Abstract
Lac has been attracting the scientific community’s attention since time immemorial, aided by its wide-ranging applicability in the textile, plastic, metallurgy, and food industries. Lac exudate comprises three primary constituents: wax, resin, and dye. Lac dye, a mixture of polyhydroxy anthraquinones, is emerging as an up-and-coming candidate for various biological activities. A dearth of systematic reviews substantiating the chemical aspects, biological activities, and therapeutic potential of lac dye in treating human ailments propelled the need to comprehensively review its multifaceted prospects. The historical significance of lac dye, along with the chronological development of lac exudates, has been thoroughly discussed in this review. Additionally, the global abundance and distribution of lac was scaled, emphasizing its cultivation, especially in the context of Indian terrain. This review also sheds light on the chemistry of various constituents of lac dye. Further, the methods of isolation and characterization were extensively elaborated. More importantly, the biological importance of lac dye was systematically presented, discussing its role as an antioxidant, anticancer, antimicrobial, and antifungal agent. This review highlights the disconnect about the research of lac and attempts to instigate the inquisitive minds of academia and the scientific world in further exploring and bridging the gaps relating to lac research.
Introduction
Globally, there has been an increasing trend among the scientific community aimed at exploring and acknowledging the tremendous potential of natural products that offer a vast repository for uncovering novel therapeutic agents (Atanasov et al., 2015; Halagali et al., 2024). Amongst these natural sources, animal-derived products have garnered attention due to their unique and often uncharted pharmacological properties (Hemant et al., 2022; Newman & Cragg, 2016; Tehzeeb & Dhanabalan, 2020). Lac is one such intriguing animal-derived substance that has come to light in the pharmaceutical sector.
Lac is a sticky exudate secreted by the female lac insects Laccifer lacca and Kerria lacca. This resinous secretion is processed into various forms, mainly comprising waxes, resins, dyes, and pigments (Bashir et al., 2022). Some of the constituents of lac are known to possess certain bioactivities, while others can be used as versatile materials with multifaceted applications. The exact composition may vary depending on the source and processing methods, but collectively, they provide a fascinating subject for scientific inquiry (Panwar & Author, 2020). Furthermore, amongst the primary constituents of lac, “lac dye” has been particularly riveting because of the presence of laccaic acids. Laccaic acids are derivatives of complex anthraquinone moieties that might be responsible for imparting the dyes with their potential therapeutic properties (Hong et al., 2011). Preliminary research suggests that lac dye possesses numerous therapeutic activities that can benefit human health, including antimicrobial, antioxidant, and anti-inflammatory properties. However, it is essential to emphasize that despite its intriguing prospects, research on lac dye remains scarce and requires further exploration.
The scarcity of comprehensive research on lac dye is a compelling reason to conduct a thorough review of this natural product. A search was performed on PubMed and Web of Science Core Collection to identify published articles relating to lac and lac dye. The search revealed a dearth of articles relating to the search query, with no systematic review to report and substantiate the chemical aspects, biological activities, and therapeutic potential of lac constituents to treat human ailments. Correspondingly, it felt indispensable to review these critical prospects, and the following objectives were charted out: (a) presenting a brief background to summarize the historical chronology, cultivation, and global distribution of lac, majorly focusing on the Indian context; (b) discussing significant chemical aspects of lac dye more comprehensively, incorporating the methods of isolation and characterization of laccaic acids; (c) elaborating crucial biological aspects of lac dye encompassing the different investigations relating to anticancer, antimicrobial, and antioxidant activities, and more; (d) furnishing critical research gaps and future perspectives to guide various research groups in performing additional scrutiny. Such a review would not only consolidate existing knowledge but also shed light on its unexplored facets, potentially leading to the discovery of new pharmaceutical applications. By examining the major constituents, extraction methods, and recent advancements in lac research, this review aims to provide substantial information that can be of immense value to the advancement of pharmaceutical science and the development of innovative healthcare solutions.
Methodology
This review systematically explores the pharmaceutical potential of lac dye, specifically focusing on the anthraquinone-based constituents known as laccaic acids. Utilizing a targeted search strategy on the Web of Science database, relevant literature is curated using key terms such as “lac dye,” “laccaic acids,” “life cycle of lac,” “isolation of laccaic acids,” and “instrumental techniques for laccaic acid structure elucidation.” Inclusion criteria prioritize articles discussing medicinal aspects, the life cycle of lac, methods for isolating laccaic acids, and instrumental techniques for structural elucidation. The selected literature is synthesized to provide a comprehensive overview, and the quality of the studies is assessed based on relevance and methodological rigor. The resulting review aims to inform researchers about the pharmaceutical potential of lac dye and laccaic acids, emphasizing their diverse medicinal applications and the methodologies involved in studying them.
Historical Development
Indians have been familiar with the use of lac since the Vedic period (Table 1). One of the earliest references was found in the Atharva Veda (verse 5 of 5th volume), where the insect and its habitat were termed Laksha, along with its brief description and medical applications (Dave, 1950). A brief mention of lac as “Laksha Griha,” an inflammable house of lac, was made in the great Indian epic “Mahabharata” (2000 years old), constructed by descendants of Kuru, the Kauravas, as a part of the conspiracy to burn down their enemy “Pandavas,” alive (Roy, 1978). In the medieval ages, “Abu Hanifa” mentioned its use as a medicinal product (Perveen et al., 2013).
Chronological Account of Lac Usage and Research.
