Abstract
Background
Glechoma longituba (Nakai) Kuprian (GL) is primarily used to treat urinary tract infections and traumatic injuries in traditional medicine in East Asia. Various components of GL are reported to show beneficial activities on brain diseases, including cerebral infarction and brain trauma. We hypothesized that GL extract may potentially have a beneficial effect against traumatic brain injury (TBI) animal model.
Objectives
This study aims to investigate the effects of GL extract on TBI mouse model.
Materials and Methods
The water extract of GL (GLDW) was obtained by boiling with water. The TBI model was established using ICR mice with controlled cortical impact injury. The experimental groups were divided into sham, control, and three GLDW groups (30–300 mg/kg p.o.). Brain damage, neuronal density, and activated microglial density were evaluated. Motor functional deficits were evaluated using Rotarod test and Balance beam test, and cognitive functional deficits were done using novel object recognition (NOR) test and Y-maze test.
Results
Oral administration of GLDW did not reduce brain damage or neuronal death. However, it improved motor and cognitive functions in a dose-dependent manner in both motor tests and NOR test except Y-maze test.
Conclusion
GLDW can improve the impairment of motor and cognitive functions induced by TBI, but it does not have a protective effect against brain damage and neuronal death. Further studies are necessary in order to investigate the mechanisms GLDW’s functional recovery effect on TBI.
Introduction
Glechoma longituba (Nakai) Kuprian (GL), a perennial herb of the Lamiaceae family, is widely distributed throughout Asia (Jang et al., 2016). The aerial part of GL has been mainly used to treat urinary tract infection, urinary stones, jaundice, and traumatic injuries, including bone fractures, in traditional medicine in East Asia (State Pharmacopoeia Committee, 2015). Glechoma species have been studied for their various pharmacological activities, including anti-inflammatory (An et al., 2006; Kim, Song, et al., 2011), antioxidant-related cardio-protective (Zhu et al., 2013), antithrombotic (Ouyang et al., 2019), cytotoxic (Kim, Lee, et al., 2011), anti-urolithic (Liang et al., 2016), and hypoglycemic (Lee et al., 2008) activities.
GL has been reported to contain various chemical components, including flavonoids, triterpenoids, monoterpenoid glycosides, rosmarinic acid, norlignans, chlorogenic acid, and caffeic acid (Liang et al., 2016; Ouyang et al., 2019). Among its various components, caffeic acid (Kerman et al., 2012; Zhang et al., 2007), dihydro-caffeic acid (Lee et al., 2015), and chlorogenic acid (Lee et al., 2012) have been reported to have pharmacological activities in brain diseases, including focal cerebral ischemia and brain trauma.
The major cause of brain trauma is external physical force (Xiong et al., 2013) that occurs for various reasons, including motor vehicle crashes and sports activities. At the beginning of traumatic brain injury (TBI) with skull fracture, external forces such as shearing, tearing, and stretching cause direct injury to blood vessels, neurons, axons, and glia, and result in bleeding. Secondary damage persists for hours to months after the trauma, leading to additional damage, including blood–brain barrier damage, edematic change, and hypoxia-ischemia (Wurzelmann et al., 2017; Xiong et al., 2013). Additionally, sensori-motor function and cognitive memory impairments occur following brain damage (Luo et al., 2019).
To date, various methods have been established for experimental TBI, including shockwave, controlled cortical impact (CCI), weight drop, and fluid percussion, and it is well documented in a previous report (Xiong et al., 2013). Among these, CCI-induced TBI animal model is one of the most popular models for showing motor and cognitive deficits similar to that of TBI patients. Additionally, CCI models have low mortality rates and high reproducibility, and allow for precise adjustment of impact settings, such as velocity, dwell time, and depth. However, it has the disadvantage of requiring invasive craniotomy (Yu et al., 2009).
