Abstract
Background
Inflammation mediated by hyperglycemia, lipid metabolism disorders, abnormal myocardial energy metabolism, myocardial cell damage, and changes in ventricular structure are important mechanisms in the occurrence and development of diabetic cardiomyopathy (DCM). Cinnamon polyphenols (CP) are extracted from the traditional Chinese medicine cinnamon, which is believed to have cardioprotective effects and has been used in China for hundreds of years.
Objectives
This study aimed to investigate the therapeutic effects of CP on DCM and its possible mechanisms.
Materials and Methods
The DCM model was established by a single intraperitoneal injection of 85 mg/kg 1% streptozotocin in rats. The treatment group was orally administered 135 mg/kg CP for 28 consecutive days, a dosage verified by experiments on AC16 myocardial cells and detecting liver and kidney injury markers.
Results
CP could reduce high blood sugar, high blood lipids, left ventricular mass index, levels of myocardial injury markers, adenosine diphosphate (ADP), and cyclic adenosine monophosphate (cAMP) in the model group. It also improved weight loss, and increased myocardial adenosine triphosphate (ATP), adenosine monophosphate (AMP), phosphocreatine (PCr), and ATP/ADP, AMP/ATP, and PCr/ATP ratios. The expression of p-mTOR/mammalian target of rapamycin (mTOR), p62, and peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC1α) in the myocardium of the CP group was enhanced, while the expression of LC3II/LC3I was inhibited.
Conclusion
CPs can alleviate myocardial damage by reducing blood sugar, blood lipids, and abnormal autophagy levels while promoting the restoration of myocardial energy metabolism homeostasis.
Keywords
Introduction
Diabetic cardiomyopathy (DCM) is a condition in which individuals with diabetes experience structural and functional abnormalities in their hearts. This condition is not related to other common cardiovascular risk factors like coronary artery disease and hypertension. DCM is a significant contributor to the high mortality rates observed in diabetic patients (Jia et al., 2018; Ogrotis et al., 2023). During the initial phases of DCM, notable characteristic features include left ventricular hypertrophy and abnormal cellular signal transduction, which can result in impaired cardiac diastolic function. However, as the disease progresses, the heart deteriorates further, eventually leading to systolic dysfunction (Murtaza et al., 2019).
The pathogenesis of DCM involves a range of factors, including metabolic abnormalities, autophagy dysregulation, oxidative stress, inflammation, mitochondrial dysfunction, and endothelial dysfunction. These factors collectively exert their effects at systemic, myocardial, and molecular levels (Marwick et al., 2018; Ritchie & Abel, 2020). Among the above factors, dysfunction in energy metabolism is vital to the development of metabolic disorders.
Adenosine triphosphate (ATP) is produced by mitochondria, which serve as the primary energy source for cellular functions, as well as regeneration processes (Herzig & Shaw, 2018). The heart, being the most energy-demanding organ in the body, heavily relies on ATP and other energy substrates for its proper function (Doenst et al., 2013). Even a slight decrease in ATP synthesis efficiency can significantly impair myocardial cell function.
The dysregulation of energy metabolism is recognized as a significant factor in the pathogenesis of various metabolic diseases including diabetes. In DCM, a distinctive characteristic of cardiac energy metabolism is the impaired oxidation of glucose accompanied by an increased uptake of fatty acids. This metabolic shift results in reduced energy production efficiency and exacerbates lipid accumulation, ultimately leading to myocardial damage (Jia et al., 2016).
A cellular degradation pathway called autophagy is regulated by nutrient levels and cellular metabolic balance in a tight manner (Mizushima et al., 2010). Maintaining cellular homeostasis and ensuring normal physiological conditions in myocardial cells are crucial functions (Dong et al., 2010). However, excessive autophagy can destroy cytoplasm and organelles, releasing apoptotic factors and causing myocardial damage (Nishida et al., 2008).
The mammalian target of rapamycin (mTOR), a serine/threonine protein kinase, is a significant contributor to energy metabolism and cell growth (Yarahmadi et al., 2021; Zoncu et al., 2011). mTOR can promote mitochondrial metabolism by mediating the coactivation of peroxisome proliferator-activated receptor gamma coactivator 1-alpha (PGC1α) (Laplante & Sabatini, 2013). mTOR can also prevent autophagy by inhibiting the UNC5-like 1 (ULK1) complex (Filomeni et al., 2015). Changes in markers related to autophagy are strongly linked to the energy requirements of the heart. mTOR signaling pathway, which acts as a key energy-sensing element, connects cellular energy levels with the autophagy machinery. By establishing a connection, autophagy can effectively regulate cellular metabolic robustness and maximize cellular viability by optimizing ATP availability and improving energy efficiency (Kanamori et al., 2015).
Traditional Chinese medicine offers multiple pharmacological effects, targets multiple pathways, and is cost-effective, making it important in the treatment of chronic illnesses (Atale et al., 2020).
