Abstract
Background
Osteoarthritis (OA) is a chronic disease worldwide. With resatovir usually used as an inhibitor, resatovir and glycyrrhizic acid have several pharmacological activities.
Objectives
We intend to explore the mechanism underlying resatovir combined with glycyrrhizic acid in OA.
Materials and Methods
After the establishment of an animal model of OA through anterior cruciate ligament transection and destabilization of the medial meniscus, the rats were subjected to intramuscular injection of resatovir combined with glycyrrhizic acid (60 mg/kg) or 5% glucose (60 mg/kg) every day for 4 weeks. During the progress, the rat movement and the bend score of knees were observed through behavioral studies. Additionally, the SKP2 gene expression and toll-like receptor 4 (TLR4)/nuclear factor-κB (NF-κB)-related signaling pathways were examined, as apoptosis and autophagy were evaluated by flow cytometry.
Results
With increased inflammatory cells in the model group, treatment with resatovir combined with glycyrrhizic acid significantly decreased the number of inflammatory cells. Meanwhile, the treatment resulted in decreased SKP2 expression in OA cells when hindering the migration of preosteoclast cells. Interestingly, it was observed that the bend score increased over the treatment with resatovir plus glycyrrhizic acid and TIPPI% declined dramatically. Resatovir treatment enhanced autophagy and apoptosis in OA cells and decreased pP65, P65, and LC3 expression.
Conclusion
Resatovir combined with glycyrrhizic acid can inhibit the inflammatory response through decreasing SKP2 expression and induce autophagy activation in OA cells through TLR4/NF-κB signaling, thereby attenuating OA progression.
Introduction
Osteoarthritis (OA) is a leading cause of pain and physical disability, especially among older adults (Cui et al., 2020). It is reported that 33.6% of people over 65 years old suffer from OA, and also 13.9% among the youth (over 25 years) (Neogi, 2013). Up to now, it has been proven that abnormal lipid metabolism can accelerate the development of OA (Gkretsi et al., 2011). People with a high-fat diet may increase the risk of OA in offspring (both generations), which leaves problems about the pathogenesis and treatment strategies of OA in the future (Masuyama et al., 2016). In our previous work, macrophages were cultured with 5 µg/mL oxidized low-density lipoprotein (LDL) and polarized into M1 phenotype when activating the release of inflammatory factors and enhancing inflammatory response (van Tits et al., 2011). Several studies have shown higher serum LDL levels in OA patients compared with those in healthy controls (de Munter et al., 2016). LDL can be oxidized in an inflammatory environment, and therefore, under pathological conditions with free radicals present, high levels of LDL can lead to elevated levels of oxidized LDL (oxLDL) (Levitan et al., 2010). Coculture of chondrocytes and synovial fluid can increase the content of inflammatory factors and promote cell apoptosis (Hoff et al., 2013). Nuclear factor-κB (NF-κB) pathway is involved in autophagy and the inflammatory response of chondrocytes, as increased LC3 expression in synovial fluid macrophages in OA can promote the binding ability with other inflammatory factors and deoxyribonucleic acid (DNA) (Ni, Kuang, et al., 2019). In the synovial fluid of LC3 mutant rats, as a fatty acid transporter, the expression of CD36 increases with a decline in the level of fatty acids, high-density lipoprotein cholesterol, and blood glucose (Febbraio & Silverstein, 2007). The findings indicate that it relates to the transcription regulator p35 and the metabolism of lipids. LC3 might regulate autophagy signal transduction, stimulating the release of intracellular inflammatory factors and the expression of inflammatory pathway-related factors. Therefore, LC3 may affect immune function by regulating abnormal lipid metabolism of macrophages, thereby inhibiting vascular proliferation in OA (Heckmann et al., 2017). However, it is unclear whether LC3 can downregulate the activity of NF-κB and other inflammatory cytokines to achieve therapeutic effects.
