Abstract
Background
A protozoan disease termed coccidiosis is common worldwide and affects both domestic and wild animals. New alternatives must be discovered because traditional coccidiostats are becoming less effective. More recently, nanomedicine has been a better option for treating a wide range of diseases.
Objectives
This study aimed to investigate the role of biosynthesized selenium nano-particles (Bio-SeN) utilizing neem leaf extract on the outcome of renal damage caused by murine coccidiosis.
Materials and Methods
In this study, 25 mice were employed, and five groups (five mice/group) were formed: Group 1, this group is considered as control; Group 2, this group was noninfected and received Bio-SeN for 5 days; and Groups 3, 4, and 5: infected groups received 103Eimeria papillata oocysts. Following that, for 5 days, Group 4 was administered Bio-SeN, while Group 5 was given amprolium (120 mg/kg). On the fifth day post-infection (p.i.), oocyst output, as well as body weight, was recorded, and mice were euthanized so that blood could be drawn for biochemical examination. The kidneys were also removed to prepare histological sections and homogenate for the determination of oxidative damage markers.
Results
The results demonstrated that by day 5 p.i., mice treated with Bio-SeN after infection with E. papillata had significantly reduced oocyst output in their feces. Body weight gain was significantly reduced by Eimeria infection to −6.07%, and it improved to −3.64% with Bio-SeN treatment. Significant degenerative alterations in the form of vacuolation and inflammatory infiltration in the renal tubules, together with the presence of erythrocytes in some blood sinusoids, were linked to infection. Furthermore, the infected mice’s protein and carbohydrate contents were disturbed by the infection. Urea, creatinine, and uric acid levels in the kidneys significantly increased as a result of the Eimeria infection. In the infected mice, these alterations result in oxidative damage. During the Bio-SeN treatment, all of these infection-induced parameters show significant changes.
Conclusion
Overall, our findings show that Bio-SeNs significantly exhibit a significant in vivo renal response against murine coccidiosis.
Introduction
A common intestinal infection known as coccidiosis is brought on by unicellular eukaryote parasites belonging to the Eimeria genus (Apicomolexa, Eimeriidae) (Pakandl, 2009). It represents a continuous problem for wild and domestic animals. This parasite species infects hosts through the fecal-oral pathway, when its highly host-specific oocysts are released (Madlala et al., 2021). Rapid intestinal epithelium multiplication by Eimeria species can result in decreased feed intake and the rate of absorbing nutrient materials, and decreased body-weight gain, which is associated with dehydration and an increased risk of developing additional diseases (Abbas et al., 2017). Eimeria papillata is a model coccidian parasite that causes numerous pathological alterations in mice’s jejunum (Alajmi et al., 2023). Anticoccidial medication is often required for conventional disease control to avoid the spread of infectious oocyst stages (Tewari & Maharana, 2011). Medications for coccidiosis management include amprolium, nicarbazin, nitrofurazone, and sulfaquinoxaline (Lan et al., 2017). Due to the widespread use of these medications, adverse effects on the health of animals and drug resistance have emerged (Philippe et al., 2014).
Recently, research studies focused on the usage of nanotechnological tools for the treatment and control of infectious diseases (Abdel-Gaber, Hawsah, Al-Otaibi et al., 2023; Abdel-Gaber, Hawsah, Al-Shaebi et al., 2023; Alkhudhayri et al., 2018, 2020; Chen et al., 2023; Rezaei et al., 2019). Selenium (Se) is a micronutrient necessary for the appropriate functioning of all tissues and the healthy body (Kieliszek, 2019). It is also a cofactor of several enzymes (e.g., glutathione peroxidase (GPx) and thioredoxin reductase) (Zoidis et al., 2018). Selenium nanoparticles (SeN) represent one of the nanotechnological approaches that are used in the utilization of biological agents (i.e., plant extracts) that have been developed as eco-friendly alternatives to chemical methods (Hosnedlova et al., 2018). SeN shows promising potential as a drug carrier in biological applications (Forootanfar et al., 2014). SeN has also been reported to have antiparasitic activities against protozoan infections such as Eimeria papillata (Alkhudhayri et al., 2018, 2020).
