Abstract
The detrimental effect of nuclear accidents due to localized or whole body radiation exposure results in severe cellular damage. The current study was carried out to evaluate radiation-mediated variability in blood components of metal scrap workers exposed accidently to cobalt-60 source. Blood samples collected initially from five hospitalized patients, coded P1–P5, were processed for total leukocyte counts (TLC), platelet (PLT) counts, haemoglobin, estimation of DNA double strand breaks by measuring phosphorylated form of H2AX (γ-H2AX) and chromosomal aberrations (dicentrics). Blood cells count (TLC), in all the patients except P2, was found decreased. Dicentrics increased in all the five patients. γ-H2AX was found significantly elevated in patients P2 and P4. After 3 days, 21 subjects working in close vicinity of accident site were evaluated for the above-mentioned markers to confirm their possibility of radiation exposure; however, all the parameters in these subjects were found within normal limits. Blood from patients P1–P5 was collected again after 11 days. Studies revealed exorbitant increase in γ-H2AX in lymphocytes and monocytes of patients P1, P4 and P5. TLC and PLT count in these patients had fallen further. Dicentrics declined with time in all the five patients. Based on the studied blood biomarkers, we conclude that the five subjects showed signs of radiation exposure. Measurement on radiation dose could not be performed in the current study; however, the generated data particularly on dicentrics provide ample evidence of radiation exposure.
Introduction
Nuclear accidents and their catastrophic effects are widely reported in the literature.1,2 To minimise the loss of lives and infrastructure, there is an urgent need to have preparedness at local, state, national and international levels. Medical preparedness to handle such incidences is the most critical challenge. 3 Biodosimetry is considered as an essential tool before precise and specific medical intervention.4,5 Besides, extended psychosocial support to minimize the distress specifically to those who were not in risk zone, biodosimetry significantly reduces burden on health-care system. 6
Skin burns, bone marrow stem cells depletion, gastrointestinal damage and other organ failure leading to cumulative effects may result in long-term implications of radiation exposure. 7 Acute radiation syndrome (ARS) has been categorized in prodromal, latent, manifest and recovery/death depending on dose, dose rate, exposed area and type of radiation activity (α, β, γ and neutron). Before initiating treatment, data regarding physical dosimetry, biodosimetry, clinical symptoms, lymphocyte depletion kinetics and assays indicative of DNA damage are considered imperative. 8 This approach helps in assessing patient’s risk of prognosis, probabilities of developing ARS and urgency for hematopoietic and gastrointestinal supportive therapies. 9 , 10
Blood cells count is considered as a sensitive biomarker, but larger individual variation and less informative in case of low and partial body exposures make this marker less effective 11 in assessing radiation dose. Cytogenetic studies (dicentrics) from peripheral lymphocytes are reported to correlate well with the dose of radiation and are considered as gold standard for the measurement of radiation exposures. 12 , 13 The International Organization for Standardization (ISO) has recognized dicentrics assay as an international standard biodosimeter in 2004. This test, however, has its limitation during mass casualty screening and in severe medical condition due to complex methodology and time required for evaluation of slides. 14 Increase in radiation-induced H2AX phosphorylation, the marker for double strand break (DSB) formation, has recently been considered for radiation absorbed dose estimation, although with a limitation of maximum intensity within 24 h of sampling. 15 , 16 Alexander et al. have reviewed strategies for biodosimetry and emphasized on the need of multi-parametric approach for radiation dose assessment, broadly by personal and area monitoring, clinical signs and symptoms, blood cells count, time of onset of nausea and vomiting, cytogenetic assay, impaired cognition and neurological deficit and other emerging biodosimetry techniques. 17
The present study provides data on blood parameters, dicentrics, γ-H2AX and appearance of clinical symptoms in five subjects accidentally exposed to radiation. Delay in awareness about radiation accident led to variations in frequency of vital biomarkers due to which the radiation dose could not be estimated.
Methodology
News related to accidental radiation exposure at Mayapuri, Delhi, India, broke out on 14 April 2010 after five patients admitted in a hospital showed symptoms related to radiation sickness. After thorough investigations, it was reported that a cobalt container (purchased almost three decades ago for academic purpose) was sold to a scrap dealer who broke it open on 12 March 2010. Initially blood samples were collected from five hospitalized persons on 14 April 2010 (30 days after the opening of radiation source container) for confirmation of radiation exposure through various biodosimetry assays. All the five patients were working in the close vicinity of the radiation source and patient P4 was actively involved in dismantling the cobalt container. Information collected from the hospital where all the five patients were admitted was not suggestive of any chronic disease; however, they were from lower economic strata. The symptoms reported after initial clinical observations were indicative of radiation exposure. 3 days after, blood samples were collected from 21 persons, who either had their business in the close vicinity of affected area or deputed temporarily for security reasons. All the studied subjects were males in the age group of 22–60 years.
