Abstract
Background
One of the most effective anticancer medications for treating a range of cancers is cisplatin (Cis). Its use is limited because of its nephrotoxicity.
Objectives
This study aims to evaluate the role of linarin (LIN) in protecting rats from Cis-induced nephrotoxicity.
Materials and Methods
The animals were split up into four groups: Group I consisted of control animals; group II consisted of Cis-treated animals; group III consisted of Cis-treated animals that received a 25 mg/kg dosage of LIN; group IV consisted of Cis-treated animals that received a 50 mg/kg dose of LIN. We measured the kidney and body weights of each experimental group. Blood samples were taken to measure the blood urea nitrogen (BUN) and Cr levels in the test animals. To further evaluate the effects of LIN therapy in a dose-dependent manner, an investigation of antioxidant enzymes, oxidative stress indicators, plasma sodium and potassium levels, inflammatory markers, and histopathology was conducted.
Results
We found that giving LIN to Cis-treated rats helped to enhance renal function. The animals’ body weight, histopathological conditions, and amount of antioxidant enzymes were all markedly elevated by LIN in the animals. Additionally, it decreased kidney weight, BUN and Cr levels, oxidative stress indicators, and inflammatory markers.
Conclusion
Therefore, we conclude that LIN can be utilized to protect the kidneys from damage caused by Cis therapy. Nurses can promote well-being and recovery in patients at risk of Cis-induced nephrotoxicity by encouraging gentle physical activities and providing emotional and psychological support, leveraging LIN’s protective effects.
Introduction
The negative impact of chemicals on the activity of the kidneys is known as nephrotoxicity. Metals like mercury, lead, and arsenic and abuse drugs like cocaine are a few examples of these substances (Barnett & Cummings, 2018). Numerous functional outcomes are possible, such as any combo of blood pressure regulation impairment, renal endocrine function, and/or glomerular or tubular failure (Skinner, 2011).
One effective anticancer medication is Cis. Combination chemotherapy regimens based on Cis are presently the first line of treatment for a variety of malignancies. An increase in dosage greatly enhances the therapeutic benefits of Cis. Nevertheless, Cis’s cumulative nephrotoxicity and neurotoxicity restrict high-dose treatment (Hanigan & Devarajan, 2003). Many biological processes in human cells depend on metals, and the medication Cis contains metallic platinum. It is thought to have an impact on several signal transduction pathways, which in turn trigger the onset of apoptosis. Studies have also revealed that, similar to the majority of metals, contributes to the production of reactive oxygen species (ROS). Influences the tumor-suppressor protein p53, triggers apoptosis through intrinsic caspases and the death receptor–tumor necrosis factor (TNF) interaction, results in mitochondrial dysfunction, and influences calcium signaling within the cell by placing stress on the endoplasmic reticulum (ER) (Manohar & Leung, 2018).
One indicator of nephrotoxicity involves alterations in renal function as determined by the serum creatinine (sCr), blood urea nitrogen (BUN), glomerular filtration rate (GFR), or urine output; however, nephrotoxic ants can harm the kidneys without affecting any established clinical marker of renal function (Zhou et al., 2008). As of right now, no medication can be used therapeutically to prevent or treat nephrotoxicity caused by Cis. Numerous low-toxicity, high-efficacy medicines derived from natural materials have been created to guard against kidney injury caused by Cis. For instance, curcumin, pomegranate, and ginseng have anti-inflammatory and antioxidant properties. They may also be able to prevent oxidative stress by raising the levels of antioxidant enzymes. These organic compounds might be used as supplements to lessen the nephrotoxicity caused by Cis since they may have anti-inflammatory and antioxidant qualities (Fang et al., 2021).
Flavonoids have been identified to be one of the most significant naturally occurring phenolic compounds with a wide range of beneficial biological qualities among the several phytochemical classes. A glycosylated flavone called linarin (LIN) has been found in several plant species, mostly in the Asteraceae and Lamiaceae families. LIN demonstrates antioxidant, anti-inflammatory, antiosteoporosis, antiapoptosis, antidiabetic, antipyretic, and antimicrobial activities (Mottaghipisheh et al., 2021). In the present study, the objective was to determine the ameliorative effect of LIN on Cis-induced nephrotoxicity in rats via determining body weight (BW), kidney weight (KW), level of BUN and Cr, antioxidant markers, level of sodium and potassium in plasma, proinflammatory cytokines, histological observations. LIN, an antioxidant drug, has been found to protect against Cis-induced nephrotoxicity through its anti-inflammatory and antioxidant properties. It reduces inflammation by lowering proinflammatory biomarkers and decreases oxidative stress by neutralizing free radicals. Nursing care for patients undergoing similar treatments should include monitoring renal function, administering may be as prescribed, managing hydration, providing nutritional support, preventing and managing complications, and managing pain and symptoms.