Neem, or Azadirachta indica, is a tree in the Meliaceae family of mahogany. As one of the most promising therapeutic plants, it possesses a variety of biological properties, particularly those of antifungal, antibacterial, anti-inflammatory, and anti-ulcer ones (Herrera-Calderon et al., 2019; Lee et al., 2017; Pillai et al., 1984; Sudan et al., 2020). Neem leaf aqueous crude extract has been reported to exhibit significant hepatoprotective, cardioprotective, hypoglycemic, and hypolipidemic properties (Bopanna et al., 1997; Chattopadhyay et al., 1992; Peer et al., 2008). Furthermore, neem leaf extract has been shown to have a protective effect against diabetic nephropathy in rats (Oluwole Busayo et al., 2011). In addition, neem has good antiparasitic action against some protozoans like Plasmodium falciparum (Khalid et al., 1986), Plasmodium berghei (Habluetzel et al., 2019), Leishmania amazonensis (Carneiro et al., 2012), Trypanosoma brucei (Mbaya et al., 2010), and Eimeria papillata in mice (Dkhil et al., 2013).
It has not yet been documented, meanwhile, how SeN, either by itself or in conjunction with neem, protects against kidney damage brought on by coccidiosis infection. Moreover, few studies are available until now about the effects of coccidiosis on the renal tissues of infected animals (Abollo & Pascual, 2005; Bennett et al., 2007; Franson & Derksen, 1981; Garijo et al., 2022; Gruber et al., 1996; Hagag et al., 2020; Jankovsky et al., 2017; Leighton & Gajadhar, 1986; Metwaly et al., 2012; Oksanen, 1994). To assess the potential protective effect of biosynthesized SeN generated from neem leaf extract on changes in oxidative stress markers and E. papillata-induced kidney injury in mice, this study was conducted.
Materials and Methods
Plant Collection and Extract Preparation
Neem leaves were collected from Riyadh, Saudi Arabia’s botanical parks. The leaves were identified botanically in the herbarium at King Saud University (Department of Botany and Microbiology, College of Science) with authentication no. 11740. Manikandan et al. (2008) developed a 70% methanolic extract of leaves. Briefly, the collected leaves were ground into a powder (~500 g) with an electric blender. One hundred of the produced powders were mixed with 1,000 mL of 70% methanol at 4°C for 24 hour and then filtered from insoluble impurities. In a Yamato RE300 rotary vacuum evaporator, the extract was dried and concentrated for 2 hours and then kept for later use at −20°C.
Biosynthesized Selenium Nanoparticles (Bio-SeN) Preparation
The synthesis of SeN was performed by using sodium selenite (Nanocs Inc., Boston, MA, USA) as the source for materials, which was subsequently reduced with ascorbic acid and stabilized using polysorbate, according to the method of Vahdati and Tohidi Moghadam (2020). This experiment involved the dissolving of 30 mg of Na2Se2O3 in 90 mL of Milli-Q water. After complete dissolution, 10 mL of 56.7 mM ascorbic acid was added dropwise while being vigorously stirred. Every 2 mL of ascorbic acid formed SeN, and after that, 10 µL of polysorbate was added. Initially, the colloidal solution was mixed by a ratio of 9:1 with neem extract. After 15 minutes of stirring at 45°C, the mixture was cooled to 27°C to precipitate. The color change in the solution showed the production of biosynthesized selenium nano-particles Bio-SeN.
Parasite Passage
Eimeria papillata is a laboratory strain obtained from Prof Heinz Mehlhorn (Heinrich-Heine-Universitat, Germany). The oocysts were propagated through periodic passage in five laboratory mice, Mus musculus with 103 sporulated E. papillata oocysts by oral gavage. On the 5th day post-infection (p.i.), oocysts were isolated from the feces of laboratory mice and suspended in the solution of potassium dichromate (K2Cr2O7) of 2.5% (wt/vol) (Long et al., 1976). To use, K2Cr2O7 was removed by washing thrice in 3 mL of phosphate-buffered saline (pH 7.4) (Sigma Aldrich, Taufkirchen, Germany), and the oocyst suspension was diluted to provide the recommended oral dose for experimental animals.