Guidelines for managing TBI have been established (Carney et al., 2017); however, their application is inconsistent, and the research supporting certain recommendations is often insufficient (Theadom et al., 2023). There is ongoing development of pharmacological treatments, such as neurostimulants (Galgano et al., 2017), but the evidence is mixed, leading to diverse practices in prescribing these drugs postinjury (Barra et al., 2020; Zorowitz et al., 2005). Although various agents showing neuroprotective effects in TBI have been reported in preclinical studies, most studies focusing on single-target therapy have not succeeded in clinical trials because complex pathophysiological processes are involved in TBI (Wurzelmann et al., 2017). Currently, there are no pharmacotherapies approved by the Food and Drug Administration (FDA) to mitigate the neurological impairments resulting from TBI.
In Asia, traditional medicine uses various therapeutics from single-crude extracts to prescriptions, to treat TBI patients and several researches have shown the effects of these drugs, such as Astragali radix (Liu et al., 2018; Xie et al., 2018), Drynariae Rhizoma (Wang et al., 2016), Dangguisusan (Jung et al., 2018), and Chunghyul-Dan (Choi et al., 2017) on TBI models.
We hypothesized that GL may have beneficial effects in the TBI model for two reasons. First, the traditional usage of GL has been closely related to the pathology of TBI with the effects of controlling hemorrhagic change and traumatic injury and reducing inflammatory reaction. Second, it contains various pharmacological compounds that are reportedly effective against brain damage, such as chlorogenic acid and its derivatives including caffeic acid and dihydro-caffeic acid.
Therefore, we administered GL extract to mouse CCI-TBI model and investigated its effects on brain injury, deficits of sensori-motor and cognitive functions.
Materials and Methods
Sample Preparation
GL was harvested and collected from the medicinal plant garden in Global campus of Kyung Hee University (Suwon, Gyeonggido Province, Korea) from May to June 2019, and its plant origin was cross-identified by Professors Youngmin Bu and Kyungjin Lee of Kyung Hee University. GL was washed, dried under shade for a day (20–24°C), and further dried in a convection oven (60°C). The voucher specimens (GL006) were kept in a herbarium at the College of Korean Medicine, Kyung Hee University. After the drying process, GL (dry weight = 639.8 g) was reflux extracted with boiling distilled water (DW) for 2 h (90–100°C). Water extract of GL (GLDW, yield = 27.25%) was obtained after evaporation (40°C), and freeze drying and stored at refrigerator (4°C).
TBI Induction and Grouping
All the procedures of the current animal experiments were approved by the Institutional Animal Care and Use Committee of Kyung Hee University (KHUASP(SE)-16-135). Seven-week-old male ICR mice weighing 31–33 g (Daehan Biolink, Seoul, Korea) were acclimated for a week in a cage with constant conditions (temperature (22–24°C), humidity (50–60%), and a 12/12-h light/dark cycle) with food and water ad libitum.
The TBI animal model was induced by a CCI protocol based on our previous method (Joo et al., 2021). Mice were anesthetized with 1–2% isoflurane in nitrous oxide/oxygen (70%/30%) and then fixed tightly in a stereotactic frame using ear bars. A circular craniotomy with a diameter of 4 mm was performed with a hand drill to prepare an impacting area between the lambda and bregma (−2 mm anteroposterior, 2.5 mm mediolateral from bregma). Mechanical impact was induced using an electromagnetic impactor (Stereotaxic Impactor, Leica, Germany) with depth of 2.5 mm, dwell time of 300 ms, and velocity of 2 m/s. The isolated-skull bone was tightly recovered to avoid bleeding immediately after the impact using super glue. The body temperature of mice was maintained (37 ± 0.5°C) during all the induction procedures.
The mice were divided into five groups at random: Sham-operated group (sham), control, and three GLDW-treated groups (30, 100, and 300 mg/kg). The control and GLDW-treated mice were orally administered with water (3.3 mL/kg) and same volume of GLDW twice daily, respectively, for 8 days after TBI induction. The mice of sham group were made by only craniotomy without impact and received DW in the same manner. Body weight of the mice was measured once a day at 9
Motor Function Tests
Rotarod and Balance beam tests were applied to evaluate sensori-motor function at 0, 3, and 8 days after TBI induction in a quiet soundproof booth with dim light, and the mice were acclimated for 30 min in this booth before testing.
