Abstract
Background
The immune system plays a crucial role in protecting the host by regulating biological activities in multiple organs and circulatory fluids. The phytoextracts are evident to modulate a series of biological activities and play a protective role in managing healthy conditions through interaction with the immune system and exhibit antioxidant, antitumor, anti-inflammatory, antimicrobial, and immunomodulatory activities.
Objectives
Understanding the function and regulation of immunomodulation may provide potential strategies for the development of natural extracts for the management of adverse conditions.
Materials and Methods
In the present study, we have investigated the immunomodulatory activity of proprietary polyherbal formulation (Phytocee™) containing Emblica officinalis, Ocimum sanctum, and Withania somnifera using in vitro macrophage phagocytosis and ex vivo natural killer (NK) cell activity. The effect of water and methanolic extracts of Phytocee on NK cell activity was performed using mice splenocytes as effector cells and YAC-1 fibroblast as target cells. The effect of Phytocee on phagocytosis in murine macrophage (J774A.1) cells against fluorescein-labeled bioparticles was also investigated.
Results
The significant increase in NK cell activity of 35% at 200 µg/mL of water extract and dose-dependent increase with methanol extract confirmed the NK cell-mediated immunomodulation of Phytocee. A phagocytic accumulation of bioparticles within macrophages at 100 and 200 µg/mL of extract group suggests a macrophage activating role and immuno-effectiveness of Phytocee herbal formulation.
Conclusion
Based on the current data and the historical use of ingredients in the polyherbal formulation support the immunoprotective role of Phytocee.
Introduction
Plants and their components, as natural sources, have provided relief to several trauma conditions since their existence. Engaging with the conventional practices of using plants in medical practice with the advancement of modern science and phytochemistry knowledge enhanced the application and significant use of plants in diverse fields (Yuan et al., 2016). In any organism, the role of dietary constituents primarily aims to provide nutritive and medicinal benefits. The plant-based diets are often made with a combination of multiple plant materials in specific ratios to achieve enhanced bioactivity for healthy outcomes. It is a well-established fact that phytoextracts are known to participate in and modulate a plethora of biological activities and play a protective role in managing a healthy condition (Hossain, 2018). Different agents of plant origins and their polyherbal formulation are reported to interact with the immune system in a complex way and exhibit a wide range of biological effects including antioxidant, antitumor, anti-inflammatory, antimicrobial, hepatoprotective, and immunomodulatory activities (Nemmani et al., 2002; Singh et al., 2013).
The immune system has a pivotal role in protecting the host by activating various immune cells and producing a variety of cytokines and inflammatory mediators. These mediators are released by specialized white blood cells when an infection or inflammatory condition occurs to help fight the infection (Alamgir & Uddin, 2010; Amirghofran, 2012). The phagocytic macrophages and lymphocytes such as natural killer (NK) cells, B cells, and T cells function under the roads of immunological barriers in both normal and pathological conditions. NK cells are effector lymphocytes and are important defense cells in the initial response against viral and microbial infection and in controlling tumor formation (Liu et al., 2006). To kill an infecting agent, NK cells release perforin and granzymes, where perforin opens the target cell and helps embed granzymes to destroy the toxic cell. This process activates the NK cells and releases cytokines that also signal other white blood cells to assist in eliminating the infecting cell to safeguard the host (Haller et al., 2000; Voskoboinik et al., 2006). On the other side, T-helper type 1 cells and activated T cells release interferon-gamma (IFN-γ), which activates the macrophages with a phagocytic nature innate immune cells involved in scavenging/destroying pathogenic organisms and foreign bodies and clearing cellular debris by the production of reactive oxygen and nitrogen metabolites (Hirayama et al., 2017; Jantan et al., 2015). These macrophages also participate in the wound healing process and maintain cellular homeostasis along with controlling infections (Watanabe et al., 2019). These immune cells are activated regularly and function normally as immunological barriers in body cells. However, when the immune system is disrupted, it requires activation by pharmaceutical drugs or natural agents to boost the immune system by immunomodulation. Natural agents are the preferred choice over other options/drugs due to their nonsynthetic production from natural sources and can be consumed as dietary components with minimum or no side effects (Di Sotto et al., 2020; Jantan et al., 2015).
