Abstract
The importance of replacing synthetic pigments with natural types is increasing day by day in the food industry due to the harmful effects of some synthetic pigments. Microorganisms are a major source of natural pigments, which nowadays have attracted the attention of researchers. In this study, carotenoid pigments were produced by
Keywords
Introduction
Nowadays, food consumers and patients are seeking natural additives and/or traditional medicines in foods instead of synthetic types. For instance, one in three people in the US has used at least one form of the alternative drug. Plants and microorganisms still represent a large source of natural bioactive compound that might serve as leads for the development of novel drugs. 1
In recent years, numerous studies have been done for the development of isolation methods of known metabolites and reducing the price of novel compounds isolated from microorganisms. It is revealed that natural product chemists and microbiologist are interested in less investigated drug sources, such as some microorganisms, which turned out to be a large unutilized supply of metabolic diversity. Hence, the study of biological activity of natural products derived from microorganisms has increased remarkably in recent years due to the demand for the substances having potential pharmaceutical applications or economic value as food additives, cosmetics, drugs, and functional personal-care products.2,3
Carotenoids are one of the important pigments, which are achieved from plant and microbial sources. These pigments that belong to the chemical group known as isoprenoid polyenes are lipid-soluble and yellow-orange-red in color.3,4 Carotenoids derived from plants have various issues, such as high sensitivity to heat, light, pH, and chemicals, and also have some practical difficulties for industrial use, such as low solubility and inconvenient access during the year. Nowadays, researchers seek pigment production from microorganisms in various industries. The rapid growth of pigment-producing microorganisms, inexpensive mediums, relatively easy procedure to extract pigment, independent to the weather conditions, and the wide variety of colors are the benefits of pigment production from microorganisms, in addition to benefits in human and animal health.5,6 Pigments producing microorganisms are completely ordinary in nature. Several microorganisms such as
The risk of the food-borne diseases caused by microbial contamination, the incidence of some chronic diseases such as cancer, sensitivity and inflammation, and common side effects of traditional chemotherapy, indicating a crucial need for new approaches to the control microbial contamination.7,8
Carotenoids have been studied comprehensively and verified to show different beneficial effects on human health through serving as precursors of vitamin A, anti-inflammatory effect, antimicrobial and antioxidant activity (AA), etc.8–12 Also, it has been proven that carotenoids derived from plants (e.g. α-carotene,
13
β-carotene,
14
β-carotene, canthaxanthin, and astaxanthin
15
) and carotenoids extracted from fruits of red paprika (
In recent years, several studies focused on the production and characterization of pigments from
Materials and methods
Microorganism and materials
Condition of pigment production
M. roseus
For inoculum preparation, a loopful of
R. glutinis
A single colony was transferred from the stock culture on YPG (10 g yeast extract, 20 g peptone, and 20 g galactose in 1 L water) agar to 50 mL YPG broth followed by incubation at 27°C for an overnight period. A total of 3 mL of YPG broth was used for inoculation of 100 mL semi-synthetic medium in a 500 mL flask, then incubated in a shaker incubator at 150 rpm and 27°C for 72 h. 25
Pigment extraction and spectrophotometric analysis
Microorganism cells were harvested by centrifugation at 10,000 rpm for 20 min, followed by washing with distilled water and centrifuged again. To extract carotenoid pigments, the method described by Bhosale and Gadre 26 was used with some modification. Briefly, cells were ruptured twice with 10 mL of acetone and broken by homogenizer (Scilogex D500, USA). The suspension was then centrifuged and the supernatant was gathered. The extracts were collected and carotenoid pigments were extracted with the same volume of petroleum ether.
Total carotenoid (TC) was measured spectrophotometrically using the method described by Hornero-Méndez et al. 27 In practice, after dissolving the pigment concentrates in 10 mL acetone, the absorbance was measured by spectrophotometer at 472 and 508 nm and TC was calculated using the following formulas:

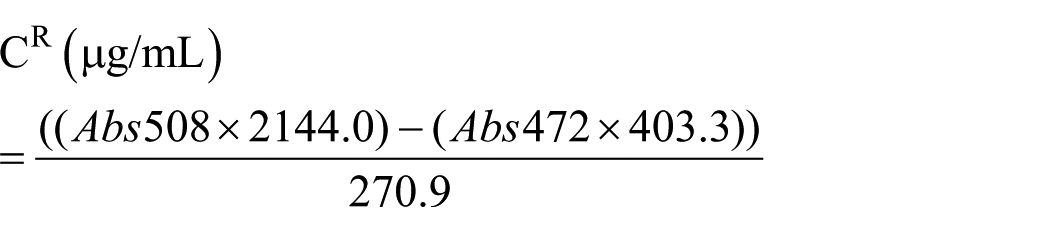

where CY and CR represent the yellow and red isochromic families, respectively.
