Abstract
Background
This study aims to explore the use of polystyrene nanoparticles as a carrier to encapsulate astragalus polysaccharides and regulate the glycolytic pathway of the DNAX-activating protein of 12 kDa/spleen tyrosine kinase (DAP12/SYK) axis through miR-370, thereby affecting the development mechanism of prostate cancer.
Materials and Methods
Polystyrene nanoparticles coated with astragalus polysaccharide nanoparticles (APS/PDA NPs) were prepared, and PC-3 cell experiments were used for culture and further detection of miRNA and other genes, detection of glycolysis-related proteins and genes, and observation of the biology of PC-3 cells behavioral changes, exploring the mechanism of action of APS/PDA NPs.
Results
We successfully prepared composite materials of APS/PDA NPs, and found that APS/PDA NPs could inhibit glycolysis and induce apoptosis in PC-3 cells. Further experiments showed that APS/PDA NPs could increase miR-370 expression, inhibit glycolysis, and induce apoptosis. APS/PDA NPs play a strong role in inhibiting the DAP12/SYK axis, inhibiting glycolysis, and causing apoptosis. Combined with biological information and multiple experiments, it was confirmed that miR-370 targeted DAP12, and the inhibition of PCa progression by APS/PDA NPs was achieved through targeting miR-370 and negatively regulating the DAP12/SYK axis.
Conclusion
As a drug carrier, polystyrene nanoparticles can effectively deliver astragalus polysaccharides to prostate cancer cells, thereby increasing the local concentration of drugs and enhancing the therapeutic effect. More encouragingly, this drug carrier not only performed well in inhibiting glycolysis but also significantly upregulated the level of miR-370, which further inhibits the expression of DAP12, providing a new therapeutic idea for inhibiting the development of prostate cancer.
Keywords
Introduction
The glycolytic mechanism of prostate cancer is currently a hotspot that urgently needs to be broken through (Kelpsiene et al., 2022). It is of great significance to further explore the therapeutic targets of this disease and improve prognosis. Tumor cells are generally highly dependent on glucose, and based on their active growth, there is a great demand for glucose uptake and metabolic rate (Chi et al., 2022). This abnormal phenomenon of glucose metabolism is called “along the glycolytic pathway.” During this process, prostate cancer cells can preferentially upregulate PFKFB2 and PFKFB3 to some extent, so that glucose can be transformed into lactic acid or precursor substances required for tumor metabolism. However, due to excessive glucolysis, the production rate of ATP increases, leading to an increase in the ratio of ATP/ADP in the cell, inhibiting AMPK signaling, thereby promoting glycolysis pathway, and further affecting the development of cancer (Zhu et al., 2022). In general, prostate cancer is related to glycolysis, abnormal glucose metabolism, and lactic acid acidification, which enable cancer cells to survive and proliferate rapidly under hypoxic conditions (Malinowska et al., 2022). Therefore, targeting the regulation of the glycolytic pathway may be an essential strategy for the treatment of prostate cancer. By interfering with key enzymes or signaling pathways in the glycolytic pathway, it is expected to block the energy supply and metabolic pathways of cancer cells, thereby inhibiting tumor growth and progression.