The Rotarod test was performed by the method described in a previous study with minor modifications (Tucker et al., 2016). Mice were put vertically on a rotating rod (10 rpm) facing the dark side, and it was gradually accelerated to 70 rpm within 120 s. First latency to fall was measured and running time (%) was calculated as a percentile of the current latency relative to the baseline latency. Among five trials repeated the average of three median values was used as data.
The Balance beam test was performed by the method described in a previous study with minor modifications (Choi et al., 2017). Mice were put vertically on the middle of a round wooden beam (diameter 0.5 cm, length 100 cm) facing the dark side. The six scores were given as follows: mouse could not hold on and fell off the beam in less than 30 s, 0 points; mouse could remain on the beam for 30 s without any movement, 1 point; mouse could turn sideways but could not walk, 2 points; mouse could turn sideways and walk even one step, 3 points; mouse could turn and walk with the affected hind limb slipping more than 50%, 4 points; mouse could turn and walk with the affected hind limb slipping less than 50%, 5 points; and mouse could turn and walk without foot slipping, 6 points.
Cognitive Function Tests
Novel object recognition (NOR) and Y-maze tests were applied to assess cognitive functional deficits at 7 and 8 days, respectively, after TBI induction in a quiet soundproof booth with dim light. The mice were acclimated for 30 min in this booth before testing.
The NOR test was performed by the method described in a previous study with minor modifications (Joo et al., 2019). Mice were habituated for 30 min in a black open field box (28 cm × 37 cm and 24 cm in height) containing two blue-colored familiar objects (BFOs) placed in fixed locations. The mice were returned to the cage for 4 h and then placed in the same box for 5 min acclimation. After 30 min break in their cage, the mice were put in the box to explore a BFO and a yellow-colored novel object (YNO) for 5 min. Cognitive function was calculated as a recognition index (%): [(Time (sec) YNO − Time (sec) BFO)/(Total time (sec) YNO and BFO)] × 100.
The Y-maze test was performed by the method described in a previous study with minor modifications (Baratz et al., 2015). The Y-maze consisting of three arms (9 cm × 30 cm and 14.5 cm in height) with the angle of 120° was made of black acryl. The arms were randomly divided into the start arm (SA), novel arm (NA), and familiar arm (FA). Mice were placed in SA and allowed to freely explore SA and FA for 5 min, with NA closed by a door. After 2 min break, NA was opened, and mice were then placed in SA and allowed to freely explore all the arms for 2 min. Cognitive function was calculated as a preference index (%): (Time (sec) NA − Time (sec) FA)/(Total time (sec) both NA and FA).
Histological and Immunohistochemical Analysis
Histological processes including perfusion-fixation, postfixation, brain isolation, and brain sectioning were performed by the method of a previous study (Joo et al., 2021). Briefly, 9 days after TBI, mice were transcardially perfusion-fixed. Brains were isolated and went through 24 h of postfixation with paraformaldehyde. Brains were frozen-sectioned (40 µm) using Cryocut (HM440; Carl Zeiss, Germany) after 30% sucrose cryoprotection. Five sections of the brain were selected and stained with cresyl violet (Joo et al., 2019). The non-stained area, empty area, and astrogliosis area of cerebral cortex were considered damaged area. The damaged area (mm2) of each brain section was measured by ImageJ software (1.43 u, NIH, USA), and the total damaged area (mm2) was obtained by summing up all the damaged areas of five sections. The data was calculated as relative percentile of the total damaged area to the intact hemisphere of five sections.
After floating, the brain sections were reacted with primary antibody (anti-goat ionized calcium-binding adapter molecule-1 (IBA-1) antibody (1:1,000; Abcam, UK) and anti-rabbit NeuN antibody (1:500, Cell Signaling, USA)) and followed by reaction with anti-goat and anti-rabbit secondary antibody (1:200; Vector Laboratories, USA). They were identified using 3,3-diaminobenzidine (Sigma, USA) after 60 min reaction with an avidin-biotin-peroxidase complex kit (Vector Laboratories). Data were obtained by counting the IBA-1-positive activated microglia and NeuN-positive intact neurons in the thalamic region located between 1.5 mm and 2 mm lateral, 2.5 mm and 3.0 mm ventral from dorsal top of brain section (−1.82 mm bregma) (Paxinos et al., 2001).