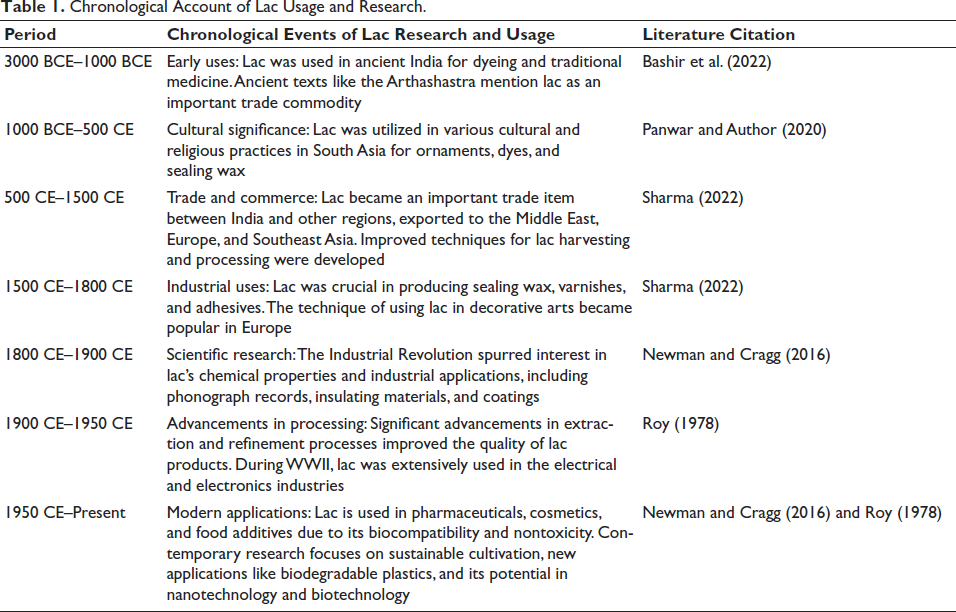
The first (quasi)-scientific work on lac was started by a missionary priest, Father Tachard, at Pondicherry in 1709, who dispatched his work on lac as his memoir to France, describing the occurrence and properties of lac (Gibson, 1942). Later, James Kerr, a medical officer, published an article in 1781 entitled “Natural history of the insect which produces the gum lacca,” depicting the morphology of the lac insect and its host plants (Kerr & Banks, 1781). William Roxburgh published his articles in Philosophical Transactions of the Royal Society of London in 1791, where he named the insect Chermes lacca and described its life history (Roxburgh, 1791). The life history of the lac insect was further elucidated by H. J. Carter, which corrected the errors of existing writings profoundly (Carter, 1861).
Based upon the monograph of lac insects and its supplement, the taxonomy of lac insects was developed by Chamberlin (1923, 1925). Further work was carried out by researchers Kapur and Varshney (Varshney, 1976, 2009). K. lacca yields the maximum amount of resin among several species and is the foremost choice of cultivators. So far, 90 species under 9 genera have been reported worldwide. In India, the majority of the lac is produced by the insect species K. lacca with two subspecific forms—Kusumi and Rangeeni, depending upon the host preferences, life cycle, and quality of lac resin (Sharma, 2022).
Distribution and Cultivation
The cultivation and distribution of lac, as well as lac dye, are a significant industry with deep-rooted traditions in several countries, particularly India. Lac cultivation is predominantly found in regions such as India, Myanmar, Thailand, and some Chinese provinces, where the female lac insect (L. lacca) thrives on specific tropical trees (Pal et al., 2010). India, in particular, has emerged as a leader in lac production, with Jharkhand being the top lac-producing state, followed by Chhattisgarh, Madhya Pradesh, and others (Pal et al., 2010, 2011; Sharma, 2016).
In India, stick lac, the raw material for the lac industry, is produced at an average annual volume of 20–22 kt. Jharkhand leads the nation’s lac production, contributing 55% of the total production, with the top five states collectively producing 95% of the country’s lac. A significant portion of the lac products is exported, while a smaller percentage, around 20–25%, is used domestically (Sharma, 2016, 2022). Lac cultivation often takes place in wasteland or forest areas, involving the participation of approximately 3–4 million tribal communities living in these states. These tribal populations play a vital role in caring for, nurturing, cultivating, and harvesting lac, which in turn significantly contributes to their economic well-being. The lac industry accounts for a substantial portion of their agricultural income, ranging from 20 to 38% in states like Jharkhand (Bashir et al., 2022). Additionally, the processing of raw lac also provides employment opportunities to the local population.
Furthermore, India earns foreign exchange worth 120–150 crore per annum by exporting lac to nearly 45 countries, including Germany, Italy, Egypt, Indonesia, and the United States of America. Beyond being a source of income for millions of lac farmers, lac cultivation plays a crucial role in conserving the rich flora and fauna in the areas where it is practiced (Ranjan et al., 2011; Yogi et al., 2021).
The cultivation of lac follows a consistent process across India, with minimal variation due to local climatic conditions. The cultivation process begins by placing broodlac on designated host plants, and twigs containing broodlac are cut, bundled, and tied to suitable locations on fresh host plants to encourage the swarming and colonization of emerging lac insect larvae on succulent shoots. The choice of host plant and the availability of healthy broodlac at the right time are critical factors considered during cultivation. High yield depends on effectively managing host stocks, using pest-free brood, inoculating host plants at the right time and in the correct manner, and minimizing attacks from pests and predators (Ghosal et al., 2018; Subbarayudu & Ram, 1997).
Two strains of lac insect, Rangeeni and Kusumi, are preferred based on factors such as the time needed for the insect’s life cycle, desired resin quality, and the insect’s preference for host plants. Both strains produce crops twice a year, known as jethwi and aghani from the Kusumi strain, and kartiki and baisakhi from Rangeeni. The inoculation period for these strains varies, with kartiki and baisakhi being inoculated in June/July, aghani in June/July, and jethwi in January/February. The harvested lac is obtained by cutting the lac-coated twigs, referred to as sticklac, which is stored in dry conditions (Bhattacharya et al., 2002). During harvesting, it is essential to manage and minimize the impact of key pests of lac insects, with the most destructive pests including Eublemma amabilis and Pseudohypatopa pulverea (Sarvade et al., 2018). This longstanding practice of lac cultivation in India not only contributes to the livelihoods of millions of individuals but also maintains a harmonious balance between economic development and the conservation of the unique ecosystems in which it thrives. Additionally, lac and lac dye play a crucial role in various industries, including pharmaceuticals, textiles, and cosmetics, making this natural product a valuable and versatile resource for both traditional and modern applications (Mohanta et al., 2013).