Microorganisms and inoculum preparation
Antimicrobial activities of PEM and PER were assayed against
Antimicrobial activity
Kirby–Bauer method
The disk diffusion method was carried out to evaluate the antimicrobial activity of PEM and PER. Briefly, an amount of inoculums (0.1 mL) was spread on Mueller–Hinton agar (MHA) (for bacteria) and SDA (for fungi). Sterile paper discs (6 mm in diameter, Sigma-Aldrich Company, USA) were soaked in PEM and PER at concentrations of 0.5, 1.5, 2.5, 3.5, and 5 mg/mL. After incubation of plates at 37°C for 24 h (for bacteria) and 27°C for 72 h (for fungi), the halo of inhibition was measured. Penicillin or gentamicin discs were placed in the plates as a comparative standard. The result was obtained by measuring the microbial free zone area diameter. The experiment was performed three times to minimize the error and the mean values are presented.11,29
Determination of minimum inhibitory concentration (MIC)
Agar dilution method was used to determine MIC values of PEM and PER against some microorganisms. In practice, the pigment at concentrations of 2, 4, 8, 16, 32, 64, 128, and 256 mg/mL was mixed with sterile MHA (for bacteria) and SDA (for fungi), and then 0.1 mL of microbial suspension (0.5 McFarland) was plated using the pour plate method. These plates were incubated at 37°C for 24 h (for bacteria) and 27°C for 72 h (for fungi), and then the MIC of the pigments was evaluated. The MIC was defined as the lowest concentration of PEM and/or PER that prevents visible growth of a bacterium in comparison with positive (medium + microorganism) and negative (medium) control groups. 30
Determination of minimum bactericidal concentration (MBC) and minimum fungicidal concentration (MFC)
The agar dilution method was also used to determine MBC and MFC of PEM and PER. With this difference, bacteria and molds were sub-cultured on nutrient agar (NA) and yeast extract glucose chloramphenicol (YGC) agar, respectively, from the plate containing MIC of the pigment. If the sub-cultured microorganisms cannot grow, the MIC and MBC and/or MFC concentrations will be equal. However, in contrast to the situation, the grown bacteria were sub-cultured in MHA (for bacteria) and SDA (for fungi) containing further pigment concentrations (8, 16, 32, 64, 128, and 256 mg/mL) and the MBC was considered as a concentration of the pigment that bacteria did not grow.11,29
Antioxidant and free radical scavenging activity
Radical scavenging activity
PEM and PER were evaluated for their radical scavenging capacity. In practice, 1 mL of DPPH+ solution (0.3 mM) in ethanol: hexane (equal proportions) was mixed with 1 mL pigment extract (100 µg/mL) in ethanol: hexane. This blend was agitated and kept at 21°C in a dark place for 15 min and then the absorbance was measured spectrophotometrically at 540 nm using an ultraviolet visible (UV-Vis) spectrophotometer (Shimadzu, Japan). The inhibition activity was calculated by the following formula: 30

Abssample: absorbance value of pigment extract plus DPPH
Abscontrol: absorbance value of DPPH solution
Abscontrol: absorbance value of pigment solution
β-carotene bleaching test
Evaluation of AA by β-carotene bleaching test was carried out using the method described by Conforti et al. 31 In practice, emulsion of β-carotene was prepared using the above method and 5 mL of this emulsion was transferred to test tubes containing a 0.2 mL sample of ethanol (70%) at various concentrations. The emulsion containing 0.2 mL ethanol was used as the control. A solution of propyl gallate was used for comparison. After keeping the tube at 45°C for 60 min, the absorbance of the samples was measured spectrophotometrically at 470 nm using a UV-Vis spectrophotometer against a blank (emulsion without β-carotene). The practice was done at the first time and then at 30 and 60 min. The AA was expressed as successful bleaching β-carotene using the following formula:

where A0 and A0 represent the absorbance values at the first time for sample and control, respectively. “t” is time of incubation.