Statistical Analysis
Data were expressed as mean ± standard error of mean (SEM). Statistical significances were analyzed by one-way analysis of variance (ANOVA), followed by Dunnett’s test (vs. control group) using GraphPad Prism v.5.0 (GraphPad Software, USA). p values under 0.05 were considered statistically significant.
Result
Effect of GLDW on Body Weight
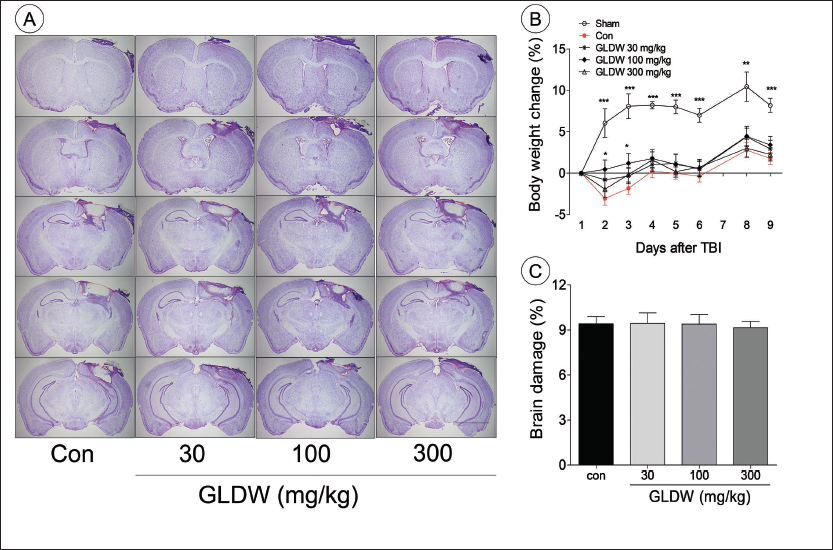
The body weight gradually increased in the sham group. However, the body weight in the other groups decreased after TBI induction and then gradually increased. All the GLDW-treated groups tended to lose less body weight until 9 days post-TBI. In particular, the GLDW 100 mg/kg group showed significantly less weight loss than the control group at 1 day and 2 days post-TBI (Figure 1B).
The Effect of GLDW on Histological Damage and Body Weight Change After Traumatic Brain Injury (TBI). (A) Shows Brain Tissue Stained with Cresyl Violet of Each Group. The Red Dotted Line shows the Damaged Regions of the Brain. (B) Shows Brain Damage (%) of Each Group. (C) Shows Body Weight Change (%). Con and Sham are the Control Group and the Sham Group, Respectively. Scale Bar (A) is 400 µm. The Results were Presented as Means ± Standard Error of Mean (SEM) (n = 8). One-way Analysis of Variance (ANOVA) with Dunnett’s Test was used to Analyze Statistical Significance (*p < 0.05, **p < 0.01, ***p < 0.001 vs. the Control Group).
Effect of GLDW on Brain Injury
The main injured area of cresyl violet stained brain sections was located between the third and fourth sections (bregma −1.82 mm and −2.46 mm). The brain sections from each group showed that the most damaged regions were hippocampus and parietal cortex (Figure 1A).
The control group showed 9.41 ± 0.48% brain damage. GLDW-treated groups showed 9.44 ± 0.70% (30 mg/kg), 9.40 ± 0.62% (100 mg/kg), and 9.15 ± 0.42% (300 mg/kg) of brain damage (Figure 1C).