Cinnamon, a traditional Chinese medicine, derived from the dried bark of Cinnamomum cassia Presl, belonging to the Camphoraceae family, has the potential to protect the cardiovascular system (Ziegenfuss et al., 2006). Cinnamon polyphenols (CP), as water-soluble components in cinnamon, possess pharmacological effects such as antidiabetic and antioxidant properties. Nevertheless, there has been a scarcity of studies specifically examining their influence on DCM.
Therefore, in this experimental study, we selected CP as the active ingredient from cinnamon to investigate its protective effects on DCM and explore its influence on mechanisms related to energy metabolism.
Materials and Methods
Materials
Streptozocin (STZ) (cat# S17049) and CP extracts [the polyphenol content in the CP extract was analyzed by Professor Xu Xu from Shanghai Institute of Technology, with an effective content of 30%, cat# S29915; the corresponding analysis methods and results can be found in the published article (Shi et al., 2023)] were purchased from Shanghai Yuanye Bio-Technology Co., Ltd.
Mrs Weiliang Gu provided AC16 human cardiomyocytes from Shanghai University of Traditional Chinese Medicine.
Cell Culture and Treatments
AC16 human cardiomyocytes were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and incubated at 37°C in a cell culture incubator with 5% CO2. During the treatment period, AC16 cells were cultured in a normal complete medium for 24 h. Then the cells were divided into groups: The control group (0 mM glucose) and the high-glucose group (11–77 mM glucose). Glucose solutions of different concentrations were prepared using serum-free medium, and the cells were cultured for 24 h. Cell viability was measured using the CCK-8 assay. After determining the optimal intervention concentration of glucose, AC16 cells were cultured in a normal complete medium for 24 h. Then the cells were divided into groups: The control group (0 mM glucose), the high-glucose group (66 mM glucose), and the CP intervention group (0.016–1,250 µg/mL). Different concentrations of CP solutions were prepared using a serum-free medium containing 66 mM glucose, and the cells were further cultured for 24 h. Cell viability was measured using the CCK-8 assay.
Animals
Eighteen 8-week-old male Sprague-Dawley rats with a body weight of 200–220 g (Shanghai SLAC Laboratory Animal Co., Ltd) were applied in this experiment. Rats in the experiment were kept in a controlled environment with a temperature range of 22–24°C, humidity levels between 40% and 45%, and a 12-h cycle of light and darkness. They were given unrestricted access to food and water. In our study, animal experiments were conducted by the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996) and approved by the Animal Care and Use Committee of Shanghai University of Traditional Chinese Medicine (Approval No. PZSHUTCM221017002). On the last day of the experiment, all the rats were deeply anesthetized by urethane to measure electrocardiography (ECG) and take blood. When blood sampling was complete, all the rats were euthanized by rapid intravenous injection of 75 mg/kg of potassium chloride before subsequent sampling operations were performed.
Experimental Design
The rats were divided into two groups: The normal control (NC) group (n = 6) and the model control (MC) group (n = 6), using random assignment. Diabetes was induced in rats by a single intraperitoneal injection of STZ (dissolved in sodium citrate buffer with pH = 4.5) at a dose of 85 mg/kg body weight. Rats in the NC group received an injection of sodium citrate buffer at the same volume. The levels of fasting blood glucose (FBG) were monitored 3 days after the STZ injection. Diabetic rats with FBG levels > 16.7 mmol/L were selected for the MC group (n = 6) and CP group (n = 6) for subsequent experiments. Rats in the CP group were orally administered CPs via gavage at a dose of 135 mg/kg body weight daily.
Sample Collection
At the end of 28 days of administration, all the rats were anesthetized with an intraperitoneal injection of 20% urethane (5 mL/kg), the body mass was measured, and ECG was recorded. Blood was taken via the abdominal aorta, and serum was obtained by centrifugation of the blood at a speed of 3,500 revolutions per minute for a duration of 10 min. The left ventricles were separated, and weights were recorded. The left ventricles were separated, and weights were recorded. The serum and left ventricles were stored at a temperature of −80°C.
Assessment of ECG
ECG was assessed by a multichannel biological signal system (RM6240C, Chengdu Instrument Factory) to evaluate cardiac damage in DCM and the protective effect of CP.
Histological Analysis
Rat heart tissues were obtained and rinsed with chilled saline solution, followed by fixation in 4% paraformaldehyde and embedding in paraffin. Hematoxylin and eosin (H&E) were used to stain the tissue slices. A light microscope was utilized to conduct morphological examinations. Pathological alterations, including hypertrophied cardiomyocyte interstitial, disrupted organization of myocardial fibers, infiltration of inflammatory cells, and fibrotic characteristics, are indicated by a single marker for each occurrence. The ultrastructure of the myocardium was analyzed using transmission electron microscopy (Tecnai G2 Spirit BioTWIN).