Bovine brain peroxidation
The lipid peroxidation activity was evaluated using the thiobarbituric acid (TBA) test. PEM and PER were evaluated for their antioxidant activity versus liposomes that were supplied from bovine brain extract in phosphate-buffered saline (PBS) (5 mg/mL). The absorbance was measured at 532 nm and the inhibition of lipid peroxidation was calculated using the following formula:
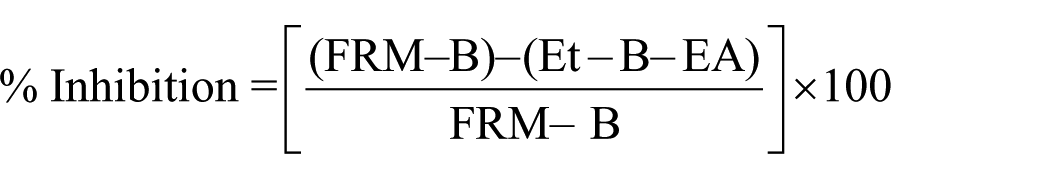
Anticancer activity
In vitro Epstein–Barr virus (EBV) early antigen
The experiment was performed using the method described by Maoka et al.
16
with some modifications. In brief, Raji cells (indicator cells) were grown to a concentration of 106 cell/mL, gently centrifuged, and the pellet was resuspended in medium containing n-butyrate (4 mM), as inducer, 12-
In vivo carcinogenesis
This test was carried out on mice skin papillomas promoted by TPA. The mice used in this study were divided into three experimental classes (each including 10 mice aged 6 weeks). Mice were locally treated with 100 µg 7,12-dimethylbenz[a]anthracene (DMBA) as a promoter in 0.1 mL acetone through a shave in their backs. One week after beginning, papilloma forming was promoted twice a week by using 1 µg TPA in acetone to the skin of mouse. Classes I and II were locally treated with PEM (85 nmol) and PER (85 nmol) in acetone (0.1 mL), respectively, 1 h before the TPA treatment. Class III was locally treated alone with TPA. Papillomas and the incidence were evaluated weekly for 15 weeks. 16
Anti-inflammatory activity
The topical anti-inflammatory activity was studied as inhibition of the TPA-induced ear edema in mice. Mice (25–30 g aged 6 weeks) were held for 1 week before the experiment at 21±°C. In practice, a solution of TPA (1 µg) in acetone (20 µL) was applied to the inner surface of the right ear (1 cm2) by a micropipette. Control mice received only TPA, whereas other animals received the irritant together with PEM or PER. Ear thickness was gauged before and 6 h after the treatment. The inhibitory ratio (%) was calculated by the following formula: 32

Edema A is induced by TPA alone (
Edema B is induced by TPA plus sample (
Each of the above amounts was the average of individual measuring from five mice. ID50 values were determined by the method of probit-graphic interpolation.
Statistical analysis
All experiments and measurements were carried out in triplicate. All statistical analyses were performed using Minitab® version 16.1.1 (Minitab Inc., USA. 2010). Data from the experiments were subjected to Student’s
Results
Pigment production
Table 1 shows TC, yellow, and red isochromic fractions contents of PEM and PER. A significant difference (
TC, yellow, and red isochromic fractions contents of PEM and PER (μg/g).

Means (± SD) within a column with the different sign letters are significantly different (
Means (± SD) within a column with the different sign letters are significantly different (
Antimicrobial activity
Kirby–Bauer method
Given the results, PEM and PER had an antimicrobial effect on all examined bacteria, so that the effect was increased by increasing pigment concentration, as is shown in Table 2. The antimicrobial activity of PER against all pathogenic bacteria was higher than PEM.
Average diameter (mm) of microbial free zone area of PEM and PER.

G, antibiotic use of gentamicin; P, antibiotic disc of penicillin.
Determination of MIC
MIC of both pigments for molds was remarkably more than bacteria, so that the highest MIC was observed for
MIC of the PEM and PER.