Life Cycle, Synthesis, and Harvesting of Lac
The production of lac, a resinous substance derived from the lac insect, involves a complex life cycle, synthesis, and cultivation process. The life cycle of the lac insect begins with the laying of eggs by the female lac insect on the host tree’s twigs. These eggs are usually laid in large clusters that cover a large surface area on the twigs. After hatching from the eggs, the larvae, known as “crawlers,” are highly mobile and spread across the host plant. They are equipped with piercing-sucking mouthparts, which they use to extract sap from the host tree. During this stage, the larvae secrete a protective cover known as broodlac as they continue to feed and grow. At a certain point in their development, the larvae cease feeding and undergo metamorphosis, transforming into pupae within the protective broodlac. This stage is characterized by the formation of a hard, resinous shell around the pupae, which protects them from environmental factors. The mature pupae metamorphose into adult lac insects. The adult females are wingless and remain attached to the host tree, where they secrete a resinous substance, which accumulates as lac resin. The males, on the other hand, are tiny and have wings, allowing them to disperse in search of females. The female lac insects continue to secrete resin while mating with the male lac insects. After mating, the females lay eggs and start the life cycle anew. This process can occur multiple times, with several generations of lac insects produced in a single year (Bhattacharya et al., 2002). The life cycle of the insect has been illustrated in Figure 1.
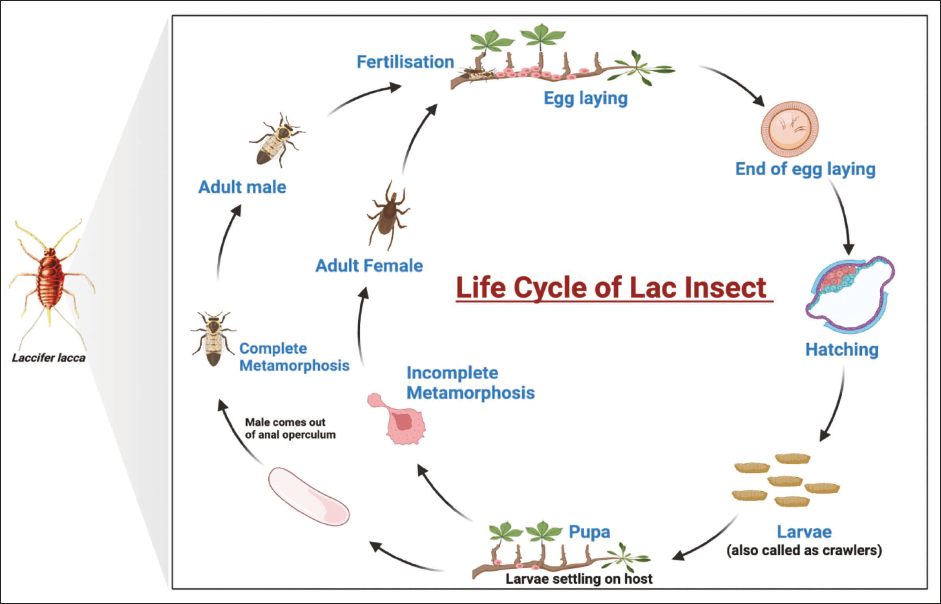
The synthesis of lac resin occurs primarily during the adult female stage of the insect’s life cycle. The insect secretes the resin as a protective covering, both for herself and her eggs. This secretion is rich in a mixture of resins, waxes, pigments, and other bioactive compounds, which collectively form the lac resin. The cultivation of lac involves the following key steps: Lac insects infest specific tropical trees, with some of the common host plants including ber (Ziziphus mauritiana), kusum (Schleichera oleosa), and palas (Butea monosperma). The selection of appropriate host plants is crucial for successful lac cultivation. To initiate lac cultivation, broodlac, which is a bundle of twigs containing eggs, larvae, and pupae, is attached to the selected host plants. The broodlac is carefully tied to the host tree’s branches, ensuring that the emerging larvae have access to succulent shoots for feeding and development. Lac cultivators must ensure the healthy growth of host plants, protect them from pests and predators, and manage the broodlac to facilitate desirable swarming and colonization of the lac insect. Lac is harvested after the swarming phase, usually when the broodlac-covered twigs are cut from the host plants. This harvested material is referred to as stick lac. After harvesting, stick lac undergoes further processing, including cleaning, refining, and separating the resin from impurities. This processed lac can be used in various industries, including pharmaceuticals, textiles, and cosmetics (Chen et al., 2013; Kumar & Bhatnagar, 2013).