Examination of General Blood Biomarkers
Serum levels of glucose (GLU), total cholesterol (TC), triglyceride (TG), creatine kinase (CK), lactate dehydrogenase (LDH), high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C) were determined with the corresponding kits (cat# A154-1-1, A111-2-1, A110-2-1, A032-1-1, A020-1-2, A112-2-1, A113-2-1, Nanjing Jiancheng Bioengineering Institute, Nanjing, China). TC to HDL-C (TC/HDL-C) and LDL-C to HDL-C (LDL-C/HDL-C) ratios were computed.
Measurement of Serum AGEs and CRP
The contents of serum advanced glycation end products (AGEs) (cat# HLE20290) and C-reactive protein (CRP) (cat# HLE97152) were measured by ELISA with kits from Shanghai Haling Biological Technology Co., Ltd. (Shanghai, China).
Measurement of Myocardial Energy Metabolism
The perchloric acid method was used to handle the heart tissues to acquire the extracts. The energy levels in the extracts were measured with high-performance liquid chromatography (HPLC, America, Ultimate 3000, Thermo Fisher Scientific). Detection indexes included ATP, adenosine diphosphate (ADP), adenosine monophosphate (AMP), phosphocreatine (PCr), and cAMP. PCr/ATP, ATP/ADP, and AMP/ATP ratios were computed.
Western Blotting Analysis
Total protein was obtained by collecting and homogenizing the left ventricular tissues in RIAP lysis buffer, followed by a 30-min ice lysis and centrifugation of the lysates at 4°C and 12,000 rpm for 10 min to collect the supernatants. A bicinchoninic acid (BCA) Protein Assay Kit (cat# P0012S, Beyotime Biotechnology Co., Ltd., Shanghai, China) determined the total protein concentration. Afterward, the entire proteins were segregated through SDS-PAGE electrophoresis and then transferred onto a PVDF membrane. After blocking with 5% nonfat milk for 2 h, the membranes were individually incubated with primary antibodies overnight at 4°C (cat# 2983T mTOR 1:1,000, cat# 5536S Phospho-mTOR (Ser2448) 1:1,000, cat# 2178S PGC1α 1:1,000, cat# 8025S SQSTM1/p62 1:1,000, cat# 12741T LC3A/B 1:1,000, Cell Signaling Technologies, Danvers, Massachusetts, USA, cat# AF2835 β-Tubulin Mouse Monoclonal Antibody, Beyotime Biotechnology Co., Ltd., Shanghai, China). Then, the membranes were washed with PBST buffer five times for 5 min each and subsequently incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies for 1 h at room temperature (cat# A0216 HRP-labeled Goat Anti-Mouse IgG 1:1,000, cat# A0208 HRP-labeled Goat Anti-Rabbit IgG 1:1,000, Beyotime Biotechnology Co., Ltd., Shanghai, China). Specific proteins were visualized using an enhanced chemiluminescence (ECL) reagent (cat# P0018AS, Beyotime Biotechnology Co., Ltd., Shanghai, China). The membranes were washed with stripping buffer three times for 10 min each to dislodge primary and secondary antibody conjugates. Then, the membranes were washed with PBS buffer three times for 5 min each and blocked again.
Statistical Analysis
The average ± standard error of the mean (SEM) is used to represent all numerical data. Statistical significance among the three groups was determined by one-way ANOVA and LSD post hoc tests. Statistical analysis was performed using SPSS 26.0 software. A value of p < 0.05 was considered significant, and p < 0.01 is considered more significant.
Results
CP Improves AC16 Cardiomyocyte Survival in Hyperglycemia Environment
After different concentrations of GLU were applied to AC16 myocardial cells, with the increase of GLU concentration, the cell viability showed a gradual decrease compared to the 0 mM GLU group. Furthermore, when the GLU concentration reached 33 mM, with the increase of GLU concentration, the cell viability of each group decreased significantly compared to the 0 mM GLU group. When the GLU concentration was 66 mM, the cell viability was significantly lower compared to the control group and was below 80%. Although the decrease in cell viability became more significant with the continuous increase of GLU concentration, considering the osmotic effect produced by high-concentration GLU could also have adverse effects on myocardial cells, hence, 66 mM GLU was chosen as the concentration for subsequent experiments inducing high sugar (Figure 1A).
Effects of Different Concentrations of Cinnamon Polyphenols (CP) on the Survival of AC16 Cardiomyocytes under Hyperglycemia Induction. The Effects of Glucose (GLU) Ranging from 11 to 77 mM (A) and CP Ranging from 0.016 to 1,250 µg/mL (B) on the Cell Viability of AC16 Myocardial Cells were Measured. Data are shown as the Mean ± Standard Error of the Mean (SEM). n = 6 in Each Group. *p < 0.05, **p < 0.01 in Contrary to Normal Cultured Cell Group; #p < 0.05, ##p < 0.01 in Contrary to 66 mM GLU Group.