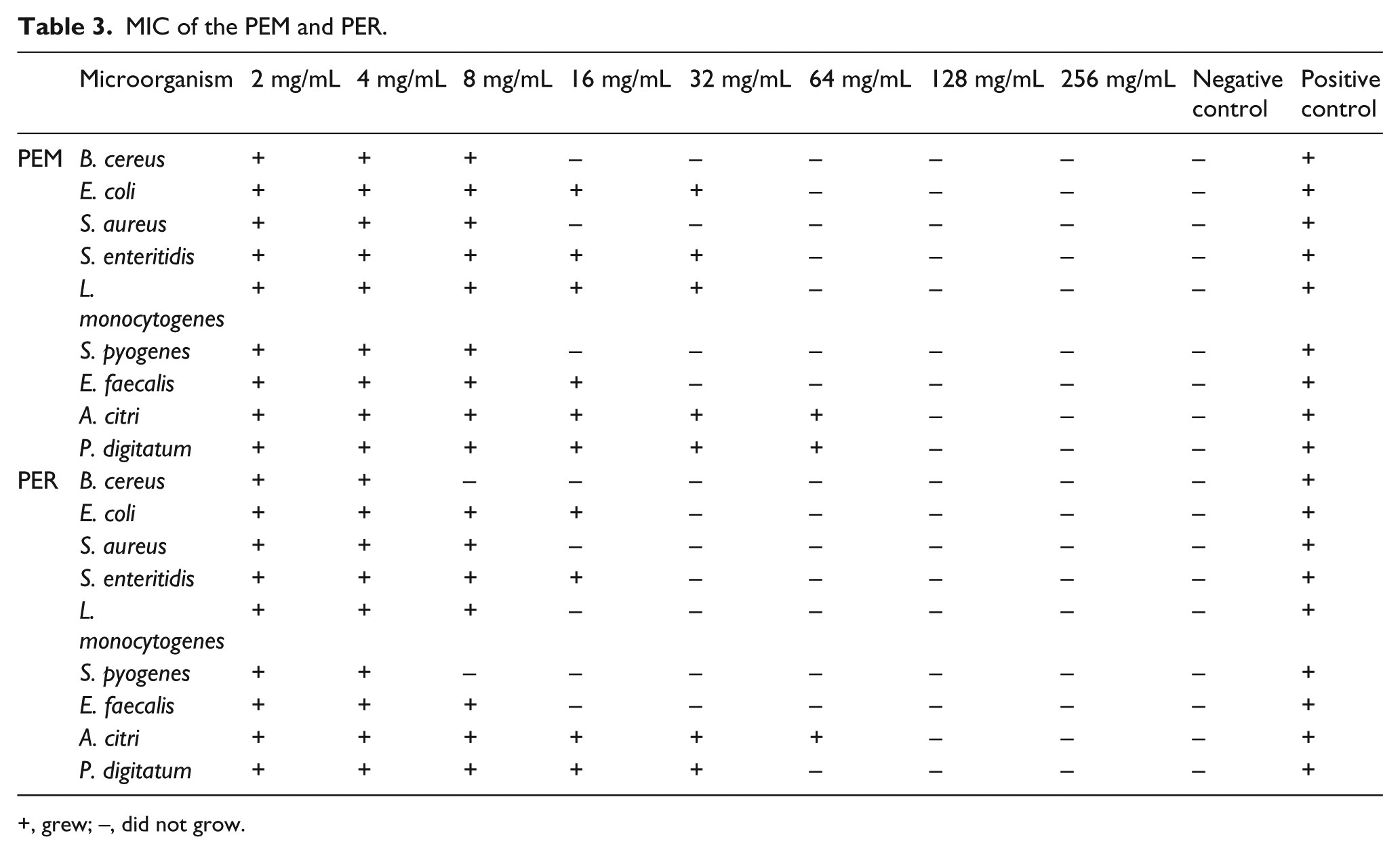
+, grew; −, did not grow.
Determination of MBC and MFC
MBC and MFC of PEM and PER are shown in Table 4. At the used concentrations of PEM and PER, a bactericidal effect was observed for both pigments against all bacteria. At the applied concentrations of PEM and PER, a fungicidal effect was observed for PER against
MBC and MFC of PEM and PER.

G, antibiotic dose of gentamicin; P, antibiotic disc of penicillin.
Antioxidant and free radical scavenging activity
Table 5 shows IC50 values of antioxidant activities of PEM and PER. The antioxidant and free radical scavenging activities of PER were higher compared to PEM.
IC50 values of antioxidant activities of PEM and PER.

Means (± SD) within column with the different sign letters are significantly different (
Means (± SD) within column with the different sign letters are significantly different (
In vitro and in vivo anti-tumor-promoting
Table 6 shows the inhibitory effects of PEM and PER on the activation and viability of the Raji cells so that the inhibitory effect was increased by increasing concentration (mol ratio/TPA). Figure 1 shows the inhibition of TPA-induced tumor promoting by PEM and PER.