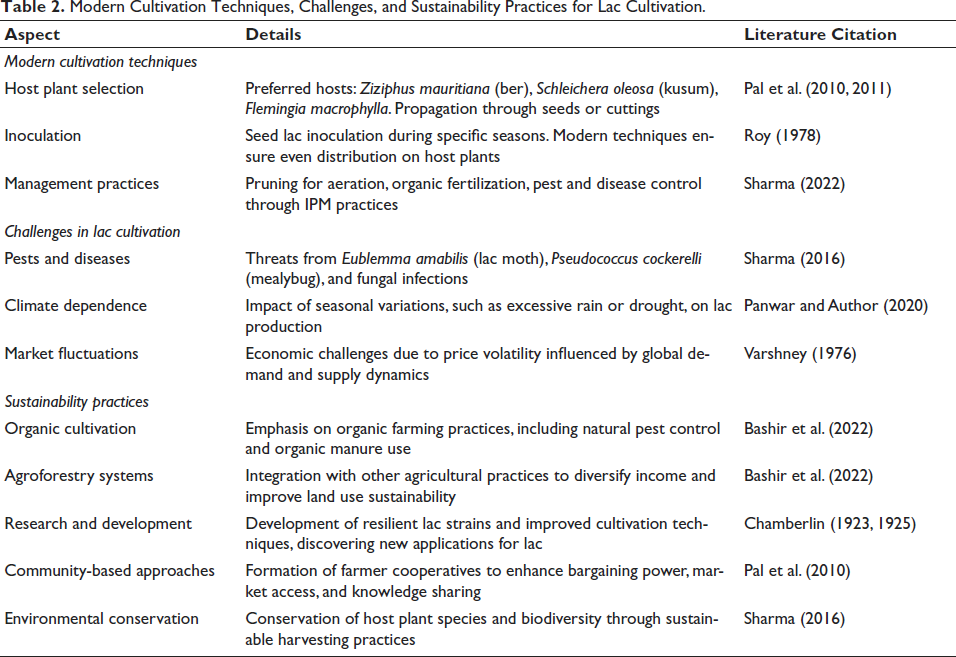
The cultivation of lac involves a well-coordinated process that takes advantage of the lac insect’s life cycle, primarily focusing on the resin-producing activity of adult female lac insects (Table 2). This practice has been a source of livelihood for many communities, particularly in regions like India, and provides a valuable natural resource with diverse applications (Wang et al., 2006).
Modern Cultivation Techniques, Challenges, and Sustainability Practices for Lac Cultivation.
Lac Composition
Sticklac harvested from the crop contains a number of entities, including insect body debris, soluble salts, proteins, woody matter, and others. The strain of the insect, the climate, and the host plant directly affect the composition of lac and its quality (Bose, 1963). The main constituents obtained from sticklac are resin, dye, and wax, among which resin is the principal constituent. Lac contains 68% w/w resin, 6% wax, 1–2% dye, and 25% other components like pigments (Srivastava et al., 2013). Resin is the primary component of lac, which accounts for the bulk of the material. It is a complex polyester consisting of a long chain of aliphatic hydroxy acids (C14–C18 carbon chain), such as butolic acids, aleuritic acid, and terpenic hydroxy acids possessing the structure of cedrene. Resins provide lac with its adhesive and glossy properties. Apart from resin, the minor constituents of lac are dyes and waxes. Long-chain esters, alcohols, and hydrocarbons comprise the wax, whereas the dye extracted from sticklac, commonly known as lac dye or lacquer dye, is made up of a range of anthraquinone derivatives called laccaic acids (Yuan et al., 2021). Lac dyes have been used for centuries in the production of textiles, cosmetics, and food products (Srivastava et al., 2010). Overall, lac is a versatile natural material that finds applications in the production of cosmetics, textiles, and pharmaceuticals. We shall delve further into the extraction, isolation, and applications, mainly focusing on lac dyes.
Extraction of Lac Dye
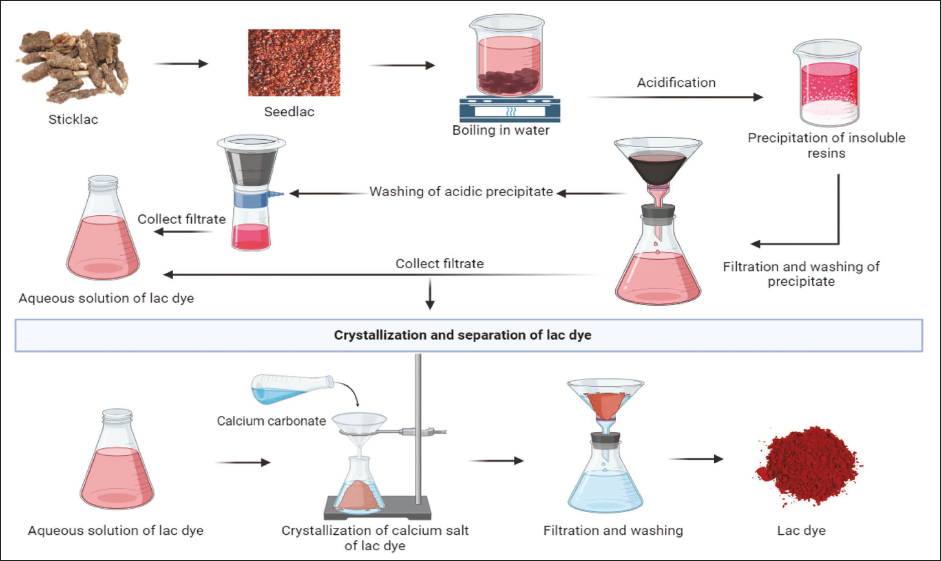
The process of extraction and purification of lac dye from sticklac consists mainly of solvent extraction, precipitation, and crystallization. Sticklac is harvested from the branches of the host trees infested with the lac insect. The raw lac contains impurities, such as wood and insect remnants, that need to be removed. The stick lac is crushed into smaller pieces and washed to remove the impurities. The crushed and washed sticklac is then boiled in water. This causes the resinous portion of the lac to melt, while the dye solubilizes in water. Subsequently, the resins and waxes are precipitated by acidification using sulfuric or hydrochloric acid (0.1% v/v). The precipitate is then removed by filtration, while the supernatant liquid containing the dye is collected for further processing. The acidic precipitate is further extracted separately with boiling water for the removal of any remaining dye. The washings are then mixed with the previously collected supernatant (Ghosh & Sengupta, 1977).
Crystallization of the dye in the form of calcium salts is then carried out by treating the filtered liquor with calcium carbonate or quicklime till the solution becomes colorless. The calcium salt of the dye is left to settle. The salt suspension is washed and acidified, then it is left undisturbed for 8–10 days at room temperature until the calcium salt of dye completely crystallizes out. The dye is filtered, washed, and dried to obtain the purified lac dye (Sharma et al., 2020). The process of obtaining lac dye from crude sticklac has been depicted in Figure 2.
Process of Extraction of Lac Dye from Sticklac.