In the present study, Phytocee™, a polyherbal formulation developed by Natural Remedies Private Limited, Bengaluru, which contains the extracts of Emblica officinalis, Ocimum sanctum, and Withania somnifera, is used to explore the immunomodulatory properties. These polyherbal formulations are reported to provide a wide range of pharmacological and clinical benefits for the preservation and restoration of health, including cell-mediated immune response and antioxidant properties (Chandrasekaran et al., 2010; Hirayama et al., 2017; Jantan et al., 2015; Jeba et al., 2011; Sadekar et al., 1998; Vaghasiya et al., 2010). Hence, the current study aims to demonstrate the immunoprotection enhancing activity of polyherbal formulated Phytocee extract through the in vitro phagocytic and ex vivo NK cell activity.
Materials and Methods
Materials
The test substance, Phytocee™, was developed by M/s Natural Remedies Private Limited, Bengaluru, India. Phytocee is a polyherbal combination of E. officinalis (70% w/w), O. sanctum (20% w/w), and W. somnifera (10% w/w).
The J774A.1 murine macrophage cell line (ATCC, # TIB-67™), YAC-1 Mus musculus lymphoma cell line (ATCC, # TIB-160™), carboxymethyl cellulose (CMC) (HiMedia Labs, India), 1× phosphate-buffered saline (PBS) (Sigma), RPMI-1640, DMEM medium (Gibco®, Thermo Fisher Scientific, Inc., USA), 10% fetal bovine serum (FBS), penicillin (100 U/mL), streptomycin (100 µg/mL), NH4Cl, KHCO3, Na2EDTA (HiMedia) and NaHCO3, C3H3NaO3, levamisole, Trypan blue (Sigma), and Vybrant Phagocytosis Assay Kit (Molecular Probes # V-6694) were procured and stored at specified temperature.
Preparation of Test Substance
The 100 mg of test substance (Phytocee Water Extract and Methanolic Extract) was weighed and suspended in 1 mL of dimethyl sulfoxide (DMSO), followed by sonicating for 10 minutes before the start of the experiment procedures. It was filtered using a 0.22 µm syringe filter. Further dilution to final concentrations was prepared using RPMI 1640 medium.
Isolation of Splenocytes
The BALB/c mice were obtained from the Central Animal Facility, Natural Remedies Private Limited. The spleens were aseptically removed and placed in RPMI 1640 medium. Single-cell suspensions were prepared and passed through a cell strainer (BD Biosciences, Durham, NC, USA). Lymphocytes were obtained by density gradient centrifugation using Ficoll–Histopaque and used as NK cells (crude population). All the animal experimental procedures were executed as per the Committee for Control and Supervision of Experiments on Animals (CCSEA) guidelines.
Cell Viability Determination in Splenocytes
The cell viability was performed in 96-well plates using calcein-AM method (Neri et al., 2001). In brief, calcein-AM-labeled mouse splenocytes were adjusted at 5 × 106 cells/well in RPMI 1640 supplemented with 10% HI FBS, 50 µM β-mercaptoethanol, and 1% antibiotics, and 200 µL of the cell suspension was plated into a 96-well culture plate with various concentrations of test items. Further, the cells were incubated for 28 hours at 37°C with 5% CO2. After 28 hours, the fluorescence was assessed in a fluorescence reader (FLUOstar, BMG-Labtech, Germany).
Cell Viability Determination in YAC-1 Cells
The cell viability was performed in 96-well plates using the calcein-AM method (Neri et al., 2001). In brief, calcein-AM-labelled YAC-1 cells were adjusted to be 1 × 105 cells/well in RPMI 1640 supplemented with 10% HI FBS and 1% antibiotics, and 200 µL of the cell suspension was plated into a 96-well culture plate with various concentrations of test items. These cells were incubated for 4 hours at 37°C with 5% CO2. After 4 hours, the fluorescence was assessed in a fluorescence reader (FLUOstar, BMG-Labtech, Germany).
Cytotoxicity was determined by a decrease in the fluorescence readings compared to the control.
NK Cell Activity
The NK cell activity was performed using isolated spleen cells (effector cells) and YAC-1 M. musculus lymphoma cells (target cells) following cell viability, which was determined by the calcein-AM method. In brief, 200 µL of the splenocyte suspension containing 5 × 106 cells/well in RPMI 1640 containing 10% FBS and 1% penicillin-streptomycin were plated onto the flat bottom 96-well plate with various concentrations of test items. These cells were incubated for 24 hours at 37℃ with 5% CO2 and prepared as effector cells. Further, the 100 µL of calcein-AM-labelled YAC-1 cell (target cells) suspension containing 1 × 105 cells/well was added to effector cells and incubated for 4 hours at 37℃ in a CO2 incubator. NK cells alone (E) and YAC-1 cells alone (T) were also incubated as controls. After 4 hours of incubation, the NK cells mediated lysis of YAC-1 cells was assessed in a fluorescence reader (FLUOstar, BMG-Labtech, Germany) and the percent of NK cell activity was calculated (% NK cell activity = [1 − (RFU(effector + target) – RFU(effector))/RFU(target)] × 100) (Hwang et al., 2012; Madaan et al., 2015; Moore et al., 2014).
