Abstract
Background
Natural compounds with radioprotective properties have gained attention due to their potential to reduce health risks associated with radiation exposure. Galangin (GA), a flavonoid recognized for its antioxidant activity, has been investigated for its capacity to alleviate oxidative stress.
Objectives
This study aimed to explore the radioprotective effects of GA against gamma radiation-induced double-strand breaks (DSBs), dicentric chromosomes (DCs), and cytokinesis-block micronuclei (CBMN) in human blood lymphocytes.
Materials and Methods
Human blood peripheral lymphocytes (HBPLs) were treated with varying concentrations of GA (1, 5, and 10 µM) for 24 h before being exposed to gamma radiation (3 Gy). Following radiation exposure, the cells were incubated for another 24 h to evaluate deoxyribonucleic acid (DNA) damage. Cytogenetic assays were performed to measure DC and CBMN frequencies, while DSBs were assessed using γ-H2AX analysis. Additionally, the phosphorylation of ATM (p-ATM) was analyzed through Western blotting, and DNA repair gene expression was examined using a polymerase chain reaction (PCR) array. Statistical methods were employed to analyze the data and determine the significance of GA’s protective effects.
Results
Gamma radiation exposure markedly increased the incidence of DCs and CBMN in HBPLs in a dose-dependent manner. Pretreatment with GA, however, significantly reduced these effects in a concentration-dependent fashion. GA also decreased radiation-induced DSBs, as demonstrated by reduced levels of γ-H2AX and p-ATM. Furthermore, GA-treated cells exhibited a downregulation of DNA repair-related gene expression compared to cells exposed solely to radiation. Among the tested concentrations, 10 µM GA provided the greatest protective effect.
Discussion
GA effectively mitigates gamma radiation-induced DNA damage in human lymphocytes by lowering the incidence of DSBs, DCs, and CBMN. The protective effects appear to be mediated through modulation of the DNA damage response and the reduction of oxidative stress.
Conclusion
This study highlights GA’s potential as a radioprotective agent and suggests its relevance in radiation therapy and preventive applications. Further research is warranted to fully elucidate its mechanisms and therapeutic potential.
Introduction
Radiotherapy is a widely utilized treatment method, employed in managing over 50% of all cancer subtypes. The effectiveness of this approach relies on delivering the optimal radiation dose while maximizing tumor cell sensitivity to the treatment (Baskar et al., 2012). However, the radiation dosage used in therapy is often limited by the sensitivity of surrounding healthy tissues. The primary goal of radiotherapy is to maximize cancer cell destruction while minimizing harm to adjacent normal tissues. To achieve this, various strategies have been developed to protect normal cells from the harmful effects of ionizing radiation during treatment. Insights into deoxyribonucleic acid (DNA) damage response mechanisms, genetic modifications, and molecular signaling pathways have the potential to pave the way for improved treatment strategies, particularly when combined with pharmacological interventions (Bhattacharya & Asaithamby, 2017). Several adjuvant compounds have been proposed to enhance the sensitivity of tumor cells to radiation while safeguarding normal tissues from its adverse effects (Zhang et al., 2023).
Radiation-induced double-strand breaks (DSBs) are often evident as chromosomal aberrations (CAs). These include three primary types: dicentric chromosomes (DCs), acentric fragments (AFs), and acentric rings (ARs), which arise from chromatid breakages and improper exchanges (Begum et al., 2012). CAs serve as biomarkers for radiation exposure and can be identified in cultured human blood lymphocytes (Prasad et al., 2005). Furthermore, CAs resulting from ionizing radiation are often observed as micronuclei (MN) in affected cells (Vral et al., 2011). The cytokinesis-block micronucleus (CBMN) assay is a widely utilized method for assessing DNA damage caused by radiation (Beinke et al., 2016). Another technique for detecting radiation-induced DNA strand breaks is the comet assay, or modified single-cell electrophoresis, which offers a rapid means of identifying single-strand breaks (SSBs) and DSBs at the individual cell level (Møller, 2018; Olive, 2009). In response to DSBs, mammalian cells recruit specific proteins to the damaged sites. Phosphorylated histone H2AX (γ-H2AX) and phosphorylated ataxia-telangiectasia mutated protein (p-ATM) can be detected at these sites using specialized antibodies (Lee et al., 2019). Specifically, phosphorylation of ATM at serine 1981 (p-ATM) and H2AX at serine 123 (γ-H2AX) are early indicators of cellular responses to DSBs induced by ionizing radiation (Murray et al., 2016).
