Abstract
Background
Fracture healing involves multiple growth factors and the interaction between multiple signaling pathways. Pue (puerarin) is a natural compound with multiple biological activities, but its effect on fracture healing and its relationship with the bone morphogenic protein (BMP)-Smad pathway is not yet clear.
Objectives
This study intends to assess the effect of puerarin on fracture healing.
Materials and Methods
Seventy rats were used for the study to establish a tibial shaft fracture rat model. The experimental rats were divided into a tibial fracture group, Pue dose group (low, medium, and high), BMP-2 group, Noggin group, Pue + SY-LB-35 group, and Pue + Noggin group through the random number method. We observed the fracture healing through micro-CT and histology, observed the tibial stump tissue with hematoxylin and eosin (HE) staining, and detected the expression of genes related to the BMP-Smad pathway through quantitative real-time polymerase chain reaction (qPCR) and Western blot.
Results
We found that puerarin can promote tibial shaft fracture healing in rats in a dose-dependent manner; through micro-CT and histological analysis, we observed increased trabecular bone formation during fracture healing in rats treated with puerarin. Callus formation is accelerated, and bone density increases. In addition, puerarin also significantly increased the expression of genes related to the BMP-Smad pathway, including BMP2, Smad1, Smad5, and so on.
Conclusion
Puerarin can promote tibial shaft fracture healing in rats, and its mechanism is related to upregulating the expression of genes related to the BMP-Smad pathway. It further improves the fracture-healing mechanism and provides candidate drugs for the treatment of tibial shaft fracture healing in clinical medicine.
Introduction
Although traditional treatment methods can achieve specific therapeutic effects in tibial shaft fracture treatment in some cases, the effects are sometimes not ideal, and there are problems such as long healing time and many complications (Mick & Fischer, 2022), so it is necessary to find more effective treatments.
Studies believe that the participation of the bone morphogenic protein (BMP)-Smad pathway is involved in fracture healing (Ma et al., 2020). BGN is expressed in bone and skeletal connective tissue (Liu et al., 2021) and regulates bone development (Zhang et al., 2023). It can induce mesenchymal stem cell/stromal cell (MSC) differentiation into osteoblasts and can further recruit substances such as Smad1, regulate gene expression, and promote osteoblast differentiation (Caddy et al., 2020; Fu et al., 2020). The BMP-Smad pathway can promote phosphorylation of Smad protein and induce the expression of Runx2 and Sp7 by upregulating the levels of BMP-2, BMP-4, and BMP-6 genes, and further regulate the expression of alkaline phosphatase (ALP), osteocalcin (OCN), Col, and so on, thereby promoting bone. The formation of tissue plays a role in improving diseases such as osteoporosis and osteoarthritis (Sun et al., 2021; Zhou et al., 2019). At the same time, BMP-Smad can also promote osteoblast proliferation during fracture healing to a certain extent by activating genes such as Smad1, Smad5, and Smad8 (Maung et al., 2021). In some research reports, promoting the expression of BMP-2, BMP-4, and BMP-6 during the fracture-healing process can activate the BMP-Smad pathway and further promote the proliferation of osteoblasts (Wang et al., 2020). To a certain extent, fracture healing can be promoted by regulating the BMP-Smad pathway, and it can also exert anti-inflammatory, antioxidant, and other effects to improve the quality and speed of fracture healing (Fan et al., 2023). Therefore, studying the mechanism of the BMP-Smad pathway plays an important role in studying tibial shaft fracture healing in rats.
Studies have reported that traditional Chinese medicine has good efficacy and mechanism of action in treating tibial shaft fracture healing (Hao et al., 2022). Pue (Puerarin) is isolated from Pueraria lobata. It is an isoflavone derivative with a coronary expansion effect. It can improve blood circulation, reduce myocardial oxygen consumption, and have a certain protective effect on acute myocardial hemorrhage (Kulczyński et al., 2021). In recent years, studies have shown that puerarin can weaken the promoting effect of SONFH on rabbit tissue pathological abnormalities, offset the inhibitory effect of SONFH on the expression of ALP, RUNX2, COL1A1, and miR-34a in rabbits, and improve osteoblast viability (Jiang et al., 2021). At the same time, it can also stimulate cell migration by regulating growth factors such as FGF, EGF, and NGF, and it plays an important role in promoting fracture healing (Tong et al., 2022). Xiao et al. (2020) found through mouse experiments that puerarin can inhibit the occurrence of osteoclasts by inhibiting MAPK/NF-κB signaling, thereby reducing osteoclasts in ovariectomy-induced osteoporosis model mice associated bone loss. In addition, puerarin can promote fracture healing to a certain extent by promoting the proliferation of osteoblasts and inhibiting the differentiation of osteoclasts. Its anti-inflammatory and antioxidant effects can also reduce complications during the fracture-healing process (Yang et al., 2022). However, it is currently unclear whether puerarin can regulate the BMP-Smad pathway to promote tibial shaft fracture healing, and further research is needed.
