Abstract
Background
Ionizing radiation can induce bystander effects (RIBE), and Astragalus polysaccharides (APS) have a protective effect against RIBE. It has been proved that RIBE occurred and APS played the inhibitory effects on the RIBE when radiation was carried out only one time. However, whether RIBE happened by multiple irradiations and the effects of APS on the RIBE are unclear. This study aimed to investigate whether APS suppress RIBE damage induced by multiple irradiations.
Materials and Methods
A549 cells were irradiated with 2 Gy X-rays to obtain a conditioned medium. Bone mesenchymal stem cells (BMSCs) was incubated with the conditioned medium 5 times every 3 days or APS. Cell proliferation was detected by CCK-8 and colony formation assay, and genomic instability and DNA damages were detected by the micronucleus and immunofluorescence assay. The protein associated with methylation and cancer-associated proteins in BMSCs were detected by western blot.
Results
After five stimulations with a conditioned medium, the proliferative capacity of BMSCs decreased; APS pre-intervention promoted the proliferation of BMSCs. And conditional medium intervention increased the micronucleus rate and a number of 53BP1 foci; APS pre-intervention reduced the micronucleus rate and number of 53BP1 foci. In addition, the conditional medium intervention reduced the expression of methylation-related proteins and increased the expression of cancer-related proteins, while APS pre-intervention reversed this trend.
Conclusion
Irradiated A549 conditioned medium can induce RIBE of BMSCs, which might be related to methylation and cancer-associated proteins and APS may block RIBE by multiple irradiations in BMSCs.
Introduction
Radiotherapy is a common treatment for tumors. Number of studies have demonstrated that X-ray can destroy single or double chains of DNA in a cell, which surely caused DNA damage, induced cell apoptosis, and suppress cell growth to kill tumor cells (Kong et al., 2018). However, radiation-induced bystander effects (RIBE) also occur in neighbor cells or outside tissue and have the same effect with direct irradiation (Depta et al., 2017; Yeles et al., 2017). So, it is inevitable to damage the normal tissues and cells around the tumor even far away resulted by RIBE when radiotherapy is carried out.
Human bone mesenchymal stem cells (BMSCs), derived from the early embryonic mesoderm, is a pluripotent stem cells with the capacity for powerful proliferation and differentiation (Zhang et al., 2017). BMSCs have a strong ability to migrate towards tumor, inflammation, and injury sites. It has been found that tumor cells can secrete a variety of chemokines to recruit BMSCs to gather around the tumor cells and participate in the repair of tissue damage around the tumor (Li et al., 2017; Xiao et al., 2016). Up to now in the process of radiation only one time, X-ray will cause RIBE damage to BMSCs around it has been reported in our study (Zhang et al., 2018), but the effects of the RIBE induced by multiple irradiations were unclear. Therefore, it is of great significance to explore the RIBE affected by multiple radiations.
Natural products can be used as natural radiosensitizers and radioprotectors, and in combination with radiotherapy, they can effectively kill tumor cells and have a protective effect on normal cells/tissues (Yi et al., 2021). Some natural radiosensitizers such as paclitaxel, curcumin, genistein, and papaverine have entered clinical trials, and resveratrol has been repeatedly shown to have radiation protection effects in many independent studies (Komorowska et al., 2022). Astragalus polysaccharide (APS) is one of the main components of Astragalus membranaceus (AM), which has one part effect of AM. And many studies found that APS has the effect of anti-radiation, anti-oxidation, and alleviating the adverse reaction of radiotherapy (Liu et al., 2014; Xie et al., 2016; Zhang et al., 2017). APS has been reported to have anti-apoptotic effects during radiotherapy for lung cancer (Mao et al., 2019), and APS inhibits the RIBE by regulating MAPK/NF-κB signaling pathway in BMSCs when radiation happened only once. However, the protective effects of APS against multiple radiation-induced RIBE damage in BMSCs have not been reported.