Ionizing radiation generates reactive oxygen species (ROS), which cause severe oxidative damage, leading to redox imbalance and the activation of cellular signaling pathways (Yi et al., 2020). Key endogenous antioxidants, such as reduced glutathione (GSH), catalase, superoxide dismutase (SOD), and GSH peroxidase, play a critical role in protecting normal tissues from radiation-induced oxidative stress. Several phytochemicals enhance the activity of these antioxidant enzymes, thereby shielding cellular components from oxidative damage caused by ionizing radiation (Materska et al., 2015; Shyam et al., 2018; Wu & Dhanasekaran, 2020). Nevertheless, when cells experience severe radiation damage, endogenous repair mechanisms may not suffice. Supplementing with exogenous natural antioxidants can help mitigate radiation effects. Epidemiological studies suggest that a diet rich in fruits and vegetables, which are abundant in natural antioxidants, reduces the risk of radiation-induced DNA damage and CAs (Rostami et al., 2016). Radioprotectors, which are chemical agents designed to protect normal tissues during radiotherapy, are one approach to minimizing such damage. For example, amifostine is a Food and Drug Administration (FDA)-approved radioprotector that shields normal cells during radiotherapy (King et al., 2020).
Natural compounds with free radical scavenging activity offer significant potential for enhancing radioprotection in normal cells during cancer treatment (George et al., 2021). Galangin (GA), a flavonol found in Alpinia officinarum, Alpinia galanga, and propolis, is one such compound (Tuli et al., 2022). It exhibits a variety of pharmacological properties, including the inhibition of alpha-glucosidase activity, which reduces glycation product formation (Zeng et al., 2019). GA also prevents airway remodeling by suppressing ROS generation and modulating mitogen-activated protein kinase (MAPK) signaling pathways (Liu et al., 2015). Additionally, it induces apoptosis in gastric cancer cells (Liang et al., 2021) and inhibits epithelial-mesenchymal transition in renal cell carcinoma (Cao et al., 2016). Despite its potential as a functional food component (Mak et al., 2018), GA against radiation-induced cellular damage has not been previously reported. The current study aims to investigate the radioprotective properties of GA, focusing on its ability to mitigate ionizing radiation-induced damage in human blood lymphocytes.
Materials and Methods
Chemicals
The chemicals and reagents used in this study included GA, cytochalasin-B (Cyt-B), colchicine, phytohemagglutinin-M (PHA-M), and fetal bovine serum, all sourced from Sigma Co., St. Louis, USA. Additional materials, including RPMI-1640 medium,
GA Preparation
A stock solution of GA (1 mM) was prepared using 0.05% dimethyl sulfoxide (DMSO) and diluted to working concentrations for experimental use. Control experiments included 0.05% DMSO as a vehicle control.
Study Plan
Human blood peripheral lymphocytes (HBPLs) were obtained from BioIVT Scientific Company. The lymphocytes were freshly isolated from healthy donors and delivered within 24 h of collection. The cells were divided into the following experimental groups: Group I—Control: HBPLs treated with 0.05% DMSO. Group II—GA only: HBPLs treated with 10 µM GA. Group III—Radiation only: HBPLs exposed to gamma radiation doses of 1, 2, or 3 Gy. Group IV—Radiation + 1 µM GA: HBPLs pretreated with 1 µM GA for 1 h, followed by gamma radiation exposure (1, 2, or 3 Gy). Group V—Radiation + 5 µM GA: HBPLs pretreated with 5 µM GA for 1 h, followed by gamma radiation exposure (1, 2, or 3 Gy). Group VI—Radiation + 10 µM GA: HBPLs pretreated with 10 µM GA for 1 h, followed by gamma radiation exposure (1, 2, or 3 Gy).
Gamma radiation was delivered at doses of 1, 2, and 3 Gy using a cobalt-60 teletherapy unit, with a dose rate of 1.6 Gy/min. The GA treatments (1, 5, and 10 µM) were administered to whole blood samples 1 h before irradiation. After exposure, the samples were held at room temperature for 1 h and then transported to the laboratory in an icebox for further analysis.
Dicentric Aberration (DC) and CBMN Assay
The International Atomic Energy Agency (IAEA) established the guidelines for the cytogenetic analysis (International Atomic Energy Agency, 2011). A 0.5 mL sample of whole blood was cultured in 4.5 mL of RPMI-1640 medium; then, PHA-M, 200 µL was added to stimulate cell division, and the cultures were incubated for 72 h in a 5% CO2 atmosphere.