Recent studies have highlighted the role of innate immune system activation and induction of proinflammatory cascades through toll-like receptor (TLR) activation. Ligand-induced TLR signaling activates NF-κB, which mediates inflammatory pathways, thereby leading to OA and dysfunction (Baker et al., 2011; Oeckinghaus et al., 2011; Turner et al., 2014). NF-κB is crucial to the inflammatory phase of OA. Through NF-κB, interleukin (IL)-18, tumor necrosis factor (TNF)-α, MYD88, and intercellular adhesion molecule-1 can be transcriptionally processed, and they are all involved in the progression of OA. When the inhibitor of nuclear factor-κB-α (IkBα) is degraded by the immunoproteasome, IkBα activates nuclear factor-κB p50/p65 heterodimer (Bharti & Aggarwal, 2002; Oeckinghaus & Ghosh, 2009). Treatment with toll-like receptor 4 (TLR4) inhibitor resatovir or transfection with TLR4 siRNA can block the downregulation of CYP3A4 and P-gp by outer membrane vesicles (OMVs). Resatorvid (TAK-242, CLI-095, TAK242, CLI095) is a selective inhibitor of TLR4 signaling with anti-inflammatory activity (Matsunaga et al., 2011). Glycyrrhizic acid, an important component of licorice, is an ingredient of traditional Chinese medicine characterized by various functions such as anti-inflammatory, anti-infective, and antioxidant activities. It is noted that resatorvir might directly bind to the Cys747 residue in the intracellular region of TLR4, disrupting the interaction between TLR4 and the adapter molecules TIRAP and TRAM, thereby inhibiting TLR4 signaling. S-phase kinase-related protein 2 (SKP2) is a protein-coding gene affecting the cell cycle process (Indovina et al., 2015; Whitfield et al., 2002). SKP2 promotes tumorigenesis in breast cancer and radiation tolerance by degrading PDCD4 (de Seny et al., 2013). Previous studies have also shown that the osteogenic transcription factor Runx2 affects osteoblast differentiation, and SKP2 hinders osteogenesis through degradation of Runx2 (Krock et al., 2018). Therefore, we hypothesize that resatovir affects chondrocyte autophagy in knee OA by regulating Skp2. The glycyrrhizic acid extraction diagram is shown in Figure 1.
Glycyrrhizic Acid Extraction and Research Idea Diagram.
Materials and Methods
General Materials
A total of 50 male rats (weight: 180–220 g) were raised, of which 40 were taken for the animal model, and 10 were taken as a control group (Group A).
Thirty model rats were successfully established and assigned to the model group (Group B) and the p35 intervention group (Group C) (n = 15). For the rats in group A, their joint capsule was only torn and then sutured horizontally. For the model group, surgical destabilization of the medial meniscus was performed on the knee joint under sterile conditions. After the rat was fixed, the lateral skin of the rat’s knee joint was removed with eye scissors, followed by patellar valgus fixation. The joint capsule was ruptured, and the patellofemoral ligament was not damaged. The diseased joint fatty tissues were removed, and the knee joint was exposed. The medial meniscal collateral ligament was separated after patella valgus fixation. The rats were fed normally, and samples were collected 12 weeks after the model was successfully established. Pathological examination was carried out, and synovial fluid was extracted. In Group C, after the establishment of model rats, the animals received an intramuscular injection of resatovir combined with glycyrrhizic acid (60 mg/kg) daily for 4 weeks. Glycyrrhizic acid is a natural plant component with great anti-inflammatory and anti-infective effects, with its molecular formula shown in Figure 2. The total modeling time is 12 weeks. After terminating the modeling and treatment, the rats were anesthetized and sacrificed. The synovial fluid and joint tissue were collected under sterile conditions.
Glycyrrhizin Plant Map.

Evaluation Methods
(a) Normal cartilage: Joints are smooth without specific and atypical hyperplasia. (b) Type I regression: The joint surface is slightly unfair with a slightly wavy texture. (c) Type II regression: Rough joint surface. (d) Type III regression: Bony damage to the articular surface with an obvious degradation process. (e) Type IV regression: Severe joint shipments with structural damage. Negative for cartilage radiata. The frozen cartilage synovial fluid was taken for the detection of the content of TNF-α and IL-6. The fluid was centrifuged for 20 min to collect supernatant, which was assimilated into the synovial fluid. The expression of cytokines was detected by ELISA, and the expression of NF-κB p65 in cartilage synovial fluid by immunohistochemistry. With a tube sealed tightly with surrounding fluid, sections were incubated for 15 min and with NF-κB p65 primary antibody for 1 h. The sections were counter-stained and then observed, and the number of positive cells was calculated under a microscope.
Reagents and Materials
LC3 (ab1872), human GSDMD-N (ab210070), and mouse GSDMD-N (ab219800) were all purchased from Abcam. FAM-FLICA Caspase 1 detection kit came from the American ICT Co., Ltd. Other reagents were obtained from Shanghai Uniview Co., Ltd.