Materials and Methods
Experimental Materials
Experimental Animals
Ninety male SD rats were purchased from Tianqin Biotech. This study was approved by the ethnic committee of Hunan Provincial Brain Hospital.
Instruments
Protein transfer system and electrophoresis instrument were purchased from BioRad; a small animal microtomography scanner was purchased from Guangzhou Laishang Biotechnology LTD; surgical microinstruments were purchased from Suzhou Medical Equipment Factory; ABI7900 HT real-time polymerase chain reaction (PCR) purchased from ABI Company of the United States; fully automatic biochemical analyzer (model: Xuzhou Meikang Electronic Equipment LTD; model: BIOBASE BK200).
Reagent
Puerarin (purity: ≥98%; CAS number: 3681-99-0; Shanghai Baishun Biotechnology Ltd; mainly an isoflavone derivative extracted from Pueraria lobata with crown-expanding effect, and also has therapeutic effects cardiovascular and cerebrovascular diseases, liver protection, and enhancement of human immunity); BMP-specific agonist SY-LB-35 (specification: 10 µg, purity >97%) was purchased from Merck; BMP-specific inhibitor Noggin (specification: 10 µg, purity >97%) were purchased from Abbexa Company; rat ALP, type I procollagen N-telopeptide (PINP) and OCN ELISA kits were purchased from Sangon Biotechnology Company; Mouse anti-BMP-2 and Runx2 were purchased from Aimeiji Company (Product No.: ANT-183); goat antirabbit horseradish peroxidase IgG was purchased from Yisheng Biotechnology (Shanghai) (product no.: 33101ES60) and β-actin Antibody [UBIA (Shanghai), product number: UBI6009]; rabbit anti-SMAD1/5/8 was purchased from Aimeijie Company (product number: 10821-1); rabbit anti-p-SMADI/5/8 was purchased from Hubei Apti Biology (CAS: IPD-ANP13221).
Method
Preparation and Grouping of Tibial Fracture Models
Eighty healthy male SD rats were selected and intraperitoneally injected with 8.0% sodium pentobarbital (20 mg/kg) to anesthetize the rats and fix them on the operating table. The surgical site was disinfected with iodophor and placed in the anterolateral tibial diaphysis of the rats. Cut the skin 1.5–2 cm at the junction of the upper 1/3 to expose the diaphysis of the tibia. After drilling, use a 0.8 mm Kirschner wire (about 17 mm in length) to retrogradely drive the Kirschner wire upward, and then drive it antegrade into the tibial diaphysis. After fixation, use a scalpel to cut back and forth transversely about 1 cm below the tibial tubercle to cause fractures, which were washed and sutured layer by layer. Penicillin was injected intraperitoneally every day after surgery to prevent infection. The rats were X-rayed immediately after the operation, and obvious fracture lines were visible in the rats after successful modeling. Eighty tibia fracture rats were divided into tibia fracture group, Pue low-dose, medium-dose and high-dose group, BMP-2 group, Noggin group, Pue + SY-LB-35 group, Pue + Noggin group, 10 animals in each group, were given intragastric administration of normal saline, intragastric administration of 10 ng/mL Pue solution, intragastric administration of 20 ng/mL Pue solution, intragastric administration of 30 ng/mL Pue solution, and transcutaneous injection of 50 ng/mL SY-LB-35 solution at the fracture site. Percutaneous injection of 50 ng/mL SY-LB-35 solution at the fracture site, intragastric administration of 30 ng/mL Pue solution + transcutaneous injection of 50 ng/mL SY-LB-35 solution at the fracture site, intragastric administration of 30 ng/mL Pue solution + Percutaneous injection of 50 ng/mL Noggin solution at the fracture site. The intragastric volume was 10 mL/(kg·d), and the injection volume was 2 mL/(kg·d). Another 10 normal rats were selected without surgery and were only intragastrically administered and injected with an equal amount of normal saline as the control group (Cont). Administration was given for a total of 4 weeks.