Effect of GLDW on Motor Function
Rotarod Test
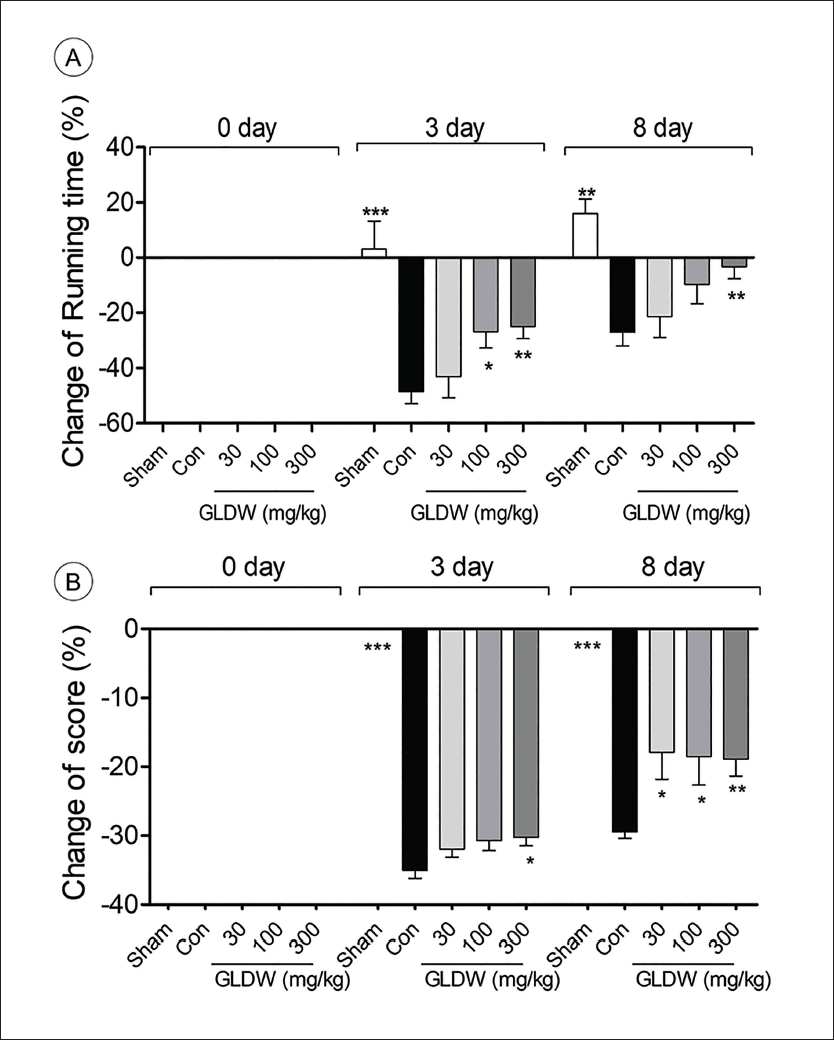
The running time of the sham group gradually increased, while the control group decreased to −48.6 ± 4.3% on day 3 compared to day 0 and recovered to −27.1 ± 4.9% on day 8. GLDW treatment dose-dependently prolonged running time. Running time of GLDW-treated groups decreased by −26.9 ± 5.1% (100 mg/kg, p < 0.05) and −25.0 ± 4.36% (300 mg/kg, p < 0.01) on day 3. The running time of GLDW 300 mg/kg group recovered to −3.27 ± 4.35% (p < 0.01) on day 8 (Figure 2A).
The Effect of GLDW on Motor Functional Deficits Assessed by Rotarod Test (A) and Balance Beam Test (B) Evaluated at 0-, 3-, and 8-Days Post-Traumatic Brain Injury (TBI). Con and Sham are the Control Group and the Sham Group, Respectively. The Results were Presented as Means ± Standard Error of Mean (SEM) (n = 8). One-way Analysis of Variance (ANOVA) with Dunnett’s Test was used to Analyze Statistical Significance (*p < 0.05, **p < 0.01, ***p < 0.001 vs. the Control Group).
Balance Beam Test
The motor functional score of the sham group was highest (6 points) until day 8, while the control group decreased to −35.0 ± 1.2% on day 3 and then slightly recovered to −29.5 ± 0.9% on day 8. The score of the GLDW 300 mg/kg-treated group was −30.3 ± 1.4% (p < 0.05) on day 3. On day 8, the score of all the GLDW-treated groups recovered to −17.9 ± 3.9% (30 mg/kg, p < 0.05), −18.5 ± 4.1% (100 mg/kg, p < 0.05), and −18.9 ± 2.4% (300 mg/kg, p < 0.01) (Figure 2B).