Mice and Coccidia Infection
Twenty-five male mice (C57BL/6), aged 9–12 weeks and weighing 20–22 g, were acquired from King Saud University’s Pharmacy College’s animal facilities (Saudi Arabia). Mice were kept in specific pathogen-free conditions at a controlled temperature (23 ± 5℃), maintained under a 12/12 hours light–dark cycle. Mice were given water ad libitum and a standard diet. The experiment used mice following the institution’s requirements for the care and use of research animals (approval number KSU-SE-22-66). Mice were randomly allocated into five experimental groups with five mice per group, as follows:
Group 1: This group represents non-infected-non-treated mice gavaged with 100 µL distilled water. Group 2: This group represents noninfected daily orally treated mice group with Bio-SeN with a dose of 0.5 mg/kg for 5 days. Group 3: This group represents infected-non-treated mice inoculated orally with 103E. papillata of sporulated oocysts. Group 4: This group represents infected and daily orally treated mice with Bio-SeN with a dose of 0.5 mg/kg for 5 days; this was done after 60 minutes of infection at the beginning of the experiment with 103E. papillata of sporulated oocysts (according to Abdel-Gaber, Hawsah, Al-Otaibi et al., 2023; Abdel-Gaber, Hawsah, Al-Shaebi et al., 2023). Group 5: This group represents infected and daily orally treated mice with Amprolium with a dose of 120 mg/kg for 5 days; this was done after 60 minutes of infection at the beginning of the experiment with 103E. papillata of sporulated oocysts (according to Abdel-Gaber, Hawsah, Al-Otaibi et al., 2023; Abdel-Gaber, Hawsah, Al-Shaebi et al., 2023).
Both the treated and untreated infected groups’ fresh fecal pellets were obtained. The McMaster chamber was utilized to determine the oocysts per gram of wet fecal pellets (Dkhil et al., 2013). Weight changes in mice were estimated by subtracting the beginning body weight (at day 0 p.i.) from the end body weight (at day 5 p.i.) after each mouse was weighed individually.
Sample Collection
All mice were euthanized with CO2 asphyxiation. Heparinized tubes were used to collect the heart’s blood via cardiac puncture, and plasma was then separated and kept at −20°C for biochemical analysis. Mice kidneys were removed and divided into small pieces. Parts were fixed in 10% buffered formalin for histology, while others were used in the oxidative stress assays and preserved at −80°C.
Histological and Histochemical Preparation
Freshly prepared mouse kidneys were fixed for 24 hours at 27°C in 10% neutral buffered formalin. Following serial dilutions of ethyl alcohol (70%, 80%, 90, 95%, 10 minutes for each) to dehydrate the specimens, they were embedded in paraffin. After that, hematoxylin and eosin (H&E) were used to stain paraffin sections of 5 µm (Drury & Wallington, 1980). Histological damages in renal tissue were scored according to Metwaly et al. (2012). The periodic acid-Schiff’s method developed by Hotchkiss (1948) for total carbohydrates and the mercuric bromophenol blue method developed by Mazia et al. (1953) for total proteins were used to stain the remaining sections of renal tissue. A Leica DM 2500 microscope was used to examine and photograph the slides using NIS ELEMENTS software, version 3.8.
Biochemical Analysis
Plasma biochemical parameters associated with kidney functions (i.e., urea, creatinine, and uric acid) were estimated using commercial kits purchased from Human Gesellschaft (Biochemica und DiagnosticambH, Germany). Urea, creatinine, and uric acid levels were determined using the Berthelot (1859), Schirmeister et al. (1964), and Fossati et al. (1980) methods, respectively. Absorbance was evaluated with an Ultrospec 2000 UV spectrophotometer (Amersham Pharmacia Biotech, Cambridge, UK).
Preparation of Tissue Homogenate
Pieces of the freshly expelled renal tissue were weighed, homogenized in ice with (1:10 w/v) phosphate buffer (0.1 M), and centrifuged for 10 minutes at 4°C at 5,000 rpm. The 10% w/v supernatant was refrigerated until it was used for biochemical assays.