Materials and Methods
Materials
LIN and Cis were purchased from Sigma–Aldrich (USA). We bought TNF-α, interleukin (IL)-1β, and IL-6 enzyme-linked immunosorbent assay (ELISA) kits from R&D systems (Minneapolis, MN). The investigations employed only analytical-grade chemicals and all other chemicals were purchased from standard suppliers.
Animals
We obtained 24 Wistar male rats weighing between 200 and 220 g from the approved animal center. Groups of six rats per cage were housed with unlimited access to food and water, and they were maintained at a constant temperature of 23 ± 2.0°C with a 12-h light and dark cycle. The Animal Ethics Committee’s Guidelines for the Use of Experimental Animals were followed when conducting the investigations.
Experimental Design
The animals were divided into four groups, each with six animals. Group I received oral administration of saline water as the control group. Group II: also called the Cis-induced single dosage of 7.5 mg/kg i.p. Groups III and IV were administered Cis with LIN at doses of 25 and 50 mg/kg i.p., respectively.
Morphometric Analysis
The experimental animals’ BW was measured on days 0th and 21st of the study and compared across all experimental groups. After the investigation, the KW of the animals was measured and compared across all experimental groups.
Blood Sample Collection and Serum Separation
Before their execution at the end of the investigation, every animal received intramuscular injections of xylazine and ketamine hydrochloride. After the blood was drawn, it was transferred to dry, anticoagulant-free tubes and allowed to clot for 15 min at room temperature (22 ± 2°C) while tilted. The serum was then stored for further biochemical analysis at −80°C.
Preparation of Tissue for Homogenate and Microscopic Observation
Upon completion of the study, the kidneys of the entire experimental rat were promptly removed and weighed. The kidneys were used to create the homogenate after being cleaned with normal saline. The renal homogenates’ supernatants were centrifuged and then kept at −80°C until they were needed to measure oxidative stress and antioxidant indicators.
Estimation of BUN and Creatinine
BUN and serum Cr levels were measured. They were calorimetrically measured using commercially accessible specific kits and the protocol provided with the product (Larsen, 1972; Tiffany et al., 1972).
Estimation of Antioxidant Biomarkers
The levels of the antioxidant biomarkers, such as malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), glutathione reduced (GSH), and glutathione peroxidase (GPx), were measured calorimetrically using kits that were available commercially and by following the manufacturer’s instructions (Aebi, 1984; Habig et al., 1974; Nielsen et al., 1997; Nishimura et al., 2000).
Estimation of Sodium and Potassium Level
To determine the amounts of sodium and potassium, samples were kept at a temperature of −30°C. Sodium and potassium levels in plasma samples were measured with a flame photometer. To quantify potassium and sodium in plasma, samples were diluted 1:200 (Mayer & Starkey, 1977).
Estimation of Proinflammatory Cytokines
Using an ELISA kit, the levels of IL-6, TNF-α, and IL-1β on homogenized renal tissues were measured to investigate the proinflammatory cytokines generated in the kidneys as a result of Cis poisoning (Grassi et al., 1991; Wong & Clark, 1988).
Histoarchitecture Study
The kidneys that were removed from the experimental animals were cleansed using isotonic saline and left in 10% neutral buffered formalin for a duration of 48 h. Paraffin wax was used to enclose the kidneys after the procedure. The parafilm blocks were cut into small slices that were 5–7 µm thick using a microtome. The thin slices were stained with hematoxylin and eosin and then examined under a light microscope (Olympus, Tokyo, Japan) using 10× magnification (scale bar indicates 10× magnification).
