Abstract
The current study investigated the γ-aminobutyric acid (GABA) levels and GABA metabolic enzymes (GABA transaminase (GABAT) and glutamate decarboxylase (GAD)) activities at 2 and 4 h after treatment, using a high-performance liquid chromatography with ultraviolet detectors and colorimetric assay, in the cerebral cortex of mice treated with 20, 40 or 80 mg/kg β-cypermethrin by a single oral gavage, with corn oil as vehicle control. In addition, GABA protein (4 h after treatment), GABAT protein (2 h after treatment) and GABA receptors messenger RNA (mRNA) expression were detected by immunohistochemistry, Western blot and real-time quantitative reverse transcriptase polymerase chain reaction, respectively. β-Cypermethrin (80 mg/kg) significantly increased GABA levels in the cerebral cortex of mice, at both 2 and 4 h after treatment, compared with the control. Also, GABA immunohistochemistry results suggested that the number of positive granules was increased in the cerebral cortex of mice 4 h after exposure to 80 mg/kg β-cypermethrin when compared with the control. Furthermore, the results also showed that GABAT activity detected was significantly decreased in the cerebral cortex of mice 2 h after β-cypermethrin administration (40 or 80 mg/kg). No significant changes were found in GAD activity, or the expression of GABAT protein and GABAB receptors mRNA, in the cerebral cortex of mice, except that 80 mg/kg β-cypermethrin caused a significant decrease, compared with the vehicle control, in GABAA receptors mRNA expression 4 h after administration. These results suggested that attenuated GABAT activity induced by β-cypermethrin contributed to increased GABA levels in the mouse brain. The downregulated GABAA receptors mRNA expression is most likely a downstream event.
Keywords
Introduction
Pyrethroids are synthetic insecticides derived from pyrethrins that have higher efficiency, lower toxicity, environmental stability and are used in a wide array of indoor and outdoor applications, including veterinary, agricultural usages and home pest control. 1 On the basis of the chemical structure and signs of acute toxicity, pyrethroids have been classified as type I, which induces whole body tremor and type II, which induces choreoathetosis with salivation (CS) intoxication syndrome. 2,3 However, not all pyrethroids fall neatly into either of these two categories. 4 Pyrethroids are a class of neurotoxic insecticides, in which the mechanism of action on mammal is complex due to the multiple factors and links. 5 –7 The molecular basis for pyrethroid neurotoxicity has been attributed to their actions on voltage-gated and receptor-regulated ion channels. 1,8 –11 Moreover, some studies showed the alterations regarding pyrethroid-induced neurotransmission. 12,13 It has been reported that direct effects of pyrethroids on ion channels have been explored as a possible explanation for the enhanced central nervous system (CNS) neurotransmitter release that accompanies intoxication. 4
γ-Aminobutyric acid (GABA) is the most important inhibitory neurotransmitter and actively participates in maintaining the excitation/inhibition balance in the CNS. There are differential presynaptic actions of pyrethroid insecticides on GABAergic neurons in the hippocampus 14 as well as distinct effects of pyrethroids on the GABA receptor–chloride ionophore complex. 15 –17 Therefore, further information concerning the effects of pyrethroids on GABA neurotransmitter and GABA receptors will be required to clarify their potential roles in the development of pyrethroids neurotoxicity.
In the brain, glutamate is derived from α-ketoglutaric acid, an intermediate of the tricarboxylic acid cycle. Then, glutamate is converted to GABA by glutamate decarboxylase (GAD). GABA is metabolized by the successive action of GABA transaminase (GABAT). Thus, GAD and GABAT play important roles in the metabolism of GABA in the CNS. Ji et al. 18 have already reported pyrethroid-induced alterations in GABAT. However, it needs to be further discussed as to which one the earlier site is regarding the effects of pyrethroids on GABA and GABAT. Furthermore, the effects of pyrethroids on GAD in the mammalian CNS are still unclear.
Cypermethrin, a class II synthetic pyrethroid pesticide, is used commonly to control insects globally for agricultural and domestic purposes. 19 As a mixture of the α and θ forms of cypermethrin, β-cypermethrin has higher activity than cypermethrin and thus is used widely. Here, we investigate the effects of β-cypermethrin on the GABA neurotransmitter and its metabolic enzymes and receptors, in order to better understand the downstream effects and toxic symptoms of pyrethroid-induced acute neurotoxicity. These types of studies supply information that can be the basis for additional research and will provide an important theoretical basis for the treatment of acute pyrethroid poisoning.
Materials and methods
Chemicals
β-Cypermethrin (molecular formula: C22H19C12NO3; molecular weight: 416.3 g/mol; purity: 97.16%; isomers mixture: 1R, S-cis and 1R, S-trans) was a generous donation by Shenyang Research Institute of Chemical Industry (Shenyang, China); adenosine diphosphate, nicotinamide adenine dinucleotide (NADH), dansyl chloride, pyridoxal phosphate and GABA were purchased from Sigma Chemical Corporation (St Louis, Missouri, USA); goat anti-mouse GABA polyclonal antibody, goat anti-mouse GABAT polyclonal antibody and goat anti-mouse β-actin polyclonal antibody were obtained from Santa Cruz Biotechnology (Santa Cruz, California, USA). Immunohistochemistry kit was purchased from Beijing Zhongshan Biotechnology (Beijing, China). All other reagents were of the highest purity available and from commercial sources.