Western Blot
Cellular proteins were collected through the kit, followed by protein quantification. Each lane contained 20 µg. Electrophoresis was carried out by direct current for 90 min and then switched to alternating current for 30 min. The protein was transferred to the membrane and incubated with primary and secondary antibodies. After washing, the blot was developed with a luminescent solution, and GAPDH was quantified as a standard to correct for total protein loading.
Flow Cytometry
After treatment, cells were stained with fluorescein isothiocyanate (FITC) and propidium iodide to measure apoptosis by flow cytometry.
Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR)
With GAPDH as endogenous control, cDNA was utilized for real-time PCR with primers shown in Table 1, and the expression was calculated with the 2−∇∇Ct method.
Primer Sequences.

Statistical Method
SPSS23.0 software processed data assessed by one-way ANOVA and expressed as mean ± standard deviation. p < 0.05 refers to a difference.
Results
Intraperitoneal Injection of Resatovir Combined with Glycyrrhizic Acid Reduces Exercise Injuries in OA Animals
To establish the OA model, resatovir plus glycyrrhizic acid was injected into a rat’s left knee joint with knee flexion, and a CatWalk test was conducted to evaluate nociception caused by exercise. As expected, over 3 days, we noticed increased knee flexion scores and decreased percentage of total ipsilateral paw print intensity (%TIPPI) during the CatWalk test on day 3 (before resatovir injection). This indicated that OA induction caused exercise-induced injury (Figure 3A).
Intraperitoneal Injection of Resatovir Combined with Glycyrrhizic Acid Attenuates Exercise Injuries in Osteoarthritis (OA) Animals. (A) Knee-Bend and (B) CatWalk Tests to Assess Injuries Related to Joint Movement and Loading.
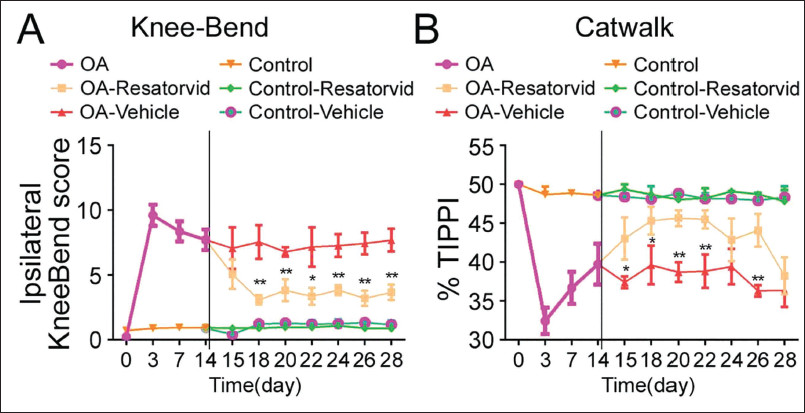
On the 14th day, animals were injected with resatovir plus glycyrrhizic acid and saline, respectively, for 14 days. After receiving intraperitoneal injections of resatovir combined with glycyrrhizic acid, the knee flexion scores had decreased on the 15th day, and exercise-induced nociception also began to decrease since the 18th day (Figure 3A, p < 0.05 or p < 0.01). Regarding the CatWalk trial, resatovir plus glycyrrhizic acid administration induced elevation of %TIPPI until day 22 (Figure 3B, p < 0.05). However, TIPPI values began to decrease during the 2nd week. The rats receiving daily drug injections did not show any differences in knee flexion scores and TIPPI, maintaining the exercise-induced nociception before intervention (Figure 3). Control animals injected with saline in both tests had similar behavioral responses as before treatment.
The Mechanism of Resatovir Combined with Glycyrrhizic Acid
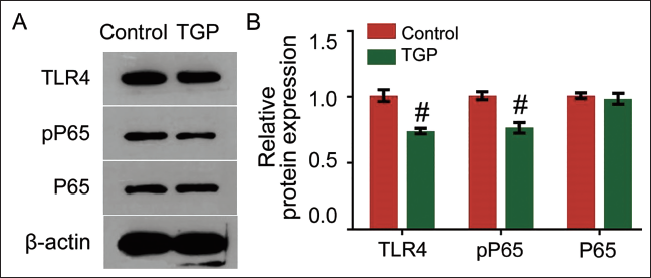
The expression of TLR4, P65, and pP65 was found to be consistent in the kidney tissue of rats, with the most serious impact at 12 weeks. However, pP65 was lowly expressed in the experimental group. Its expression was inhibited after treatment with resatovir plus glycyrrhizic acid. This meant that resatovir treatment suppressed the P65 signaling pathway and then slowed down the pathogenesis of OA, as shown in Figures 4A and B.