Oxidative Stress Assays
The renal homogenate was used to determine oxidative stress biomarkers such as GPx (Ellman, 1959), malondialdehyde (MDA) (Ohkawa et al., 1979), and nitric oxide (NO) (Green et al., 1982) using recommended protocols of the commercial kits (Bio-Diagnostic Co., Egypt). A spectrophotometer (Ultrospec 2000) was used to measure the absorbance.
Statistical Analysis
A one-way analysis of variance (ANOVA) was used for the analysis. Statistical comparisons across groups were performed using a post hoc test in SigmaPlot® (Systat Software ver. 11, Inc., Chicago, IL, USA). The data had a significant p value of less than 0.05 and was displayed as Mean ± Standard Deviation (SD).
Results
On day five, the number of oocyst shedding was reduced by the Bio-SeN treatment, from 5.130 ± 0.38 × 106 to 1.42 ± 0.87 × 105 oocyst/g feces (Table 1). Similar to amprolium, Bio-SeN exhibited excellent efficacy against E. papillata. No contamination was observed between infected and control mice which was confirmed by the absence of oocysts in their fecal pellets. At 5th-day p.i., weight gains in mice were reduced by Eimeria infection with −6.07% as compared with the control group. Following Bio-SeN treatment, the reduction in body weight was −3.64% (Table 1).
Oocyst Output and Weight Change in Mice at Day 5 p.i. with E. papillata.
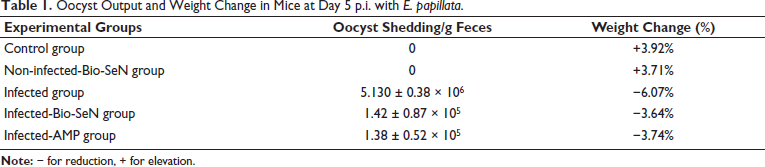
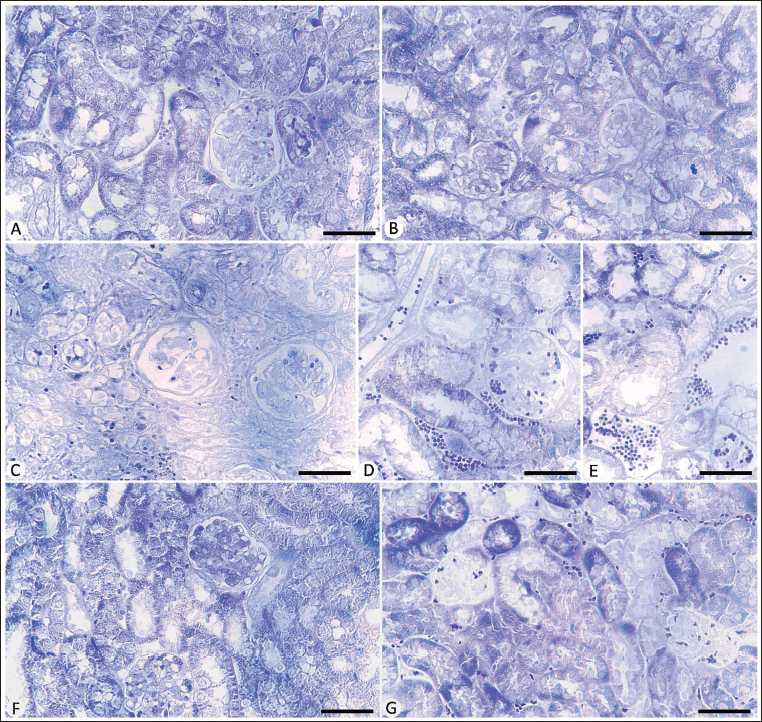
As shown in Figure 1, microscopic examination of kidney sections for the control mice group revealed normal renal tubules and glomeruli. The infected group with E. papillata oocysts showed a moderate pathological change in the renal tissue as indicated by degenerative alterations in the renal tubules, such as vacuolation and inflammatory infiltration (Figure 1 and Table 2). Moreover, it appears certain blood sinusoids were erythrocyte-filled. This renal damage was diminished when mice were treated with Bio-SeN (Figure 1 and Table 2).