It is important to note that the choice of the precipitating agent and specific processing conditions will influence the desired properties of the separated components. Additionally, optimization of the extraction process is necessary for maximizing the efficiency, yield, and purity of the final extracted product. Ensuring the purity of the dye is crucial, especially for applications in textiles, cosmetics, food, and pharmaceuticals (Hu et al., 1997).
Chemistry of Lac Dye
Lac dye is composed of various laccaic acids (Table 3), namely, A (IA), B (IB), C (IC), D (ID), E (IE), and F (IF), along with erythrolaccin (IG), isoerythrolaccin (IH), and deisoerythrolaccin (II) (Pandhare et al., 1966; Wadia et al., 1969). These laccaic acids are derived from phenyl anthraquinones containing methoxyl or carbomethoxyl substituents. The laccaic acids desoxyerythrolaccin, erythrolaccin, and isoerythrolaccin are responsible for imparting the reddish-yellow color of lac (Burwood et al., 1967).
Constituents of Lac Dye with their Name and Structures.
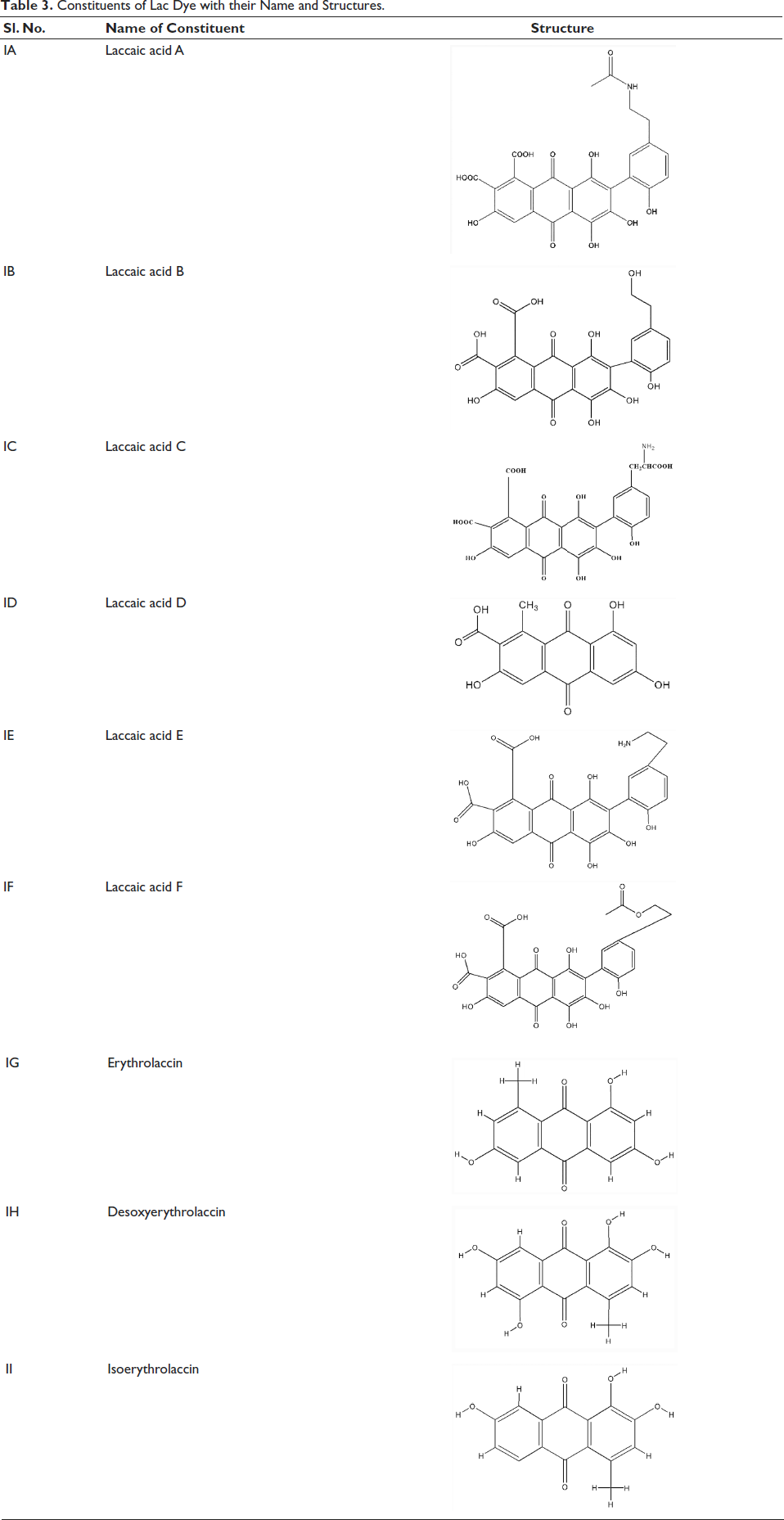
The 1H-NMR analysis confirmed that laccaic acid A is 1,3,4,6-tetrahydroxy-2-[2-hydroxy-5-(β-acetamidoethyl)-phenyl]-9,10-anthraquinone-7,8-dicarboxylic acid. Correspondingly, laccaic acid A was reported to have the molecular formula of C26H19NO12. The UV–VIS absorption spectrum of laccaic acid A in ethanol showed peaks at 290 (log ε 4.8), 340 (log ε 4.31), 500 (log ε 4.32), and 530 (log ε 4.12) nm, while the infrared absorption spectrum showed characteristic transmission peaks at 1,715, 1,692, 1,667, and 1,626 cm–1. Peaks at 1,692 and 1,715 cm–1 indicated the presence of carboxylic acid groups; 1,626 cm–1 corresponds to chelated quinone carbonyl groups as in quinizarin and purpurin; and the 1,667 cm–1 peak indicated an additional carbonyl group in amide. Potentiometric titration showed laccaic acid A to be a dibasic acid with pKa at 4.0 and 6.4 (Burwood et al., 1965; Pandhare, 1969).