microRNAs are widely recognized by the oncology community as potential anticancer therapeutic targets this year. Studies have shown that (Steckiewicz et al., 2022) miR-143/145 inhibits key glycolytic enzymes such as hexokinase 2 (HK2) and regulates the glycolytic pathway by downregulating HK2, thereby reducing the level of glycolysis in tumor cells. In addition, miR-155 is upregulated in breast cancer, lymphoma, and others, and affects the glycolysis pathway by regulating glucose transporter 1 (GLUT1) and pyruvate kinase M2 (PKM2). Moreover, in breast cancer, miR-370 can target and inhibit Wnt pathway activity. It can also target the PIK3CA gene, thereby activating PI3K/Akt and participating in the development of gastric cancer (Suga et al., 2022). Another study reported (Park et al., 2022) that downregulating miR-370 can reduce the expression of IKKβ (IκB kinase β) gene, thereby inhibiting NF-κB signaling and achieving the purpose of treating liver cancer. However, whether miR-370 also affects glycolysis has a great research space. DAP12 (DNAX activating protein 12) and SYK (tyrosine kinase) are key molecules in intracellular signaling pathways that are closely related to cancer progression and therapy resistance. But whether it can help in the treatment of prostate cancer still needs to be further explored. Therefore, studying whether miR-370 can affect prostate cancer by regulating glycolysis of DAP12/SYK axis will help to gain insight into the pathogenesis of prostate cancer and find new therapeutic avenues. Several studies have shown that multiple medicinal plants possess many cellular activities and might be used for the treatment of several diseases (Açar et al., 2023; Cerqua et al., 2022; D’Avino et al., 2023; Fernández et al., 2021; Ferrarini et al., 2022; Şahin et al., 2023). Polysaccharides are complex in structure and have certain biological activities. In recent years, studies have shown that astragalus polysaccharide (APS) has potential anticancer effects in cancer treatment (Gonçalves et al., 2022), especially the inhibitory effect on prostate cancer has attracted much attention. As a natural product, APSs have antioxidative, immunomodulatory, and anti-inflammatory effects that make it an ideal candidate for therapeutic drugs. In addition, its application in the treatment of tumors and glycolysis may involve multiple signaling pathways. Studies have shown that APSs are considered to have anti-inflammatory effects (Perini et al., 2022), which may reduce inflammatory factors by inhibiting NF-κB signaling and PI3K/Akt signaling to interfere with the proliferation and survival of tumor cells, thereby inhibiting tumor growth and inflammatory response and growth. Another study found that (Sang et al., 2021) APSs may activate the AMPK pathway to promote the consumption of ATP and the accumulation of glycolysis products in cells, thereby interfering with the glycolysis process of tumor cells. In addition, in a hypoxic environment, APSs may affect the glycolysis of tumor cells by regulating the HIF-1 pathway and affecting the expression or function of GLUT (Zhang et al., 2021). However, the application of APS in the treatment of prostate cancer still faces some challenges, one of which is its bioavailability and targeting in vivo. As a commonly used carrier, polystyrene nanoparticles have good biocompatibility and stability and are widely used in the field of drug delivery. Therefore, encapsulating APS in polystyrene nanoparticles is expected to improve its drug delivery and biological effects in the treatment of prostate cancer, as well as the regulation mechanism of miR-370 in it. We hope to reveal the underlying mechanism of prostate cancer development, provide a basis for the development of new treatment strategies, and bring new hope for the treatment of prostate cancer patients.
Materials and Methods
Research Materials
Human prostate cancer (PCa) cell line PC-3 cells were purchased from Auragene Company in Changsha, Hunan; APS (CAS, product number: B20562-20mg, specification 20mg, Shanghai Yuanye); LDHA antibody (Wuhan Huamei); GLUT antibody (Shenzhen Haodi Huatuo); MCT1 antibody (Wuhan Yipu); scanning electron microscope (TEM, Shanghai Carl Zeiss).
Preparation and Characterization of Nanoparticles
Preparation of astragalus polysaccharide nanoparticles (APS/PDA NPs)
Add an appropriate amount of APS into distilled water, stir to dissolve it fully, and obtain a uniform APS solution with a concentration of 20 mg/mL; then pour the polystyrene solution into an ultrasonic cleaner to remove air bubbles and insoluble impurities to form a uniform polystyrene solution. Styrene precursor solution, transfer the cleaned solution to a water bath, keep the temperature at 70–80°C, and continue stirring to further diffuse the polystyrene molecular chains and form nanoparticles. Add the APS extract into the prepared polystyrene solution and mix evenly. Unreacted substances were removed by ultracentrifugation, and finally, the nanoparticle samples were dried to obtain composite samples of APS/PDA NPs (Cao et al., 2022).
Characterization of Nanoparticles
Dip a small amount of nanoparticle dispersion, drop it evenly on the conductive adhesive, spray gold for 1 minute after natural drying, then observe the morphology and structure of nanoparticles with TEM, and analyze their potential by dynamic laser scattering.
Cell Culture
PC-3 cells were cultured and the well-grown cells were taken as the PC-3 group for subsequent experiments. APS was added to the PC-3 cell culture medium and incubated to make it compatible. The cells were fully contacted, which was set as the APS group. APS/PDA NPs were added to the PC-3 cell culture medium for incubation, which was set as the APS/PDA NPs group, and the effect of APS/PDA NPs on the glycolysis of PC-3 cells was analyzed. In addition, set APS/PDA NPs+anti-miR-370 group and APS/PDA NPs+anti-miR-NC group, APS/PDA NPs+si-DAP12 group, APS/PDA NPs+pc-DAP12 group; analyze APS/PDA whether NPs affect the glycolysis of PC-3 cells by regulating DAP12/SYK through miR-370 to inhibit the development of prostate cancer.