In this experiment, A549 cells were irradiated with X-ray by multiple times and collected its medium every time. The medium was transferred separately to BMSCs, named bystander cell. Then, APS was used against RIBE induced by multiple radiations between BMSCs and A549 cells. We studied the effects of APS on genome damage, methylation level, and deterioration risk of BMSCs caused by RIBE induced by multiple radiations to explore the drugs that could reduce the radiation damage to BMSCs and provide a feasible suggestion for reducing the side effects of radiotherapy.
Materials and Methods
The sources of the reagents and instruments used in this study are shown in Table 1.
Reagents and Resources.
Cell Culture
BMSCs were cultured in a mesenchymal stem cell medium and then maintained at a constant temperature in an incubator with 5% CO2.
Human non-small cell lung cancer cells (A549) were cultured in DMEM/F-12 medium containing 10% fetal bovine serum supplemented with 100 units/mL penicillin and 100 mg/mL streptomycin, followed by constant maintenance in an incubator containing 5% CO2.
Irradiation
A549 were irradiated laboratory RX-650 X-ray source operated at 100 kVp energy. The dose rate was about 0.6 Gy/min (100 KeV, 5 mA). The total absorbed dose was 2Gy each time, and five times of X-ray radiation were carried out in each group. The irradiation was carried out at room temperature.
Drugs
APS was purchased as a standard with a purity of up to 98%. According to the conditions of our early experimental exploration (Zhang et al., 2018), the final concentration of APS used in this experiment was 50 µg/mL.
Conditioned Medium Transfer
When A549 was cultured to 80%, the culture medium was refreshed with mesenchymal stem cell medium before the fifth irradiation. The medium was collected 3 h after irradiation and centrifuged at 94g for 5 min to exclude cells and cellular fragments, and the medium received after centrifugation was the conditioned medium.
Groups
BMSCs were divided into four groups. BMSCs cultured with mesenchymal stem cell medium served as a blank control group (Ctrl). BMSCs cultured with mesenchymal stem cell medium rich in 50 µg/mL APS for 3 days served as APS medium group (APS). BMSCs cultured with conditioned medium served as a conditional medium group (RIBE). BMSCs cultured with conditional medium rich in 50 µg/mL APS for 3 days served as APS and conditioned medium group (APS + RIBE).
Cell Viability Assessment
Cell proliferation levels were assessed by the CCK-8 assay for viability. Experiments were performed according to the manufacturer’s instructions in 96-well format in triplicate. Cells at the logarithmic growth stage were taken, treated with radiation and different concentrations of APS, and then prepared into single-cell suspensions. Cells were inoculated into 96-well plates at a density of 2000 cells per well and incubated at 37°C in a 5% CO2 incubator until the cells were plastered, then incubated at 1, 2, 3, 4, and 5 d with 20 µL CCK-8 and continued to incubate for 4 h. The absorbance values of the cells were measured at 450 nm using a full wavelength enzyme marker.
Cell Colony Formation Assay
Cell survival was measured by the colony formation assay. BMSCs were first counted for cells and then 100 BMSCs were inoculated in each 60 mm dish and incubated at a constant temperature in an incubator containing 5% CO2. The total number of cells was 100 in each dish. The treated cells were incubated in the corresponding medium for 14 days. Colonies were fixed in 1.0% crystal violet and 0.5% glacial acetic acid in ethanol. Colonies of at least 50 cells were scored as surviving cells. The image of each group was collected. The cell area of BMSCs cells in each group was analyzed by Image Pro software, and the proliferation rate of BMSCs in each group was calculated as the following formula: Colony rate = (Clone multiplication number/Planting cell number)%.
Micronucleus (MN) Assay
The micronucleus number of BMSCs in each group was measured by Cytochalasin B assay.
After treatment with radiation and APS, each group of cells was treated with 1 µL of cytochalasin B at a final concentration of 4 µg/mL, and the culture was terminated after 48 h. The cells were fixed in glacial acetic acid and methanol (1:9, v/v), dried in a fume hood for 20 min, and stained with acridine orange dye; 1000 binucleated cells were observed by fluorescence microscopy, and the cellular micronuclei rate (MNF) was counted according to the criteria for determining MN and calculated according to the following formula. MNF = (total number of MN counted/1000) × 100%.