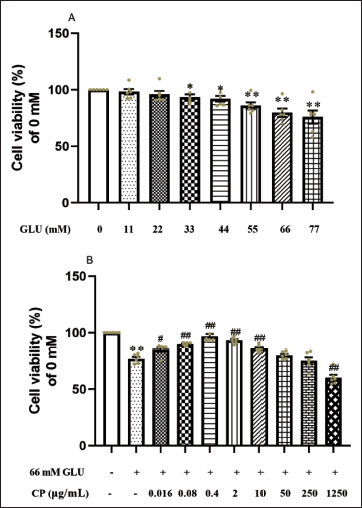
After 24 h of treatment with CP at concentrations ranging from 0.016 to 1,250 µg/mL on myocardial cells induced by 66 mM GLU, compared to cells in the hyperglycemia environment group, the cell viability of the 0.016, 0.08, 0.4, 2, and 10 µg/mL groups all showed significant increases, while there was no significant change in cell viability when the concentration was 50 µg/mL, and cell viability continued to decline with increasing concentrations. Among them, when the CP concentration was 0.4 µg/mL, the cell viability was the highest (Figure 1B).
CP shows No Harm to the Liver and Kidneys of DCM Rats
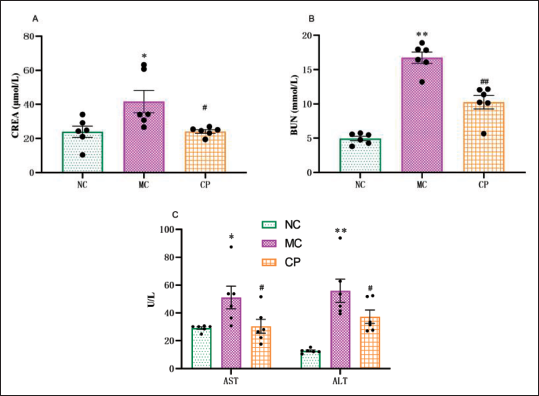
The levels of BUN, CREA, AST, and ALT in the MC group rats were significantly elevated compared to those in the NC group rats. However, after intervention with CP, the levels of BUN, CREA, AST, and ALT in the CP group rats were significantly reduced compared to those in the MC group rats (Figure 2A–C).
Effects of Cinnamon Polyphenols (CP) on Markers of Liver and Kidney Injury in Diabetic Cardiomyopathy (DCM) Rats CREA (A), BUN (B), AST and ALT (C) were Determined in NC, MC, and CP Groups using the Appropriate Kits. Data are shown as the Mean ± Standard Error of the Mean (SEM). n = 6 in Each Group. *p < 0.05, **p < 0.01 in Contrary to NC Group; #p < 0.05, ##p < 0.01 in Contrary to MC Group.
CP improves General Parameters in DCM Rats
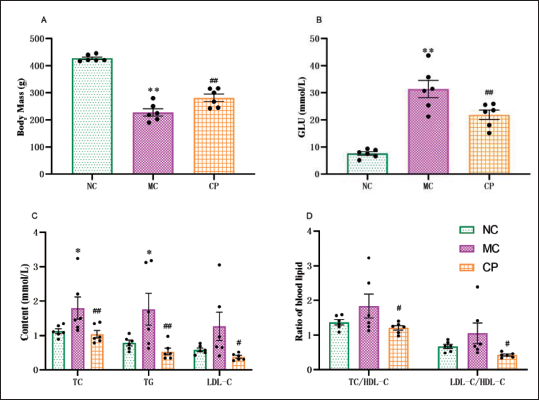
Compared to the NC group, the body mass of STZ-induced DCM rats exhibited a noteworthy decrease. However, the CP group tended to increase body mass in comparison to the MC group (Figure 3A). The blood glucose levels in MC group rats exhibited a substantial increase, whereas, after CP treatment, the phenomenon of high blood glucose levels was considerably improved (Figure 3B). In contrast to the NC group, the MC group showed a lift in the levels of TC, TG, and LDL-C in blood lipids, while after CP treatment, this trend was reversed (Figure 3C). As opposed to the NC group, the MC group showed an upward trend in the TC/HDL-C and LDL-C/HDL-C ratios, whereas the CP group demonstrated a downward trend about MC group (Figure 3D).
Effects of Cinnamon Polyphenols (CP) on Body Mass, Blood Glucose, and Blood Lipids in Diabetic Cardiomyopathy (DCM) Rats. The Statistical Graphs of Body Weight (A) and Blood Glucose (B) were Performed. Total Cholesterol (TC), Triglyceride (TG), and Low-Density Lipoprotein Cholesterol (LDL-C) were Determined in NC, MC, and CP Groups using the Appropriate Kits (C). The TC/High-Density Lipoprotein Cholesterol (HDL-C) and LDL-C/HDL-C Ratios between Groups were also shown (D). Data are shown as the Mean ± Standard Error of the Mean (SEM). n = 6 in Each Group. *p < 0.05, **p < 0.01 in Contrary to NC Group; #p < 0.05, ##p < 0.01 in Contrary to MC Group.