Histological Section for Renal Tissue in Different Experimental Mice Groups Stained with Hematoxylin and Eosin (H&E). (A) Noninfected Mouse Kidney. (B) Noninfected-treated with Bio-SeN Mouse Kidney. (C) Infected Mouse Kidney on Day 5 p.i. (C) Infected-treated Kidney with Bio-SeN. (D) Infected-treated Kidney with AMP. Sections Stained with Hematoxylin and Eosin. Scale Bar = 50 µm.
Histopathological Score for the Renal Tissue Damages Induced by E. papillata.
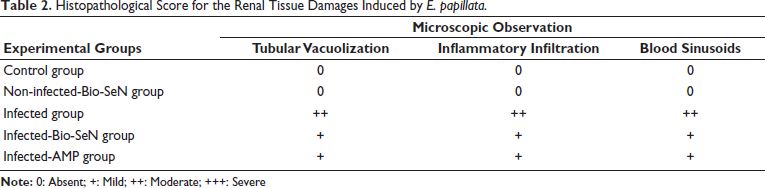
Microscopic examination revealed a depletion in cytoplasmic glycogen within the renal tissue after E. papillata infection, with reduced density of the basement membranes, and brush margins of the renal tubule (Figure 2). Also, the glomeruli showed lower positivity levels than the control mice group. Oral Bio-SeN administration caused improvement in the renal tissue structure (Figure 2).
Carbohydrate Content in Kidney Sections Stained with Periodic Acid Schiff’s (PAS) Method. (A) Control Noninfected Kidney with Normal Content. (B) Noninfected-treated Kidney with Normal Content. (C) E. papillata Infected Kidney with Depletion in Their Carbohydrate Content. (D and E) Infected Treated Mice (Bio-SeN and AMP, Respectively) with Improvement in Their Level. Scale Bar = 50 µm.
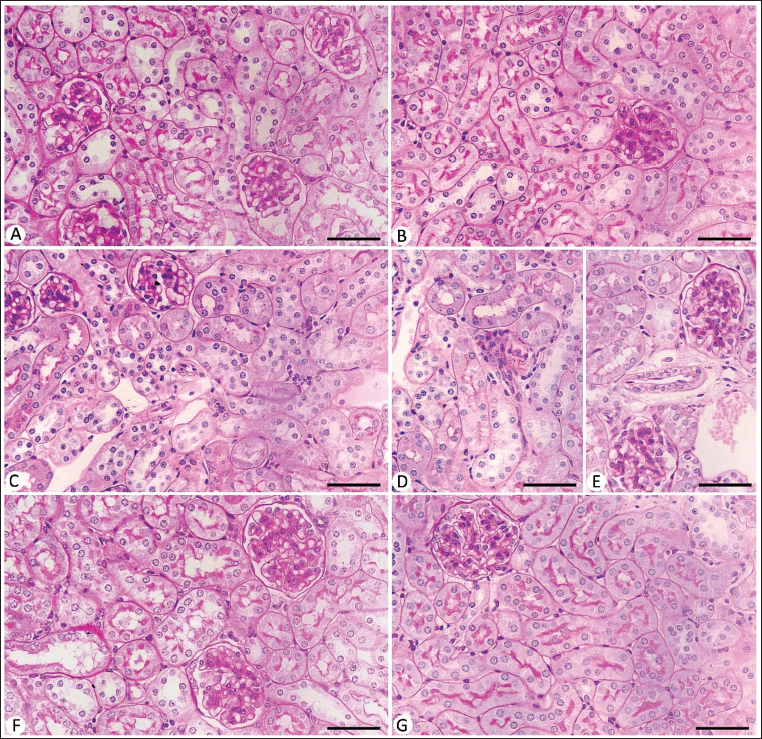
Because of a positive affinity for bromophenol blue staining, the control kidney tubules and glomeruli proteins displayed uniformly packed blue-colored granules (Figure 3). The total protein content in the kidney tubules as well as glomeruli was reduced in the infected mice group. Treatment with Bio-SeN showed improvement in the histological structure of the renal tissue (Figure 3).