Phagocytosis Assay
The assay was performed as per the Vybrant Phagocytosis Assay Kit method (Molecular Probes # V-6694). Briefly, murine macrophage J774A.1 cells were plated in 96-well plates on day 0 (0 hours) at 1 × 105 cells/well in DMEM medium containing 10% FBS. The plates were incubated for 1 hour at 37°C in a CO2 incubator (5% CO2). Samples, at various concentrations, were added to the cells, and the cells were incubated for 1 hour at 37°C (5% CO2). After 1 hour, 25 µL of fluorescein-labeled Escherichia coli (K-12 strain) bioparticles were added to the macrophages, and cultures were re-incubated for 2 hours at 37°C in a CO2 incubator (5% CO2). At the end of 2 hours, the supernatant was discarded. Trypan blue was added to the cells, and there was a further incubation of 1 minute. Trypan blue was removed, and the fluorescence was measured in a fluorescent plate reader (FLUOstar, BMG-Labtech, Germany) at an excitation of 485 nm and an emission of 520 nm. The efficacy of the compound as an immunostimulant was determined by comparison of the percentage phagocytosis effect with the drug untreated macrophage set (Foukas et al., 1998).
Statistical Analysis
The experimental data are reported as mean ± standard deviation (SD), and statistical analysis was performed by using a one-way analysis of variance (Dunnett’s test) using the GraphPad Prism statistical software. Values were considered statistically significant if *p < 0.05 compared to control.
Results
Immunomodulatory Potential of Water and Methanolic Extracts of Phytocee on NK Cells
The effect of Phytocee water and methanolic extract on NK cell activity compared to control cells for reflection of the immunomodulatory potential of test items was examined. Prior to analyzing NK cell activity, the murine splenocytes and YAC-1 cells’ individual cell viability were determined to set the treatment concentration of both extracts. In the dose range-finding study, the murine splenocytes and YAC-1 cells cultured separately in 96-well plates were incubated with test items at the indicated concentrations (Phytocee water extract (3.125, 6.25, 12.5, 25, 50, 100, and 200 µg/mL), Phytocee methanol extract (3.125, 6.25, 12.5, 25, 50, 100, and 200 µg/mL)) in triplicates. The murine splenocytes were treated for 28 hours, and YAC-1 cells were incubated for 4 hours. Cell viability was evaluated using the calcein-AM method.
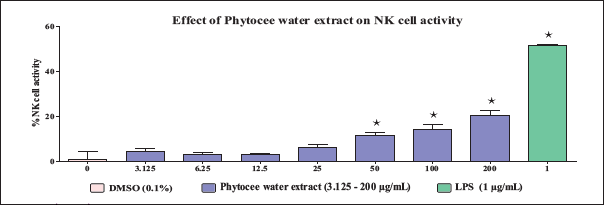
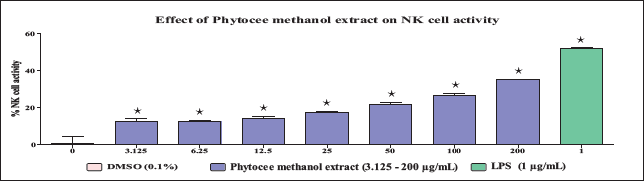
The results of the dose range-finding study revealed that Phytocee water extract and Phytocee methanol extract did not exhibit significant differences in murine splenocytes as well as in YAC-1 cell viability at the tested concentrations ranging from 3.125 to 200 µg/mL. Hence, based on the results of the dose range-finding study, the above concentrations were selected for NK cell activity evaluation. The results revealed that Phytocee methanol extract exhibited a significant increase in NK cell activity at the tested concentrations ranging from 3.125 to 200 µg/mL with a maximum activity of 35% at 200 µg/mL (Figure 1), and Phytocee water extract exhibited a significant increase in NK cell activity at the tested concentrations ranging from 50 to 200 µg/mL in comparison to control (Figure 2). The significant increase in lipopolysaccharide (LPS)-treated reference control confirmed the NK cell activity-mediated immunomodulation.