Clinical signs and symptoms
All the five patients hospitalized initially and 21 persons reported later were reviewed by the clinicians for appearance of radiation-related symptoms, such as, skin desquamation nausea-vomiting-diarrhea (NVD syndrome), weakness and haematological parameters. Performa with detailed clinical history was filled in each case.
Haematological parameters
Whole blood (2 ml) samples were collected from age- and sex-matched controls and five radiation victims along with 21 more subjects working in the close vicinity of radiation exposure. Samples were taken in heparinized BD vacutainers (BD, Franklin Lakes, NJ, USA) of 4 ml capacity. Written consent was collected from all the control individuals. Counting of white blood cells was performed on fully automated blood cell counter (Beckman Coulter, LH 500, California, USA). Simultaneously, for reconfirmation, the cells were also counted manually by spreading blood mixed with Turk’s fluid (2% acetic acid and 2–3 drops of methylene blue) on Neubauer chamber by following the standard protocol. Leukocyte counts were expressed as cell number per cubic millimetre of blood. Platelets (PLT) were counted on the same blood cell counter as mentioned above and values were expressed as cell number per cubic millimetre of blood. Haemoglobin (Hb), expressed as gram per decilitre of blood, was also measured on fully automated blood cell counter.
Cytogenetic assay
Whole blood collected from controls (five samples) and radiation victims (five samples) was cultured in replicates using the technique described by Moorhead et al. 18 Heparinized blood (0.3 ml) was added to sterile disposable plastic vessels containing 5 ml of prepared culture medium (4ml RPMI 1640,1ml heat inactivated fetal calf serum, antibiotics 1000U/ml of penicillin and 100μg/ml of streptomycin, 2% Phytohaemagglutinin M). After 46 h of incubation at 37°C, colchicine (100 μl of 25 μg/ml/culture) was added in each vessel for 2 h before harvesting. On termination, cultures were centrifuged at 1200 r/min for 5–7 min. Supernatant was removed and replaced with hypotonic solution (0.075 M potassium chloride), and after incubation for 30 min at 37°C, the solution was centrifuged and the pellet was resuspended in a freshly prepared fixative (3:1, methanol: acetic acid). Cells were washed thrice with the fixative. Slides were prepared by air drying method. Dried slides were stained with 2% Giemsa stain diluted with phosphate buffer (pH 6.8) for 5 min. Washed, dried and mounted slides were observed under 1000× oil immersion (Olympus, BX 50, Model BX50F4, Olympus Optical co. LTD, Japan) for well-spread metaphase plates (minimum 50 per case or depending on availability).
Measurement of γ-H2AX fluorescence intensity
γ-H2AX fluorescence intensity was measured in the blood cells (lymphocytes and monocytes) according to the method of Chow et al. 19 with some modifications. Blood samples from radiation exposed patients and from age- and sex-matched healthy human volunteers were collected in heparinized vacutainers and stored at 4°C till further experimentation. Briefly, 1 ml of blood was fixed with 10% formaldehyde at room temperature for 10 min followed by 30 min incubation in 0.1% Triton X-100 for permeabilization at the same temperature. After centrifugation, 50% methanol was added to the pellet and the cells were washed once with buffer (4% foetal bovine serum (FBS) in phosphate buffered saline (PBS)). Reaction mixture was incubated overnight with the primary antibody anti-phospho-histone H2AX, Ser 139 (1:50 dillution in 4% FBS in PBS, Upstate cat. No. 05-636, NY, USA). Buffer-washed cells were further incubated for 1 h with secondary goat anti-mouse IgG (H + L) fluorescein isothiocynate (FITC)-conjugated (1: 400 dilution) antibody (Millipore cat.no. 12-506, EMD Millipore corporation, Temecula, CA, USA). Cells obtained after centrifugation for 10 min were washed twice with buffer and analyzed using a flow cytometer (Becton Dickinson Bioscience, USA, FACS CALIBUR 3CB, Becton Dickinson Biosciences, USA). Radiation dose response measurement was also generated from blood samples collected in heparinized vacutainers from three healthy human volunteers. For this study, 1 ml whole blood was exposed individually to 5 and 10 Gy in gamma irradiator (Cobalt Tele therapy, Bhabhatron II, Panacea Medical Technologies Pvt. LTD, Bangalore, India) at the dose rate of 2.56 Gy/min. After 60 min, the exposed blood samples were processed as per the earlier described protocol for γ-H2AX fluorescence intensity measurement.