To assess DC aberrations, colchicine (200 µL, 0.04 µg/mL) was introduced to the cultures at the 70th h to arrest cells in metaphase. At the 72nd hour, the cells were harvested using a hypotonic solution of 0.56% potassium chloride (KCl). After harvesting, the cells were washed and resuspended in Carnoy’s fixative (3:1 methanol:acetic acid). Fixed cells were then spread onto prelabeled microscope slides, air-dried, and stained with 4% Giemsa solution. The prepared slides were mounted in distyrene plasticizer xylene (DPX) and examined under a microscope at 100× magnification using oil immersion to detect CAs across the experimental groups.
The protocol for the CBMN assay was similar to that of the DC assay, with the addition of Cyt-B (6 µg/mL) at the 44th hour of incubation to prevent cells at the cytokinesis stage of the cell cycle. At the 72nd hour, the cells were harvested using a hypotonic 0.56% KCl solution, washed, and fixed with Carnoy’s fixative. The fixed cells were subsequently distributed on prelabeled slides, air-dried, and stained with 4% Giemsa solution. Slides were examined under a microscope to evaluate mono-, bi-, and multinucleated cells under different treatment conditions, as described by Prasad et al. (2005).
Measurement of Intracellular ROS Levels
The dichloro-dihydro-fluorescein diacetate (DCFH-DA) probe was employed to evaluate the impact of GA on ROS generation induced by 3 Gy gamma radiation. In this procedure, HBPLs were treated with 1 µM DCFH-DA dye and incubated for 1 h to allow oxidation of the dye. Following the incubation period, the cells were washed with phosphate-buffered saline (PBS) to remove any excess unbound dye remaining. Dichlorofluorescein (DCF), the oxidized fluorescent product, was subsequently quantified through fluorescence microscopy and a spectrofluorometer. For the purpose of detection, the excitation and emission wavelengths were established at 485 and 535 nm, respectively (Rastogi et al., 2010).
Analysis of Cellular Antioxidants System
Irradiated blood lymphocytes were suspended in 0.1 M dithiothreitol and subjected to gentle sonication in a solution containing 130 mM KCl and PBS. The resulting supernatant was collected to assess cellular antioxidant levels. SOD activity was determined by measuring the inhibition of the nicotinamide adenine dinucleotide (NADH)-phenazine methosulfate-nitroblue tetrazolium formazan reaction. The assay was initiated by adding NADH to the reaction mixture, and the reaction was stopped after 90 s by introducing glacial acetic acid. The color formed during the reaction was extracted into the n-butanol layer and measured spectrophotometrically at 520 nm (Kakkar et al., 1984). Catalase activity was analyzed by monitoring the decomposition of hydrogen peroxide (H2O2) over varying time intervals. The reaction was terminated by adding a dichromate-acetic acid mixture, which forms perchromic acid that subsequently converts to chromic acetate when heated in the presence of H2O2. The amount of chromic acetate produced was measured at 620 nm using a microplate reader (Sinha, 1972). The activity of GSH peroxidase was assessed by allowing a known amount of the enzyme to react with H2O2 and reduced GSH for a defined duration. The remaining GSH in the reaction was then quantified to determine glutathione peroxidase (GPx) activity (Rotruck et al., 1973).
Determination of DSBs by Neutral Comet Assay
The neutral comet assay was implemented in accordance with the methodology established by Olive and Banáth (2006) to assess the severity of DNA DSBs. 50 µL of control or irradiated HBPL and 200 µL of 0.5% low melting point agarose (LMPA) was poured onto the slides. The cells were lysed using a prechilled lysis solution and incubated at 4°C for 1 h. The cells were transferred to an electrophoresis chamber that was filled with buffer at potential of hydrogen (pH) 7 after lysis. The electrophoresis was conducted for 25 min at a voltage of 1.33 V/cm. The DNA was visualized by staining the specimens with 10 µg/mL EtBr after electrophoresis. An epifluorescence microscope was employed to acquire comet images, with an excitation wavelength of 510–560 nm and an emission wavelength of 590 nm. The CASP image analysis software (Kumaravel et al., 2009) was employed to analyze the comet assay parameters, including tail length, tail moment, Olive tail moment, and % tail DNA.