Tibia Histomorphometric Score
After the tibial tissue was decalcified, paraffin sections were prepared. After dehydration and dewaxing in xylene and 100% ethanol, they were stained with hematoxylin, differentiated, and counterstained with eosin. After treatment, slides were finally sealed with neutral resin and observed under a microscope. The histological fracture-healing score is used as the judgment standard: 0 point means there is fibrous tissue proliferation; 1 point means normal fibrous tissue appears with a small amount of cartilage; 2 points means fibrous tissue and cartilage are mixed; 3 points means there is a lot of cartilage and fibrous tissue; 4 points are cartilage tissue; 5 points are mainly cartilage with small proportion of immature bone; 6 points are a mixture of cartilage and immature bone; 7 points are mostly immature bone with cartilage; 8 points are only immature bone tissue; 9 points only mature bone tissue.
Tibial Imaging-Healing Score
The existence and blurring degree of the fracture line: a clear fracture line can be observed, and the fracture end has not begun to heal, and the score is 0; the fracture line is slightly blurred, and the score is 1; the fracture line is partially blurred, and the score is 2; the fracture line is mostly blurred, and the score is 3; fracture line is completely blurred, the score is 4. Presence and quantity of callus: no callus is formed, scored as 0; a small amount of callus is formed, scored as 1; callus is formed more but does not surround the fracture end, scored as 2; callus surrounds most of the fracture end, score is 3; callus surrounds the fracture end, score is 4. Restoration of the continuity of the bone cortex: if the bone cortex at the fracture end is discontinuous, the score is 0; if part of the bone cortex is continuous, the score is 1; if most of the bone cortex is continuous, the score is 2; if the bone cortex continuity is restored, the score is 3; if the bone cortex is continuous, the score is 3. Continuity is fully restored, score 4. Presence and density of trabecular bone: no trabecular bone is formed, a score of 0; a small amount of trabecular bone is formed, a score of 1; more trabecular bone is formed, but does not surround the fracture end, a score of 2; trabecular bone is surrounded most of the fracture end, and the score is 3; trabeculae surround the fracture end, and it scores 4.
Detection of Serum Osteogenesis-related Factors
Twenty-four hours after the last administration, the experimental rats were killed by decapitation, and blood was taken from the abdominal aorta. After mixing serum with the corresponding kit reagents, a fully automatic biochemical analyzer was used to detect bone mineral density (BMD), number of bone trabeculae, bone thickness, and bone density. Volume fraction, ALP, OCN, levels. The muscles and other soft tissues were removed from the rat’s left tibia, and then the Kirschner wire in the rat tibia was removed to facilitate subsequent detection.
Western Blot Detection of BMP2, p-Smad1, Smad1, and Runx2 Protein Expression
Take the rat tibia tissue, clean it with physiological saline, and then use precooled RIPA to lyse cells and extract protein, which was quantified by the BCA method, transfer the total protein to NC membrane after electrophoresis and incubate it, then administer primary antibodies BMP2 (ab284387, 1:1,000), p-Smad1 (ab126761, 1:1,000), Smad1 (ab126761, 1:1,000), and Runx2 (ab92336, 1:1,000), incubated at a certain temperature. Wash the NC membrane with TBST washing solution to remove unbound primary antibodies. Add a secondary antibody (goat antirabbit 1:2,000) and incubate for 1 h. Wash the NC membrane again with TBST washing solution to remove unbound secondary antibodies. Place the NC membrane into the chemiluminescent substrate solution and perform a color development reaction in a dark room. Imaging was performed using a chemiluminescence imager. Based on the brightness, position, and size of the colored bands, the protein expression levels of BMP2, p-Smad1, Smad1, and Runx2 were quantitatively and qualitatively analyzed.