Detection of Glucose and Lactic Acid Consumption
Each group of PC-3 cells was planted on a six-well plate (1 × 106 cells/mL) and cultured for 24 hours. After that, supernatants were collected and processed, and the glucose content and lactic acid content in the culture solution of each well were calculated according to the calculation formula given in the instructions. Consumption is calculated.
Apoptosis Detection
After culture for 24 hours, the supernatant was discarded, washed with PBS solution, digested with 0.25% trypsin, and centrifuged to collect cell pellets. Wash the cells three times with PBS solution, and then add 500 µL of binding buffer solution (cell density is 1 × 106 cells/mL). Then add 5 µL of Annexin V-FITC (the kit was purchased from Wuhan Punuosai) under dark conditions and incubate. Then add 5 µL of propidium iodide (PI) and continue to incubate for 10 minutes under the same conditions. Finally, a flow cytometer purchased from BD Company was used to detect apoptosis of cells in each group immediately.
PCR Assay
Take the PC-3 cells used in the experiment, extract the total RNA, prepare the PCR reaction solution, including template DNA primers (forward primer and reverse primer), and transcribe it into cDNA. Prepare the PCR amplification reaction mixture and make the PCR reaction (primers in Table 1) into a PCR tube or 96-well plate, put it into a PCR machine for reaction, including pre-denaturation (95°C, 3–5 minutes), cycle denaturation (95°C, 15–30 seconds), annealing (primer-specific temperature, 15–30 seconds), extension (72°C, fragment length dependent, 1 minute/kb), and other steps, cycled 20 times. The relative level was calculated using the 2-∇∇Ct method.
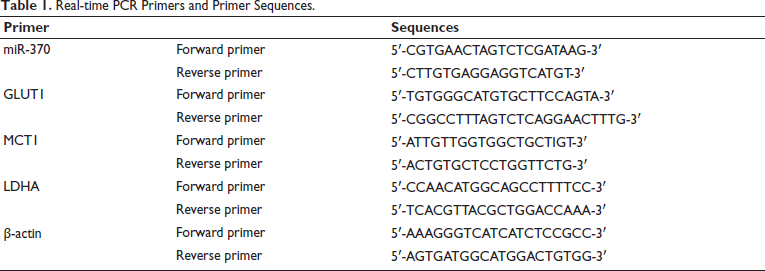
Real-time PCR Primers and Primer Sequences.
Dual Luciferase Assay
Using RegRNA2.0 bioinformatics analysis software, it can be predicted that miR-370 regulates specific targets. Co-transfect T/GHA-VSMC with DAP12-WT plasmid, DAP12-MUT plasmid, SYK-WT plasmid, SYK-MUT plasmid, and negative control sequence or miR-370 mimic, and use the dual luciferase reporter gene kit method for comparison.
Statistical Analysis
SPSS26.0 and GraphPad Prism processed data which were expressed as mean ± SD. All data calculations were performed using the F-test, and the comparison of data between groups was performed using the LSD method. p < 0.05 indicates a difference.
Results
Composite Materials of APS/PDA NPs Successfully Prepared
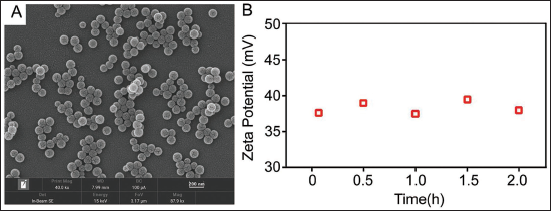
TEM images show that the prepared APS/PDA NPs composite has a spherical shape (Figure 1A), an average particle size of 30 nm, and a low degree of aggregation. During the polymerization process, due to the hydrophilic nature of methacrylic acid. The carboxyl groups on the surface will be introduced to the surface of the microsphere first, thus endowing it with negatively charged characteristics, and the zeta potential is –34.5 mV (Figure 1B).
Characterization of Composites of APS/PDA NPs. (A) TEM Image of APS/PDA NPs; (B) Zeta Potential Distribution of APS/PDA NPs.
