easy-to-use products were formulated using ATTEB and evaluated. The formulation qualified the standard evaluation parameters. The stability of the formulation was assessed as per ICH guidelines, and formulations were found to be stable. The study findings suggest that the effervescent tablet formulation of ATTEB is well-stable and has good immunomodulatory activity in mice animal models.
Footnotes
Abbreviations
ALT: Alanine transaminase; ARE: Asparagus racemosus extract; ATTEB: Asparagus racemosus, Tinospora cordifolia, and Trigonella foenum-graecum extract blend; ATTEB H: Asparagus racemosus, Tinospora cordifolia, and Trigonella foenum-graecum extract blend high dose; ATTEB L: Asparagus racemosus, Tinospora cordifolia, and Trigonella foenum-graecum extract blend low dose; CPCSEA: Committee for the Purpose of Control and Supervision of Experiments on Animals; CTR: Control group; DLC: Differential leukocyte count; HPTLC: High-performance thin layer chromatography; IAEC: Institutional Animal Ethics Committee; ICH: International Council for Harmonisation of Technical Requirements of Pharmaceuticals for Human Use; IPA: Isopropyl alcohol; NFTB: Nylon fiber-treated blood; NI: Neutrophil index; OD: Optical density; PMN: Polymorphonuclear; PRR: Pattern recognition receptor; PVP: Polyvinylpyrrolidone; RES: Reticuloendothelial system; SD: Standard deviation; STD: Standard drug group; TCE: Tinospora cordifolia extract; TFGE: Trigonella foenum-graecum extract; TLC: Total leukocyte count; UB: Untreated blood.
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animal studies designed for the immunomodulatory activity of ATTEB were approved by the Institutional Animal Ethics Committee-Committee for the Purpose of Control and Supervision of Experiments on Animals (IAEC-CPCSEA) (approval number: IAEC/2019/01/01).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