The presence of a minor fraction, laccaic acid B, was reported by Pandhare (1969), which is chemically 1,3,4,6-tetrahydroxy-2-[2-hydroxy-5-(β-hydroxyethyl)-phenyl]-9,10-anthraquinone-7,8-dicarboxylic acid and shows similarity to the electronic spectrum of laccaic acid A (Bhide, 1969). Laccaic acid C is present as a minor component in lac dye, and its UV–VIS spectra were comparable to those of laccaic acid A and B. Nuclear magnetic resonance (NMR) research revealed the presence of an additional amino acid side chain (Rama Rao, 1969). Laccaic acid D, with a molecular formula of C16H10O7, has been reported to be identical to xanthokermesic acid with Vmax 1,665 and 1,631 cm–1, corresponding to nonbonded and bonded carbonyl groups (Mehandale et al., 1968). According to 2D-NMR spectroscopy findings from a study, the structure contains three aromatic carbons (CH), as evidenced by positive peaks at δ 123.957, 119.156, and 113.770. A positive signal at δ 13.832 in the aliphatic region indicated the existence of the CH3 group, supporting the structure (Dagur & Ghosh, 2022). Laccaic acid F is represented as C26H18O13 or terminal O-acetate of laccaic acid Β, depicting structural similarity. Even in neutral water at 60°C, laccaic acid F, an exceptionally unstable component of laccaic acids, hydrolyzes to produce laccaic acid B after 2 hours (Hu et al., 1997). So far, the chemistry of laccaic acid A has been explored to a certain extent since it is the most prominent component found in lac dyes. However, further exploration of the chemistry of the minor fractions is required.
Isolation Methods for Laccaic Acids
For the first time, lac dye was subjected to the column of cation exchange resin of “cashew nut shell liquid” and “polyamide column of 1-butanol (saturated)” with 0.3 M hydrochloric acid as eluant for its separation into two major fractions (Burwood et al., 1967). Fractions were designated as “laccaic acids.” Later, the structure of one fraction was established through NMR and found as laccaic acid A. The molecular formula was similar to Burwood’s laccaic acid A1 (Haque & Faruq, 1999). Laccaic acid A1 was isolated from sticklac of Bangladeshi origin by using silica gel column chromatography and thin-layer chromatography (TLC) with a molecular formula of C20H14O10 (Burwood et al., 1967).
Laccaic acid B was isolated and identified from the polyamide column as a minor fraction, which reportedly contained a chelated quinine group (Bhide, 1969). Laccaic acid C was isolated as the significant fraction by using cellulose columns and laccaic acid E as the minor fraction. Laccaic acid C differs from laccaic acids A and B due to the presence of α-amino acid side chains (Rama Rao, 1969). Laccaic acid D was isolated from sticklac obtained from rangini (host) by using silica gel column chromatography and acetone as the eluant (Mehandale et al., 1968). Laccaic acid F was isolated from Thai stick lac by a combination of silica gel column chromatography and preparative TLC (Hu et al., 1997). Erythrolaccin, the major pigment of seed lac, was extracted by concentrating and diluting the ethanolic extract after the removal of waxes from seedlac. Further subjection of the residue to column chromatography gave desoxyerythrolaccin from methanol (Mehandale et al., 1968).
Instrumental Methods for Separation and Identification of Laccaic Acids
For the separation and identification of laccaic acids, various instrumental techniques can be used, such as high-performance liquid chromatography (HPLC), high-speed countercurrent chromatography (HSCCC), HPLC/mass spectrometry (MS), and others. One of the methods was developed using HPLC in combination with a diode array detection (DAD), and electrospray ionization mass spectrometry (ESI-MS) was used for the detection of laccaic acids A and B from historical antiques. Chromatographic separation was carried out on a Xterra C18 5 µm 3.0 × 250 mm HPLC column (Waters Co., USA), thermostatted at 40°C, by a gradient elution program that utilized two solvents: solvent A: H2O-0.01% trifluoroacetic acid (TFA) and solvent B: CH3CN 0.01% TFA. Gradient elution program: initial 95%A evolved to the final 5%A within a time period of 33 minutes and a flow rate of 0.6 mL/min. Correspondingly, at Rt 9.8 and 12.9, separation peaks for laccaic acids B and A were detected and identified using liquid chromatography–mass spectrometry (LC–MS) (Karapanagiotis & Chryssoulakis, 2006).
Another method was developed by Oka et al. (1998) using high-speed countercurrent chromatography for separating lac dye components by using HSCCC-1A prototype apparatus with a multilayer coil planet centrifuge (10 cm orbital radius). The two-phase solvent system, thoroughly equilibrated in the separation funnel, constituted of tert-butyl methyl ether, n-butanol, acetonitrile, and water (2:2:1:5). A 25-mg quantity of the sample was separated, and fractions were analyzed by HPLC and electrospray tandem mass spectrometry. A yield of 9.5 mg of 98.1% pure laccaic acid A, 3.6 mg of 98.2% pure laccaic acid B, and 2.6 mg of 97.2% pure laccaic acid C was obtained.
Hirata et al. (2001) developed an HPLC method for the separation of main pigments in commercial lac color using an RP-18 column with 0.1 mol/L citric acid buffer solution: methanol (16:5) as mobile phase and photodiode array (PDA) detector set at 280 and 490 nm. The pigments detected using HPLC were laccaic acids A, B, C, and E and confirmed by ESI-MS. To determine the anthraquinone components found in red natural dyes, a reversed-phase liquid chromatography method was devised hyphenated with diode array detection and electrospray mass spectrometric methods, where it was concluded that the latter was more apt for such analysis (Ackacha et al., 2003).