Reports generated based on multi-parametric approach were regularly communicated to the concerned physicians for treatment planning. After 40 days of hospitalization, four of the five patients admitted were successfully treated and discharged. Depending on the need, patients were put on cytokine (granulocyte-macrophage colony-stimulating factor) therapy in addition to other symptomatic treatments. Patient P4 unfortunately succumbed to multi-organ failure after 12 days of hospitalization.
Statistical analyses
Analysis of variance (ANOVA) was applied to evaluate the statistical significance in Hb, PLT and total leukocytes counts (TLC) of controls. The analysis was carried out on patients on day 30 and repeated on day 40. Paired t test was used to compare γ-H2AX fluorescence intensity in lymphocytes and monocytes of controls and patients.
Fischer’s exact test was applied to compare dicentrics in controls versus patients studied on day 30. Comparison in dicentric data obtained from patients on days 30 and 40 was also done using the same test (Fischer’s exact test). A value of p < 0.05 is considered as statistically significant.
Results
Report on hospitalized patients (P1–P5)
Clinical signs and symptoms from all the five patients were recorded at the time of hospitalization. To ascertain the extent of radiation exposure, blood samples from these patients were collected, initially and after a gap of 10 days to analyze the haematological parameters, frequency of dicentrics and DNA DSBs in the form of γ-H2AX fluorescence intensity.
Clinical signs and symptoms
Signs of NVD syndrome were not reported in any of the patient. Dark blue patches on hands, legs and nails of these individuals were observed on the very first day of hospitalisation however, faded gradually with passage of time. All the five patients had complained of severe fatigue and anorexia. They were given symptomatic treatment and transferred next day to a super speciality hospital. During repeat study after 10 days, of the five patients studied, three (P1, P4 and P5) were suggestive of bone marrow suppression.
Haematological parameters
On day 1 of the study, TLC and PLT counts in patients P1, P3 and P5 were in the range of 3100–3300 per mm3 and 33,000–91,000 per mm3, respectively. In patient P2, TLC was 5600 per mm3 and PLT count was 43,000 per mm3 (Figure 1). P4 had shown severely decreased TLC (500 per mm3) and PLT counts (19,000 per mm3). All the above five patients were working in the close vicinity of cobalt-60 source; however, patient P4 was actively engaged in dismantling the container containing cobalt-60 source.

TLC, PLT counts and Hb content in the blood of unexposed and patients (P1–P5) exposed accidentally to cobalt-60 at Mayapuri, Delhi, India. Blood sample (2 ml) from all the subjects was collected in heparinized BD vacutainers on 30th and 40th day of radiation accident. Samples were analyzed using Beckman Coulter automatic blood cell counter. Data are expressed individually for all the patients. In controls, bars are indicating mean ± SD of blood samples collected from five healthy individuals. TLC: total leukocyte counts; PLT: platelet; Hb: haemoglobin.
Blood samples analyzed after 10 days of initial report showed further depletion in total blood cell counts (Figure 1). In patient P1, TLC depleted to 820 per mm3 from 3300 per mm3 analyzed during initial studies. TLC had fallen in P4 from 500 per mm3 during the first report to 130 per /mm3 at the time of repeat sampling. In patient P5, TLC had depleted to 1400 per mm3 from 3100 per mm3. Patients P2 and P3 also showed significantly reduced TLC but fall was not so severe. PLT count also depleted notably in all the five patients. The fall deepened further in the second study. In patient P4, the PLT count (1700 per mm3) was minimum.
During initial sampling (Figure 1), Hb in patients P1–P3 and P5 was in the range of 11.5–14.0 g/dl, while in P4 it was 8.4 g/dl. Trend in Hb fall at the time of second study (after 10 days) was almost parallel to TLC depletion pattern (P1: 9.3; P2: 10.2; P3: 11.9; P4: 6.1; and P5: 9.6 g/dl).
Figure 2 depicts the mean ± SD of the data collected on PLT, TLC and Hb from five patients exposed to radiation and equal number of healthy controls. ANOVA applied for TLC and PLT in controls and patients studied on day 30 and repeated on day 40 have demonstrated the difference as significance (p < 0.05). The Hb content was also significantly less (p < 0.05) in patients observed on day 30 and day 40 of exposure when compared with the controls. Statistical significance using ANOVA test on day 30 versus day 40 post exposure patients have shown p < 0.05 in TLC, PLT and Hb content.