Effectof Different Concentrations of Phytocee Water Extract on Natural Killer (NK) Cell Activity. The Spleen Cells and YAC-1 Cells were Incubated Together and Treated with or without Different Concentrations of Phytocee Water Extract (3.125, 6.25, 12.5, 25, 50, 100, and 200 µg/mL) and Lipopolysaccharide (LPS) (1 µg/mL) in a 96-well Plate and the Cell Viability was Measured by Calcein-AM Method. Results were Expressed as % NK Cell Activity as Compared to Control (mean ± SD). Values are Significantly Different from Control at *p < 0.05 Using One-way Analysis of Variance (ANOVA) Followed by Dunnett’s Test.
Effect of Different Concentrations of Phytocee Methanol Extract on Natural Killer (NK) Cell Activity. The Spleen Cells and YAC-1 Cells were Incubated Together and Treated with or without Different Concentrations of Phytocee Methanol Extract (3.125, 6.25, 12.5, 25, 50, 100, and 200 µg/mL) and Lipopolysaccharide (LPS) (1 µg/mL) in a 96-well Plate and the Cell Viability was Measured by Calcein-AM Method. Results were Expressed as % NK Cell Activity as Compared to Control (mean ± SD). Values are Significantly Different from Control at * p < 0.05 using One-way Analysis of Variance (ANOVA) Followed by Dunnett’s Test.
Phytocee Induces Phagocytosis of Fluorescein-labeled E. coli in J774A.1 Murine Macrophages
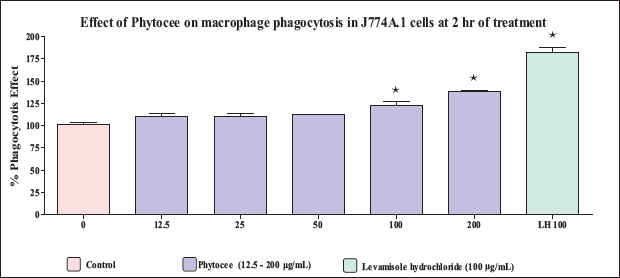
The murine macrophage J774A.1 cell line is incubated with Phytocee extract (12.5, 25, 50, 100, and 200 µg/mL) and internalization of fluorescein-labeled E. coli (K-12 strain) bioparticles is measured after 2 hours of coculture to determine whether a substance can increase the phagocytic capacity of macrophages. Phytocee exhibited a significant increase in macrophage phagocytosis at the highest tested concentration of 100 and 200 µg/mL (Figure 3). Levamisole, the positive control for the assay, also demonstrated a significant increase in macrophage phagocytosis at the tested concentration of 100 µg/mL. The above observations suggest a probable mild macrophage activating role for Phytocee.
Effect of Phytocee on Phagocytosis of Fluorescein-labeled E. coli by J774A.1 Murine Macrophages. The Murine Macrophage J774A.1 Cells were Treated with or without Different Concentrations of Phytocee Extract (12.5, 25, 50, 100, and 200 µg/mL) and Levamizole (100 µg/mL) in a 96-well Plate and the % Phagocytosis was Measured Using Vybrant Phagocytosis Assay Kit Method. Results were Expressed as % Phagocytosis Effect as Compared to Control (mean ± SD). Values are Significantly Different from Control at * p < 0.05 Using One-way Analysis of Variance (ANOVA) Followed by Dunnett’s Test.
Discussion
The participation and specific stimulation of organs and cells which are involved in the functional behavior of the organism determine the outcome of a healthy condition. In livestock management, the growth parameter of animals and birds depends on the activation or suppression of many biological activities to maintain normal conditions and to overcome pathological adversity. The notable biological properties such as antioxidant, anti-inflammatory, antimicrobial, antitumor, hepatoprotective, and immunomodulatory activities are significantly involved in the management of overall health (Nemmani et al., 2002; Singh et al., 2013). Unveiling the above properties by suitable agents through symptom-based therapy can prevent infection and help in the restoration of a healthy system. In several instances, the immune system can be a target of several pharmaceutical drugs and herbal constituents through the process of immunostimulation or immunosuppression events for exploring immunomodulator activity (Amirghofran, 2012; Di Sotto et al., 2020).