Analysis of Messenger Ribonucleic Acid (mRNA) Expression Pattern of DNA Repair Markers by PCR Array
Radiation-induced DNA damage often leads to cell cycle arrest, allowing for stabilization and repair of the compromised cellular genome. The mRNA expression profiles of DNA repair enzymes in response to GA treatment and/or radiation exposure were analyzed using the RT2 Profiler™ system (SA BioSciences). Total mRNA was extracted using the Qiagen RNeasy Mini Kit, and the purity of the ribonucleic acid (RNA) was assessed using a Nanodrop spectrophotometer. The integrity of the RNA was confirmed through agarose gel electrophoresis. Complementary DNA (cDNA) was synthesized using a cDNA Synthesis Kit, and the expression of DNA repair-specific genes was examined through PCR amplification using the SYBR Green Qiagen kit. The relative expression levels (RQ) of several key DNA repair genes, including BRCA1, GADD45A, RAD21, ATM, ATRX, NBS1, ATR, XRCC6, XRCC6BP, OGG1, ERCC2, DDB1, and XRCC1, were analyzed using an RT2 Profiler™ PCR array. Fold changes in gene expression were calculated from Ct values obtained during the PCR. The fold change data were then used to create a clustergram with the aid of an online heatmap generation tool (
Western Blot Analysis of γ-H2AX and p-ATM
Protein concentrations were determined, and 20 µg of protein was deposited onto a 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel for separation. The Bio-Rad semidry transfer method was employed to transfer the proteins to a polyvinylidene fluoride (PVDF) membrane following electrophoresis. The membrane was subsequently occluded with 5% nonfat milk at 4°C for the night. Immediately after blocking, the membrane was incubated with primary antibodies against p-ATM (1:1,000), γ-H2AX (1:400), and β-actin (1:800) at 37°C for 2 h. The membrane was subsequently incubated at room temperature (37°C) for 1 h with horseradish peroxidase (HRP)-conjugated secondary antibodies (1:2,000). The Bio-Rad chemiluminescence substrate reagent was employed to detect the protein bands. Using ImageJ software, the band intensity was quantified, and the protein expression levels were normalized to β-actin (Johnson, 2013).
Statistical Analysis
Statistical analysis was performed using Statistical Package for the Social Sciences (SPSS) software, and the data were presented as Mean ± Standard Deviation (SD), unless otherwise noted. Differences between experimental groups were assessed using one-way analysis of variance (ANOVA) followed by Duncan’s Multiple Range Test (DMRT). A p value of less than 0.05 was considered statistically significant between the groups.
Results
Effect of GA on Gamma Radiation-induced DC Frequencies
Significant dose-dependent increases in the frequency of DCs were observed following gamma radiation exposure, with the highest number of DCs detected at 3 Gy irradiation (Figure 1). In contrast, pretreatment with GA effectively reduced the formation of DCs induced by gamma radiation in a concentration-dependent manner. Even at the lowest concentration of 1 µM, GA pretreatment significantly mitigated DC formation induced by 1 Gy radiation. The highest GA concentration (10 µM) showed a marked reduction in DC formation in HBPLs exposed to 3 and 4 Gy of radiation.
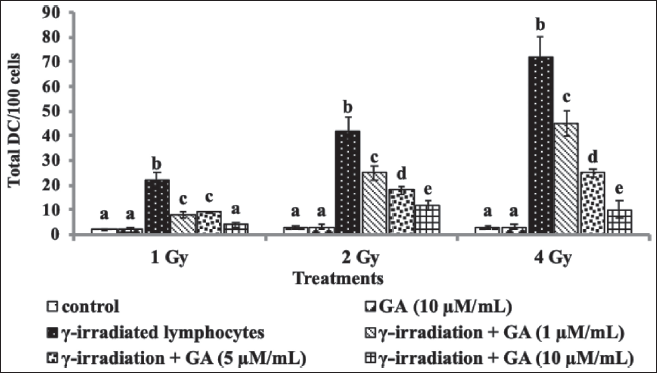
Effect of GA on Gamma Radiation-induced MN Frequencies
Pretreatment with GA significantly reduced the frequency of CBMN in a concentration-dependent manner (Figure 2). The highest concentration of GA (10 µM) was particularly effective in preventing the increase in CBMN frequencies induced by gamma radiation, compared with the lower concentrations (1 and 5 µM). Additionally, treatment with GA alone did not result in any significant increase in DC or MN frequencies in HBPL, indicating that GA does not exhibit toxicity at the concentrations used for radioprotection.