Resatovir inhibits the Toll-like Receptor 4 (TLR4)/Nuclear Factor-κB (NF-κB) Signaling Pathway and Thereby Slows Down the Occurrence of Osteoarthritis (OA). (A) Western Blot Results. (B) Quantification of Protein Expression. # p < 0.05.
Resatovir Combined with Glycyrrhizic Acid Regulates the Expression of SKP2
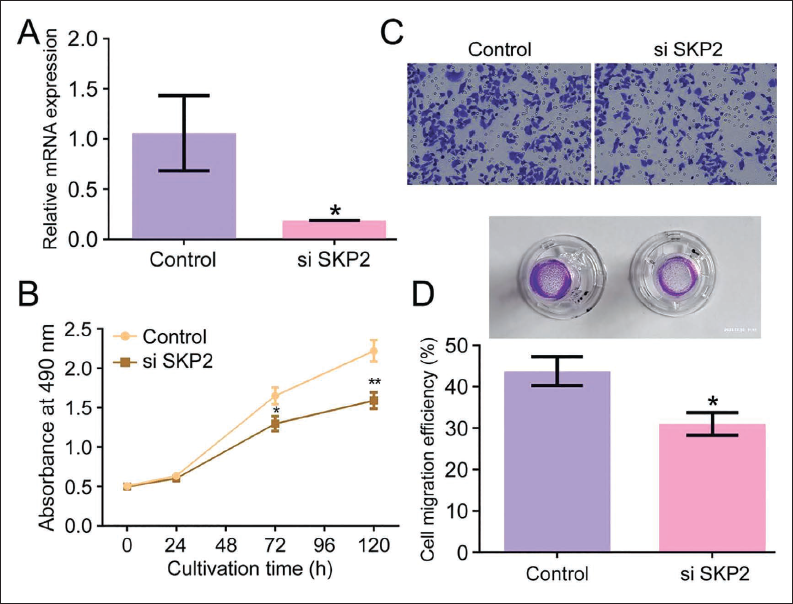
The results showed that SKP2 inhibition successfully suppressed osteoclast differentiation after transfection. SKP2 was transfected into RAW264.7 cells, and its expression was detected (Figure 5A). Under the interference of the SKP2 inhibitor, osteoclast cell proliferation was significantly blocked (Figure 5B). In addition, the migration ability of preosteoclast cells also decreased when the expression of SKP2 was reduced (Figure 5C).
Resatovir Combined with Glycyrrhizic Acid Regulates the Expression of SKP2. (A) Representative Images of Cell and Reverse Transcription Quantitative Polymerase Chain Reaction (RT-qPCR) Result. (B) Analysis of Cell Apoptosis. (C) MTT Test Result. (D) Migration Ability. # p < 0.05.
Inhibiting TLR4/NF-κB Signaling Increases Expression of LC3 in OA Cells
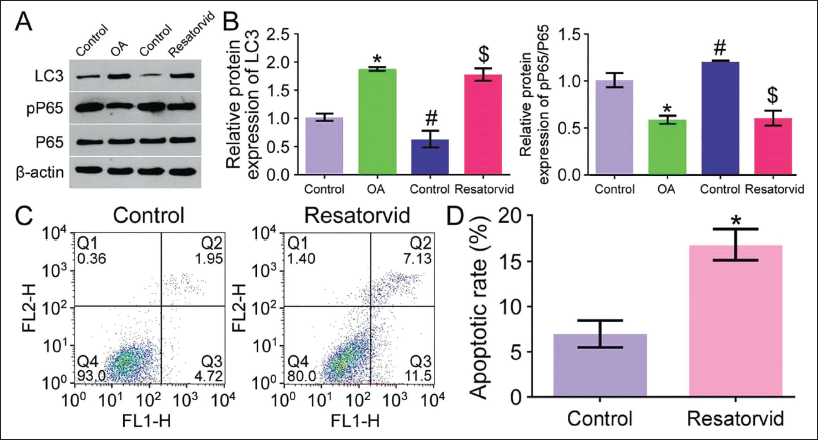
Since upon resatovir plus glycyrrhizic acid, we observed similar changes in TLR4 and NF-κB in OA cells, we next explored apoptosis-related proteins and found that levels of pP65 and LC3 were elevated in OA cells upon resatovir treatment. As such, inhibition of TLR4 or NF-κB caused decreased pP65 and LC3 expression in OA cells stimulated by resatovir combined with glycyrrhizic acid (Figures 6A and B). Flow cytometry results activated autophagy in OA cells upon treatment with TLR4/NF-κB signal inhibition (Figures 6C and D).