W. somnifera, commonly known as ashwagandha, is extensively studied and is reported to have anti-inflammatory, antioxidant, and antitumor activities (Alam et al., 2012; Shi et al., 2021). The extract of E. officinalis (amla), with a history of medicinal value, has long been used in traditional systems of plant medicine against several harmful conditions and is known to exhibit a wide range of biological effects including antioxidant, antitumor, anti-inflammatory, antibacterial, and hepatoprotective properties (Singh et al., 2013). O. sanctum, commonly known as Tulsi or holy basil, has been proven for health management effects through its antioxidant, anti-inflammatory, antimicrobial, antidiabetic, hepatoprotective, and wound healing effects (Jeba et al., 2011; Vaghasiya et al., 2010). Based on the traditional practice and convincing background of previous reports, the polyherbal combination of E. officinalis, O. sanctum, and W. somnifera extract is formulated (Phytocee™) for immune health management of livestock. Since the immune system is susceptible to the unfavorable environment and invading pathogens, additional protection through the external source is advised for normal productive growth in animal and bird farming (Chandrasekaran et al., 2010; Sadekar et al., 1998).
In our study, the effect of Phytocee water and methanolic extracts on immunomodulation is investigated through NK cell activity and phagocytosis assay. NK cell activity was performed using mice splenocytes and YAC-1 fibroblast cells, whereas the phagocytic assay was evaluated by murine macrophage J774A.1 cells engulfing activity against fluorescein-labeled E. coli (K-12 strain) bioparticles exposure. The results of NK cell activity are concomitant with the earlier reports evaluated using these plant extracts for immunomodulatory function (Nemmani et al., 2002). The Phytocee showed a significant increase in NK cell activity with a maximum activity of 35% at 200 µg/mL of water extract, and a dose-dependent increase with methanol extract in comparison to control and LPS-treated reference control confirmed the NK cell activity mediated immunomodulation by Phytocee. The extract of Phytocee also displayed enhanced phagocytic properties on par with data reported using similar herbal formulations (Davis & Kuttan, 2000; Hasan et al., 2016; Maheshwari et al., 2022). A significant increase in the accumulation of fluorescence-labeled bioparticles within J774A.1 cell was observed at 100 and 200 µg/mL concentrations compared to the control and the positive control (levamisole), suggesting a macrophage-activating role and immuno-effectiveness of Phytocee herbal formulation.
Studies from different laboratories showed a positive influence of W. somnifera treatment on enhanced NK cell activity and have demonstrated the immunomodulatory potential of W. somnifera with an increase in macrophages phagocytic activity (Davis & Kuttan, 2000, 2002; Harikrishnan et al., 2012; Ziauddin et al., 1996). These observations suggest that W. somnifera may be used as an immune adjuvant with multiple therapeutic benefits in pathological conditions. In an in vitro study, the fruit extracts of E. officinalis treated rat lymphocytes demonstrated the capacity to overcome the heavy metal-induced immunosuppression along with cytoprotective properties through modulation of interleukin-2 (IL-2), IFN-γ production, and apoptosis (Sai Ram et al., 2002). Hasan et al. (2016) demonstrated the antioxidant and anti-inflammatory activity of amla extract with favorable immunomodulation capacity through adequate stimulation of the activity of macrophages, NK cells, and white blood cells.
O. sanctum is a medicinally important Ayurvedic herb proven for its role in multiple biological activities. The extract of Tulsi is well documented as a natural therapeutic agent to fight infections and diseases by stimulating the production of T-helper cells, NK cells, and immune cells (Jeba et al., 2011; Nemmani et al., 2002). O. sanctum has been found to have protective effects against induced (chemo and radiation) immunosuppression through the involvement of cytokine signaling molecules, which play a key role in the anti-inflammatory response (Mediratta et al., 2002). In O. sanctum-administered Wistar albino rats, the enhanced production of RBC, WBC, and hemoglobin levels was observed and suggested a significant immunomodulatory effect (Chandrasekaran et al., 2010; Jeba et al., 2011; Vaghasiya et al., 2010).
Conclusion
Based on the above-reported studies and current data, the polyherbal-formulated Phytocee has been confirmed to have immunoprotective-enhancing properties. From the long traditional background, these polyherbal formulations are evident for their safe use as dietary constituents and for the overall health of livestock. Further studies are required to elucidate the exact mechanism of action and to explore the individual and combinational bioactive potency of these polyherbal ingredients responsible for the modulation of the immune system.
Footnotes
Abbreviations
NK cells: Natural killer cells; CMC: Carboxymethyl cellulose; PBS: Phosphate-buffered saline; FBS: Fetal bovine serum; CCSEA: Committee for Control and Supervision of Experiments on Animals; SD: Standard deviation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Ethical Approval and Informed Consent
Not applicable as this article does not contain any studies with human participants performed by any of the authors.