APS/PDA NPs Can Inhibit Glycolysis and Induce Cell Apoptosis
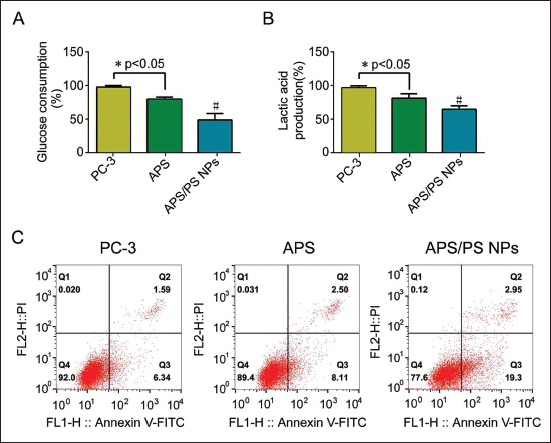
The glucose consumption and lactate production of PC-3 cells are at a high level. After the intervention of APS and APS/PDA NPs, the above levels were significantly reduced, especially in the APS/PDA NPs group (p < 0.05, see Figure 2A and B); the results of apoptosis detection showed that the APS group and the apoptosis rate in the APS/PDA NPs group were all upregulated, especially in APS/PDA NPs group (p < 0.05, see Figure 2C).
APS/PDA NPs Can Inhibit Glycolysis and Induce Apoptosis in PC-3 Cells (n = 12). (A) Glucose Consumption of PC-3 Cells in Three Groups; (B) Lactic Acid Content of PC-3 Cells in Three Groups; (C) Cell Apoptosis in Three Groups, Compared with APS Group, #p < 0.05.
APS/PDA NPs Increase miR-370 Expression to Inhibit Cell Glycolysis and Induce Apoptosis
miR-370 expression in PC-3 cells was low. After the intervention of APS/PDA NPs, the expression of miR-370 was increased, and GLUT1, MCT1, and LDHA were significantly downregulated (p < 0.05, Figure 3A–D), and the apoptosis rate was increased.
APS/PDA NPs Can Increase the Expression of miR-370 in PC-3 Cells to Inhibit Glycolysis and Induce Apoptosis. (A) The Expression of miR-370 in PC-3 Cells of Each Group; (B) The Expression of GLUT1 in PC-3 Cells of Each Group; (C) The Expression of MCT1 in PC-3 Cells of Each Group; (D) LDHA Expression in PC-3 Cells in Each Group; (E) Apoptosis of PC-3 Cells in Each Group; Compared with APS/PDA NPs Group, #p < 0.05.
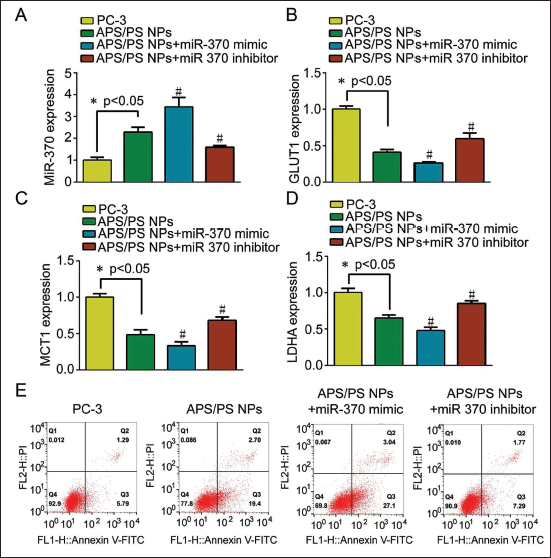
On the basis of APS/PDA NPs intervention, miR-370 mimic and miR-370 inhibitor were transfected, respectively, and it was found that GLUT1, MCT1, and LDHA in APS/PDA NPs+miR-370 mimic group further decreased (Figure 3A–D) with increased apoptosis, consistent with the trend of APS/PDA NPs. After transfection of miR-370 inhibitor, effect of APS/PDA NPs on inhibiting PC-3 cell glycolysis and inducing apoptosis was reversed (Figure 4E). This suggested that APS/PDA NPs inhibited glycolysis and induced apoptosis by upregulating miR-370.
