Abstract
Background
Modern research has proved that the volatile oil (VO) of Citrus × aurantium L. [Rutaceae] has the effect of treating depression, but the mechanism of treating depression by sniffing has not been studied.
Objectives
To study the mechanism of VO on depression.
Materials and Methods
Part I: Except for the blank group, the sniffing group and nasal drip group were continuously administered for 7 days to investigate the optimal administration method. Part II: Except for the blank group, the model, positive group, high, medium, and low dose group of VO were established by chronic unpredictable stress test. The VO was given on the 7th day of modeling for 17 consecutive days. The expression of neurotransmitters and receptors in the brain was determined. The medicinal ingredients in rat brain tissue and blood were detected by gas chromatography–mass spectrometry (GC–MS).
Results
Compared with nasal drip, sniffing administration has less irritation to the nasal mucosa. The VO could reduce the forced swimming immobility time, adjust the activity state, increase the content of γ-aminobutyric acid (GABA), reduce the content of glutamate, and upregulate the expression of GABAα1, GABAγ2, and their mRNA. The main pharmaceutical component is d-limonene.
Conclusion
d-limonene in VO upregulates GABAα1, GABAγ2 expression to treat depression.
Introduction
Depression is a very common condition in modern life. With the rapid development of the economy and the increase in social pressure, emotional factors have become the key factors of depression (McLean, 2020). Life and work pressure, negative life events, negative emotions, and irritability are the risk factors for depression (Wu et al., 2021). Depression is currently mainly treated through medication and psychological therapy. The drugs for depression mainly include selective serotonin reuptake inhibitors, serotonin modulators, 5-HT norepinephrine reuptake inhibitors, dopamine norepinephrine reuptake inhibitors, melatonin antidepressants, and others. The pathogenesis of depression is complex, mainly including monoamine, HPA axis, excitatory and inhibitory neurotransmission, epigenetics, and inflammation. Depression is associated with neurotransmitter disorders, including the γ-aminobutyric acid (GABA) and glutamatergic systems. Due to the side effects of depression medication and patient noncompliance, these patients gradually rely on nonpharmacological treatments (Bo et al., 2017; Zheng et al., 2020).
Since hundreds of years ago, there were records of aromatherapy. It can achieve the functions of regulating the spirit and improving the function of the spleen and stomach by smelling fragrance (Agatonovic-Kustrin et al., 2020; Gürler et al., 2020; Kawabata et al., 2020; Kemper, 2020). The characteristic of aromatic lies in its rich aromatic component—the volatile oil (essential oil). Due to its richness and varied physiological activities, essential oil is widely used to manage conditions involving the nervous system, thereby playing an important role in the treatment of depression, insomnia, Alzheimer’s disease, and many other diseases (Manganiello-Terra et al., 2020; Gong et al., 2020). Volatile substances from the essential oil can enter the brain circulation through the sense of smell. Its stimulating and inhibiting effects can relieve depression and help sleep problems by soothing the liver and spleen and regulating the nervous system (Kennedy et al., 2018; Wang & Heinbockel, 2018). By means of olfaction, the aromatic molecules of essential oil are transmitted to the brain through the nose, which can promote the release of chemical substances in the nervous system, producing calming and relaxing effects and achieving the purpose of treating depression.
The volatile oil of Citrus × aurantium L. [Rutaceae] is an important pharmacodynamic component, mainly including d-limonene, γ-terpinene, β-laurene, and α-pinene. According to the report, the Citrus × aurantium L. [Rutaceae] essential oil has antidepressant effects. The component limonene can maintain low concentrations of corticosteroids in the serum and monoamines in the brain, thereby reducing physical and psychological stress. For patients with depression, it is possible to stimulate the parasympathetic nervous system while reducing sympathetic activity. In this study, we found that the volatile components can enter the brain and blood of rats. The irritation of nasal mucosa was also evaluated to provide the basis and reference for the rational administration of volatile oil. Taking the rats, with liver depression as the model, we studied the effect of essential oil on neurotransmitters and the mechanism of treating depression (Figure 1).
A Schematic Diagram of the Research.