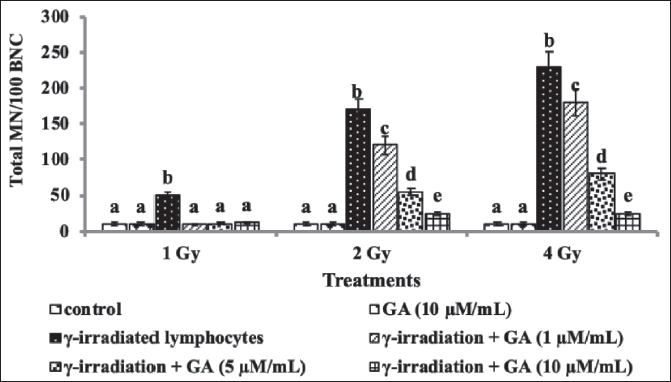
Effect of GA on Radiation-induced ROS Generation in HBPL
To evaluate the protective effect of GA against radiation-induced oxidative damage, we measured intracellular ROS generation by analyzing the two-electron oxidation of DCFH to the fluorescent product DCF under different treatment conditions. Our results showed that irradiated HPBLs exhibited significantly higher ROS levels compared with the unirradiated sham-control. However, pretreatment with GA (10 µM) for 1 h prior to 3 Gy gamma irradiation effectively prevented the increase in intracellular ROS levels in HPBL. The ROS levels in cells treated with GA alone were similar to those of the sham control group (Figure 3).
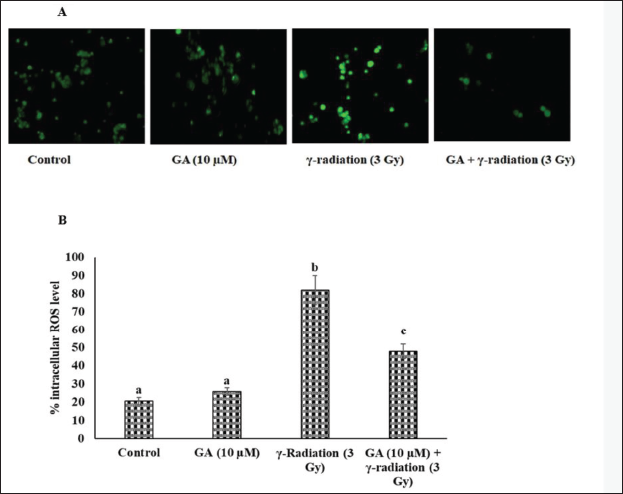
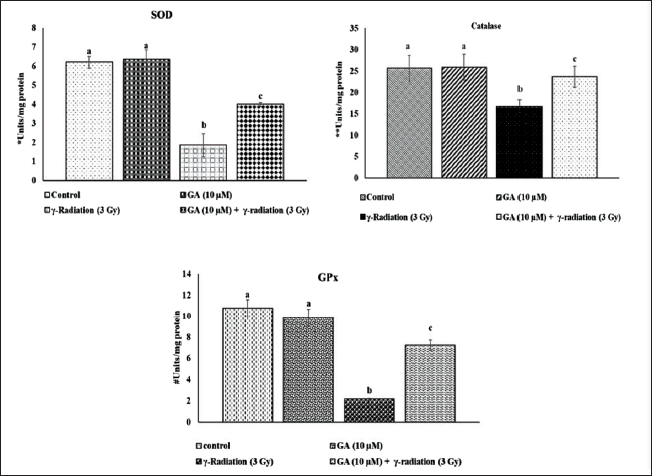
Effect of GA on Radiation-induced Antioxidants Depletion in HBPL
Antioxidant enzymes play a crucial role in protecting cells from radiation-induced damage. Figure 4 illustrates the activities of key antioxidant enzymes (SOD, catalase, and GPx) in HPBLs following GA and/or radiation treatment. The results revealed a significant decrease in the activities of these enzymes in HPBL exposed to 3 Gy radiation. However, pretreatment with 10 µM GA effectively prevented the radiation-induced reduction in SOD, catalase, and GPx activities. No significant changes in antioxidant enzyme activity were observed in the GA-alone treated control group.
Effect of Galangin (GA) on γ-Irradiation (3 Gy) Mediated Enzymatic Antioxidants Depletion in Human Blood Peripheral Lymphocyte (HBPL). The Activities of Superoxide Dismutase (SOD), Catalase and Glutathione Peroxidase (GPx) were Measured by Spectrophotometer and the Values are Expressed as Mean ± Standard Deviation (SD). (n = 6). The Enzyme Activity were Expressed as Units/mg Protein. For SOD *U: the Amount of Enzyme Inhibiting 50% Nitroblue Tetrazolium (NBT) Reduction/min. For Catalase **U: µmol of Hydrogen Peroxide Consumed/min. For GPx #U: µg of Reduced Glutathione Consumed/min.
Effect of GA on Radiation-induced DNA DSBs in HBPL
To further investigate the protective effect of GA against radiation-induced DNA damage, we performed a neutral comet assay to assess DNA DSBs. The comet parameters, including % tail DNA, tail length, tail moment, and Olive tail moment, were evaluated in HPBL. Irradiated HPBL exhibited increased comet parameters, indicating a higher level of DNA damage. However, GA pretreatment (10 µM) for 1 h before exposure to 3 Gy gamma radiation significantly reduced these comet parameters, indicating protection from DSBs. The DNA damage levels in GA-treated cells were similar to those observed in the sham control group (Figure 5).