Protein Content in Kidney Sections Stained with Mercuric Bromophenol Blue Method. (A) Control Noninfected Kidney with Normal Content. (B) Noninfected-treated Kidney with Normal Content. (C) E. papillata Infected Kidney with Depletion in Their Protein Content. (D and E) Infected Treated Mice (Bio-SeN and AMP, Respectively) with Improvement in Their Level. Scale Bar = 50 µm.
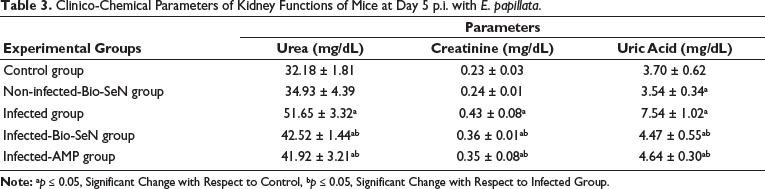
Levels of kidney function including urea, creatinine, and uric acid were significantly elevated due to Eimeria infection and reached 51.65 ± 3.32 mg/dL, 0.43 ± 0.08 mg/dL, and 7.54 ± 1.02 mg/dL, respectively. Upon treatment, Bio-SeN inhibited these elevations to be 42.52 ± 1.44 mg/dL (urea), 0.36 ± 0.01 mg/dL (creatinine), and 4.47 ± 0.55 mg/dL (uric acid) (Table 3). So, the Bio-SeN ameliorated the levels of kidney functions from 1.60 to 1.32 fold (urea), 1.86 to 1.56 (creatinine), and 2.03 to 1.20 fold (uric acid), which were normalized to the control group.
Clinico-Chemical Parameters of Kidney Functions of Mice at Day 5 p.i. with E. papillata.
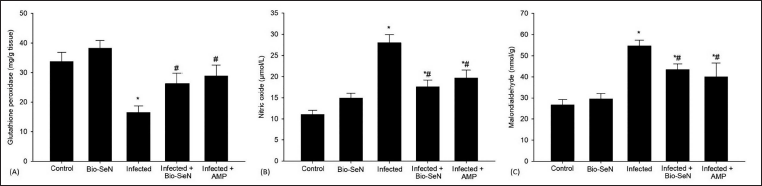
The antioxidant system was significantly altered by Eimeria infection. When mice were given Bio-SeN orally, the antioxidant system in their kidney tissue significantly improved. Renal tissues showed a higher GPx of 26.26 ± 3.50 mg/g tissue (Figure 4). Also, following Bio-SeN treatment, there was a significant decrease in the oxidative damage indicators in the infected renal tissue. The level of NO was reduced to 17.57 ± 1.62 µmol/L (Figure 4). MDA content was decreased to 433.93 ± 26.70 nmol/g after Bio-SeN treatment (Figure 4).
Discussion
Among the intestinal protozoan diseases that impact several animal species is coccidiosis (Shivaramaiah et al., 2014). Previous studies, with particular attention to the intestine location of infection, examined the potential of natural resources to combat mice coccidiosis instead of using coccidiostats (Metwaly et al., 2015). Nonetheless, there is a reciprocal interaction between the gastrointestinal (GI) tract and the kidneys. The effects of coccidiosis on the renal tissues of infected animals are the subject of very few research studies (Abollo & Pascual, 2005; Bennett et al., 2007; Franson & Derksen, 1981; Garijo et al., 2022; Gruber et al., 1996; Hagag et al., 2020; Jankovsky et al., 2017; Leighton & Gajadhar, 1986; Metwaly et al., 2012; Oksanen, 1994). Thus, this study assessed the protective function of Bio-SeN, employing an extract from neem leaves, against the renal damage resulting from murine coccidiosis.