Mean ± SD of Hb, TLC, PLTs, dicentrics and γ-H2AX fluorescence intensity in blood collected from cobalt-60 radiation-exposed individuals (days 30 and 40 post exposure) and age and sex matched controls. ANOVA has been applied for statistical comparison in PLT, TLC and Hb in between control and radiation-exposed groups. Paired t test has been applied for statistical comparison in γ-H2AX fluorescence intensity in between control and radiation-exposed groups. Fischer’s exact test has been applied to compare number of dicentrics in different groups.*p < 0.05 considered as statistically significant; @controls versus 30th day post exposure; §controls versus 40th day post exposure; †30th versus 40th day post exposure; Ωdicentrics not seen. ANOVA: analysis of variance; PLT: platelet; TLC: total leukocyte counts; Hb: haemoglobin.
Chromosomal aberrations
Analysis of chromosomal aberrations in blood samples of the patients exposed to radiation revealed significant increase in the number of dicentrics. Presence of scorable metaphases was found low in all the five patients. In patients P1, P2, P4 and P5 out of 35, 30, 43 and 57 metaphase plates studied, respectively, two dicentrics were found in each case. In patient P3, there was one dicentric out of 62 plates scored (Table 1). No ring chromosome was seen in any of the patients during the current study. Figure 3 represents the metaphase plates from (a) control and (b) radiation-exposed blood samples indicating the presence of dicentric.

Measurements of γ-H2AX fluorescence intensity in lymphocytes and monocytes and dicentric assay in blood collected from healthy controls and from five persons exposed accidentally to cobalt-60. For γ-H2AX study blood (3 ml) was collected in heparinized BD vacutainers. γ-H2AX fluorescence intensity was measured on a flow cytometer using primary antibody anti-phospho-histone H2AX, pSer-139 and FITC-conjugated secondary goat anti-mouse IgG (H + L). Data are expressed individually for all the five patients. In controls, observations are represented as mean ± SD. Dicentrics were counted in metaphase plates obtained after culturing the blood for 48 h using standard techniques. Ω = 0 dicentrics; Φ = during second analysis (40th day post exposure) γ-H2AX fluorescence intensity and dicentrics could not be detected in patient P4 due to non-availability of required numbers of lymphocytes.
Dicentrics in lymphocytes of persons accidentally exposed to cobalt-60 source at Mayapuri, Delhi, India.

aWhite blood cell counts in patient P4 was too low. Not even a single metaphase could be located and the culture was full of debris. Patient P4 died the next day of blood collection due to multiple organ failure. Data representation from five individuals exposed to radiation source.
Cytogenetic studies carried out again after 10 days gap showed fall in dicentrics yield (Table 1) in all the four patients (P1–P3 and P5). In patient P4, culture processed for cytogenetic studies was found full of debris (Table 1) and not even a single metaphase plate could be located for the study. This may be because of severely depleted (TLC: 130/mm3) lymphocytes due to their extensive DNA damage, also confirmed by flow cytometric observations. Unfortunately, this patient died the next day of sample collection. Fischer’s exact test applied to compare the mean values of dicentrics in controls versus patients studied on day 30 and patients studied on day 30 versus day 40 post exposure (Table 1, Figure 2) has demonstrated the difference as statistically significant (p < 0.05).
Measurement of γ-H2AX fluorescence intensity
Flow cytometric measurement of γ-H2AX level in peripheral blood cells (lymphocytes and monocytes) of all the five patients demonstrated increase in γ-H2AX fluorescence intensity when compared with age and sex matched controls (Figure 4). Mean fluorescence intensity of γ-H2AX in lymphocytes was found 1.3-fold elevated in patients P2 and P4 (Figure 5(a)). γ-H2AX in monocytes was also found enhanced in all the cases except patient P1, where it was similar to the controls. In patient P4, monocytes have shown 4.5-fold increase in γ-H2AX and this was the maximum in comparison with all the other cases studied (Figure 5(b)).

Metaphase from unexposed and radiation exposed human blood samples. Blood collected in heparinized BD vacutainers was cultured in replicates. Colchicine was administered 2 h before the termination of culture (48 h). Giemsa-stained slides were scored at 1000X (oil immersion) for chromosomal aberrations. (a) Metaphase from the blood of unexposed human subject. (b) Metaphase with chromosomal aberrations: dicentric chromosome (d) from the person exposed to cobalt-60.