Inhibiting the Toll-like Receptor 4 (TLR4)/Nuclear Factor-κB (NF-κB) Signaling Pathway Increases the Expression of LC3 in Osteoarthritis (OA) Cells. (A) Western Blot detects the Protein Expression of pP65, P65, and LC3. (B) Quantification of LC3 and pP65 Expression by Western Blot Analysis. (C) Flow Cytometry detects Apoptosis Expression. (D) Quantification of Cell Apoptosis. *, #, $ p < 0.05.
Discussion
In this study, we found that a combination of resatovir and glycyrrhizic acid could slow down the pathological process of OA by inhibiting the TLR4/NF-κB signaling pathway and activating autophagy. We have proved the efficacy of the treatment is closely related to the repair of bone damage and is a key factor in the osteoautophagy process. When these patients develop OA, inflammation usually takes place, which affects patients’ normal life activities. The chronic inflammatory disease slowly evolves into OA and then develops into bone tumors (Wang et al., 2015). Drug intervention is central to this process, such as the common small molecule drugs that can interfere with inflammatory intermediates. Previous studies have depicted the clinical and research value of resatovir. Recent studies have reported that apart from alleviating inflammation, resatovir treatment also can play a role in improving the condition of OA. This study mainly adopted resatovir to treat genetic model rats and found that the effect of resatovir was significant. This finding might change people’s understanding of TLR4 inhibitors (Xu et al., 2021). We wondered whether small-molecule drugs such as resatovir would impact the occurrence of OA, so we constructed OA model rats and found that resatovir treatment with glycyrrhizic acid significantly improved the inflammatory pathology.
SKP2 is an important inflammatory molecule in the extracellular matrix (Suzuki et al., 2013). Therefore, the occurrence of OA must be inseparable from SKP2. Early inflammation in diabetic OA influences some signaling pathways (Tsalamandris et al., 2019). TLR4/NF-κB contributes to the regulation of cell cycle replication. TLR4/NF-κB inhibits cell proliferation by silencing the expression of CDK. Previous studies have found that in intestinal inflammation models, TLR4 silencing leads to the activation of the NF-κB and NLRP3 signaling pathways, inducing excessive secretion of IL-1β (Wu et al., 2017). Some studies have shown that SKP2 affects TLR4/NF-κB signaling by changing the polarity of cells as well as cell mitosis (Ni, Hao, et al., 2019). Our study demonstrated that fibronectin changed significantly in tissues after OA treatment, so we speculate that resatovir combined with glycyrrhizic acid may affect TLR4/NF-κB signaling, thereby changing the pathological progress. Immunohistochemistry staining indicated that compared with a higher level of SKP2 in the glomeruli of control rats, its expression of SKP2 in the experimental group of rats decreased upon resatovir treatment, and some studies have shown that inflammatory signals are attenuated in TLR4-deficient transgenic mice (Yu et al., 2017). In addition, some evidence suggests that activation of TLRs can trigger the formation of inflammasomes and autophagy, dependent on the expression of LC3 (Shi et al., 2012). These findings noted the necessity of examining the expression of TLR4 in OA patients and analyzing its relevance in osteoblasts. In vitro and in vivo experiments, resatovir treatment-induced suppression of NF-κB signaling induced autophagy activation in OA cells, which was partially reversed by TLR4 and NF-κB inhibitors.
Conclusion
SKP2, an important indicator of inflammation, plays a large role in inflammation, as its content probably determines the degree of inflammation. In the present study, we found that resatovir combined with glycyrrhizic acid was significantly effective during the treatment process. Resatovir treatment greatly decreased the expression of SKP2 and the reduced SKP2 blocked TLR4/NF-κB signaling, thereby reducing the development of OA. We revealed for the first time that resatovir combined with glycyrrhizic acid could affect TLR4/NF-κB signaling pathways in OA.
Footnotes
Abbreviations
LDL: Low-density lipoprotein; NF-κB: Nuclear factor-κB; OA: Osteoarthritis; PDCD4: Programmed cell death protein 4; SKP2: S-phase kinase-associated protein 2; TLR: Toll-like receptor.
Acknowledgments
The authors would like to thank Changshen Yuan and Kan Duan for their valuable contributions to this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethics committee of the Graduate School of the Guangxi University of Chinese Medicine.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Research Project of Guangxi University of Traditional Chinese Medicine in 2022 (No. 2022MS039).