APS/PDA NPs Can Inhibit the Expression of DAP12 in PC-3 Cells to Inhibit Cell Glycolysis and Induce Apoptosis. (A) The Expression of miR-370 in PC-3 Cells of Each Group; (B) The Expression of GLUT1 in PC-3 Cells of Each Group; (C) The Expression of MCT1 in PC-3 Cells of Each Group; (D) LDHA Expression in PC-3 Cells in Each Group; (E) Apoptosis of PC-3 Cells in Each Group; Compared with APS/PDA NPs Group, #p < 0.05.
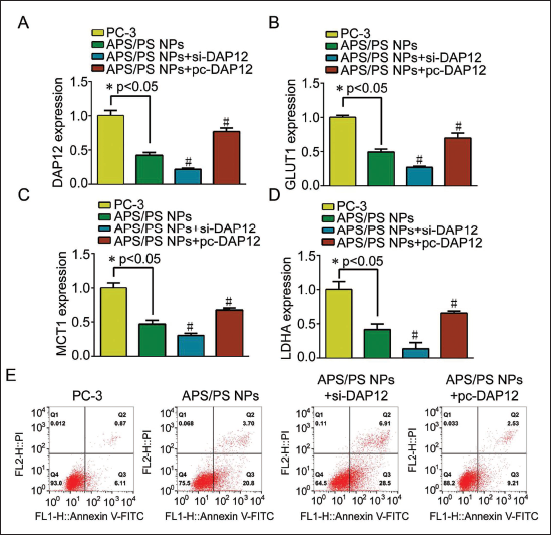
APS/PDA NPs Can Inhibit DAP12 Expression, Inhibit Cell Glycolysis and Induce Apoptosis
DAP12 was highly expressed in PC-3 cells. The results after APS/PDA NPs intervention showed significantly inhibited DAP12 expression in APS/PDA NPs group cells. At the same time, the expressions of GLUT1, MCT1, and LDHA levels were also significantly inhibited (p < 0.05, see Figure 4A–D), and the apoptosis rate was significantly increased (Figure 4E). On the basis of APS/PDA NPs intervention, DAP12 was overexpressed/inhibited, respectively, and GLUT1, MCT1, and LDHA in APS/PDA NPs+si-DAP12 group were further decreased (p < 0.05) and the apoptosis rate was further increased (Figure 4E), which was consistent with the trend of APS/PDA NPs. After transfection with pc-DAP12, APS/PDA NPs inhibited the glycolysis of PC-3 cells and the effect of inducing its apoptosis was reversed. This suggested that APS/PDA NPs inhibited glycolysis and induced apoptosis by inhibiting DAP12 expression.
miR-370 has Base Sequence Complementarity with DAP12 Gene mRNA, Which Can Target and Negatively Regulate DAP12/SYK
The relative luciferase activity of miR-370 and DAP12-WT co-transfection group was significantly lower than miR-370+DAP12-MUT co-transfection group; the relative luciferase activity of miR-370 and SYK-WT co-transfection group was significantly lower than miR-370+SYK-MUT co-transfection group (p < 0.05) (Figure 5A).
There is a Base Sequence Complementarity between miR-370 and DAP12 Gene mRNA, Which Can Target and Negatively Regulate DAP12/SYK. (A) Relative Luciferase Activity Detected by Dual Luciferase Reporter Gene; (B) Compared with miR-370+DAP12-MUT Co-transfection Group, *p < 0.05; Compared with miR-370+MYK; Compared with MUT Co-transfection Group, #p < 0.05.
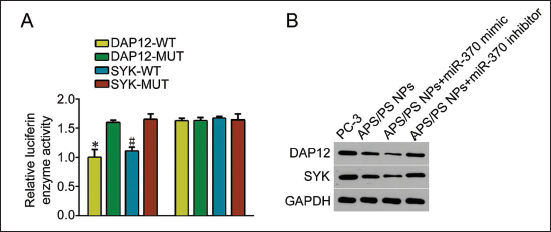
In the results of Western blot detection, when PC-3 cells were intervened with APS/PDA NPs, transfected with miR-370 mimic or APS/PDA NPs+miR-370 mimic, the expression of DAP12 was significantly downregulated, and APS/PDA downregulation trend was the most significant after NPs+miR-370 mimic intervention. On the contrary, after miR-370 inhibitor transfection, DAP12 expression was abnormally active (Figure 5B). It is suggested that miR-370 can target and negatively regulate the expression of DAP12.