Immunofluorescence Staining
The number of 53BP1 foci in each group was measured by immunofluorescence staining assay as reported (Nelson et al., 2018). BMSCs were inoculated into 12-well plates, cells were fixed in 4% paraformaldehyde for 15 min, and permeabilized by incubation with 0.5% Triton-X 100 for 10 min. After closure with 5% skimmed milk for 1 h, the cells were incubated with mouse monoclonal anti-53BP1 antibody for 2 h at room temperature. Cells were subsequently incubated with Alexa Fluor® 594 anti-mouse antibody for 1 h at room temperature. Nuclei were re-stained with 4′,6-diamidino-2-phenylindole (DAPI) and filmed, observed under a fluorescent microscope, and photographed. Further, 100 cells per sample were counted and the mean number of foci was calculated.
Chromosomal Aberration Experiment
Cells were cultured in a medium with colchicine for 4 h, low permeability treated with KCL for 50 min in a temperature tank at 37°C, stained with Giemsa for 15 min with Glycerol sealing tablets, and then observed under a microscope with an oil lens. The chromosome number of split figures was recorded. The number of aberrant cells and the number of chromosome aberrations were recorded. The total aberration rate and aberration rate of chromosomes were calculated as follows: Total aberration = (number of chromosomes in each type/total number of cells) × 100%. Distortion ratio = chromosome aberration/ chromosome number.
Detection of Whole Genome Methylation in Each Group
The whole genome methylation in each group was detected by Methyl Flash Methylated DNA Quantification Kit. Preparation of cell suspensions with a density of 1 × 106. First, adjust the total DNA of the extracted DNA, so that the total DNA content reaches the same level. Second, prepare buffer, the diluted positive control ME4, 1 × TE, and the resulting ME4 concentration as per the instructions. Third, do the combination and the capture of methylation of DNA following the instructions. At the last, absorbance at 450 nm was read using an enzyme marker and we can use the following formula for calculating the DNA ratio of 5-mC
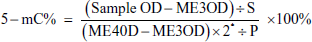
Western Blot
BMSCs were lysed in RIPA buffer to extract the proteins. They were then separated by electrophoresis in 12% SDS-PAGE separation gel, followed by membrane transfer with PVDF membranes. At the end of the membrane transfer, the membranes were closed in 5% skimmed milk powder for 2 h, followed by incubation with DNMT3A, DNMT3B, MECP2, MBD2, P53, P16, c-Myc, H-Ras, and GAPDH antibodies at certain concentrations for 2 h, respectively. Final exposure development was performed and protein intensities were quantified using Adobe Photoshop software.
Statistical Analysis
All data were statistically analyzed using SPSS 23.0 software. The data obtained from the independent experiments were analyzed by analysis of variance (ANOVA) with post hoc analysis for multiple comparisons. Values represented mean ± SD. p < 0.05 was considered a statistically significant difference. All figures are done by the Origin software.
Results
Optimum Concentration of APS
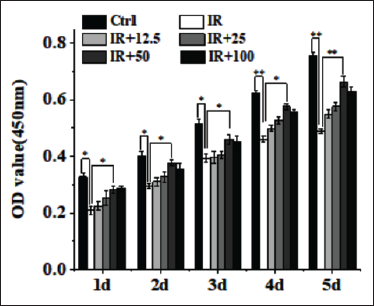
As shown in Figure 1, CCK-8 results showed that compared with the Control group, the proliferation of BMSCs was significantly reduced after radiation at different times (p < 0.05). Compared with the radiation group, the proliferation of BMSCs treatment with 12.5 µg/mL APS was promoted on the fourth day and the fifth day (p < 0.05); the proliferation of BMSCs treatment with 25 µg/mL APS was promoted on the third day, the fourth day, and the fifth day (p < 0.05); the proliferation of BMSCs treatment with 50 and 100 µg/mL APS was obviously promoted in each day (p < 0.05) and the proliferation of BMSCs treatment with 50 µg/mL APS is obviously higher than treated with 100 µg/mL, so the optimum concentration of APS is ultimately 50 µg/mL.