Statistical Analysis
The statistical analysis was carried out with GraphPad Prism. The data is presented as mean ± standard deviation (SD). To determine if there were any differences between the groups, an analysis of variance (ANOVA) and the Tukey’s post hoc test were employed. Differences between means were considered statistically significant if p < 0.05.
Results
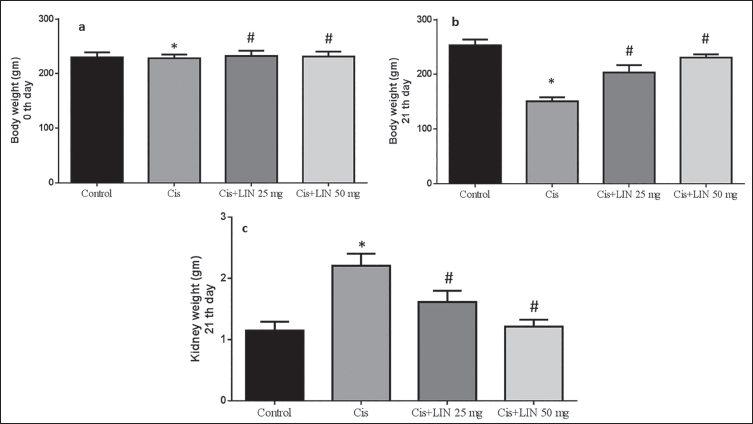
Effect of LIN Administration on the BW in Cis-treated Rats
The impact of LIN on the BW and KW of rats receiving Cis-treatment is seen in Figure 1(a–c). The experimental animals’ BWs were the same in each group on the 0th day. The BW of the animals treated with Cis alone was found to be significantly decreased than that of the normal control group. The BW of rats (groups III and IV) that were given LIN in a dose-based manner after being induced with Cis toxicity significantly increased.
The KW of the animals treated with Cis alone was significantly greater than that of the normal control group. The KW of rats (groups III and IV) that were given LIN in a dose-based manner after being induced with Cis toxicity significantly decreased.
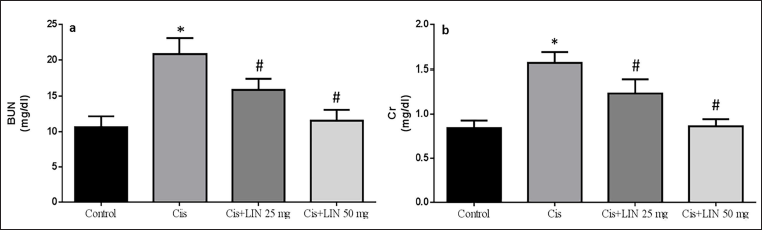
LIN on Serum Levels of BUN and Creatinine in Rats with Cis-induced Nephrotoxicity
The effects of LIN on the serum levels of Cr and BUN in Cis-treated rats are shown in Figure 2a and b, respectively. The results showed that the rats exhibited acute nephrotoxicity 24 h after administering Cis, as seen by a significant increase in BUN and Cr levels. BUN and creatinine levels were significantly lower in rats treated with LIN in a dose-dependent manner (groups III and IV) than in rats treated with Cis (group II) for nephrotoxicity produced by Cis.
Effect of LIN on the Activity of Antioxidant Enzymes in Cis-treated Rats
The effect of LIN on kidney antioxidant enzyme expression is seen in Figure 3.
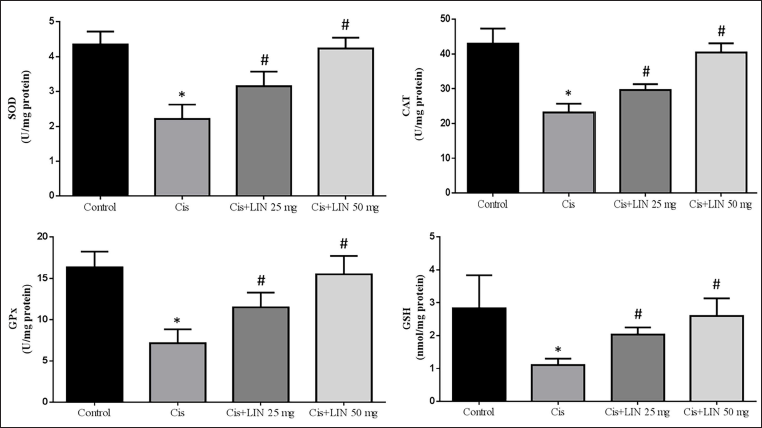
In Cis-treated animals (group II), the expression of SOD, CAT, GPx, and GSH content was significantly downregulated than in the control group. Treatment of Cis-treated rats with LIN in a dose-based manner (groups III and IV) markedly increased these antioxidant enzymes compared to the Cis-treated group (group II).