Effect of GLDW on Cognitive Function
NOR Test
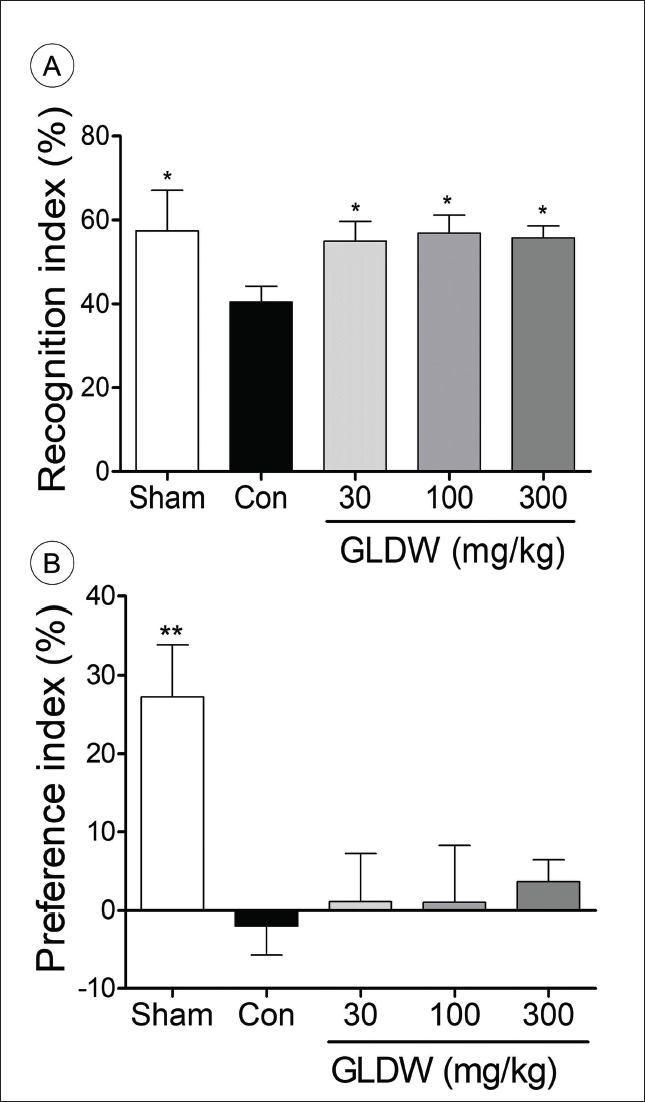
In the NOR test, the recognition index of the sham group was 57.4 ± 9.7%, while that of the control group was 40.4 ± 3.8% (p < 0.05). All the doses of GLDW showed the similar recognition index to each other (55.0 ± 4.7% (30 mg/kg), 56.9 ± 4.2% (100 mg/kg), and 55.7 ± 2.9% (300 mg/kg), p < 0.05). GLDW treatment showed significant improvement in the recognition index (p < 0.05), which was similar to that of the sham group (Figure 3A).
The Effects of GLDW on Cognitive Functional Deficits Assessed by Novel Object Recognition (NOR) Test (A) and Y-Maze Test (B) at 7- and 8-Days Post-Traumatic Brain Injury (TBI), Respectively. Con and Sham are the Control Group and the Sham Group, Respectively. The Results were Presented as Means ± Standard Error of Mean (SEM) (n = 8). One-way Analysis of Variance (ANOVA) with Dunnett’s Test was used to Analyze Statistical Significance (*p < 0.05, **p < 0.01 vs. the Control Group).
Y-Maze Test
The preference index of the sham group was 27.3 ± 7.0%, while the preference index of the control group was −1.9 ± 4.1% (p < 0.01). In the GLDW-treated groups, the preference index tended to increase by 1.1 ± 6.2% (30 mg/kg), 1.0 ± 7.2% (100 mg/kg), and 3.61 ± 2.9% (300 mg/kg). However, all the doses of the GLDW treatment did not show any significant difference (Figure 3B).