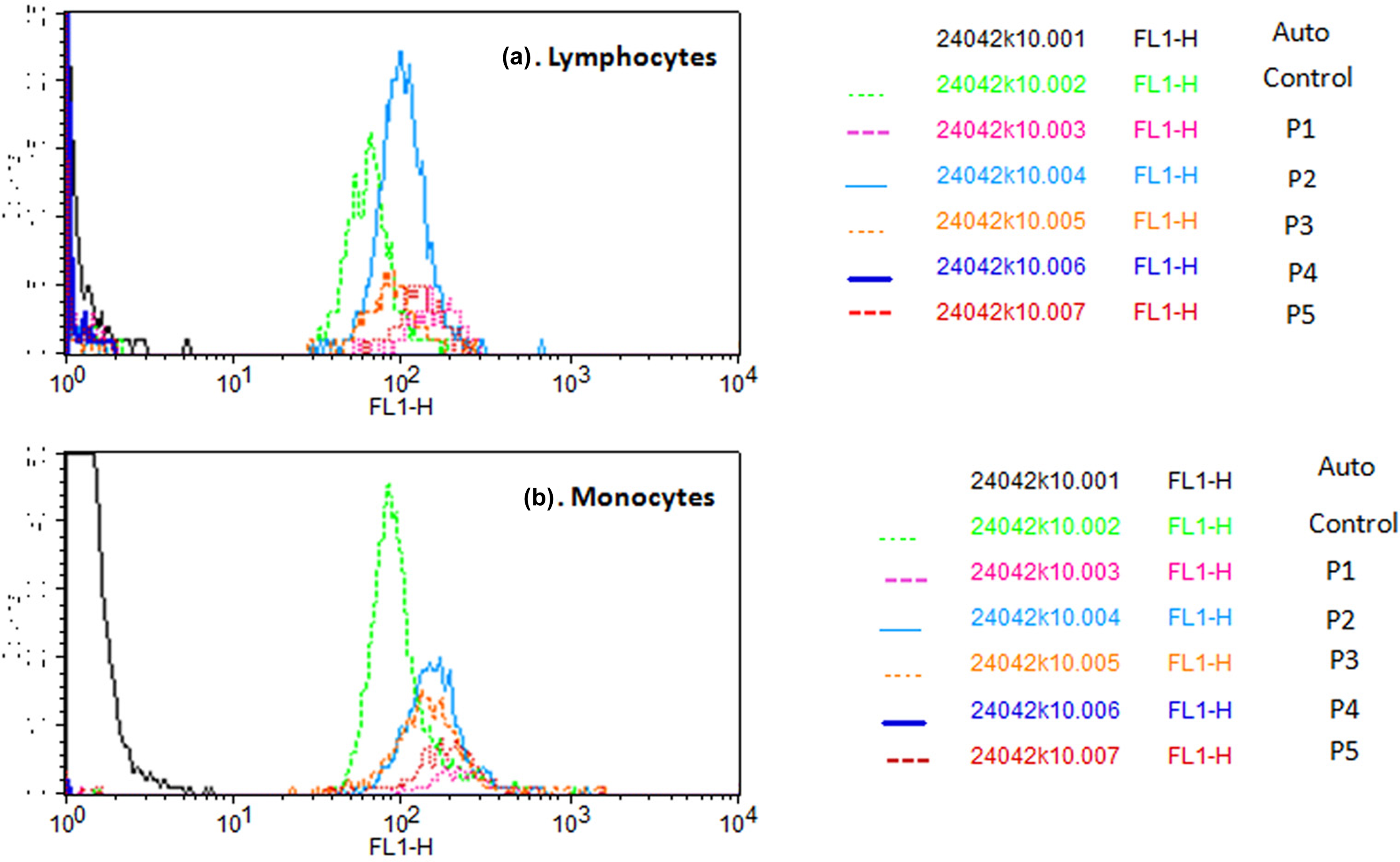
Overlay of flow analysis of γ-H2AX fluorescence intensity in (a) lymphocytes and (b) monocytes of blood collected from unexposed controls and five patients exposed accidentally to cobalt-60. Blood was collected in heparinized vacutainers on 30th and 40th day of radiation exposure. Samples fixed with 10% formaldehyde and permeabilized with 0.1% Triton X-100 were centrifuged at 1200 r/min to remove red blood cells. The separated leukocytes incubated with primary anti-mouse γH2AX and secondary goat anti-mouse IgG antibodies were analyzed using flow cytometer. In patient P4, the fluorescent intensity in lymphocytes could not be detected as majority of these cells were found dead.