HE Staining
After the tibia samples were fixed, they were placed in 0.5 mol/L EDTA solution for 4 weeks for decalcification. After decalcification and routine dehydration, they were embedded in paraffin, sectioned coronally from the fracture end (5 µm/section), incubated with hematoxylin stain for 10 min, differentiated, destained with PBS for 40 s, and incubated with eosin stain. After 1 min, the slide was sealed, and the tissue morphology of the broken end was observed under a light microscope.
RT-PCR Detection
Ribonucleic acid (RNA) was extracted, and complementary deoxyribonucleic acid (cDNA) was used to reverse the transcription. The results of this method were verified using SYBR fluorescent PCR technology. The BMP2. p-Smad1. Smad1, Runx2, and GAPDH primers (Table 1) were used. Relative levels were estimated using the 2−∇∇Ct method.
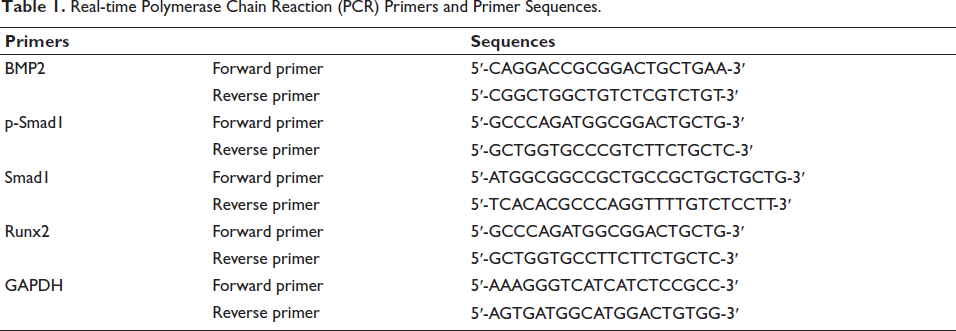
Real-time Polymerase Chain Reaction (PCR) Primers and Primer Sequences.
Statistical Method
SPSS 21.0. processed data analyzed by t test, and chi-square (χ2) test with p < 0.05 as the test standard.
Results
Successful Modeling
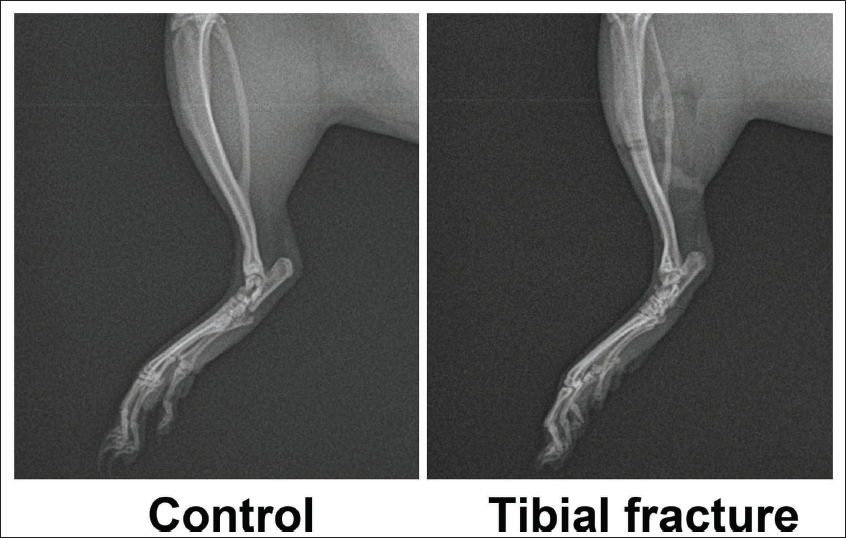
In the comparison between the control group and the tibial fracture group, it was found that the fracture line was not visible in the control group. Still, an obvious fracture line was visible in the tibial fracture group (Figure 1). Therefore, the tibia fracture rat model was successfully created with a success rate of 100%.