APS Promoted the Growth of BMSCs
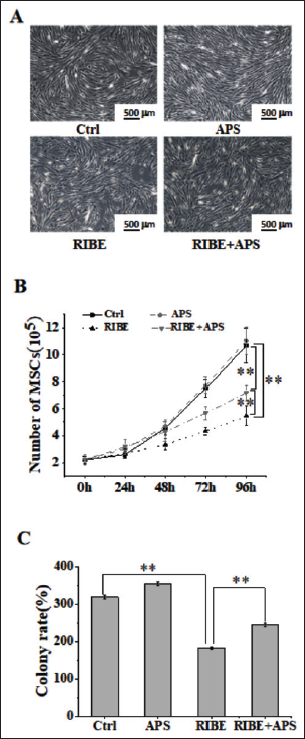
To assess the effect of CM and APS on bystander cells, BMSCs, we first observed the morphological changes of BMSCs by inverted microscopy. These results (Figure 2A) suggest that BMSCs showed vorticity growth, the cell contour was full and the size was uniform, no differentiation in the Control group and the APS group. Cells were sparse and bifurcated in RIBE group and the number was little than in Control group. In RIBE+APS group BMSCs, the number of cells increased obviously. To evaluate the effect of CM on bystander effect cells quantitatively, the number of BMSCs (Figure 2B) and the cell colony formation assay (Figure 2C) were conducted. They showed that CM reduced the number of BMSCs as well as the clonal formation of BMSCs, and APS promoted the proliferation and growth of BMSCs.
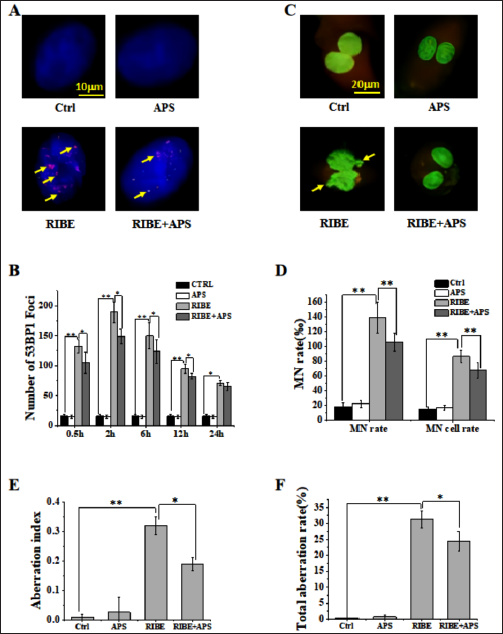
APS Reduced the Genomic Instability of BMSCs Induced by CM
To verify whether CM affects the genomic stability of BMSCs, we characterized typical markers associated with genomic stability and DNA damage. We measured expression levels of 53BP1 Foci, MN, and chromosome aberration analysis of BMSCs by immunofluorescence staining, acriorange dyeing, and colchicine method, respectively. As shown in Figure 3B, D, E, and F, the expression of DNA damage marker, 53BP1 Foci, MN, the total aberration rate, and the aberration index of BMSCs chromosomes were upregulated treatment by CM and APS downregulated them. Moreover, in Figure 3A and C, the image of 53BP1 Foci and MN of BMSCs chromosomes showed the same result as Figure 3B and D, respectively.