Both lymphocytes and monocytes, after 10 days of initial sampling, had shown further increase in DNA DSBs (enhanced γ-H2AX signal) in most of the patients with respect to the controls (Figures 4 and 5(a) and (b)). Maximum γ-H2AX fluorescence was obtained in patient P1, while in patient P4, FITC intensity was not detectable as most of the cells in this sample were found dead (Figure 6(a) and (b)). Patient P1 expressed 90% damage in DNA of both cell lineages. In patient P5, nearly 70–80% of lymphocytes and monocytes showed DNA DSBs. However, in the remaining two cases (P2 and P3) the percentage of DNA DSBs in both lymphocytes and monocytes was found less enhanced in comparison with other three patients (Figure 4). Difference in γ-H2AX values in controls versus patients at both the time intervals, using paired t test, was found statistically significant (p < 0.05) in monocytes (Figure 2). In case of lymphocytes, the p value was not significant when controls data was compared with all the five patients (P1–P5). However, after excluding patient P4, where the cell number was extremely low, the difference (paired t test) between the patients studied on days 30 and 40 was significant (p < 0.05) in both monocytes and lymphocytes (Figure 2).

Flow image demonstrating forward and side scattering of lymphocytes and monocytes population in (a) control and (b) patient P4 indicating the majority of lymphocytes and monocytes as dead cells in this patient.
Flow cytometric data, generated on five healthy human blood samples exposed to two different doses (5 and 10 Gy) of ionizing radiation, indicated dose-dependent increase in γ-H2AX level in both the cell lineages, that is, lymphocytes and monocytes (Figure 7(a–c)).
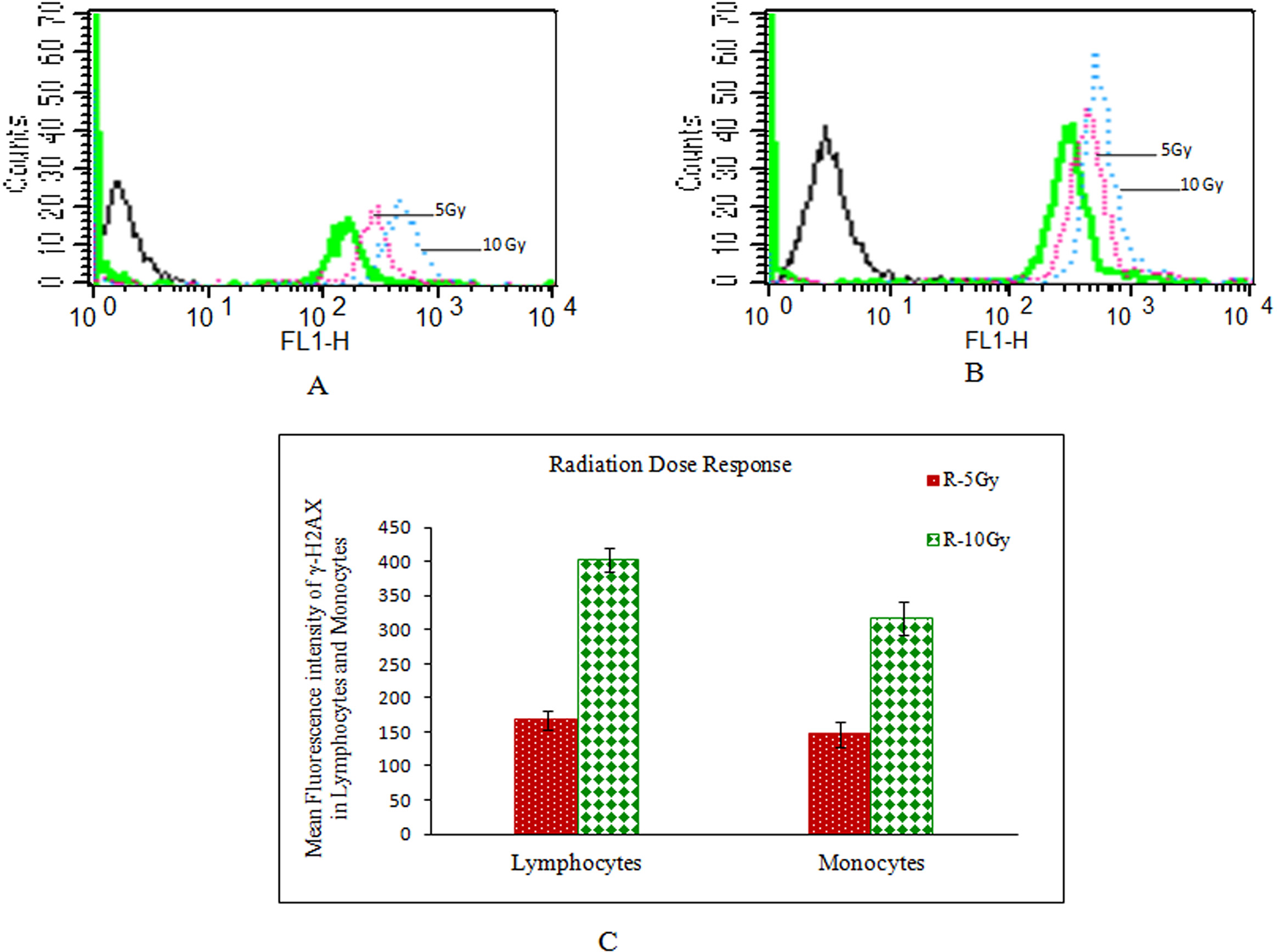
Radiation dose response of γ-H2AX in blood collected from three age and sex matched unexposed subjects. (a) Flow overlay of fluorescence intensity in lymphocytes, (b) monocytes, (c) mean fluorescent intensity of γ-H2AX in blood samples of three subjects exposed to 5 and 10 Gy. For this study, 4 ml of whole blood collected in heparin vacutainers was exposed to 5 and 10 Gy in gamma irradiator at the dose rate of 2.56 Gy/min. The exposed blood was processed for γ-H2AX fluorescence intensity measurement.