Our findings revealed that Bio-SeN demonstrates anti-coccidial activity, as indicated by the reduction in E. papillata oocysts output in the infected mice’s feces on day 5 p.i.. This lower yield indicates that oocysts, which are comparatively inactive, may develop and eventually be discharged. Bio-SeN hindered the parasites’ ability to develop in the host through the interaction with the oocyst membrane, which affected the permeability of the cellular components and ultimately caused death in Eimeria stages. The occurrence of phytochemicals in neem leaves extracts, such as alkaloids and saponins, has a significant impact on parasites as the species of Eimeria (Biu et al., 2006; Dkhil et al., 2013; Eze et al., 2020; Gotep et al., 2016; Ishaq et al., 2022; Toulah et al., 2010), and the therapeutic activity of SeN, which might be attributed to the elevation of the immune response for the infected mice against Eimeria species (Abdel-Gaber, Hawsah, Al-Otaibi et al., 2023; Abdel-Gaber, Hawsah, Al-Shaebi et al., 2023; Alkhudhayri et al., 2020; Frölich et al., 2012), has previously confirmed that Bio-SeN possesses anticoccidial activity.
In this study, the group of infected mice had a decrease in body weight gain. This result is in line with previous studies (Ahmadi et al., 2022; Alamari et al., 2024; Anwar et al., 2008; Le Sueur et al., 2009; Metwaly et al., 2012; Thagfan et al., 2017) that attributed the problem to coccidia infection, which lowers food intake and decreases nutrient digestion, causing infected animals to gain less body weight. Furthermore, as noted by Kiortsis and Christou (2012), losing weight is considered a crucial intervention to lower the prevalence of renal impairment. The mice’s body weight improved as a result of Bio-SeN restoring their nutritional status. Their mode of action is related to the sporozoite membrane’s degradation and the subsequent loss of calcium ions from the Eimeria parasite, which is essential for invasion, replication, and development. According to Abdel-Gaber, Hawsah, Al-Otaibi et al. (2023), the presence of bioactive constituents in the crude neem leaf extract is responsible for this activity.
According to the study’s histological analysis, the renal tubules’ epithelium had moderate degenerative alterations following the E. papillata infection. This outcome was in line with the findings of Hagag et al. (2020), who noted that inflammatory cells infiltrated the renal tubules and that degenerative alterations occurred following E. tenella infection in broiler chickens. Additionally, inflammatory cell infiltration of the intertubular tissues was discovered by Metwaly et al. (2012) in an experiment aimed at mitigating damage in rabbits infected with E. coecicola. Furthermore, in line with our findings, Ebaid et al. (2007) mentioned that renal tubules exhibited cytoplasmic vacuolation as a result of disruptions in lipid inclusions, with fat metabolism primarily taking place in pathological cases. As the oxidative stress status was reduced, Bio-SeN had a protective impact against renal injury. This result is in line with the outcomes of Al-Brakati et al. (2021) and Hosseini et al. (2023), who found a positive enhancement following the administration of SeN to rats for the affected renal tissue.
In this study, the renal tissue of the infected mice showed a decrease in carbohydrates. This result was in line with the findings of Ebaid et al. (2007), who suggested that the decreased carbohydrate content could result from elevated stress on the organs, which raised energy consumption and allowed equalized pressure to be applied to them. Following treatment, Bio-SeN regulates the renal tubule’s ability to actively reabsorb glucose from the blood by bringing the carbohydrate level back to normal. This hypothesis is associated with the dual action of both neem and SeN, as (a) the primary active compound, azadirachtin, in the crude neem leaves extract plays a role in the reduction of glucose-6-phosphatase enzyme activity which interrupts the feed ability for a parasite, and (b) SeN role on the losing of the essential ions for the replication of the Eimeria parasite.
There was a significant reduction in the total protein within the renal tissue of mice infected with E. papillata parasite in this study. This result is consistent with reports by Al-Saeed et al. (2017), El-Maksoud et al. (2014), and Hagag et al. (2020) indicating that hypoproteinemia is linked to malnutrition and nutritional malabsorption in chronic renal diseases. Furthermore, Ebaid et al. (2007) postulated that the target material undergoes considerable lysis and dissolution because of the disruption of lysosomal membranes by different toxicants, which release their hydrolytic enzymes into the cytoplasm, leading to a reduction in protein. Because of the active ingredients in neem leaf extract and the therapeutic activity of SeN, which work together to block the activity of the Eimeria parasite, Bio-SeN recovered renal content after treatment.