Puerarin Promotes the Healing and Repair of Tibial Shaft Fractures in Rats and is Related to the BMP-Smad Pathway
Among the tibial radiographic-healing scores of each group, the tibial fracture group was lower than the other groups. Pue intervention could improve healing scores, and the Pue high-dose group had the best improvement (Table 2). Similarly, in the bone tissue morphology-healing score, the tibia bone tissue morphology-healing score of the Pue low-dose group and Pue medium-dose group was significantly higher than tibia fracture group, and the score of the Pue-H group was more significant (Table 3). By detecting the density of tibial bone minerals, tibial BMD increased in the Pue groups, and tibial mineral density in the Pue high-dose group was best (Figure 2); compared with the tibial fracture group, the Pue groups had the best tibial mineral density. Serum superoxide dismutase (SOD) and ALP in medium- and high-dose groups both increased (Figure 3A and B), while in the malondialdehyde (MDA) test, the Pue groups showed a certain downward trend (Figure 3C). The results of HE staining showed that compared with the Cont group, the rat trabeculae in the tibial fracture group had fractures, widened gaps, and decreased numbers. After treatment, the above indicators of trabecular bone were better than those in the Cont group. The tibial fracture group was significantly improved, among which the Pue high-dose group had the most obvious improvement (Figure 4). Through further detection of BMP2. p-8mad1. Smad1 and Runx2 protein expression in tibial tissue, compared with the Cont group, the expression of BMP2. p-8mad1. Smad1 and Runx2 proteins in the tibial fracture group were reduced; the protein expression of BMP, p-Smad1, and Runx2 in the Pue groups was higher than that in the tibial fracture group (Figure 5). It is suggested that the BMP-Smad pathway is involved in the process of puerarin promoting the healing and repair of tibial shaft fractures in rats.
Comparison of Imaging Healing Scores of Rat Tibia in Each Group (x ± s, Points).

Comparison of Tibial Tissue Morphological Healing Scores in Each Group (x ± s, Points).
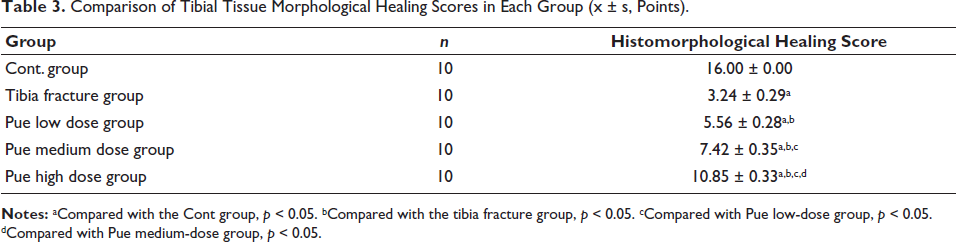
Tibia Mineral Density (n = 10).
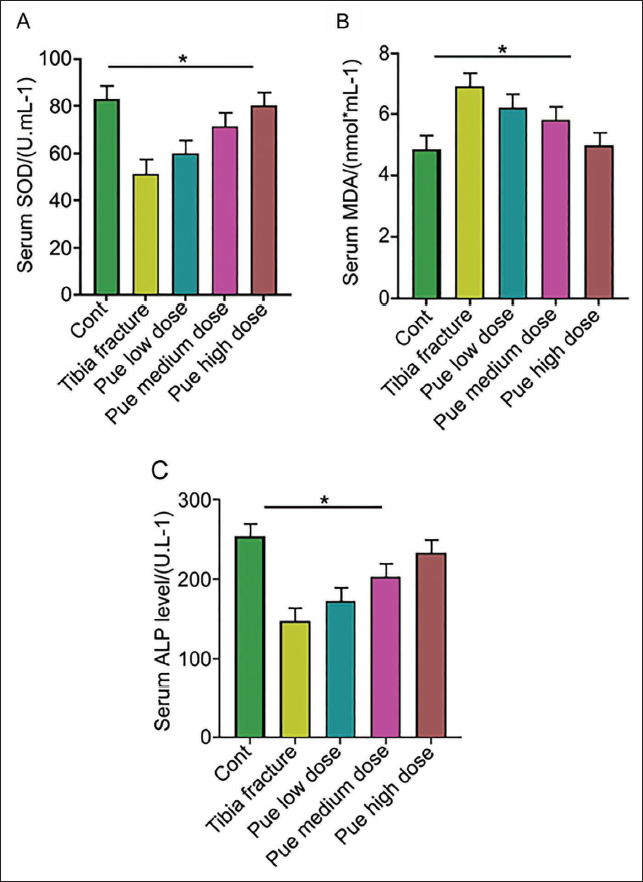
Hematoxylin and Eosin (HE) Staining Image of Tibial Stump Tissue (100×) (n = 10) (Scale Bar = 200 µm).