Study report in a batch of 21 subjects (working in the same area)
All the subjects screened in this group were male and in different age groups ranging 22–60 years. Of the 21 subjects studied, none of them had shown any apparent physical symptom that could be correlated with the direct/indirect exposure to radiation source. Blood parameters, TLC (4800–10,900 per mm3), PLT counts (1.3 × 105–3.7 × 105 per mm3) and Hb content (12.9–15.2 g/dl) in all the studied subjects were in normal range. Metaphase plates (50–70) scored for dicentrics and rings did not indicate any type of chromosomal aberration in any of the subject. Test conducted related to γ-H2AX mean fluorescence intensity in randomly selected persons from the above group did not reflect increase in γ-H2AX in any of the studied cell lineage (lymphocytes and monocytes).
Discussion
Due to enumerable constraints, biological dosimetry is rarely carried out at an initial stage of radiation accidents. Delay in sampling principally leads to decline/disappearance of some of the biomarkers with time, while others get more prominent due to cumulative effects. Delayed biodosimetry has been reported during a similar kind of radiation accident occurred in Istanbul scrap market in December 1998, where more than a month had lapsed in between the incident and its recognition by the authorities. 1 In the current study also, the cobalt container was actually broke open on 12 March 2010 but the news became public about this radiation accident on 12 April 2010 (a month after dismantling the cobalt container), and the blood for biodosimetry was drawn on 14 April 2010. Due to this delay, it was found difficult to collect the information on duration of the individual patient’s stay in the vicinity of radiation source. Exposure to protracted dose and delay in awareness about the exposure had seriously complicated the issue of establishing radiation absorbed dose.
Clinical signs and symptoms, the critical components during clinical management of radiation victims, did not show direct specificity to radiation exposure in the current study. However, the data retrieved from other evaluated biomarkers provided important information in confirming about radiation leakage. Mettler and Voelz reviewed many radiation exposure scenarios and explained various types of radiation exposures, their biological effects, multi-parametric approach, strategies for triage and specificity of radiation exposure symptoms in humans. 20 As reported earlier, we also noted that collective and comprehensive approach namely clinical symptoms, lymphocytes depletion pattern, cytogenetic aberrations and measurement of DNA DSBs by γ-H2AX estimation used in current study (Figure 2) has a great significance. 21 Outcome of these parameters has complemented each other in confirming the exposure of victims to the radiation source.
Onset of diarrhoea and vomiting soon after exposure and to the larger percentage of affected population have shown a linear correlation with radiation absorbed dose. 9 At a lower dose, however, these symptoms either do not appear or get delayed leading to misappropriation of absorbed dose. In the current study, none of the patients had any symptoms of nausea, vomiting and diarrhoea. At the time of hospitalization, they had complaint of anorexia, fatigue, hair fall and desquamation of skin. Non-occurrence of prodromal symptoms at any stage of exposure had suggested that patients may not have been exposed to a high dose of radiation.