CP improves Cardiac Damage in DCM Rats
ECGs of rats are presented in Figure 4A. The R-wave amplitude of the ECGs of MC group rats was notably higher compared to the NC group, and this elevated trend was remarkably reversed with CP treatment. Histological examination of myocardial H&E staining revealed well-aligned myocardial fibers with minimal interstitial enlargement and a small amount of inflammatory cell infiltration in the NC group. In contrast, the MC group exhibited occasional disruption of myocardial fibers, along with increased inflammatory cell infiltration and interstitial enlargement, and the pathological scores were markedly larger than those of the NC group. However, the pathological changes in the myocardium were improved, and the pathological scores were decreased in the CP group versus the MC group (Figure 4B).
Effect of Cinnamon Polyphenols (CP) on the Physiological Structure and Function of the Heart in Diabetic Cardiomyopathy (DCM) Rats. Representative Screenshots of Electrocardiogram (A) and Levels of R-wave Amplitude (A) were Performed. Representative Cross-sections of Hematoxylin and Eosin (H&E)-Stained Rat Cardiomyocytes (B) were Examined under Light Microscope (200×, Scale Bar = 250 µm). The Pathology Score Sheet was shown (B, n = 4 in Each Group). Representative Images of Myocardial Transmission Electron Microscopy (C) were shown (TEM 6,000×, Scale Bar = 2 µm). Heart Mass Index (D) was Determined. The Content of Creatine Kinase (CK) and Lactate Dehydrogenase (LDH) (E), Markers of Myocardial Injury, were Determined. Data are shown as the Mean ± Standard Error of the Mean (SEM). n = 6 in Each Group. *p < 0.05, **p < 0.01 in Contrary to NC Group; #p < 0.05, ##p < 0.01 in Contrary to MC Group.
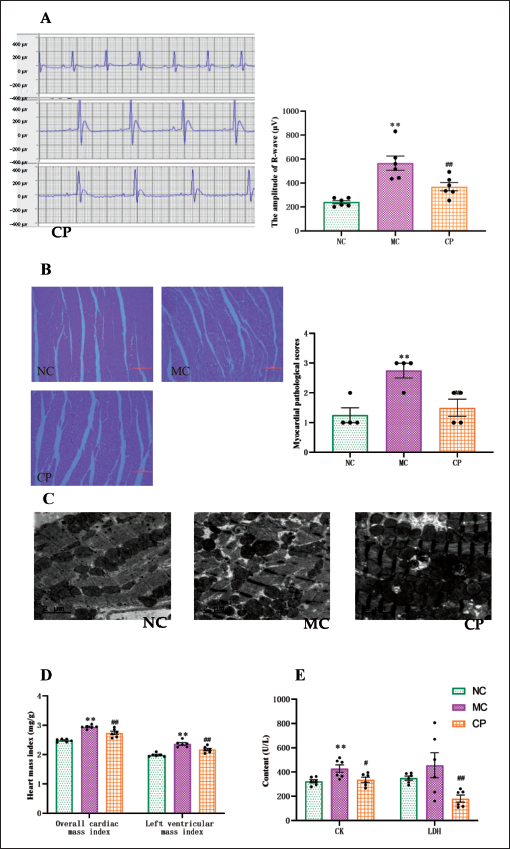
Myocardial myofilaments exhibited intact and orderly arrangement, and mitochondrial size and morphology appeared normal in the NC group. Conversely, the MC group displayed frequent myofilament breakage and disorganization, accompanied by swollen and reduced numbers of mitochondria. However, restoration of myofilament arrangement and normalization of mitochondrial number and morphology were observed after CP treatment (Figure 4C).
The MC group showed a notable rise in both the overall cardiac mass index and left ventricular index in contrast to the NC group, whereas the CP group demonstrated a momentous decline in these indexes contrasting with the MC group (Figure 4D). The levels of CK and LDH in the MC group exhibited a marked upward trend contrary to the NC group, while following CP treatment, the upward trend was reversed (Figure 4E).
CP decreases Oxidative Stress and Inflammation Levels in the Hearts of DCM Rats
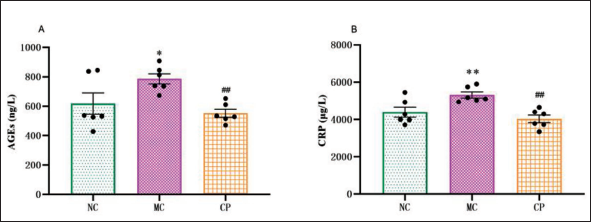
In comparison to NC group, MC group exhibited an increasing trend in AGEs level, while this increasing trend was considerably reversed in CP group (Figure 5A). MC group exhibited a substantial increase in CRP level in contrast to the NC group. After undergoing CP treatment, the level experienced a notable decrease (Figure 5B).