Discussion
APS is a bioactive component extracted from Astragalus membranaceus, which has been widely studied and used in the field of traditional Chinese medicine (Ye et al., 2023). APS not only effectively inhibits the NF-κB pathway, but also exerts the same effect on the JNK pathway. On the one hand, it reduces serum IL-1β, TNF-α, and others in the body and increases Bcl-2, NF-κB, and IκB-α proteins in the cytoplasm content, thereby reducing LPS-induced apoptosis in mouse cardiomyocytes (Liu et al., 2019). In addition, APSs can increase the number of peripheral blood cells and bone marrow cells to a certain extent, and even increase the content of IL-2 and INF-γ. Meanwhile, it can also reduce IL-10, VEGF, MMP-2, and others. The above studies have shown that APSs have positive anticancer effects. However, APSs are poorly soluble in water and have low bioavailability and targeting, which often limit their application. In order to improve the defects of traditional Chinese medicine in this aspect, researchers usually combine it with nano-drug loading technology to make it work better (Cao et al., 2022). Therefore, in this study, we used polystyrene nanoparticles as a carrier to wrap APS and successfully prepared a composite material of polystyrene nanoparticles wrapped APS.
In order to further explore the effect of polystyrene nanoparticles-coated APSs on the glycolysis of PC-3 cells, we detected the changes of glucose and lactic acid in PC-3 cells through cell culture experiments and found that APS inhibited PC-3 cell apoptosis. It has a positive effect on death and its glycolysis (Rizzo et al., 2022). However, after using polystyrene nanoparticles to encapsulate APS, it showed a better inhibitory effect.
Studies have shown (Wilt et al., 2021) the involvement of miR-370 in various cancers. In order to ascertain the mechanism of action of polystyrene nanoparticles-encapsulated APS on PC-3 cells, our results showed that miR-370 was highly expressed under APS intervention conditions, suggesting that the tumor suppressor effect of APS was related to the high expression of miR-370. Styrene nanoparticles wrapped APS strengthened this phenomenon.
Studies have found (Sha et al., 2020) that after DAP12 binds to a specific receptor, it can activate SYK kinase in the cell, further triggering phosphorylation reactions, enzyme activation, and other events, resulting in cell activation and functional regulation. In macrophages and NK cells, the activation of the DAP12/SYK axis affects the immune killing function of macrophages and NK cells to a certain extent and even the secretion of cytokines. Further results showed decreased DAP12 level in APS group, which significantly inhibited under the intervention condition of APS coated with polystyrene nanoparticles, indicating that the inhibitory effect of APS coated with polystyrene nanoparticles on PC-3 may be through upregulation of miR-370 expression, thereby inhibiting AP12 expression (Miyahira & Soule, 2022). APSs may interact with DAP12 and SYK by regulating immunoglobulin receptors (Fc receptors), triggering signal transmission, causing phosphorylation, and activating tyrosine and serine kinases, thereby making the activity and activity of macrophages and NK cells functional changes that affect prostate cancer cell metastasis. miR-370 may reduce the expression of DAP12 protein by binding to DAP12 mRNA, causing its degradation or inhibiting translation. To confirm this speculation, after using miR-370 mimic, the glycolysis of PC-3 cells was significantly reduced, and the apoptosis rate was greatly increased. Therefore, overexpression of miR-370 can significantly inhibit glycolysis and induce apoptosis. PC-3 cells also exhibited a consistent inhibitory effect after the application of DAP12 inhibitors (Washington et al., 2022), which further confirmed the hypothesis that miR-370 affects cellular glycolysis and survival through the regulation of DAP12. It was further finally confirmed that the inhibition of glycolysis under the condition of APS intervention was achieved by inhibiting DAP12, and it also strengthened the possibility that miR-370 regulates the DAP12 signaling pathway, further revealing the regulatory relationship between miR-370 and DAP12 in importance in the development of prostate cancer cells (Murgić et al., 2022). In the experiment, after we inhibited the expression of miR-370, DAP12 increased, and the apoptosis rate of PC-3 cells in the polystyrene nanoparticle-wrapped APS group decreased. It can be seen that the overexpression of miR-370 reversed the polystyrene nanoparticle-wrapped APS to accelerate PC-3 cell glycolysis and induce its apoptosis, and DAP12 is also affected accordingly. The results indicated the central role of miR-370 in regulating DAP12. Inhibition of miR-370 led to an increase in DAP12, which may be a key factor in the acceleration of glycolysis and inhibition of apoptosis (Miyahira et al., 2020). This further strengthened the verification of the regulatory role of miR-370 on the DAP12 signaling pathway.