In another study, the components of anthraquinones were detected by ESI-MS and capillary electrophoresis with UV–VIS DAD in red natural dyes, where the former proved to be more effective than latter owing to its higher selectivity and sensitivity (detection limit of 0.1–0.5 µg mL–1), with distinctive identification of carminic acid and laccaic acids A, B, and E as the coloring components in cochineal and lac-dye, respectively (Lech et al., 2014; Puchalska et al., 2003).
pH-zone-refining countercurrent chromatography (pHz-CCC) was also used on an analytical scale to separate the natural coloring agents, lac colors, along with Kaoliang. The separation of the constituents of the coloring agents was aided by the presence of the carboxylic acid group in the structure of laccaic acid. The sample solution was allowed to run through a polytetrafluoroethylene (PTFE) membrane filter, which yielded the following elution order—laccaic acids B, A, E, and C, out of which three were isolated, but laccaic acid E formed a mixture with C. The purity of the laccaic acids was determined by fractionating the eluted solvent and using LC–MS to analyze it, followed by its subjection to pHz-CCC with tridodecylamine as a ligand, yielding 790, 109, 41, and 6 mg of laccaic acid A, C, B, and D respectively, with a minimum purity of 96% (Wada et al., 2005).
The anthraquinone content of lac dye can also be determined by using a method with combination of spectrophotometry and high-performance capillary electrophoresis (HPCE) dye. The carminic acid, a multihydroxy anthraquinone, was used as a control sample to detect the anthraquinone content in the lac dye accurately, followed by dye separation aided by HPCE.
Pharmaceutical Applications of Lac Dye
Pharmaceutical Excipients
Lac dye has garnered attention for its versatile applications in the pharmaceutical industry. One of the most significant applications of lac dye is its use as a colorant. It has been approved by various regulatory bodies, such as the United States Food and Drug Administration (FDA) and the European Union (EU), for use as a natural colorant in pharmaceutical products such as tablets, capsules, and syrups. This dye is preferred over synthetic colorants as it is non-toxic, nonallergenic, and does not cause adverse reactions. Lac dye was found to be non-toxic at a dose of 200 mg/kg diet, that is, 0.02% of the diet for acute oral toxicity (Srivastava et al., 2013). It is also stable under various conditions, including high temperatures and exposure to light, which makes it suitable for use as a pharmaceutical colorant. Another application of lac dye in the pharmaceutical industry is as an excipient. Excipients are nonactive ingredients that are added to pharmaceutical products to aid in their manufacturing, processing, or administration (Patel et al., 2020). Lac dye is used as an excipient in various pharmaceuticals, cosmetics, and nutraceuticals as a colorant and improved aesthetic (Al Fahad et al., 2024).
Therapeutic Applications
Lac dye, with its array of therapeutic attributes, emerges as a compelling candidate for the exploration and advancement of novel pharmaceutical products. Its documented antioxidant, antimicrobial, and anti-inflammatory characteristics position it as a valuable resource for drug development, holding the potential to address diverse health challenges, including cancer, diabetes, and infections. Some of its reported activities have been summarized below:
Antioxidant and anti-inflammatory activity: The antioxidant activities of laccaic acids and their aluminum lake were evaluated using the DPPH assay. The assay revealed that laccaic acids demonstrated a higher antioxidative activity (EC50 = 0.38 mg/mL) compared to the aluminum lake (EC50 = 1.63 mg/mL) and butylated hydroxytoluene (EC50 = 0.57 mg/mL), but lower than ascorbic acid (EC50 = 0.14 mg/mL) and gallic acid (EC50 = 0.05 mg/mL). The study also indicated that laccaic acids had greater reducing power than the aluminum lake. However, the percentage inhibition of lipid peroxidation by laccaic acids (29.9%) was lower than that of the aluminum lake (43.8%). Overall, the findings suggested that laccaic acids possess notable antioxidant properties (Jimtaisong et al., 2013). A 25.00 g/mL lac dye concentration showed 68% DPPH scavenging capacity, implying antioxidant properties of lac dye, whereas the capacity of ascorbic acid to scrounge DPPH was about 1/5th of ascorbic acid (Zhang et al., 2012). The anti-inflammatory efficacy of laccaic acid was reported by Kuramoto et al. (1996), whereby natural colorants containing laccaic acid inhibited the production of allergen-specific immunoglobulin E by rat spleen lymphocytes at low concentrations ranging from 1 to 10 µM. Furthermore, these compounds also exhibited potential immunomodulatory properties, showing inhibition of IgG and IgM even at a low concentration of 1 mM. These findings imply that lac dye may possess the ability to regulate immunoglobulin production, which can be attributed to the presence of laccaic acids (Kuramoto et al., 1996).
Antimicrobial: In vitro evaluations by Srivastava et al. (2017) showed that methyl derivatives of lac dye demonstrated both antifungal and antibacterial efficacy. Pathogenic fungi Verticillium sp. exhibited the highest sensitivity, showing 100% inhibition at 750 and 1,000 µg/mL against methylated lac dye. The functionalized dye also demonstrated strong antibacterial properties against Ralstonia solanacearum at very low concentrations (40 and 50 µg/mL). Additionally, isolates obtained from lac dye, erythrolaccin, deoxyerythrolaccin, 2,3,5,6-tetrahydroxy-8-methyl- anthraquinone, and 1,3,4,6-tetrahydroxy-8-methyl-anthraquinone showed activity against Staphylococcus aureus, Bacillus subtilis, and Escherichia coli microorganisms with minimum inhibitory concentration (MIC) values ranging from 0.1 to 0.5 mg/mL (Lu et al., 2018; Srivastava et al., 2017). These findings suggest that lac dye has the potential to serve as a powerful antimicrobial agent that may be used in the management of multiple ailments.
Mutagenicity: The mutagenicity of lac dye was explored by a team, who reported no sign of mutagenicity in Chinese hamster lung (V79) cells when subjected to the Ames test. However, the clastogenic effect on the bone marrow cells of mice was exhibited by lac dye (Banerjee et al., 1984). Similarly, another study reported no mutagenicity for laccaic acid when subjected to two short-term assays: the Salmonella/microsome mutagenicity test and the ØX fidelity assay (Dube et al., 1984). In a study, DNA-dye complexes were prepared at different phosphate-to-dye ratio, and, like other anthraquinones, spectrophotometric studies revealed good interaction of laccaic acid with DNA (Goswami et al., 1988).