Effect of Cinnamon Polyphenols (CP) on Advanced Glycation End (AGEs) and C-Reactive Protein (CRP) Levels in Diabetic Cardiomyopathy (DCM) Rats. The Serum Levels of AGEs (A) and CRP (B) in Rats were Measured using the Appropriate Kits. Data are shown as the Mean ± Standard Error of the Mean (SEM). n = 6 in Each Group. *p < 0.05, **p < 0.01 in Contrary to NC Group; #p < 0.05, ##p < 0.01 in Contrary to MC Group.
CP improves Myocardial Energy Metabolism in DCM Rats
The MC group presented a noteworthy decrease in ATP and AMP levels in contrast to the NC group, whereas the CP group exhibited a substantial increase in these levels. Furthermore, the MC group demonstrated a notable decline in ADP levels in comparison to the NC group. Similarly, the CP group also presented a comparable decrease versus the MC group (Figure 6A). The MC group presented a notable reduction in PCr content in contrary to the NC group, whereas it displayed a substantial rise after CP treatment. Moreover, cAMP levels in the MC group exhibited no notable alteration as opposed to the NC group, whereas it exhibited a meaningful decrease in the CP group when compared to the MC group (Figure 6B).
Effect of Cinnamon Polyphenols (CP) on Myocardial Energy Metabolism in Diabetic Cardiomyopathy (DCM) Rats. The levels of Adenosine Triphosphate (ATP) (A), Adenosine Diphosphate (ADP) (A), Adenosine Monophosphate (AMP) (A), Phosphocreatine (PCr) (B), and Cyclic Adenosine Monophosphate (cAMP) (B) of Myocardial Tissue in Rats were Measured by High-Performance Liquid Chromatography (HPLC). PCr/ATP (D), ATP/ADP (D) and AMP/ATP (D) Ratios were Calculated. Data are shown as the Mean ± Standard Error of the Mean (SEM). n = 6 in Each Group. *p < 0.05, **p < 0.01 in Contrary to NC Group; #p < 0.05, ##p < 0.01 in Contrary to MC Group.
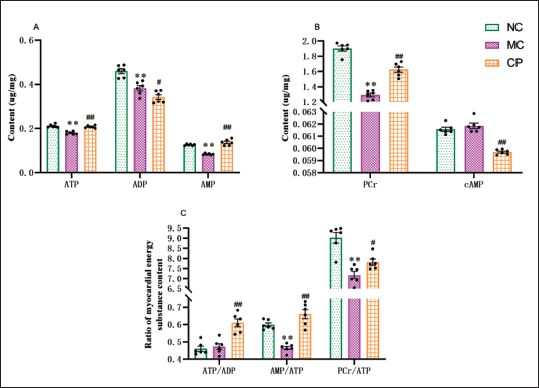
Different from the NC group, ATP/ADP ratio demonstrated no pronounced changes in MC group, while AMP/ATP and PCr/ATP ratios were robustly decreased. However, after CP treatment, all three ratios, namely, ATP/ADP, AMP/ATP, and PCr/ATP, displayed a remarkable increase (Figure 6C).
CP operates on the mTOR Signaling Pathway to Mitigate Myocardial Injury
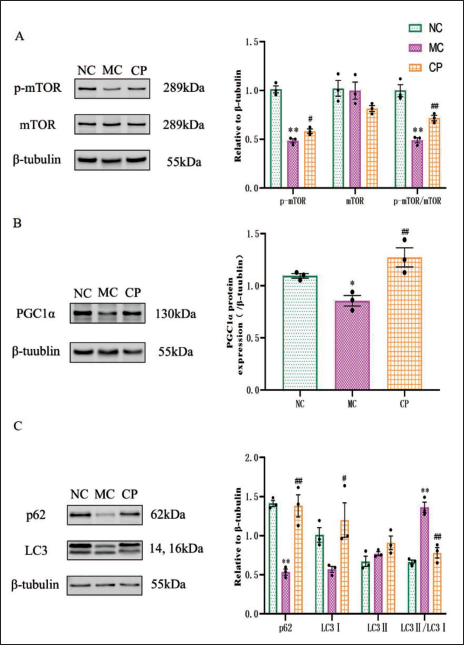
In MC group versus NC group, expression levels of p-mTOR, PGC1α, sequestosome 1 (p62), and microtubule-associated protein 1 light chain 3 (LC3) I were significantly downregulated, while expression levels of mTOR were ascended. In CP group versus MC group, expression levels of PGC1α, p62, and LC3I were considerably increased, while expression levels of mTOR were obviously diminished. LC3II levels did not show any considerable differences among the NC, MC, and CP groups. p-mTOR/mTOR ratios were considerably lower in MC group versus NC group, while they were substantially higher in CP group than MC group. But the LC3II/LC3I ratio displayed a prominent increase in MC group in contrast to NC group, but CP group substantially reversed the increasing trend (Figure 7A, B).