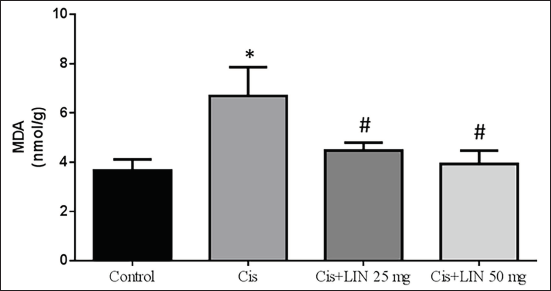
LIN on Oxidative Stress Biomarker in Cis-treated Rats
Figure 4 shows the effects of LIN on the MDA level in Cis-treated rats. The results showed that, in contrast to the control group, animals treated exclusively with Cis (group II) displayed elevated levels of oxidative stress indicators. After receiving dose-dependent treatment with LIN (groups III and IV) for rats suffering from Cis-induced nephrotoxicity, the level of MDA reduced significantly.
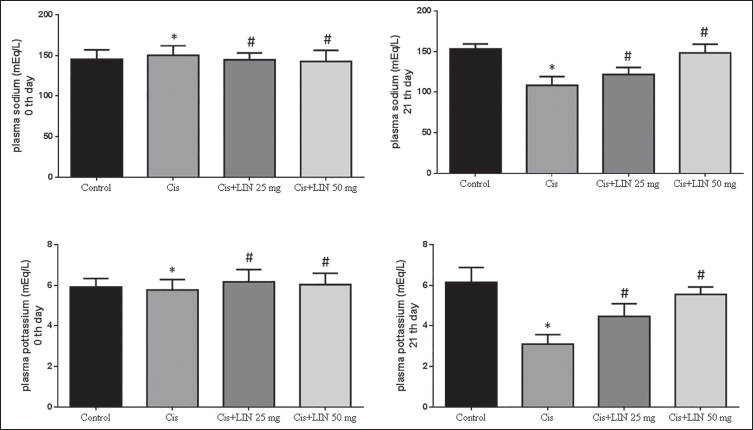
Effect of LIN on Plasma Sodium and Potassium Levels in Cis-treated Rats
The effects of LIN on the plasma sodium and plasma potassium levels in Cis-treated rats are shown in Figure 5, respectively. Compared to the 0th day and 21st day, the plasma sodium and potassium levels were elevated in the control group and LIN-treated groups. Meanwhile, in Cis alone, group II’s level was slightly reduced. The plasma potassium and sodium levels were increased in the LIN-treated animals (groups III and IV) compared to the Cis-treated group (group II).
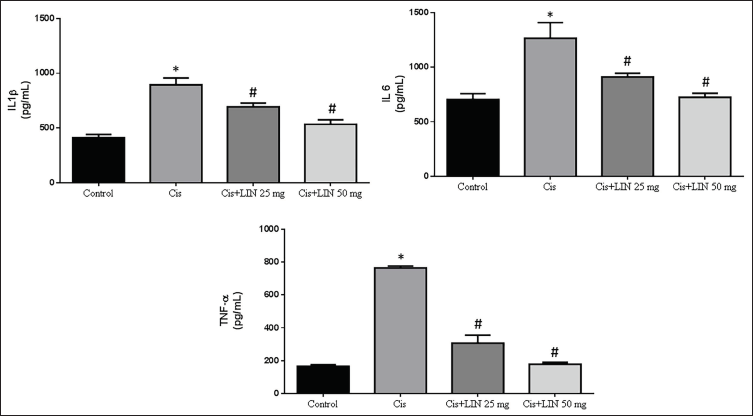
Effect of LIN on Proinflammatory Cytokines in Cis-treated Rats
The impact of LIN on the activity of proinflammatory cytokines (IL-1β, IL-6, and TNF-α) in rats receiving Cis treatment is depicted in Figure 6. The results showed that, in contrast to the control group, rats treated exclusively with Cis (group II) displayed significantly increased levels of IL-1β, IL-6, and TNF-α. The levels of IL-1β, IL-6, and TNF-α in rats with Cis-induced nephrotoxicity were significantly lower in groups III and IV treated with LIN in a dose-dependent manner than in group II treated with Cis.