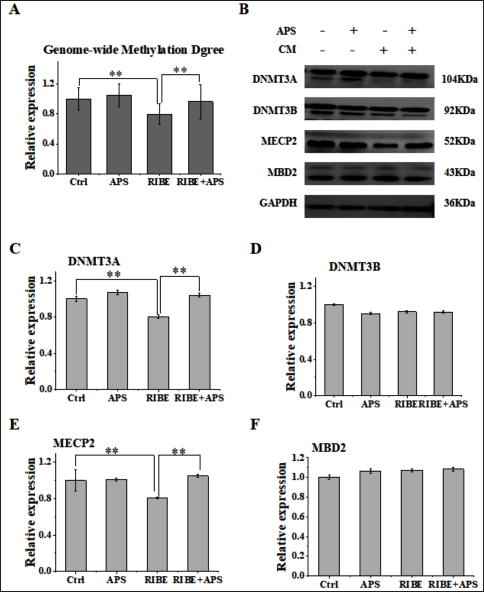
APS Promoted Methylation Level and the Related Proteins
Numerous studies have shown that exposure of cells to a conditioned medium as well as other toxic stresses can induce the methylation level to decrease. We also observed the decreased methylation level (Figure 4A) and the down expression of DNMT3A (Figure 4B and C) and MECP2 (Figure 4B and E) after being treated with CM. However, when treating cells with 50 µg APS prior to irradiation, we found that the methylation level (Figure 4A) and the expression of DNMT3A (Figure 4B and C) and MECP2 (Figure 4B and E) were upregulated, while the expression of DNMT3B (Figure 4B and D) and MBD2 (Figure 4B and F) had no difference. These results indicated that APS can modulate the bystander effect of BMSCs by improving hypomethylation levels.
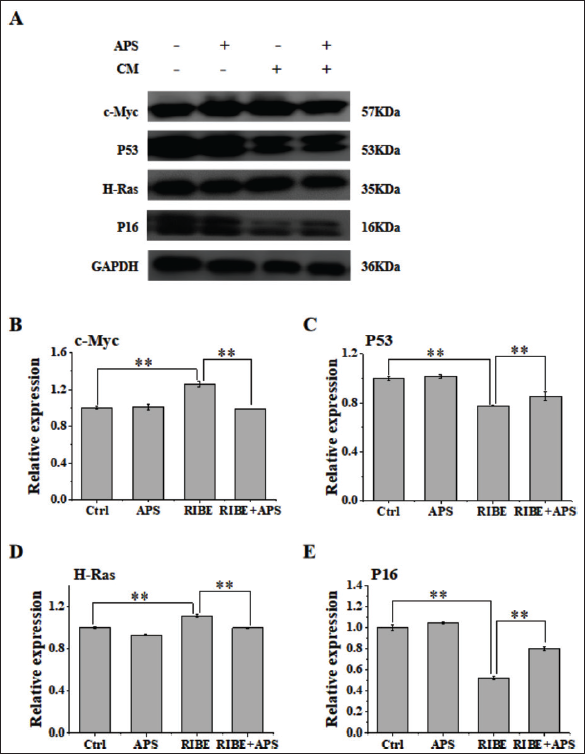
APS Inhibited Oncoprotein and Promoted the Tumor Suppressor Protein
Exposure to cells adjacent to the direct exposure field or to distant tissues has been shown to exhibit the same biological effects as radiation, such as gene mutations (Sawant et al., 2001). We also observed the oncoprotein upregulated and the tumor suppressor protein decreased after being treated with CM. The expression of oncoprotein, including c-Myc (Figure 5A and B) and H-Ras (Figure 5A and D), increased and the expression of the tumor suppressor protein, including P53 (Figure 5A and C) and P16 (Figure 5A and E), decreased invention with CM. However, when treating cells with 50 µg APS prior to irradiation, we found that the oncoprotein, including c-Myc (Figure 5A and B) and H-Ras (Figure 5A and D), upregulated and the tumor suppressor protein, including p53 (Figure 5A and C) and P16 (Figures 5A and E), downregulated. These results suggested that APS may regulate the level of cancer-related proteins.