Effect of GLDW on Neuron Density in the Thalamic Region
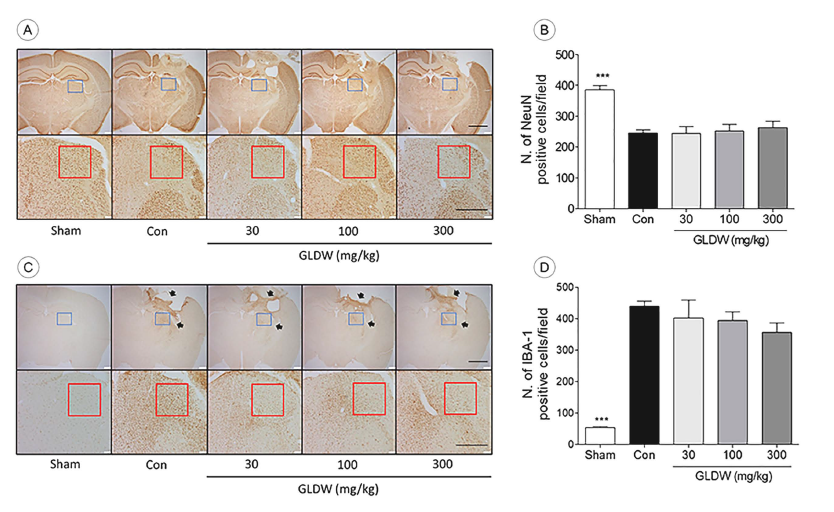
The number of intact neurons (NeuN-positive) of the sham group was 384.9 ± 14.9 ea, while that of the control group was 245.5 ± 10.3 ea (p < 0.01). All the GLDW-treated groups showed a tendency to increase the number of intact neurons without significant difference (Figure 4A and B).
The Effect of GLDW on Neuronal Density (NeuN Positive) and Microglia Activation (IBA-1 Positive) After TBI. Microscopic Photos in (A) and (C) show the Representative Entire Brain Region of Each Group (Upper Photo, ×20) and the Thalamic Region which is the Blue Square of the Upper Picture of A (Lower Photo, ×200). Scale Bars are 2 mm (upper) and 500 µm (lower), Respectively. Graph shows the Number of Intact Neurons (B) and the Number of Activated Microglia (D) in the Red Square of the Lower Photos. Arrows indicate Microglia Upregulated Area. Con and Sham are the Control Group and the Sham Group, Respectively. The Results were presented as Means ± Standard Error of Mean (SEM) (n = 8). One-way Analysis of Variance (ANOVA) with Dunnett’s Test was used to Analyze Statistical Significance (***p < 0.001 vs. the Control Group).
Effect of GLDW on Microglial Activation in the Thalamic Region
The number of activated microglia (IBA-1-positive) of the control group increased, especially near the damaged areas, including the hippocampus, thalamus, and cortex (Figure 4C, arrows). That of the sham group was 53.4 ± 3.6 ea, while that of the control group was 439.3 ± 16.0 ea (p < 0.001). GLDW treatment tended to decrease microglial activation dose-dependently, but it did not show significant difference (Figure 4C and D).
Discussion
GLDW treatment to CCI-TBI mouse model inhibited body weight loss and recovered sensori-motor and cognitive deficits in the absence of significant protective effects against brain histological damage.
GLDW dose-dependently improved motor function in the Rotarod and Balance beam tests. Generally, the CCI-TBI model has decreased motor function for 3–5 days and then recovers after 5 days after TBI induction (Shin & Dixon, 2015; Tsenter et al., 2008). Therefore, in this study, motor function was measured at 3 days when the maximal deficit occurred and at 8 days during recovery. The Rotarod test is useful for measuring motor coordination and cardiopulmonary endurance (Hamm et al., 1994; Shiotsuki et al., 2010), and the Balance beam test evaluates fine motor function of rodents involved in walking and balancing (Curzon et al., 2009). The current results indicate that GLDW may have a positive effect over motor functional deficits after TBI in terms of motor coordination, balance, and endurance, and may also be beneficial for motor dysfunction in other brain disease models.