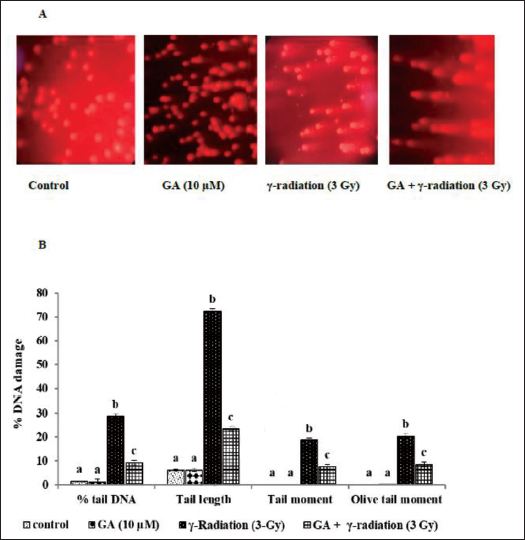
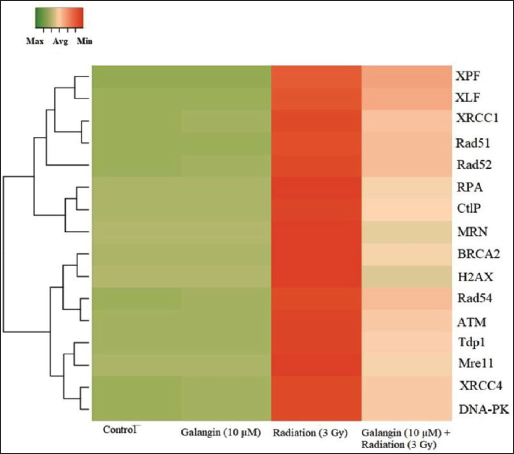
Effect of GA on Radiation-induced DNA Repair Related Gene Expression Pattern
The mRNA expression of DNA repair proteins, such as damaged DNA binding proteins, DSB repair proteins, and base excision repair enzymes (e.g., H2AX, ATM, MRN, Mre11, CtlP, RPA, Rad51, Rad52, Rad54, BRCA2, Tdp1, DNA-PK, XRCC4, XRCC1, XLF, and XPF), were significantly increased in HPBL cells that were exposed to 3 Gy radiation. Nevertheless, the overexpression of these DNA repair-related genes was normalized by pretreatment with GA. The GA-alone treatment group did not exhibit any changes in gene expression (Figure 6).
The Messenger Ribonucleic Acid (mRNA) Expression Levels of Deoxyribonucleic Acid (DNA) Repair Related mRNA Expression Pattern in Galangin (GA) and/Radiation Treated Human Blood Peripheral Lymphocyte (HBPL) by Custom Designed Reverse Transcription Polymerase Chain Reaction (RT-PCR) Array. The mRNA Expression Pattern of Genes Involved in Double-strand Breaks (DSBs) Recognition, Processing, and Ligation were Analyzed. The Total mRNA was Isolated from the Experimental Cells Analyzed as per the Manufacturer’s Instructions. The Clustergram was Constructed Based on Fold Changes of Three Independent Experiments Using Online Software Tool www.heatmapper.ca .
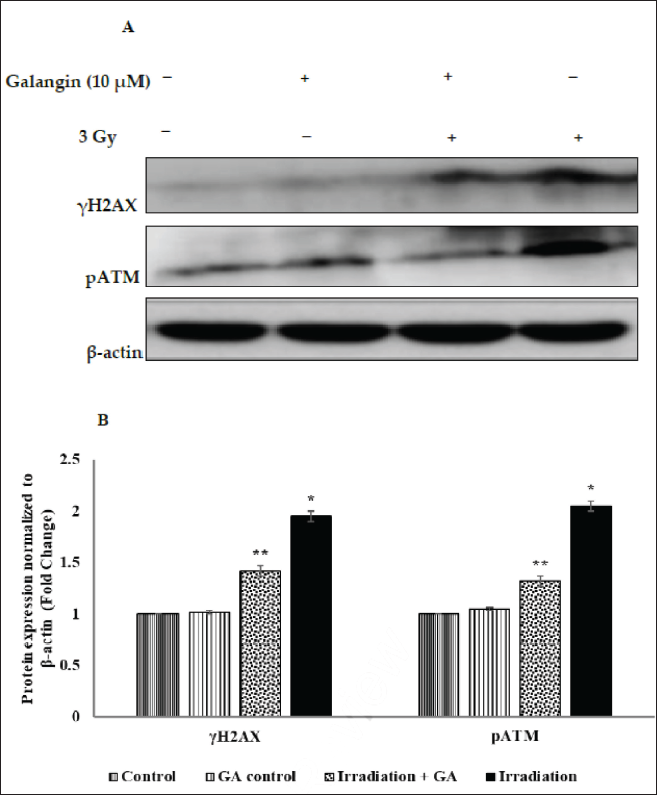
Effect of GA on Radiation-induced Activation of γ-H2AX and p-ATM Activation
In order to evaluate the influence of GA on the activation of critical DNA damage response markers, we conducted a Western blot analysis of γ-H2AX and p-ATM expression. In comparison to nonirradiated HPBL, gamma radiation exposure resulted in an elevated expression of γ-H2AX and p-ATM. Nevertheless, the radiation-induced overexpression of phosphorylated γ-H2AX and p-ATM in HPBL was significantly reduced by GA pretreatment. The overexpression of γ-H2AX or p-ATM was not induced by GA treatment alone (Figure 7).