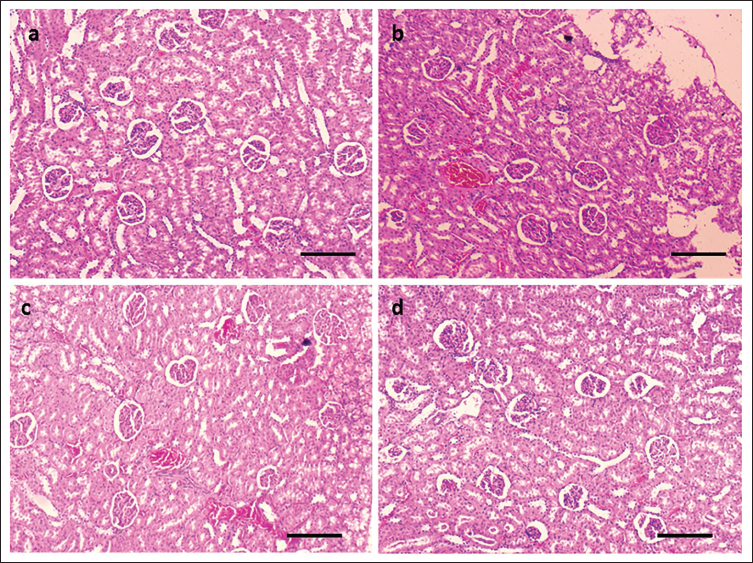
Effect of LIN on Histopathology of Cis-treated Rats
The kidney tissue of the control animals showed normal morphology (Figure 7a). The kidney tissue of the group II rat that was treated with Cis showed signs of severe damage (Figure 7b). Rats with Cis-induced nephrotoxicity (groups III and IV) who received dose-dependent treatment showed much less kidney damage and improved morphology, as seen in Figure 7c and d.
Effect of Linarin (LIN) on the Morphology of the Rat Kidneys with Cis-induced Nephrotoxicity Stained with Hematoxylin and Eosin (H&E). (a) Control Group: Healthy Kidney Structure was Seen. (b) Cis Group: The Kidney is Severely Damaged. (c and d) Cis+ LIN Group 25 and 50 mg showed Dramatic Improvement in the Morphologic Appearance under a Light Microscope (Olympus, Tokyo, Japan) using 10× Magnification (Scale Bar indicates 10× Magnification).
Discussion
One of the leading causes of acute renal damage is nephrotoxicity induced by Cis, a significant antitumor medication. Numerous investigations have demonstrated that Cis causes renal tubular cell transport mechanisms to be disrupted, ATPase activity to be reduced, cell cycle arrest to be induced, and apoptosis or necrosis to be induced in the kidney cells. Additional pathways leading to kidney damage caused by Cis include oxidative stress, fibrogenesis, and inflammation. The usage of natural treatments has increased recently to lessen the nephrotoxic impact of Cis. Among therapeutic plants, Nigella sativa is given higher weight due to its diverse pharmacologic qualities as well as its extensive historical and religious history (Hosseinian et al., 2016).
The goal of the current investigations was to test the hypothesis that consuming LIN would minimize the negative effects of Cis on the kidneys and other organs, allowing for the safe and effective use of Cis in the treatment of different types of cancer. Numerous experimental studies have demonstrated that administering nephrotoxic medicines, such as Cis, to animals resulted in a considerable reduction in the animals’ BW. One possible explanation for the weight loss caused by Cis might be its gastrointestinal toxicity and decreased food intake (Sahu et al., 2011). In line with this, we also found that, at the end of the experiment, the mean BW of the animals in group II, the animals treated with Cis, had drastically decreased. Until the conclusion of the experiment, the weight of the Cis-induced rat treated with LIN in a dose-dependent manner (groups III and IV) stayed constant. Similarly, animals treated with Cis-induced control (group II) exhibited an increase in KW associated with edema or inflammation caused by the drug’s capacity to induce tubular necrosis (Nematbakhsh et al., 2013). When compared to the group II animals, the animals treated with LIN in a dose-dependent manner (groups III and IV) showed a significant drop in KW, which may be connected to LIN’s anti-inflammatory properties.