Studies have shown that (Gourdin, 2021) in esophageal cancer cells, downregulation of SLC2A1-AS1 upregulated miR-378a-3p, thereby inhibiting glycolysis-related proteins. In addition, miR-513a3p regulates hexokinase 2 (HK2) to inhibit gastric cancer cell proliferation and glycolysis (Crawford et al., 2020; Fisher et al., 2020). miR-30a-5p inhibits gastric cancer cell growth and glycolysis by targeting the URGCP gene. We detected high expression of DAP12 and SYK in PC-3 cells. However, under the intervention of polystyrene nanoparticles coated with astragalus polysaccharide nanoparticles (APS/PDA NPs), the expressions of DAP12 and SYK were significantly inhibited, which indicated that APS/PDA NPs may affect cells by regulating the expression of DAP12 and SYK, and further found that the impressions of metabolism-related genes, such as MCT1 and LDHA, were significantly decreased, which means that APS/PDA NPs may inhibit cellular glycolysis by regulating the expression of these metabolic genes (Mahon, 2022). Further results of apoptosis rate also verified the anticancer potential of APS/PDA NPs. Through further verification experiments of overexpressing or inhibiting DAP12 experiments, after transfection of pc-DAP12, the effects of APS/PDA NPs on inhibiting cellular glycolysis and inducing apoptosis were reversed, which strongly suggested that APS/PDA NPs inhibited DAP12/SYK axis to regulate cell metabolism and induce apoptosis.
The importance of DAP12 in regulating glycolysis and apoptosis: It was observed experimentally that the upregulation of DAP12 was accompanied by the acceleration of glycolysis and the inhibition of apoptosis (Fontana et al., 2022). This suggests that DAP12 may have multiple functions in prostate cancer cells, and may not only participate in the immune response of cells but may also play a key role in metabolic regulation and cell survival. Through multiple experiments and bioinformatics predictions, the study proposed that miR-370 might be a target gene of DAP12 and negatively regulate the expression of DAP12 through miR-370. When miR-370 was inhibited, this effect was reversed. Further experimental verification showed that (Dong et al., 2020; Li et al., 2020) the inhibitory effect of APS/PDA NPs may be through increasing miR-370 expression, thereby inhibiting DAP12 and SYK, finally affecting the signal transmission of the DAP12/SYK axis, and inhibiting prostate cancer cell progression. These results strongly imply the key role of miR-370 in the inhibition of PCa progression by APS/PDA NPs, which provides important clues for an in-depth understanding of the therapeutic mechanism. It also reminds us that we should not only focus on single drugs or mediators when designing comprehensive therapeutic strategies, it is also necessary to consider their possible interactions with multiple molecules and signaling pathways in the cell to better achieve therapeutic goals (Tang et al., 2021; Zhang et al., 2022). However, the study has limitations. The PC-3 cell line used in the study represents only a small number of types of prostate cancer, so the results may not apply to other types of prostate cancer. Multiple prostate cancer cell lines could be considered to validate the broad applicability of the findings.
Conclusion
In summary, as a drug carrier, polystyrene nanoparticles can effectively deliver APSs to prostate cancer cells, thereby increasing the local concentration of the drug and enhancing the therapeutic effect. More encouragingly, this drug carrier not only performed well in inhibiting glycolysis but also significantly upregulated miR-370. The upregulation of miR-370 further inhibits the expression of DAP12, indicating that they might be a novel therapeutic target for the treatment of prostate cancer.
Footnotes
Abbreviations
APS/PDA NPs: Polystyrene nanoparticles coated with astragalus polysaccharide nanoparticles; DAP12: DNAX-activating protein of 12 kDa; SYK: Spleen tyrosine kinase.
Acknowledgments
The authors gratefully acknowledge the Second People’s Hospital of Yichang Laboratory for providing the necessary equipment for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethics committee of the Second People’s Hospital of Yichang and informed consent was obtained.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