Materials and Methods
Reagents and Instruments
The compressed air atomizer originated from Jilin Dingxiang Technology Development Co., Ltd. (Jilin, China). The gas chromatography–mass spectrometry (GC–MS) machine model used was the Agilent 7890A 5975C (Palo Alto, CA, USA). The 3-18k high-speed frozen centrifuge was from SIGMA (Germany). The enzyme marker was purchased from Labsystems Multiskan MS (Finland). The tissue fixation solution and phosphate-buffered saline (PBS) buffer were obtained from Beijing Salarbio Science & Technology Co., Ltd. (Beijing, China). The trichloromethane, sodium hydroxide, and ethyl acetate were derived from Xilong Scientific Co., Ltd. (Shantou, Guangzhou, China). The essential oil of Citrus × aurantium L. [Rutaceae] was extracted in the laboratory. The d-limonene was derived from the China Food and Drug Testing Institute (the content is 96%, Beijing, China).
Investigation and Test of Drug Delivery Method
Animal Grouping and Drug Administration
The specific-pathogen-free (SPF) grade Sprague-Dawley (SD) rats came from Hunan Slike Jingda Experimental Animal Co., Ltd. (animal license no. SCXK (Xiang) 2016-0002). All animals were kept in a pathogen-free environment and fed ad libitum. The procedures for the care and use of animals were approved by the Ethics Committee of Southwest Medical University, and all applicable institutional and governmental regulations concerning the ethical use of animals were followed. All volatile oils in this experiment were extracted in the laboratory and stored in a refrigerator at 10°C.
Divide 24 rats into 3 groups: control group, sniffing group, and nose drop group. The control group was drug-free. The sniffing group was treated with essential oil aromatherapy. The sniffing group and nasal drip group were administered oils continuously for 7 days, 3 times a day. The dose of the sniffing group was 1.25 µL/kg and that of the nasal drip group was 0.25 µL/kg (Zhong et al., 2021).
Pathological Section of Nasal Mucosa
The rat nasal tissue was fixed in 4% paraformaldehyde for 24 hours and decalcified with ethylenediaminetetraacetic acid (EDTA) decalcification solution. The tissue was harvested, dehydrated, embedded, and sliced. Then, hematoxylin and eosin (H&E) staining was performed with the nuclei and cytoplasm staining blue and red, respectively. Lastly, microscopic examinations, image acquisitions, and analyses were performed.
Effect of Volatile Oil on Rats with Depression
Preparation and Administration of Depression Model Rats
A total of 48 rats were randomly assigned to six groups, each comprising 8 animals. The groups were as follows: a control group, a model group representing depression, a positive control group treated with fluoxetine, and three experimental groups receiving different doses of volatile oil, namely the high dose (VOH), medium dose (VOM), and low dose (VOL) groups. The control group received no additional stimulation beyond the standard housing conditions. The other groups were subjected to chronic unpredictable stress test for continuous modeling for 23 days (Li et al., 2021; Yang et al., 2021). The drug was administered on the 7th day of modeling, the positive group was given fluoxetine solution (10 mg/kg). The VOH, VOM, and VOL groups were treated with essential oil aromatherapy with volumes of 1.335, 0.665, and 0.335 µL/kg, respectively.
Determination of Immobilization Time of Rats in Forced Swimming
The rats were placed into a barrel with a height of 52 cm, a diameter of 40 cm, a water depth of 25 cm, and a water temperature of 25 ± 2°C. The forced swimming experiment was carried out 1 hour after treatment. The total swimming time was 6 minutes. The first 2 minutes were not recorded, and the immobility time of rats swimming in the following 4 minutes was recorded. “Immobility” was defined as nonstruggling behavior in the water or a small body movement to keep its head out of the water (Abbasi-Maleki et al., 2019; Bombi et al., 2020; Ráez et al., 2020; Zuo et al., 2020).
An Open Field Experiment in Rats
On the 22nd day of the experiment, rats in each group were measured in the open field test after drug administration (Kajitani et al., 2020). Prior to the commencement of the experiment, the rats were acclimated to the laboratory environment for a period of 10 minutes. Subsequently, each rat was individually positioned in the center of an open field arena, with its head restrained to one side. Upon release, the video analysis system was immediately initiated to automatically capture and record the subsequent behaviors. Each rat adapted to an open box for 3 minutes, and the measurement time was 5 minutes. After the measurement, the feces particles and urine left by the rats were removed, and the next experiment was carried out.
Mechanism of Volatile Oil on Depressed Rats
The Concentrations and the Ratio of Glu to GABA were Assessed
On the 23rd day of the experiment and after the administration of the drug, all rats in each group were euthanized and their brain tissues were collected, especially the hypothalamus, cerebral cortex, and hippocampus. Brain tissues were thawed, dried, and weighed, and then a nine-times volume of PBS solution was added, followed by homogenization using an electric homogenizer. After centrifugation at 4°C for 3,500 rpm for 10 minutes, the supernatant was taken and processed for the enzyme-linked immunosorbent method (Li et al., 2020). The absorbance (OD value) was measured at a wavelength of 450 nm.