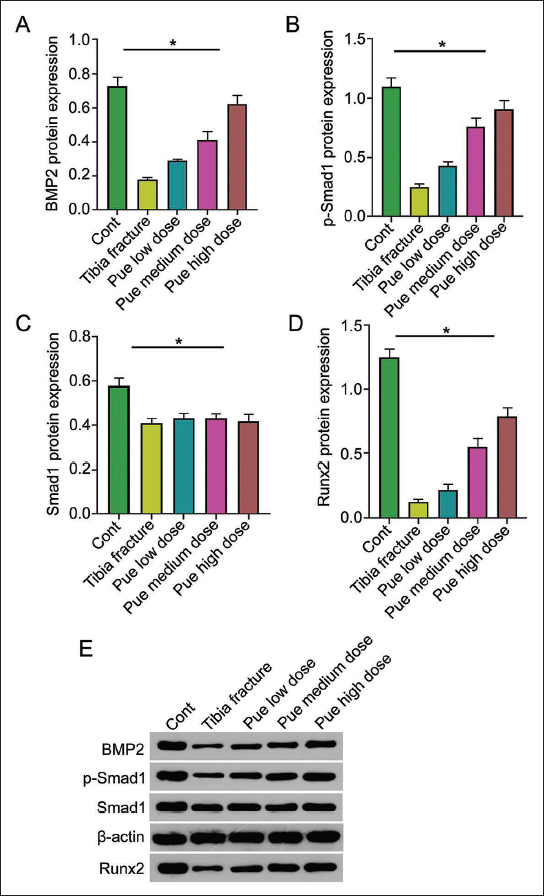
Activating the BMP-Smad Pathway can Promote the Healing of Tibial Shaft Fractures
To explore the role of the BMP-Smad pathway in tibial shaft fracture healing, we used BMP-2 and Noggin to activate and inhibit it. Compared with the tibial fracture group, the tibial BMD and serum ALP in the BMP-2 group increased to a certain extent, and the number, thickness, and bone volume fraction of trabeculae showed an increasing trend (p < 0.05); however, compared with tibial fracture group, the tibia of Noggin group BMD, serum ALP level, osteocalcin content, bone trabecular number, thickness, and bone volume fraction were all reduced (p < 0.05) (Figure 6).
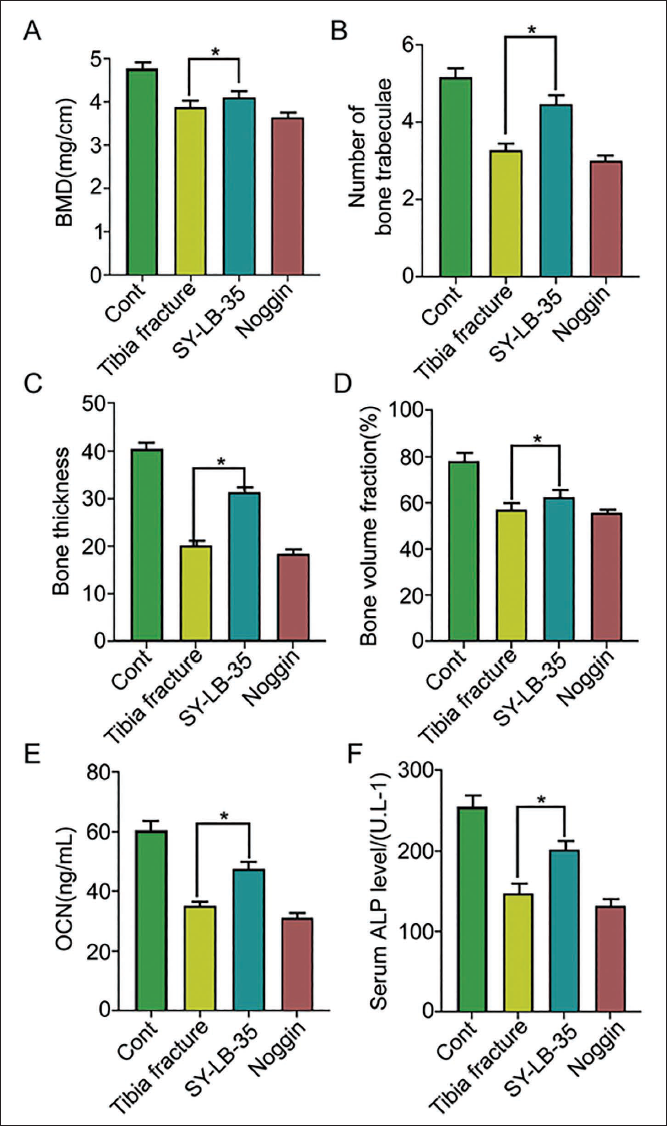
Puerarin’s Effect on the Healing and Repair of Tibial Shaft Fractures in Rats is Mainly Achieved by Activating the BMP-Smad Pathway
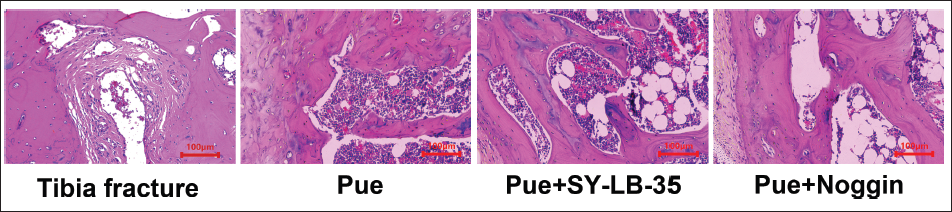
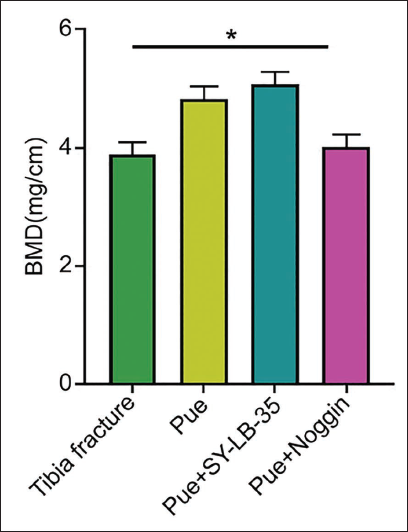
To verify whether puerarin can promote the healing and repair of tibial shaft fractures in rats by regulating the BMP-Smad pathway, we intervened in groups and performed HE staining on rat tibiae. The results found that compared with the tibial fracture group, the Pue group showed an increased number of chondrocytes and osteoblasts; in the Pue + SY-LB-35 group, dense and hypertrophic chondrocytes and newly formed bone trabeculae could be seen around the fracture end, and there were also a large number of osteoblasts in the gaps. However, in the Pue + Noggin group, the number of nonhypertrophic chondrocytes around the fracture end was reduced and accompanied by only a small number of osteoblasts in the gap between the fracture ends (Figure 7). Further observation of tibial BMD showed that compared with the Pue group, the BMD in the Pue + SY-LB-35 group increased, while the BMD in Pue + Noggin group was lower than Pue group (Figure 8, p < 0.05).