Relative ratio of EBV-EA activation with respect to positive control (100%) in the presence of PEM and PER.

Values represent relative percentage of the positive control value (100%).
TPA concentration was 20 ng (32 pmol)/mL.
Values in parentheses are viability percentages of Raji cells.
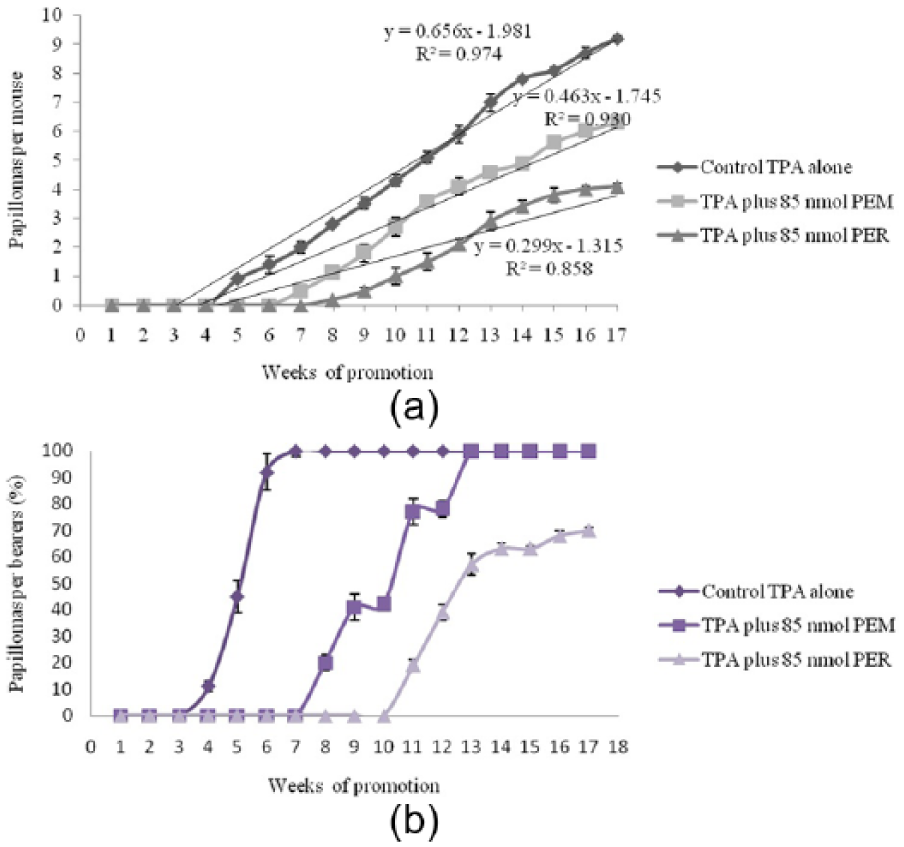
Inhibition of TPA-induced tumor promoting by PEM and PER. (a) Percentage of mice bearing papillomas; (b) average number of papillomas/mouse.
Anti-inflammatory activity
The TPA-induced anti-inflammatory activities of PEM and PER on mice are shown in Table 6. According to the result, the anti-inflammatory activity of PER was higher than PEM.
Discussion
Before the study of biochemical properties, measuring the amount of carotenoids is important. Therefore, according to Table 1, PER had significantly (
PER had more antimicrobial activity compared to PEM. This fact could be related to the type of carotenoids that were produced by each microorganism. Siva et al. 42 studied the antimicrobial properties of eight food dyes against ten bacteria and five fungal organisms and reported that red dyes showed best antibacterial activity, while yellow dyes showed better antifungal activity.
The results of MIC assay revealed that PEM and PER had more antibacterial effects against gram-positive bacteria than gram-negative bacteria, so that
PER had less MIC for all the microorganisms except
PER had bactericidal and fungicidal effects on all tested bacteria and fungi; however, the opposite is true for PEM, so that no fungicidal activity against
Carotenoids are pigments that play a major role in the protection of pigment-producing microorganisms against some environmental shocks such as decreasing temperature. This role could be due to some features of carotenoids such as AA and membrane fluidity increase.