Effects of Cinnamon Polyphenols (CP) on Mammalian Target of Rapamycin (mTOR) Signaling and Autophagy-Related Pathway Proteins. Representative Bands and Quantification of p-mTOR (A), mTOR (A), Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-Alpha (PGC-1α) (B), p62 (C) and LC3 (C) were shown. Data are shown as the Mean ± Standard Error of the Mean (SEM). n = 3 in Each Group. *p < 0.05, **p < 0.01 in Contrary to NC Group; #p < 0.05, ##p < 0.01 in Contrary to MC Group.
Discussion
In this experiment, the hyperglycemia environment caused damage to myocardial cells, resulting in a decrease in cell viability. The experiment investigated the effects of intervention with different concentrations of CP on the cell viability of myocardial cells in a hyperglycemia environment. The results showed that within the range of 0.016–2 µg/mL, the cell viability of myocardial cells in the CP intervention group increased with the concentration of CP, showing a dose-dependent relationship. However, with further increases in concentration, there was no significant improvement in myocardial cell viability. At the same time, when the intervention concentration of CP exceeded the safe range, the survival rate of myocardial cells in the intervention group significantly decreased compared to myocardial cells in the hyperglycemia environment, indicating that CP had a damaging effect on myocardial cells at this concentration. The above experimental results suggest that within a certain safe concentration range, the protective effect of CP on myocardial cells in the hyperglycemia environment exhibits dose dependence, with a concentration at which the effect is maximized. Therefore, based on the in vivo results of CP, this experiment selected the highest intervention concentration of 135 mg/kg CP from previous experiments as the optimal intervention concentration, and it is speculated to be near the concentration at which the effect is maximized.
To verify whether this concentration is near the maximum value and within the safe range of action, this experiment examined liver and kidney injury markers in DCM rats to preliminarily assess whether 135 mg/kg CP would impose a metabolic burden on DCM rats. The experimental results showed that the levels of BUN, CREA, AST, and ALT in DCM rats were significantly elevated, suggesting that DCM rats may have liver and kidney damage due to the presence of DM complications or the hepatorenal toxicity of modeling drugs. However, the levels of BUN, CREA, AST, and ALT in the CP group were significantly lower than those in the MC group, indicating that after intervention with 135 mg/kg CP, liver and kidney damage in DCM rats was alleviated, and hepatorenal toxicity did not worsen. Therefore, 135 mg/kg CP can more scientifically reflect its protective effect on DCM.
Hyperglycemia is a central factor in the pathogenesis of DCM (Sung et al., 2009). Elevated LDL-C levels can lead to the uptake of free fatty acids and the accumulation of TG and TC in myocardium, affecting myocardial energy metabolism and increasing myocardial oxidative stress (Cai et al., 2013; Zhao et al., 2019). In addition, an increase in the ratios of TC/HDL-C and LDL-C/HDL-C also increases the risk of ischemic cardiovascular and cerebrovascular damage. This experiment observed that CP significantly improved the symptoms of hyperlipidemia in diabetic rats, alleviating metabolic disorders of lipid metabolism in diabetic hearts, suggesting that CP may have the potential to reduce the risk of cardiovascular diseases such as DCM and improve the utilization of energy metabolism substrates in diabetic hearts, thereby reducing the burden of accumulation of oxidative products of free fatty acids on diabetic hearts.
One of the morphological manifestations of DCM is left ventricular hypertrophy (Falcão-Pires & Leite-Moreira, 2012). As left ventricular mass increases, cardiovascular risk also increases, indicating a poor prognosis (Fang et al., 2003). In diabetic hearts, early markers of diabetic autonomic neuropathy include an increase in R wave amplitude in the ECG (VanHoose et al., 2010). Meanwhile, DCM presents with left ventricular hypertrophy, which increases the electrical activity of the ventricular wall, thereby increasing the amplitude of the R wave (Laddha & Kulkarni, 2021). In this experiment, CP significantly reduced the elevated R-wave amplitude, left ventricular index, global cardiac index, CK, and LDH levels in diabetic rats. Additionally, results from H&E staining and transmission electron microscopy of the myocardium also showed that CP treatment improved abnormal changes in cardiac structure. This indicates that CP treatment effectively alleviated left ventricular hypertrophy and the consequent myocardial damage in DCM.
The accumulation of AGEs can trigger pathological processes such as inflammation, oxidative stress, and fibrosis, thereby exacerbating myocardial cell dysfunction (Singh et al., 2014). In DCM, overexpression of CRP exacerbates left ventricular dysfunction and remodeling of the diabetic heart through enhanced inflammation, the renin-angiotensin system, and oxidative stress (Saklani et al., 2016). In this experiment, it was found that levels of AGEs and CRP were elevated in DCM rats, suggesting the presence of myocardial inflammation. However, CP treatment reduced the levels of AGEs and CRP, thereby alleviating myocardial inflammation and protecting the hearts of DCM rats.