Tibia Mineral Density (n = 10).
Discussion
Natural product puerarin has been shown to improve postmenopausal osteoporosis with high safety. It improves OVX-induced osteoporosis and lipids by regulating phospholipid metabolism and PUFA biosynthesis in castrated rats. Metabolism, on the one hand, can reduce adipogenic differentiation by activating Wnt signaling; on the other hand, inhibition of the PPARγ pathway promotes osteogenic differentiation of BMSCs to a certain extent (Li et al., 2022). Qiu et al. (2022) found through experiments on rats that puerarin inhibited the integrin-β3-Pyk2/Cbl/Src signaling and reduced the number of trap-positive osteoclasts and serum TRAP-5b and CTx1, which can reduce the number of osteoclasts. Osteocyte bone resorption also prevented bone loss in OVX rats and further prevented osteoclast apoptosis. Li et al. (2020) found that puerarin has an antiosteoporotic effect by regulating short-chain fatty acids (SCFA) levels and repairing intestinal mucosa, and this effect can also be achieved by regulating intestinal flora disorder in OVX rats. Inhibiting inflammatory tumor necrosis factor-alpha (ITNF-α) and other inflammatory factors can increase Type II collagen content and change the expression of degeneration/bone turnover biomarkers, there by achieving recovery from osteoarthritis damage (Ma et al., 2021). It can be seen that puerarin is involved in bone disease treatment.