Discussion
Radiotherapy is one of the common treatment way for malignant tumors and fractionated multiple irradiation is a usual method in the clinic. But the damages are different between single exposure and repeated exposures to radiation. A finding suggests that repeated exposures to low doses of X-rays may increase vascular inflammation, leading to atherosclerosis (Cervelli et al., 2014). Study has demonstrated that multiple genotoxic injuries are more damaging to the germinal epithelium than a single injury due to the repetitive activation of reserve stem cells (Shah et al., 2019). RIBE is a phenomenon in that unirradiated cells respond as if they have been irradiated after they have co-functioned with the irradiated cells. The evidence has proved RIBE occurred in single exposure to radiation (Prise et al., 2009), and the nitric oxides (Jella et al., 2018), cytokines (Dong et al., 2015), inflammatory products (Chai et al., 2013), and exosomes (Yuan et al., 2016) released by radiation cells can mediate RIBE in bystander cells. Thus, exposure to cells adjacent to the direct exposure field or to distant tissues has been shown to exhibit the same biological effects as radiation, such as gene mutations, apoptosis, genomic instability, and chromosome aberrations. Here, we showed that fractionated 2Gy radiation affected A549 cell line five times, and transferred its conditioned medium to bystander cell separately, MSCs, to explore the RIBE damage in multiple exposures to radiation.
DNA methylation is an epigenetic mechanism that occurs through the addition of a methyl (CH3) group to DNA, which often alters the function of the genes and affects gene expression. The most widely characterized process of DNA methylation is the covalent addition of the methyl to the 5-carbon of the cytosine ring, producing 5-methylcytosine (5-mC), also informally known as the “fifth base” of DNA. According to epigenetic principles, hypermethylation or low methylation of DNA often leads to the silence of gene expression (Chen et al., 2015). It has been proved that radiation can cause abnormal methylation of tissues or cells, which are mainly characterized by low methylation of the whole genome and hypermethylation of specific genes (Wang et al., 2014). As far as current research advances, radiation-induced whole genome hypomethylation is often accompanied by the reduction of DNA methyltransferase, methyl binding protein (Kamstra et al., 2018). The evidence also showed that abnormal DNA methylation may expand the radiation-induced damage from a point to the whole body, even the stable heredity to the offspring, and there is a risk of inducing cancer in situ tissue and organogenesis (Li et al., 2017; Shen et al., 2017). In the present study, bystander cells treated with a conditioned medium showed lower levels of DNA methylation. And the expression of both methyltransferase DNMT3A and methyl-binding protein MECP2 were decreased in bystander cells after treatment with a conditioned medium. This indicates that repeated exposure to conditioned medium can cause low methylation of the whole genome in bystander cells. This is in accordance with other studies (Wang et al., 2014). While APS increased DNA methylation levels and was protective against DNA methylation levels in BMSCs.
Ionizing radiation can cause damage and mutation of the genome and lead to genomic instability, and genomic instability also increases the probability of cell deterioration (Santibáñez-Andrade et al., 2017). DNA is most sensitive to radiation, which shows that radiation can cut off DNA single or double strands, causing DNA to be unable to repair or repair errors, and eventually cause the tumor cell to dead (Germano et al., 2017). However, after radiation injury, the mismatch modification of DNA can also cause the recombination, mutation, and aberration of the genome, which greatly improves the risk of the occurrence of tumors (Antal et al., 2015). Micronucleus, also called the satellite nucleus, is an abnormal structure in the eukaryotic cell. The number of micronucleus reflects the degree of nuclear damage (Shi et al., 2015). Chromosome aberration is also called chromosome abnormalities, and the rate of chromosome aberration reflects the degree of chromosomal abnormalities (Kulka et al., 2017; Talan et al., 2017). Further, 53BP1 protein is one of the key DNA damage reaction proteins, and radiation can cause DNA double-strand breaks. And, after DNA double-strand breaks, the 139th-bit serine of histone γH2AX can be rapidly induced to be phosphorylated to 53BP1. Also, 53BP1 can raise a variety of DNA damage repair proteins to the DNA damage site to participate in the repair process of DNA damage, so the number of 53BP1 focus clusters is directly inverse the degree of DNA damage (Fradet-Turcotte et al., 2013; Tsvetkova et al., 2017). In this study, BMSCs treated with a conditioned medium showed lower survival and cell proliferation, along with increased micronuclei formation and 53BP1 foci, which suggests that treatment with a conditioned medium slowed the growth of BMSCs and enhanced genomic instability in BMSCs. Luckily, APS reduced the number of 53BP1 foci and micronucleus formation in BMSCs, thereby maintaining genome stability.