Immunohistochemical Method to Detect the Expression of GABAAα1 and GABAAγ2
The cerebral cortex was fixed for 48 hours and washed with running water, then embedded, and sectioned. Next, the samples were stained for immunohistochemical analysis, and images were captured using a microscope (Yang et al., 2020).
Western Blot Detection of the Expression of GABAAα1 and GABAAγ2
Cerebral cortical tissue was isolated, and lysates were prepared by homogenization. Then, the samples were centrifuged at 12,000 g at 4°C for 15 minutes and the supernatant was stored at –80°C. Protein concentrations of newly thawed protein samples were determined using the bicinchoninic acid (BCA) method. Next, the prepared PAGE glue was placed into the electrophoresis tank, which was added with the appropriate amount of electrophoresis buffer. The proteins were semi-dry transferred to a blotting membrane and then incubated with antibodies. Protein bands were detected using enhanced chemiluminescence (ECL) and scanned using an imaging system (Luo et al., 2020; Zeng et al., 2017).
Real-time Fluorescence Quantitative Polymerase Chain Reaction (PCR) to Detect the Expression of Cerebral Cortex GABAAα1 mRNA and GABAAγ2 mRNA
RNA extraction and cDNA synthesis were performed according to the kit instructions. The synthesis of cDNA was performed for PCR amplification. Reverse transcription and PCR were conducted according to the kit instructions (Fan et al., 2020). The primer sequences are as follows: GABAAα1 Primer F (5′-CGGCTCTGATTGAGTTTG-3′), GABAAα1 Primer R (5′-CAGGCTTGACTTCTTTCG-3′), GABAAγ2 Primer F (5′-TCTCTTCGTCTCTGTTTG-3′), GABAAγ2 Primer R (5′-GGTGGCATTGTTCATTTG-3′), β-actin Primer F (5′-CGGTCAGGTCATCACTATC-3′), β-actin Primer R (5′-CAGGGCAGTAATCTCCTTC-3′). The PCR procedure: 95°C, 10 minutes (95°C, 15 seconds; 55°C, 45 seconds) × 40; 95°C, 15 seconds; 60°C, 1 minutes; 95°C, 15 seconds; 60°C, 15 seconds.
The Pharmacological Substance Basis of Volatile Oil in Treating Depression
Preparation of Plasma Samples
Collect blood samples from the blank group and the sniffing group, and centrifuge for 10 minutes at 4,000 rpm. The supernatant was taken and 4 times the amount of methanol was added and then vortexed for 3 minutes. After centrifugation at 8,000 rpm for 10 minutes, the supernatant was collected (Zhang et al., 2020). The supernatant was added with 500 µL of 2 mol/L NaOH solution, and 20 mL of trichloromethane and was thoroughly vortexed. After setting it still, the chloroform layer was removed, dried, and 2 mL ethyl acetate was added to dissolve. The solution was filtered using a microporous filter membrane. The filtrate was used for GC–MS detection (Matsumoto et al., 2021).
Preparation of Brain Tissue Samples
After the blood was taken from each rat, the brain tissue was removed by rapid craniotomy. The weight of each brain was measured accurately. After adding 1.5 times normal saline, the brain tissue homogenate was prepared using an electric homogenizer. Then, 4 times the amount of methanol was added to the homogenate and vortexed for 3 minutes. Centrifuge at 8,000 rpm for 10 minutes and collect the supernatant. The method of sample preparation is the same as described above.
GC–MS Conditions
An appropriate amount of d-limonene was weighed to prepare the control solution. Further, an appropriate amount of essential oil was taken to prepare the volatile oil solution. Gas chromatographic conditions: Agilent HP-5MS (30 m × 0.25 mm × 0.25 µm) capillary quartz column; carrier gas: helium; column flow rate: 1.0 mL/min; sample volume: 1.0 µL; shunt ratio: 40:1; inlet temperature: 250°C; temperature rise procedure: 40°C; holding for 2 minutes, 10°C/min to 220°C, 20°C/min to 300°C, and holding for 5 minutes. Mass spectrometry conditions: the electron source is EI; electron energy: 70 eV; ion source temperature: 230°C; temperature of four-stage bar: 150°C; and quality scan range: full scan.