According to our research, there was a significant increase in urea, creatinine, and uric acid, which negatively impacted the kidney’s ability to secrete fluid and caused damage to the renal parenchyma as a result of the Eimeria infection. These results are consistent with those of Whealton et al. (1994) and Hochleithner (1994), who discovered urea and creatinine retention caused by glomerular filtration dysfunction and might account for elevated plasma levels in the infected group. Furthermore, Maiuolo et al. (2016) revealed that the elevated level of uric acid resulted from the catalytic reaction of protein, and then excreted by tubular excretion. The use of Bio-SeN restored normal levels of urea, creatinine, and uric acid, hence improving kidney function. This is in line with the results of Sithisarn et al. (2005), who suggested that the crude neem leaf extract’s antioxidant and free radical scavenging properties may be crucial in preventing renal damage in mice infected with PbANKA. Furthermore, Dkhil et al. (2013) found that azadirachtin indicated anticoccidial properties against E. papillata-induced infection in mice. This might be because the plant protects against renal damage after infection with Eimeria parasite. The ethanolic neem leaf extract provided a protective effect on rats’ kidney functions, as documented by Seriana et al. (2021).
According to the current study, the Eimeria infection increased NO and MDA while lowering GPx levels, causing oxidative stress and the disruption of antioxidant/prooxidant equilibrium in the renal tissue. These results validate that oxidative stress triggers a range of biochemical changes that modify the maintenance of homeostasis. This is in line with the results of Raso et al. (2001), who observed that elevated activity of the inducible nitric oxide synthase (iNOs) enzyme indicated that NO was enhanced due to the infection. Furthermore, Cedric et al. (2018) noted that the inability of the antioxidant defense mechanism to stop the formation of excessive free radicals and enhanced membrane peroxidation, which damages tissues, were the main causes of the increase in MDA levels in organs induced by infection. Furthermore, it was reported by Griss et al. (2019) and Khatlab et al. (2019) that renal coccidiosis in geese significantly increased MDA expression in the presence of several Eimeria parasite life forms. Alajmi et al. (2023) stated that GPx depletion is essential for shielding the animal body from any damage caused by free radicals during the parasitic infection with E. papillata. By bringing the values of GPx, NO, and MDA back to normal, the administration of Bio-SeN decreased the levels of oxidative enzymes and improved the antioxidant capacity of mice’s renal tissue. Previous studies have attributed the antioxidative property of Bio-SeN to active ingredients in the crude neem leaves extract (i.e., nimbin, azadirachtin, nimbidiol, quercetin, and nimbidin) (Abdel-Gaber, Hawsah, Al-Shaebi et al., 2023; Abdel Moneim et al., 2014; Dkhil et al., 2013; Manikandan et al., 2008; Xiang et al., 2018) as well as those activities of SeN (Abdel-Gaber, Hawsah, Al-Otaibi et al., 2023; Alkhudhayri et al., 2020). Furthermore, the crude neem extract could scavenge free radicals, which protects against oxidative stress, according to Kiranmai et al. (2011).
Conclusion
Our findings demonstrate that the administration of Bio-SeN led to a significant enhancement in renal damage, as evidenced by a decrease in pathological alterations and a decrease in oxidative damage indicators with a great activation of antioxidant biomarkers. Therefore, Bio-SeN should be added as a feed additive to enhance renal health status.
Footnotes
Abbreviations
Bio-SeN: Biosynthesized selenium nanoparticles; GI: Gastrointestinal; GPx: Glutathione peroxidase; H&E: Hematoxylin and eosin; iNOs: Inducible nitric oxide synthase; MDA: Malondialdehyde; NO: Nitric oxide; PAS: Periodic acid Schiff’s; p.i.: Post-infection; SeN: Selenium nanoparticles.
Acknowledgments
This study was supported by the Researchers Supporting Project (RSP2024R25), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The animal study was approved by Research Ethics Committee (REC) at King Saud University (approval number KSU-SU-22-66). The study was conducted in accordance with the local legislation and institutional requirements.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Researchers Supporting Project (RSP2024R25), King Saud University, Riyadh, Saudi Arabia.