To explore the effect of puerarin on tibial shaft fractures, we constructed a rat model of tibial shaft fractures and used low, medium, and high doses of puerarin to intervene. Subsequently, we found that high doses of puerarin promote the development of tibial shaft fractures in rats. Shaft fractures are more effective than low doses. Specifically, it improved the bone tissue morphology healing score and tibia bone tissue morphology healing score, increased BMD, SOD, and ALP levels, and reduced MDA. It shows that puerarin can promote the healing and repair of tibial shaft fractures in rats. In addition, under the intervention of puerarin, the expression of BMP2, p-8mad1, Smad1, and Runx2 proteins increased. It is suggested that puerarin promotes the healing and repair of tibial shaft fractures in rats and is related to the BMP-Smad pathway.
The BMP-Smad pathway can be activated by irisin, thereby promoting MSC-derived osteogenesis and participating in the treatment of osteoporosis and bone defects (Xue et al., 2022). Jin et al. (2020) found by knocking out miR-330-5p in mice that the Bgn-induced BMP/Smad pathway can promote BMSC osteogenesis, thereby reducing the progression of osteoporosis. In this process, Bgn increases osteoblast differentiation by activating Smad1/5/8 signaling and causes cell mineralization to a certain extent (Kusano et al., 2021; Pan et al., 2018). At the same time, other studies have pointed out that osteoblasts and osteoblast (OB) differentiation are at least partially controlled by BMP/Smad canonical signaling, and the activation of Runx2 is partly due to the mediation of BMP/Smad signaling during osteogenic differentiation, initiates the downstream osteogenic marker gene expression cascade, thereby promoting bone repair (Sautchuk et al., 2022; Wan et al., 2023). Furthermore, bone formation signaling pathways, including BMP, SMAD, and TGF-β, are upregulated in senescent MSCs, while BMP-2 is upregulated in senescent MSCs. This phenomenon explains the advantages of nonsenescent mesenchymal stem cells in bone formation and the common complications that may occur with senescent mesenchymal stem cells when using BMP-2 in orthopedic surgery (Cho et al., 2022; Lei et al., 2021; Nakashima et al., 2021).
To assess BMP-Smad’s role in tibial shaft fracture healing, we used SY-LB-35 and Noggin to activate and inhibit it and found that under the intervention of SY-LB-35, tibial BMD, serum ALP. The level and osteocalcin content increased to a certain extent, and the number and thickness of bone trabeculae and bone volume fraction also increased; however, under Noggin’s intervention, tibial BMD, serum ALP levels, osteocalcin content and Trabecular bone number, thickness, and bone volume fraction were reduced. This means that stimulating the BMP-Smad pathway is helpful for tibial shaft fracture healing.
To verify whether puerarin promotes healing and repair of tibial shaft fractures in rats by activating the BMP-Smad pathway, we combined high doses of puerarin with agonists and inhibitors of this pathway and found that in the Pue + Noggin group with the intervention of PUE, dense and hypertrophic chondrocytes and newly formed bone trabeculae appeared around the fracture end, and there were also a large number of osteoblasts in the gap. However, with the combined use of Pue and SY-LB-35, there was no hypertrophy around the fracture end. The chondrocytes are reduced and only a few osteoblasts are present in the stump space. This shows that the healing and repair of tibial shaft fractures in rats by puerarin is mainly achieved by activating the BMP-Smad pathway.
Conclusion
Therefore, studies have found that puerarin can induce the differentiation of osteoblasts by activating BMP-Smad, promoting callus formation, and further promoting tibial shaft fracture healing in rats. This will help us better understand the pathogenesis of bone diseases and provide clues for the diagnosis and treatment of bone diseases. Due to the small number of samples, the specific relevant mechanisms still need to be further improved.
Footnotes
Abbreviations
BMP: Bone morphogenic protein; HE: Hematoxylin and eosin; Pue: Puerarin; qPCR: Quantitative real-time polymerase chain reaction; Smad1: Mothers against decapentaplegic homolog 1.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethic committee of Hunan Provincial Brain Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