Results
Nasal Mucosa Irritation was Used to Optimize the Administration Mode
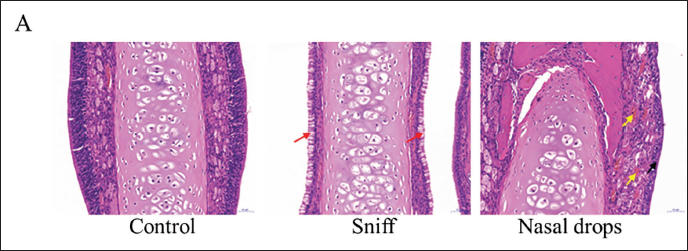
Experimental rats in the nasal drop group showed symptoms of nasal irritation, such as nasal scratching and sneezing, indicating that the administration of volatile oil drops could cause nasal discomfort and nasal mucosa irritation. Rats in the sniffing group were basically in a sleep state when they were given the drug, and occasionally they would scratch their noses. In the normal group, the external tissues of the nose were red and tender, the lips were pink, and the fur was bright and white. The rats in the sniffing group showed wetness around the muzzle after administration. In the nasal drip group, redness and swelling appeared in the external nasal tissue after administration and gradually disappeared after about 1 hour.
H&E staining was performed on the nasal mucosa to analyze the degree of irritation after different drug administrations (Figure 2). In the control group, the mucosal epithelial cells of the nasal tissue were columnar epithelium with clear contour and regular arrangement. No cell damage or inflammatory infiltration was observed in the tissue, and the overall structure was relatively normal. No inflammatory cell infiltration and cell damage were observed in the tissue of the sniffing group. In the nasal drop group, the epithelial cells of the nasal mucosa showed a significant transformation from columnar epithelial cells to squamous epithelial cells. The submucosal portion showed dilation and congestion. Altogether, H&E staining revealed that nasal drop administration was more irritating to the nasal mucosa than the inhalation route. This experiment shows that compared with nasal drip, sniffing administration is safer. Finally, animals are administered by sniffing.
Hematoxylin and Eosin (H&E) Staining of the Nasal Mucosa.
Evaluation of Depression Model
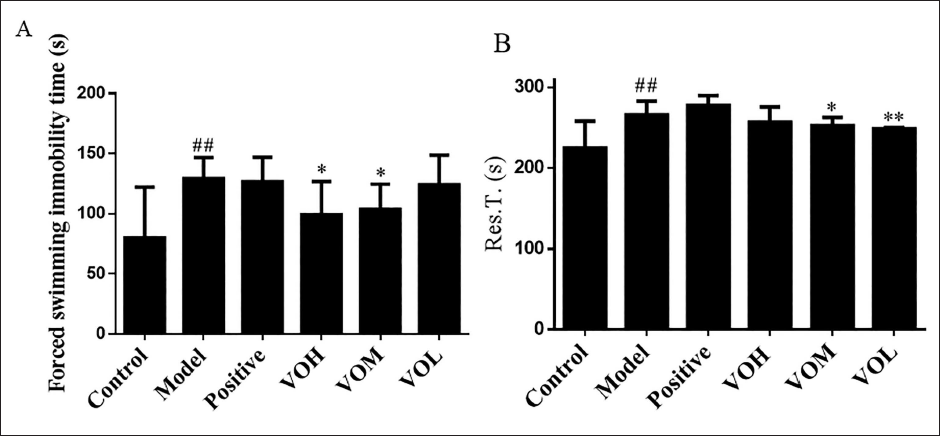
Forced swimming immobility time is an important indicator of depression in rats. The time of “immobility” in the water of each rat was recorded (Figure 3A). Compared to the control group, the duration of immobility during the forced swimming test was significantly longer in the depression group. Compared to the depression group, the duration of immobile swimming in the VOH and VOM groups decreased significantly, indicating that the volatile oil had improved the symptoms of depression in rats. On the 22nd day of modeling, open field experiments were carried out immediately after 30 minutes of administration (Figure 3B). Compared to the control group, the rest time in the depression group was significantly different (p < 0.05). Compared to the depression group, the rest time of the volatile oil group has significant differences in different degrees.
Evaluation of Depression Models. (A) Forced Swimming Immobility Time, (B) the Rest Time. #p < 0.05, ##p < 0.01 versus Control Group, *p < 0.05, **p < 0.01 versus Depression Group.