Discussion
Ionizing radiation is a commonly employed treatment for a variety of cancer subtypes; however, it can also have an impact on the rapid division of normal human cells during radiotherapy. The effectiveness of radiation therapy is contingent upon the ability to maximize the death of cancer cells while simultaneously minimizing the injury to normal tissues. This underscores the necessity of creating radioprotectors that are effective in protecting normal cells during treatment. The present investigation shows that GA has a protective effect on gamma radiation-induced DNA damage and CAs in HPBLs. The gold standard for evaluating the effects of ionizing radiation in HPBL is DC analysis (Bakkiam et al., 2015). We observed a dose-dependent increase in the frequency of DCs in HPBL that had been exposed to gamma radiation (1–3 Gy) in our study. In a concentration-dependent manner, we discovered that radiation-induced DC formation was substantially reduced by GA pretreatment. Numerous studies have demonstrated that antioxidant phytochemicals, including GA, can scavenge free radicals produced by radiation and prevent CAs that may result. In V79 cells, GA has been identified as a potent antioxidant with antimutagenic properties (Bacanlı et al., 2017). Additionally, in vitro and in vivo studies have shown that GA has an anti-clastogenic effect, which reduces CAs in mouse spleen lymphocytes (Heo et al., 1994).
MN are produced as a result of radiation exposure from AFs or damaged whole chromosomes. MN frequencies can function as an efficient biomarker for the evaluation of radiation-induced injury in cultured HPBL. In our investigation, we found that the formation of radiation-induced MNs was prevented by GA pretreatment in a concentration-dependent manner (1–3 Gy). MN formation induced by mutagens such as N-methyl-N′-nitro-N-nitrosoguanidine (MNNG) and bleomycin has been demonstrated to be reduced by GA and other flavonoids in previous research (Heo et al., 1993). Additionally, GA has been demonstrated to inhibit the clastogenic effects of N-methyl-N-nitrosourea (MNU)-induced MN formation in murine bone marrow cells (Heo et al., 2001). The antioxidant and free radical scavenging properties of GA are dependent on the structure, which comprises a C-2–C-3 double bond and a C-3 hydroxyl group (Thangaiyan et al., 2020).
The antigenotoxic activity of GA against gamma radiation may be attributed to its capacity to scavenge free radicals, which are crucial in the formation of DCs and MNs. Consequently, compounds such as GA, which possess antioxidant properties, may mitigate CAs and safeguard against radiation-induced oxidative damage. Furthermore, we observed that the generation of ROS in HPBL was efficiently prevented by the 10 µM GA treatment, which was induced by 3 Gy gamma radiation. This may elucidate GA’s anticlastogenic properties, as its antioxidant activity likely reduces the free radicals that cause DNA damage and CAs. The free radical scavenging effects of GA have been extensively documented in a variety of experimental models (Aloud et al., 2018). In addition, GA treatment has been demonstrated to enhance cellular antioxidant status and prevent oxidative damage in diabetic rats (Zeng et al., 2019), which further substantiates its potential as a radioprotective agent.
Radiation exposure leads to the generation of ROS such as superoxide (O2·−), hydroxyl radicals (HO·), hydrogen radicals (H·), and H2O2 due to the dissociation of water molecules within the cell (Islamian & Mehrali, 2015). These ROS can disrupt mitochondrial membrane potential, increase endogenous ROS levels, and cause extensive oxidative damage to cellular DNA (Kam & Banati, 2013). In our study, we observed a significant increase in intracellular ROS levels following exposure to 3 Gy of gamma radiation.