The heart is the organ with the highest energy consumption in the human body, responsible for maintaining systemic blood supply (Bhargava & Schnellmann, 2017). Energy metabolism plays a crucial role in DCM, and its disruption leads to insufficient energy supply in the heart, further affecting myocardial function (Sung et al., 2015). In this experiment, the levels of ATP, AMP, PCr, ATP/ADP, AMP/ATP, and PCr/ATP in the CP group were significantly higher compared to the MC group. These findings indicate that CP can improve energy metabolism disorders by increasing energy supply and reserves, thereby helping to mitigate existing myocardial damage. Compared to the MC group, the cAMP levels in the CP group were significantly reduced. This reduction may be due to CP’s ability to restore normal cellular signaling, enhance insulin-mediated glucose uptake and utilization, and restore normal glucose metabolism. Furthermore, the decrease in cAMP levels can attenuate the breakdown and oxidation of intracellular fatty acids, primarily relying on glucose as the main source, thus restoring energy metabolism.
Autophagic homeostasis also plays an important role in DCM. Generally, autophagy regulation helps protect myocardial cells, while excessive activation of autophagy may lead to excessive degradation of basic cellular components, resulting in cell death. In cardiac autophagy, excessive activation of autophagy is often considered a transition to heart failure (Kanamori et al., 2015). Impairment of insulin signaling in the diabetic cardiac environment leads to excessive activation of autophagy, which has adverse effects on the diabetic heart. mTOR integrates various cellular signals such as energy, nutrients, and hormones to regulate cell growth, differentiation, survival, metabolism, energy balance, and stress response to achieve cellular homeostasis (Watanabe et al., 2011). mTOR can control mitochondrial oxidative function by activating PGC1α. Different studies have shown that enhancing mitochondrial function by activating the mTOR-PGC1α pathway can compensate for insufficient energy metabolism (Chen et al., 2022; Ji et al., 2015). This experiment found that the expression of mTOR was downregulated in the myocardium of DCM rats, leading to inhibition of PGC1α expression, decreased mitochondrial metabolic capacity, and difficulty in maintaining normal energy supply to myocardial cells, further exacerbating myocardial damage. In contrast, the CP group upregulated the expression of mTOR, thereby increasing the expression of PGC1α, promoting mitochondrial biogenesis and oxidative function. This mechanism helps maintain myocardial energy supply and alleviate myocardial damage in DCM. mTOR activation negatively regulates autophagy initiation. Results from autophagy-related marker detection showed that compared to the NC group, the LC3II/LC3I ratio was increased in the MC group, and p62 levels were decreased. Compared to the MC group, the CP group had a decreased LC3II/LC3I ratio and increased p62 levels. These results suggest that autophagic flux is enhanced in type 1 DCM hearts, and excessive autophagy may exacerbate myocardial damage in DCM. CP treatment can reverse this effect of type 1 DCM by regulating autophagy to improve myocardial damage.
Conclusion
CP can protect the hearts of type 1 DCM rats by improving glucose and lipid metabolism, ameliorating abnormal myocardial structural changes, alleviating myocardial oxidative stress and inflammation levels, and restoring energy homeostasis. CP promotes mitochondrial biogenesis and oxidative function through the mTOR-PGC1α pathway, facilitating the recovery of myocardial energy homeostasis in type 1 DCM rats. Furthermore, CP reduces the abnormally elevated levels of autophagy in the hearts of type 1 DCM rats, restores autophagic homeostasis, and thereby improves cardiac injury in type 1 DCM rats.
Footnotes
Abbreviations
ADP: Adenosine diphosphate; AGEs: Advanced glycation end products; AMP: Adenosine monophosphate; ATP: Adenosine triphosphate; BCA: Bicinchoninic acid; cAMP: Cyclic adenosine monophosphate; CK: Creatine kinase; CRP: C-reactive protein; DCM: Dilated cardiomyopathy; ECG: Electrocardiogram; ECL: Enhanced chemiluminescence; GLU: Glucose; H&E: Hematoxylin and eosin; HDL-C: High-density lipoprotein cholesterol; HPLC: High-performance liquid chromatography; HRP: Horseradish peroxidase; LDH: Lactate dehydrogenase; LDL-C: Low-density lipoprotein cholesterol; LC3: Microtubule-associated protein 1 light chain 3; mTOR: Mammalian target of Rapamycin; p62: Sequestosome 1; PCr: Phosphocreatine; PGC1α: Peroxisome proliferator-activated receptor gamma coactivator 1-alpha.
Author Contribution
Xu Zhang played a role in composing the initial version, as well as being involved in project design, preliminary investigation, and manuscript editing. Xiao-Ting Zhang and Ming-Qiao Hu participated in investigation and data analysis. Shi-Yu Wu and Xu-Xu took part in the examination of energy metabolism. Jian-Ping Gao participated in conceptualization, investigation, and manuscript revision.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
In our study, animal experiments were conducted in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996) and approved by the Animal Care and Use Committee of Shanghai University of Traditional Chinese Medicine (approval No. PZSHUTCM221017002).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 82074055).