Several studies have shown that Cis treatment caused damage to rats’ tubules and glomeruli, which resulted in an increase in Cr and BUN levels in their serum (Bami et al., 2017). In the current investigation, we found that the level of Cr and BUN in the Cis-treated animals dramatically decreased with treatment with LIN in a dose-based manner (groups III and IV). Cellular antioxidants, both enzymatic and nonenzymatic, are essential for preventing tissue damage caused by ROS. The cellular radical scavenging system regulates both the generation and scavenging of ROS. Included in the radical scavenging mechanism are GPx, SOD, and CAT (Pınar et al., 2022). During detoxification, GSH acts as an endogenous nonenzymatic antioxidant, reducing the accumulation of endogenous ROS and oxidative damage caused by Cis. However, research indicates that receiving Cis reduces these enzymes’ activity (Cheng et al., 2018). Similar to this group II of the study’s Cis-triggered control animals (group II) had reduced levels of SOD, CAT, GSH, and GPx. After administering LIN in groups III and IV at different doses to Cis-treated rats, we noticed that the levels of SOD, CAT, GSH, and GPx increased, suggesting that LIN had antioxidant properties.
It is well recognized that oxidative stress contributes significantly to Cis-induced nephrotoxicity. It has been shown that Cis raises oxidative stress markers in the kidney, including MDA, NO levels, MPO activity, and PC content. It has been shown that oxidative stress may cause modifications to the kidney’s morphology and function. In line with other research, we found that Cis raises kidney MDA. The control group treated with Cis had higher levels of MDA. However, the amount of MDA was decreased when LIN was administered to the Cis-treated animals in groups III and IV according to a dose-based protocol (Allameh et al., 2020). In the current investigation, on the 21st day the level of sodium and potassium in plasma is reduced in the Cis-treated group in comparison to the control group. After the study, it was observed that treatment with LIN in a dose-based manner increased the level of sodium and potassium in plasma.
It was proposed that Cis nephrotoxicity could be related to inflammatory processes. This aligns with the findings of earlier studies of IL-1β, TNF-α, and IL-6 was observed to significantly rise upon Cis treatment (El Amir et al., 2019; Vasaikar et al., 2018). The Cis animals (group II) in this investigation showed increased levels of TNF-α, IL-6, and IL-1β, suggesting nephrotoxicity. The dose-dependently treated Cis-induced animals (groups III and IV) showed lower levels of inflammatory markers, suggesting that LIN had anti-inflammatory properties. Our histological results showed morphological alterations in Cis-treated animals (group II). These findings were consistent with the biochemical data and those from other investigations. The dose-dependent therapy with LIN (groups III and IV) resulted in a significant improvement in the morphology of the kidney. LIN, an antioxidant drug, protects against Cis-induced nephrotoxicity by reducing inflammation and oxidative stress. Nursing care should monitor renal function, manage hydration, provide nutritional support, prevent complications, and manage pain.
Conclusion
According to the current study’s findings, administering Cis caused oxidative stress-related kidney tissue damage in rats. LIN treatment protects against Cis-induced oxidative damage in a dose-dependent manner because of its antioxidant and anti-inflammatory qualities. These findings imply that LIN treatment may be applied as a prophylactic strategy to decrease renal damage caused by Cis. Nurses should educate patients on Cis-induced nephrotoxicity, encourage lifestyle modifications, and provide emotional support, leveraging LIN’s protective effects to improve patient outcomes and quality of life.
Footnotes
Abbreviations
BUN: Blood urea nitrogen; BW: Body weight; CAT: Catalase; Cr: Creatinine; ELISA: Enzyme-linked immunosorbent assay; ER: Endoplasmic reticulum; GPx: Glutathione peroxidase; GSH: Glutathione; KW: Kidney weight; MDA: Malondialdehyde; ROS: Reactive oxygen species; SOD: Superoxide dismutase; TBARS: Thiobarbituric reactive.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the Ethics Committee of Shanxi Bethune Hospital (Approval Number: SBQDL-2023-120).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