Additionally, we detected elevated comet parameters, including % tail DNA, tail length, tail moment, and Olive tail moment in HPBL irradiated with 3 Gy of gamma radiation. These increased comet parameters likely resulted from the elevated number of DNA DSBs, a key indicator of chromosomal damage. DSBs are considered a critical form of DNA damage and activate multiple repair pathways (Ivashkevich et al., 2012). However, pretreatment with GA prior to gamma radiation exposure (3 Gy) significantly reduced the comet parameters, suggesting that GA effectively enhances DNA repair mechanisms in response to radiation-induced DSBs. Similar findings have been reported for apigenin, another flavonoid, which was shown to reduce DNA strand breaks in human blood cells exposed to ionizing radiation (Begum et al., 2012).
Gamma H2AX plays a critical role in the DNA damage response following DSBs induced by ionizing radiation (Bártová et al., 2018). Activated ATM is one of the first responders at the damaged DNA site (Ui et al., 2020). The MRE11-RAD50-NBS1 (MRN) complex senses the DSBs, which activate ATM and facilitate the phosphorylation of H2AX, leading to the recruitment of nucleases such as Mre11 and CtlP, which process the DNA ends to generate single-stranded DNA (ssDNA) overhangs (Kinner et al., 2008). These ssDNA regions are then bound by RPA, followed by Rad51 binding, which is further aided by Rad52, Rad54, and BRCA2. Additionally, Ku proteins recognize the DNA ends and recruit Tdp1 or DNA-PKcs. Phosphorylated DNA-PKcs activates the DNA ligase IV/XRCC4/XLF complex to mediate the DNA end-ligation process, while gap-filling and removal of overhangs are carried out by the ERCC1/XPF nuclease, ultimately restoring DNA integrity (Moskwa, 2016).
In our study, we observed a significant upregulation of mRNA expression for DNA repair-related proteins in 3 Gy gamma-irradiated HPBLs. Furthermore, Western blot analysis revealed increased levels of gamma H2AX and p-ATM in irradiated cells. However, pretreatment with GA significantly reduced the radiation-induced expression of these DNA damage response proteins, suggesting that GA enhances DNA repair mechanisms. Similarly, apple flavonoids have been reported to promote DNA repair by modulating H2AX expression in human bronchial epithelial cells (George & Vasantha Rupasinghe, 2017). Studies also suggest that phytochemicals can protect DNA either through their antioxidant properties or by modulating DNA repair processes (Guarnieri et al., 2008). Given that GA is recognized as a powerful antioxidant (Bacanli et al., 2018), we propose that its radioprotective mechanism is likely due to its ability to enhance the DNA repair process, primarily through its antioxidant action.
Conclusion
This study demonstrates that GA provides significant radioprotective effects in HBPLs. The protective mechanism is likely due to GA’s ability to prevent DNA damage, particularly DSBs, and subsequent CAs, which is attributed to its antioxidant and free radical scavenging properties. Importantly, GA treatment did not induce any DNA damage at the concentrations effective for radioprotection. These findings suggest that GA could be a promising natural radioprotective agent for shielding normal tissues from the harmful effects of gamma radiation.
Summary
In this study, we assessed the protective effects of GA against gamma radiation-induced DNA damage, specifically focusing on DSBs, DC formation, and CBMN formation in HBPLs. Gamma radiation exposure significantly increased the frequency of DC and CBMN in a dose-dependent manner. However, pretreatment with GA (at concentrations of 1, 5, and 10 µM) effectively reduced these frequencies in a concentration-dependent manner. Additionally, GA pretreatment before radiation exposure significantly reduced the levels of radiation-induced DSBs in HBPL compared to radiation-only treated cells. These results suggest that GA protects against radiation-induced ROS generation, DNA damage, and CAs. Notably, GA (10 µM) pretreatment also inhibited the overexpression of γ-H2AX and p-ATM in response to radiation (3 Gy), indicating that GA exerts an anticlastogenic effect by modulating the DNA repair mechanisms related to radiation-induced DSBs in HBPL.
Abbreviations
CBMN: Cytokinesis blocked micronuclei; DC: Dicentric aberrations; DCFH-DA: Dichloro-dihydro-fluorescein diacetate; GA: Galangin; HBPLs: Human blood peripheral lymphocytes; ROS: Reactive oxygen species.
Footnotes
Author’s Contribution
HZ: Conception and design, drafting the manuscript, and review key revision. YX: Data acquisition, analysis, and approval publishing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent Statement
The ethical was approved by the Xián Fengcheng Hospital, Xián, China.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