Our previous study showed that the survival rate of BMSCs decreased and the level of genomic instability increased in a single radiation exposure, which is consistent with the results of multiple exposures in this paper. Through further experiments, we found that it is apoptosis and the MAPK and NF-κB signaling pathways that play a role in bystander cells (Zhang et al., 2018; Zhang et al., 2020). However, after multiple irradiations, the cell survival rate decreased more obviously and the genome damage level was higher than radiation only once. This kind of injury is more serious and difficult to repair, which easily leads to wrong repair and cancer. Therefore, the methylation level and cancer-related proteins after multiple irradiations were further explored in this paper.
It has been known that low methylation of the whole genome undoubtedly promotes genomic instability (Anzalone et al., 2018; Fukuda et al., 2017), and abnormal methylation of specific genes (such as hypermethylation of tumor suppressor gene and low methylation of oncogene) has a high correlation with the formation and the development of tumor (Dawson et al., 2012; Llinàs-Arias et al., 2017). The evidence has shown that radiation can lead to abnormal hypermethylation and low expression of P16 (Song et al., 2016). P16 is an essential gene in the cell cycle that is directly involved in the regulation of the cell cycle and negatively regulates cell proliferation and division (Yu et al., 2014). P16 is considered as a new tumor suppressor gene similar to P53. Moreover, a large number of studies have confirmed that aberrant methylation of oncogene c-Myc and H-Ras plays an important role in the process of tumor formation (Geraldes et al., 2016; Luo et al., 2010). In this work, the expression of the tumor suppressor protein, including p53 and p16, decreased when treated with a conditioned medium, and the expression of oncoprotein, including c-Myc and H-Ras, increased when treated with a conditioned medium. This indicates that multiple irradiations can cause tumor-related protein of bystander cells at high level. While APS reduced the expression of tumor-related proteins and had a protective effect on bystander cells. This is consistent with modern pharmacology that Astragalus Polysaccharides play an anti-tumor role (Lai et al., 2017).
Conclusion
In conclusion, our work confirms that APS has a protective effect against the RIBE of BMSCs after multiple radiations, which provides an experimental basis for the application of natural products in radiation protection and the development of radiation protection drugs. As natural products have unique advantages in radiation protection, we expect that our work will promote clinical transformation and provide good support for the radiation protection of clinical patients.
Summary
In summary, APS promoted the growth of RIBE-induced BMSCs, improved the genomic instability of BMSCs, and had a protective effect against RIBE, which may be related to the fact that APS alleviated aberrant genome-wide methylation and regulated the expression of methylation-related proteins as well as cancer-related proteins.
Footnotes
Abbreviations
AM: Astragalus membranaceus; APS: Astragalus polysaccharide; BMSCs: Bone mesenchymal stem cells; RIBE: Radiation-induced bystander effects; MN: Micronucleus.
Acknowledgments
We acknowledge Gansu University of Chinese Medicine, Provincial Level key Laboratory for Molecular Medicine of Major Diseases and Prevention and Treatment with Traditional Chinese Medicine Research in Gansu Colleges and Universities and Key Laboratory of Dunhuang Medicine and Transformation at Provincial and Ministerial Level for providing support and assistance for this article.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our article does not deal with human and animal experimentation, so ethical issues are not applicable here.
Funding
This study was supported by the National Natural Science Foundation of China (Nos. 81973595, 82004094 and 82260882).