Anticancer: In a study by Kemmochi et al. (2012), the effect of lac dye on an experimental thyroid cancer model in rats was studied, where the rats exposed to long-term lac dye treatment significantly reduced the development of invasive carcinomas and lung metastases, with associated downregulation of genes related to angiogenesis and tissue proteolysis. Lac dye was found targeting the activity of plasma hyaluronan-binding protein (PHBP) in macrophages and neoplastic cells, suppressing early stages of carcinogenesis (Kemmochi et al., 2012). Laccaic acid A was shown to be a direct DNA-competitive Dnmt1 inhibitor, competing with DNA substrate and manipulating the expression of methylated genes synergistically with 5-aza-2′-deoxycytidine in MCF-7 breast cancer cells (Fagan et al., 2013). An in vitro study of lac dye against six human leukemia cell lines, including U973, Raji, K562, Jurkat, MOLT 4, and HL60, showed a positive response. This study suggests that lac dye can be considered a potential anticancer agent for leukemia (Shamim et al., 2016). Due to dual inhibition of DNA methyltransferase-1 and histone deacetylase-1, phenethyl isothiocyanate and laccaic acid have demonstrated strong action against colorectal cancer (Gupta et al., 2019). Another study showed its structural similarity with Adriamycin, indicating anticancer activity. The anticancer activity of methanolic fraction (M) was evaluated, which showed promising anticancer activity against in vitro MDAMB-231 and SiHa cell lines, comparable to the standard Adriamycin (Dagur & Ghosh, 2022).
Insulin resistance: Kabeer et al. (2023) investigated the impact of laccaic acid on insulin resistance induced by a high-fat diet in mice. Chronic laccaic acid treatment showed improvements in the morphometric, biochemical, and liver function parameters of mice. The compound enhanced insulin signaling pathways, reduced inflammatory signals, and affected gluconeogenesis-related genes. Notably, laccaic acid influenced the epigenetic landscape by preventing histone methylation changes associated with insulin resistance and upregulating the H3K27 methylating enzyme EZH2. This study suggests that laccaic acid may play a role in modulating insulin resistance through multiple cellular mechanisms, making it a potential candidate for further research in metabolic disorders (Kabeer et al., 2023).
To understand the therapeutic potential of lac dye, clinical studies need to be conducted. However, no research has yet reached the clinical stage. Therefore, lac dye needs to be explored further, as it shows potential pharmaceutical applications.
Conclusion
The exploration of lac dye, a significant constituent of the lac exudate produced by the scale insect L. lacca, unfolds a multifaceted landscape that extends from its historical mentions to modern scientific inquiry. Specific habitats, cultivation conditions, and environmental factors have been shown to play a significant role in influencing the life cycle and production of lac. The isolation and structure elucidation of lac dye has unraveled its composition as a complex mixture of hydroxy anthraquinones, setting the stage for extensive exploration of its potential biological activities. Of particular interest are the laccaic acids within lac dye, which can be successfully isolated and identified through the application of sophisticated instrumental techniques such as HPLC, LC–MS, NMR, and mass spectroscopy. These analytical tools enable a comprehensive understanding of the chemical composition and structural intricacies of laccaic acids. As we delve into the wealth of knowledge surrounding lac dye and its constituents, the potential for diverse biological activities becomes apparent. The versatility of lac dye, coupled with advancements in analytical techniques, opens avenues for future research exploring its therapeutic applications. This review underscores the importance of continued investigations into the biological potential of lac dye and its components, offering insights that may pave the way for innovative applications in medicine, agriculture, and various other fields. Ultimately, the dynamic interplay between traditional knowledge and contemporary scientific methodologies positions lac as a valuable resource that warrants further exploration.
Future Perspective
The exploration of lac dye and its bioactive constituents, notably laccaic acids, presents a promising avenue for future research, particularly in the realm of therapeutics. With structural similarities to Adriamycin, laccaic acid has shown anticancer properties, warranting indepth investigation and evaluation for its efficacy in cancer treatment. The prospect of investigating lac dye for its antiangiogenic effects adds an exciting new dimension, suggesting the possibility of a multimodal anticancer approach. This area of research holds promise for uncovering novel mechanisms and therapeutic strategies for cancer treatment.
Existing literature also underscores a spectrum of other bioactivities of lac dye, including antioxidant, anti-inflammatory, antimicrobial, antidiabetic, antifungal, and antiviral properties. However, the depth of our understanding is still in its early stages, leaving substantial room for further exploration and elucidation of lac dye’s full range of potential applications. As we venture into the future, comprehensive review articles can serve as a valuable resource to consolidate existing knowledge, highlight research gaps, and guide future investigations across various medical domains. The potential of lac dye, with its rich array of bioactive constituents, awaits further exploration and holds promise for advancing our understanding of its applications in medicine. Interdisciplinary studies that integrate nanotechnology and biotechnology with lac dye research could lead to innovative products with unique properties, thereby broadening the scope and market for this natural dye. Addressing these research gaps will not only revitalize the traditional lac dye industry but also pave the way for cutting-edge applications in various sectors.
Footnotes
Abbreviations
%: Percent; °C: Degree celsius; µg: Microgram; cm: Centimeter. EC50: Half Maximal effective concentration; mg: Milligram; mL: Milliliter; mM: Millimolar; NMR: Nuclear magnetic resonance; PDA: Photodiode array; PubMed: Public/Publisher Medline; TLC: Thin layer chromatography
Acknowledgments
The author would like to acknowledge the Department of Pharmaceutical Sciences and Technology at Birla Institute of Technology, Mesra, for providing the necessary facilities to conduct this review. Additionally, we appreciate the constructive comments from the anonymous reviewers, which greatly improved the quality of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