Pharmacological Mechanism Research
The Concentrations and the Ratio of Glu to GABA
The contents of GABA and Glu in the brain tissues of each group are shown in Figure 4A. In comparison with the control group, the levels of GABA in the hypothalamus, cerebral cortex, and hippocampus of the depression group were significantly reduced (p < 0.01). Compared to the depression group, the content of GABA in the positive group and volatile oil dosing group showed a significant increase (p < 0.01). These suggest that the inhibitory neurotransmitter GABA in the hypothalamus, cerebral cortex, and hippocampus decreased after depression in rats, and the content of GABA increased after the administration of diazepam and the volatile oil.
Effect of Volatile Oil on the Content of Neurotransmitters in Rats with Depression. (A) The Content of Neurotransmitters in the Hypothalamus, (B) the Content of Neurotransmitters in the Cerebral Cortex, (C) the Content of Neurotransmitters in the Hippocampus. #p < 0.05, ##p < 0.01 versus Control Group, *p < 0.05, **p < 0.01 versus Model Group.
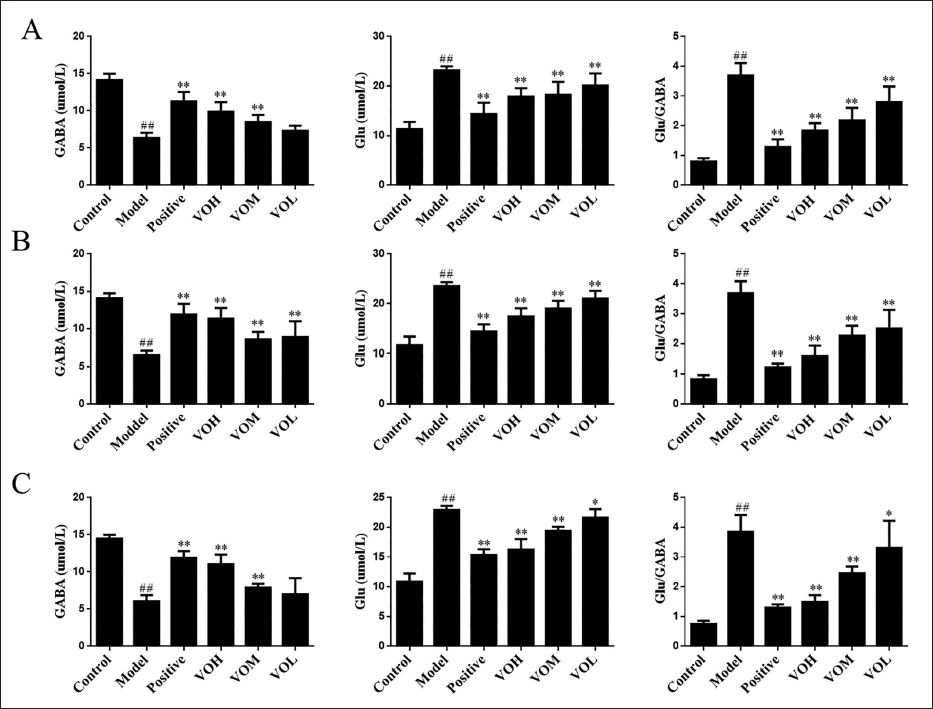
In comparison with the control group, the content of Glu in the three brain tissues of the depression group increased significantly (p < 0.01). Compared with the depression group, the content of Glu in the positive group and volatile oil dosing group showed a significant reduction (p < 0.01). These suggest that the excitatory neurotransmitter Glu in the hypothalamus increased after depression in rats, while the content of Glu decreased after the administration of positive and volatile oil.
Compared to the control group, the ratio of Glu/GABA in the depression group increased significantly (p < 0.01). Compared to the depression group, the Glu/GABA ratio in the positive group and volatile oil dosing group decreased significantly (p < 0.01). This indicates that after the modeling of depression in the rats, the excitatory neurotransmitter Glu in the hypothalamus increased, the inhibitory neurotransmitter GABA decreased, the ratio of Glu/GABA increased, and the excitatory neurotransmitter Glu became dominant so that depression occurred in the rats. After the administration of positive and volatile oil, the excitatory neurotransmitter Glu in the hypothalamus decreased, the inhibitory neurotransmitter GABA increased, the ratio of Glu/GABA decreased, and the inhibitory neurotransmitter GABA dominated, and the depression of rats was relieved, indicating that positive and volatile oil have antidepressant effects.