Haematological markers are known for not only contributing in dose estimation but also for assessing victims’ health status and the need for clinical intervention. 9,22 Fall in blood cell count (>50%) below normal values (1.3 × 109/l) in less than 12 h is reported as suggestive of radiation over exposure. Appearance of mild cytopenia without significant bone marrow damage indicate absorbed radiation dose less than 2 Gy. 9 In the current study, blood cells count although showed depletion, time taken in fall was longer (more than 40 days). This finding if correlated with non-appearance of prodromal symptoms reaffirms that radiation exposure dose was not very high. Decline in blood cell count in these cases could be probably due to delayed clinical support and cumulative effect of long and fractured radiation dose exposure. Blood cells depletion kinetics, as prescribed for radiation absorbed dose management, could not be successfully done in these cases due to non-availability of blood samples and lack of coordination. As mentioned in various reports, patients from this study were also put on cytokine therapy, although delayed, to extend all the possible support for improved survival. 23
During current investigation, cytogenetic data had revealed increased number of dicentrics in all the five patients admitted in the hospital. Varied number of dicentric yield in addition to non-homogenous depletion in blood parameters in all the five cases reflected that the victims had non-uniform radiation exposure. Subsequent sampling from the same patients after a gap of 10 days revealed expected decline in dicentrics yield during repeat study, confirming loss of biodosimetry information with the time gap. The same phenomenon of reduction in number of dicentrics with time has also been reported earlier. This information confirms that cytogenetic markers are highly informative, provided the study is conducted without losing potential time after radiation exposure.
Exposure to ionizing radiation triggers the phosphorylation of H2AX at serine 139 position and generates γ-H2AX, measured as DNA DSBs. 24 This marker is currently in practice for approximate estimation of radiation exposure during various treatment modalities. However, available literature conveys that phosphorylation starts within few minutes of radiation exposure, reaches maximum between 30 min and 2 h and disappears in 24 h depending upon the system under observation. 25
In our study, blood samples collected from patients after days 30 and 40 of cobalt-60 exposure have clearly indicated increased γ-H2AX fluorescence. The persistence of γ-H2AX, even after the lapse of such a long period, might be the result in induction of complex DNA damage which is more difficult to repair. Additionally, this could also be the result of repeated partial body exposure, where every time limited numbers of blood cells were exposed. There is also a strong possibility of inefficient detection of DNA damage by repair enzymes leading to accumulation of unrepaired DNA. In our 40th day observation, patients P1, P2 and P5 showed enormously increased γ-H2AX, while on 30th day of study, γ-H2AX values in these patients were either parallel to controls or marginally increased (Figures 4 and 5(a) and (b)). This delayed appearance of γ-H2AX also reflects the complexity of the system involved in in vivo conditions. This study also demonstrates that both the blood cell lineages (lymphocytes and monocytes) are significantly sensitive to radiation. Steep rise in γ-H2AX due to cumulative and late effect of radiation maximum up to 7 days has been confirmed earlier also. 26 However, to the best of our knowledge, our study for the first time reports the persistent occurrence of γ-H2AX even after a month of radiation exposure. It has also been conveyed here that though γ-H2AX may not be an efficient biomarker once used singly but it certainly demonstrates the severity of radiation inflicted DNA damage in white blood cells which are highly sensitive to radiation and immensely essential for survival. As explained earlier, for the significance of using multiple-assay approach for radiation exposure assessment, our report confirms that to assess the gravity of radiation exposure, single biomarker evaluation may not be meaningful; however, we find dicentric assay as the most valuable and radiation-specific biomarker. 27,28
Conclusion
Based on data collation and inter-parametric interpretation on dicentrics yield, clinical signs and symptoms, declined blood cell counts, exorbitantly increased γ-H2AX, it has been concluded in the current study that at least five patients (P1–P5, hospitalized) were exposed to ionizing radiation. During this report, we also realized that lack of awareness about management of accidental radiation exposures, infrastructure deficit, suboptimal medical intervention, insufficient command and coordination and specifically delayed information had been some of the major constraints leading to loss of vital information required for dose estimation. However, both multi-parametric approach and inter-parametric correlation have immensely helped us in confirming that the individuals had certainly been exposed to the radiation. Study has also emphasized that dicentrics is still a highly valuable biomarker to confirm radiation exposure. Current report was also a support to the clinicians in designing and reassuring their treatment plan, which was immensely valuable for victims’ health care. This may be a valuable piece of information required in strengthening our preparedness to meet the need for assessment and management of such unfortunate incidence in future.
Footnotes
Acknowledgements
M. L. Gupta, N. N. Srivastava, S. Dutta, S. K. Shukla, A. Dutta, S. Verma and M. Devi are grateful to Director, INMAS for consistent support during this study. Authors also express their gratitude to clinicians at Deen Dayal Upadhya (DDU) Hospital, National Disaster Management Authority (NDMA) and All India Institute of Medical Sciences (AIIMS) for coordination and their expert opinion. Clinical support by Col. P. J. S. Bhalla is duly acknowledged. The assistance rendered by Surabhi Dabral, Farooque Abdullah and Mrs Namita Kalra for flow cytometry measurement is highly appreciated.
Funding
The fund for this work was supported by Defence Research Development and Organisation (DRDO). SD thanks the Council of Scientific and Industrial Research (CSIR) for fund support.