Additionally, the GLDW-treated groups showed significantly improved cognitive function in the NOR test. Cognitive functional deficit is reported to become evident at 7–8 days after TBI (Baratz et al., 2015). The NOR test is widely used to assess rodent’s ability to recognize and memorize a novel object, using the natural propensity of rodents to novelty (Antunes & Biala, 2012). In the Y-maze test, however, GLDW treatment did not show any significant effects. The Y-maze test is well known to assess spatial recognition memory utilizing the natural preference of rodents on exploring novelty (Ma et al., 2007; Zhuang et al., 2001). Generally, normal memory function was reported to be closely associated with the medial temporal lobe, including the hippocampus and adjacent cortices (Baxter, 2010). The NOR test is used to evaluate visual object recognition memory and is associated with the parahippocampal regions of the temporal lobe (Hammond et al., 2004). However, the Y-maze test is known to be a test for measuring spatial memory related to the hippocampus (Aggleton et al., 1986). Therefore, the current results regarding cognitive function are thought to have resulted from differences in memory function depending on the brain region.
The clinical dose of GL dried material is known to be 9–15 g/day (Encyclopedia of Oriental Medicine, 1999). In this study, the clinical dose of GLDW is 40–68 mg/kg per day, calculated considering the yield of the extract. This corresponds to 492–836 mg/kg for mice, taking into account body surface area and metabolic rate (Reagan-Shaw et al., 2008). Accordingly, the experimental dose was set at 300 mg/kg twice a day (600 mg/kg/day). Additional doses were determined to be 100 mg/kg and 30 mg/kg at 3-fold intervals.
A significant weight loss is one of the major factors in various adverse outcomes, including motor functional deficits and neurological impairments in TBI animal model (Kahriman et al., 2022). GLDW protected the body weight loss until 3 days after TBI in the current study, which was parallel with the results of motor function tests. The motor functions of TBI mice in all the groups at day 3 were maintained or improved until day 8, which showed a similar tendency to body weight change. Thus, the effect of GLDW on body weight might positively affect functional recovery after TBI.
The thalamic region below the hippocampus is known to play a key role in information processing and transmission between various regions, including sensory, motor, and association regions, and damage to this region has been reported to affect cognitive impairment (Grossman et al., 2012). When inducing the TBI model used in this study, an impact depth of 2.5 mm caused damage beyond the hippocampus to the thalamic region. Therefore, nerve cells in this region are also damaged. Damage to the thalamic region is caused by apoptosis, in which cells gradually die and is characterized by a decrease in neuronal cells (Muessel et al., 2002). In addition, activated microglia is reportedly upregulated in case of the neuronal cell damage and involved in neuronal death (Hermann et al., 2000). The current results show the tendency of GLDW to protect against neuronal cell damage and suppress microglial activation. Although not statistically significant, it may have a partially affected cognitive function.
Although GLDW’s efficacy was not directly compared with positive control, an indirect comparison with minocycline data under the same conditions and the model revealed that GLDW did not protect against brain damage as effectively as minocycline (45 mg/kg, IP). It demonstrated approximately 70–80% effectiveness in the balance beam test on day 8, which was similar to the performance in the Rotarod test. However, GLDW’s maximal effect on cognitive function was superior to that of minocycline (45 mg/kg, IP) in the NOR test (Joo et al., 2019).
Our previous report has also shown that compound icariin showed functional recovery despite the absence of protective effects against brain damage in a CCI-TBI mice model. The mechanisms were found to be the neuroplasticity-related mechanisms, including the upregulation of the neuroplasticity and synaptic plasticity (Joo et al., 2019). Considering the above report, it could be assumed that neuroplasticity-related factors may be the main mechanism for the effect of GLDW.
Conclusion
Taken together, improved the recovery of impaired motor and cognitive functions in TBI model while it did not alleviate brain damage nor neuronal death. Further studies may investigate the mechanisms underlying the functional recovery including neuroplasticity-related mechanisms.
Abbreviations
ANOVA: Analysis of variance; CCI: Controlled cortical impact; DW: Distilled water; GL: Glechoma longituba (Nakai) Kuprian; GLDW: Water extract of GL; IBA-1: Ionized calcium-binding adapter molecule-1; NOR: Novel object recognition; TBI: Traumatic brain injury.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Animal experimental protocols were approved by the Institutional Animal Care and Use Committee of Kyung Hee University (KHUASP(SE)-16-135).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Research Foundation of Korea (NRF) grant funded by the Korean government (Ministry of Science and ICT) (No. 2020R1A2C1008603).