The Expression of GABAAα1 and GABAAγ2 Determined by Immunohistochemistry
The expression levels of the GABAA receptor subtype α1 (GABAAα1) in the cerebral cortex were assessed using immunohistochemical techniques (Figure 5A). In comparison to the control group, the expression of GABAAα1 in the depression group was found to be significantly reduced (p < 0.01). Furthermore, when compared to the depression group, the expression levels of GABAAα1 in the positive, VOH, and VOM groups exhibited a significant increase (p < 0.01). The VOL group also demonstrated a significant increase in GABAAα1 expression, albeit with a lower level of statistical significance (p < 0.05) (Figure 5B). Following the induction of depression, the expression of the GABAAα1 was observed to decrease. After administration of positive and volatile oil, the expression of GABAAα1 increased, indicating that the essential oil could upregulate the expression of GABAAα1. The expression of GABAAγ2 receptor was determined by immunohistochemistry (Figure 5C). In comparison to the control group, the expression of GABAAγ2 in the depression group was significantly reduced (p < 0.01). Compared with the depression group, the expression of GABAAγ2 in the positive, VOH, and VOM groups showed a significant increase (p < 0.01), and the expression of GABAAγ2 in the VOL group increased significantly (p < 0.05) (Figure 5D). After depression in rats, the expression of GABAAγ2 decreased, but showed an increasing level after the administration of positive and the volatile oil, indicating that the volatile oil can upregulate the expression of GABAAγ2 in the cerebral cortex of rats.
Effect of Volatile Oil on GABA Protein in Rats with Depression. (A) Immunohistochemical View of the GABAAα1 Receptor in the Cerebral Cortex. (B) The IOD Values of GABAAα1 Receptors in the Cerebral Cortex, (C) Immunohistochemical View of the GABAAγ2 Receptor in the Cerebral Cortex, (D) the IOD Values of GABAAγ2 Receptors in the Cerebral Cortex, (E) Electrophoretic Images of GABAAα1 and GABAAγ2 Expression. (F) Relative Expression of GABAAα1 and GABAAγ2 Proteins. (G) Expression of GABAAα1 mRNA and GABAAγ2 mRNA. #p < 0.05, ##p < 0.01 versus Control Group, *p < 0.05, **p < 0.01 versus Model Group.
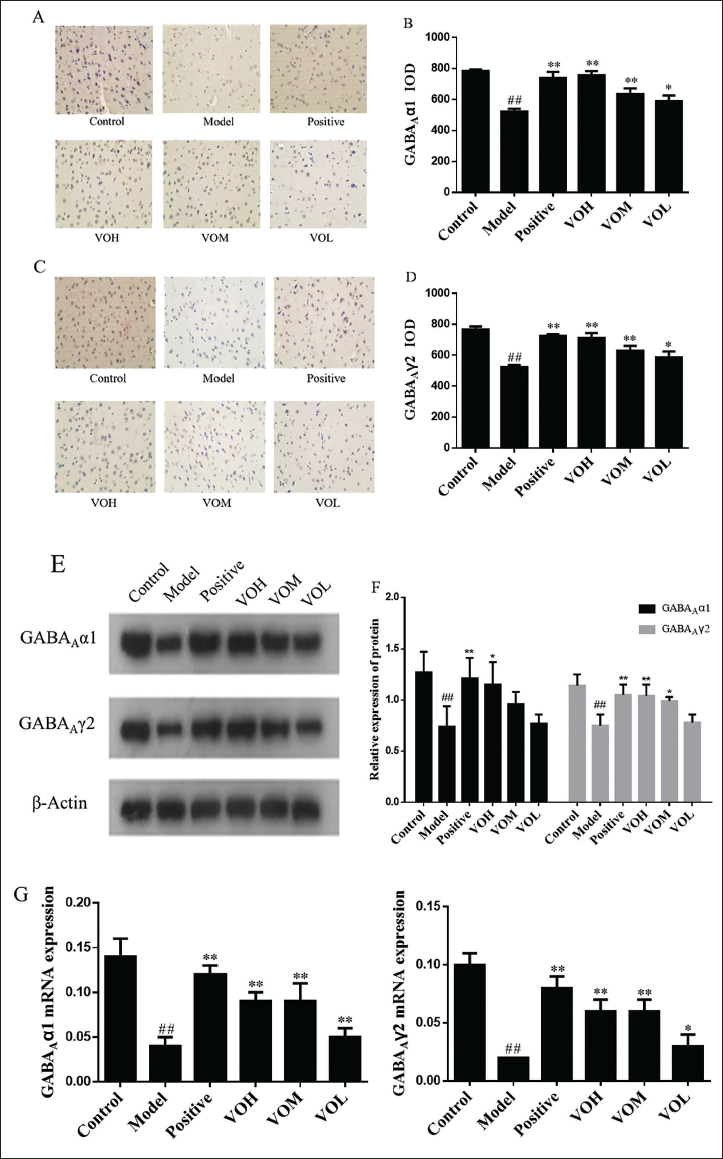
The Expressions of GABAAα1 and GABAAγ2 Detected by Western Blot
The expression of GABAAα1 and GABAAγ2 receptors in the cerebral cortex was determined by western blot (Figure 5E). Compared with the control group, the relative expression of GABAAα1 and GABAAγ2 proteins in the depression group decreased (p < 0.01). Compared with the depression group, the relative expression levels of GABAAα1 and GABAAγ2 proteins in the positive group increased significantly (p < 0.01). The relative expression of GABAAα1 (p < 0.05) and GABAAγ2 (p < 0.01) protein in the VOH group increased. Also, the relative expression of GABAAγ2 protein in the VOM group increased (p < 0.05) (Figure 5F). After depression in rats, the expressions of GABAAα1 and GABAAγ2 in the cerebral cortex decreased, and the expressions of GABAAα1 and GABAAγ2 increased after the administration of positive and the essential oil, indicating that the essential oil could upregulate the expressions of GABAAα1 and GABAAγ2, among which the VOH group had the best effect.