48
According to the results of this study, PEM and PER had antioxidant and free radical scavenging activities (Table 5). The effect of antioxidants on DPPH radical scavenging was believed to be because of their hydrogen-donating ability. PEM and PER were able to reduce the stable free radical DPPH to the yellow 1,1-diphenyl-2-picrylhydrazyl. DPPH radical scavenging of PER (IC50 = 555.5 ± 0.05 µg/mL) was measured significantly less compared to PEM (
In the β-carotene bleaching test after 30 min of incubation, PER showed the highest inhibition of linoleic acid oxidation (2.5 ± 0.02), even more than the standard sample (IC50 = 1.5 ± 0.05 µg/mL). The AA of the pigment extracts decreased during the reaction time. However, the IC50 of propyl gallate was 1.5 µg/mL after 30 and 60 min incubation. This is probably due to the thermal sensitivity of carotenoids so that the AA of carotenoid was reduced at high temperatures. 48
Using liposomes prepared from bovine brain, the highest AA was observed for PER (IC = 14.4 µg/mL), which was significantly more compared to PEM (
The differences in the AA of PEM and PER could be attributed to the higher β-carotene content of PER, since this carotenoid is known to exert significant AA.49–51 Hernández-Ortega et al.
8
studied antioxidant, antinociceptive, and anti-inflammatory effects of carotenoids extracted from dried pepper (
In vitro anti-tumor promoting activities of PEM, PER, and β-carotene as well as a strong anti-tumor-promoter, 52 were examined using the EBV activation assay in Raji cells. PEM and PER showed an inhibitory effect on EBV-EA induction without significant cytotoxicity on Raji cells in this test. PER showed notably inhibitory effect at 1000 mol/TPA ratio. Furthermore, at 10 and 100 mol/TPA ratio, PEM was more active than β-carotene, whereas the opposite is true at the high mol/TPA ratios (Table 6). Wang et al. 7 studied some biological activity of lutein and their results provided scientific evidence for the safe use and health beneficial effects of lutein.
The in vivo anti-tumor-promoting activities of PEM and PER were evaluated by a two-stage carcinogenesis method for mouse skin papillomas promoted by TPA. The incidence (%) of papilloma-bearing mice and the average number of papillomas per mouse are shown and compared with a positive control (initiated with 390 nmol of DMBA and promoted with TPA) in Figure 1. When PEM (85 nmol) and PER (85 nmol) were applied before each TPA treatment, they notably hinder the formation of papillomas and reduced the number of papillomas per mouse, as shown in Figure 1. In the positive control mice, the first papillomas emerged in the fifth week of promotion, and in mice treated with PEM and PER in the seventh and eighth weeks, respectively. After 7 weeks of promotion, the positive control mice presented a complete incidence of papillomas, while in mice treated with PEM the complete incidence was observed at the 13th week. As an interesting result, the complete incidence papilloma was not observed for the group treated with PER, so that even at week 17 only 70% of mice bore papillomas. After 17 weeks of promotion, 9.2, 6.3, and 4.1 papillomas were found per mouse in the positive control mice and the groups treated with PEM and PER, respectively. Previous researchers reported a direct relationship between the anticancer and antioxidant activities for carotenoids.7,53,54 Also in this study, a close correlation was found between these two properties for the extracted pigments. Both antioxidant and anticancer activities were higher for PER compared to PEM. Nesaretnam et al. 55 reported that rats that were fed palm-oil diets have a lower incidence of mammary cancers than those that were fed other dietary fats; these results were partly related to the presence of carotenoids or tocopherols and tocotrienols.
PEM and PER extracts were assessed with respect to their anti-inflammatory activity against TPA-induced inflammation in mice and compared with a commercially available anti-inflammatory drug (indomethacin). As shown in Table 6, both pigment extracts remarkably hinder the TPA-induced inflammation, so that ID50 of PEM and PER were measured as 0.22 and 0.09 mg/ear, respectively, which was more inhibitory than indomethacin (ID50 = 0.33 mg/ear). Nam et al. 12 studied the anti-inflammatory effects of crocin and crocetin (natural carotenoids mainly found in the crocus flower) in rat brain microglial cells and reported that crocin and crocetin provide neuroprotection by reducing the production of various neurotoxic molecules from activated microglia. Heo et al. 56 suggested that using fucoxanthin (a xanthophyll, which is one of two major divisions of the carotenoid group) could be a useful therapeutic approach for the various inflammatory diseases.
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors thanks Research’s Deputy of Baqiyatallah Medical Sciences University for providing the cost of this project and help to the implementation of this project, which adopted of the Research Council of Health and Nutrition Research Center.