The Expressions of GABAAα1 and GABAAγ2 mRNAs Detected by Real-time Fluorescence Quantitative PCR
The expressions of GABAAα1 and GABAAγ2 mRNAs in the cerebral cortex of rats were detected by real-time fluorescence quantitative PCR. Compared with the control group, the expressions of GABAAα1 and GABAAγ2 mRNAs of the model group decreased significantly (p < 0.01). Compared with the depression group, the expressions of GABAAα1 and GABAAγ2 mRNAs in the positive, VOH, VOM, and VOL groups were all increased (Figure 5G). The expressions of GABAAα1 and GABAAγ2 mRNAs decreased after depression but were both upregulated after the administration of positive and volatile oil.
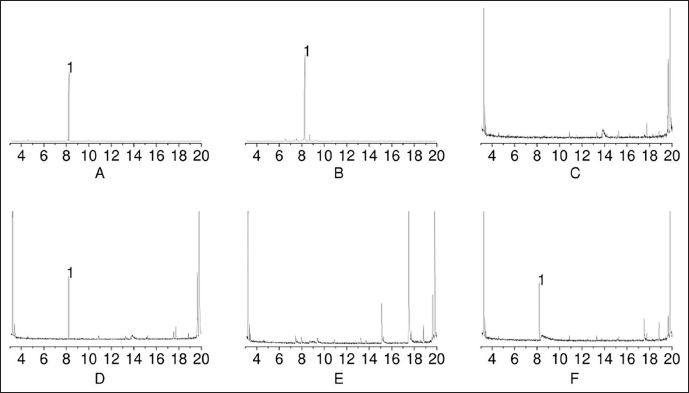
The Pharmacodynamic Substance Basis was Explored by GC–MS
The main chemical components in volatile oil are d-Limonene, γ-terpinene, myrcene, and cymeme, with relative contents of 85.896%, 6.507%, 2.203%, and 1.333%, respectively. d-limonene is the main chemical component of volatile oil. GC–MS was used to detect chemical components in plasma and brain tissue (Figure 6). Except for the blank group, d-limonene was detected in both plasma and brain tissue of the sniffing group, indicating that d-limonene can enter the blood and brain tissue through sniffing and is the main pharmacological component for alleviating depression.
Total Ion Current (TIC) Diagram of Volatile Components in Brain Tissue and Plasma Samples Determined by Gas Chromatography–Mass Spectrometry (GC–MS). (A) TIC Diagram of d-limonene, (B) Volatile Oil in Citrus × aurantium L. [Rutaceae], (C) Brain Tissue in the Control Group, (D) Brain Tissue in the Sniffing Group, (E) Plasma in the Control Group, (F) Plasma in the Sniffing Group.
Discussion
The results of nasal surface irritation and nasal mucosal pathology showed that sniffing was safer than nasal dropping, so sniffing was chosen as the administration method of volatile oil. Compared with the blank group, the model group rats showed an increase in forced swimming immobility time. After the administration of volatile oil, the duration of forced swimming immobility decreased, indicating that volatile oil improved the depressive behavior of rats.
An increasing body of research indicates that amino acid neurotransmitters, including Glu and GABA, play a role in the pathogenesis of depression. Glutamate, an amino acid neurotransmitter, is extensively distributed throughout the central nervous system and serves as the primary excitatory neurotransmitter in humans. It plays an important role in maintaining the excitability and normal activity of neurons, and its function involves processes such as sensory transmission, motor control, and advanced cortical function. GABA is an inhibitory neurotransmitter, and its synthesis mainly comes from Glu metabolism (Ogawa et al., 2014, 2018). Glutamate decarboxylase is an important biosynthetic enzyme of GABA. GABA has antianxiety and endocrine-regulating effects. Research has shown that the levels of excitatory neurotransmitter Glu in the blood and brain tissues of patients with depression are significantly higher than those of the normal control group, while the levels of inhibitory neurotransmitter GABA decrease. The increasing level of Glu and the decreasing level of GABA are positively correlated with the degree of depression, and after drug treatment intervention, Glu levels decrease and GABA content increases. This indicates that the onset of depression is related to amino acid neurotransmitters such as Glu and GABA (Torontali et al., 2019; Vanini et al., 2020; Yin et al., 2019).
In this article, we showed that Glu level increased while GABA level decreased, and the ratio of Glu/GABA increased in the hypothalamus, cerebral cortex, and hippocampus after modeling depression. The content of GABA increased, the content of Glu decreased, and the ratio of Glu/GABA decreased after the administration of volatile oil. An imbalance between excitatory and inhibitory neurotransmitters can cause depression. We also showed that the expression of GABAAα1 and GABAAγ2 proteins and mRNAs decreased significantly in the cerebral cortex after modeling depression, while increased significantly after the administration of volatile oil. d-limonene was detected in both the brain and blood by GC–MS, indicating that d-limonene is the main component responsible for the pharmacological effects of volatile oils.
This experiment has certain limitations. In terms of studying the regulation of volatile oil on depression, only the regulation of GABA receptors and glutamate was studied. In terms of experimental design, behavioral studies related to depression should be increased, and the sample size of rats should be increased to reduce individual differences and experimental errors in behavior. The results of GC–MS indicate that d-limonene is the main component responsible for the pharmacological effects of volatile oils. The next step of this experiment will be to study and explore the mechanism of action of the active ingredient of d-limonene in the treatment of depression.
Conclusion
Depression is closely related to the imbalance of excitatory amino acids and inhibitory amino acid neurotransmitters in the central nervous system. The volatile oil from Citrus reticulata can improve the behavior and neurotransmitter expression of depression rats by sniffing. The mechanism of essential oil is mainly through upregulating the expression of GABAAα1, GABAAγ2 proteins, and GABAAα1 mRNA, GABAAγ2 mRNA. The pharmacological substance base is mainly d-limonene. Next, the research group will conduct in-depth research on the pathway and explore its deeper mechanism.
Recent research has provided evidence that volatile oils may exert their effects on the brain through the nasal–brain pathway, which involves the activation of olfactory chemoreceptors and subsequent signal transduction via the olfactory system. This pathway is believed to influence the limbic system, thereby affecting neurotransmitter metabolism. Additionally, volatile oils may modulate the neuroendocrine and autonomic nervous systems, including both the sympathetic and parasympathetic divisions, leading to a range of physiological, psychological, and behavioral effects that could contribute to the alleviation of depressive symptoms.
Inhalation aromatherapy for the treatment of depression appears to be influenced not solely by the sensory perception of aromatic odors but also by the pharmacological properties of volatile oils on the nervous system. Aromatherapy, delivered via the nasal route with brain-targeting capabilities, offers a promising therapeutic approach for depression that merits additional investigation.
Footnotes
Authors’ Contributions
G. Ren and G. Ke conceived and designed the study. G. Ren, Q. Pu, and J. Zhao performed the experiments. G. Ren, R. Huang, and P. Liang analyzed all the data and wrote the manuscript. G. Ren revised the manuscript, obtained the funding, and supervised the whole study. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Sichuan Natural Science Foundation: Study on the mechanism of controlling the stable release of volatile oil from traditional Chinese medicine by porous carrier material curing technology based on the principle of “adsorption-desorption” (2022NSFSC1457), the 73rd batch project of China Postdoctoral Science Foundation, Study on the mechanism of orderly porous materials regulating the balanced release of traditional Chinese medicine essential oil (2023M732928), Scientific research project of Southwest Medical University: Study on the volatilization law of traditional Chinese medicine volatile oil and the slow-release effect of carrier material solidified volatile oil (2020ZRQNA032).
Informed Consent
Not applicable.
Institutional Review Board Statement
Not applicable.