Animals
Adult male and female albino mice (animal code SCXK 2008-1105), weighing 20 ± 2 g, were obtained from the Experimental Animal Center at China Medical University (Shenyang, China). The animals were maintained on a 12-h light/dark cycle with controlled temperature at 23 ± 3°C, relative humidity at 55 ± 15% and food and water ad libitum. The animal experiments were performed according to the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals and approved by the local authorities. All efforts were made to minimize suffering and the number of animals used.
Treatments
The mice were divided randomly into four groups (n = 20, equal numbers of male and female mice). Three groups were administered with 20, 40 and 80 mg/kg β-cypermethrin (approximately equal to one-twelfth, one-sixth and one-third of the acute oral median lethal dose, respectively) dissolved in corn oil and delivered by a single oral gavage. The dose selection was based on the observation of apparent signs of toxicity not leading to death in mice exposed to β-cypermethrin at the highest dose, while only slight signs of toxicity, or no signs of toxicity, occurred at the middle or lowest dose, respectively. The final group was given corn oil vehicle only and served as the control group. Based on the onset of toxic signs and their severity, 10 mice in each group were killed at 2 and 4 h, respectively, after treatment, and the entire cerebral cortex was collected. Half of the cerebral cortex was used for measuring GABA levels and the GABAT and GAD activity, and the other half for analyzing the expression of GABAT protein and GABA receptors messenger RNA (mRNA). Furthermore, additional 12 mice (three in each group) were selected for immunohistochemical assay to demonstrate the alteration of GABA levels in the cerebral cortex 4 h after treatment with β-cypermethrin.
GABA measurement
GABA levels assay
Brain tissue samples (0.1 g) were weighed and homogenized in 2 ml acetonitrile. The homogenate was centrifuged at 13,000 r/min for 10 min at 4°C. The supernatant was collected and evaporated to dryness at 40°C under a gentle stream of nitrogen. The dried residue was reconstituted in 400 μl sodium carbonate–sodium bicarbonate buffer solution (pH 9.5). An aliquot of 150 μl dansyl chloride (10 mmol/l), used as the derivatization reagent, was added and the sample was vortexed. The solution was incubated in the dark at 65°C for 25 min and then cooled to room temperature. GABA levels in the cerebral cortex were determined using high-performance liquid chromatography with ultraviolet detectors (Waters China, Milford, Massachusetts, USA), according to the method described by Huo et al. 20 Sample detection was performed at 254 nm with a 20-µl injection volume. The method showed good overall intraday (6.4–8.5%) and interday (6.3–10.6%) validation. The calibration curves for GABA yielded good linearity with correlation coefficients of more than 0.998 in the range of 0.0525–108 µg/g. Furthermore, 98.5–108.9% recovery was obtained by spiking known GABA standards into the brain homogenates.
Immunohistochemical analysis
Mice were deeply anesthetized and transcardially perfused with saline followed by ice-cold 4% paraformaldehyde (pH 7.4). The brains were removed, post-fixed with the same fixative overnight at 4°C and then sliced into 5-μm thick coronal sections. Sections were dewaxed in xylene and dehydrated in an alcohol row. After being cooled, they were washed thrice in phosphate-buffered saline (PBS; pH 7.2). Endogenous peroxidase activity was blocked by immersion in 3% hydrogen peroxide for 15 min. Non-specific binding of the primary antibodies was blocked by incubating the sections with diluted normal goat serum for 15 min at room temperature. Sections were incubated overnight with primary antibodies (1:200 dilution) diluted in PBS. After being washed with PBS, sections were incubated with goat anti-rabbit immunoglobulinG used as a secondary antibody for 15 min. After incubation with horseradish peroxide (HRP) avidin–biotin complex for another 15 min and a repeated washing step with PBS, the reaction products were visualized with diaminobenzidine (DBA) chromogen solution. Sections were then counterstained with haematoxylin and observed under a light microscope.
GABAT and GAD activity assays
The activities of GABA metabolic enzyme in the cerebral cortex were measured by the procedure of Sytinsky et al. 21 GAD activity is expressed as micromoles per GABA formed per milligram of fresh tissue protein per minute. A unit of GABAT activity in the cerebral cortex is defined as the amount of enzyme which catalyzes, at 37°C and pH 8.75, the formation of 1 nmol NADH in l min, and the activity of GABAT is described in units per milligram of protein. The protein content was determined using Coomassie brilliant blue by Bradford. 22
Western blot analysis
The expression of GABAT protein was determined by Western blot. Briefly, whole cell lysates were separated by polyacrylamide gel electrophoresis and then transferred to polyvinylidene fluoride membrane. After blocking non-specific binding sites with 5% skim milk, the membrane was incubated with goat anti-mouse GABAT polyclonal antibody (1:200 dilution), followed by incubation with HRP-conjugated rabbit anti-goat secondary antibody (1:5000 dilution). Immunoreactive bands were detected using enhanced chemiluminescence substrate (Walterson Chemical Co, Beijing, China), and imaged using Tanon 2500R automatic digital gel image analysis system (Shanghai, China). To confirm equal loading of protein lysates, membranes were stripped and re-probed using goat anti-mouse β-actin polyclonal antibody (1:200 dilution), and 1:5000 HRP-conjugated rabbit anti-mouse secondary antibody, and then processed as above.
Real-time qRT-PCR analysis
According to the manufacturer’s instructions, total mRNA from the cerebral cortex of mice was isolated using Trizol reagent (Invitrogen, Carlsbad, California, USA). According to the manufacturer’s instructions, reverse transcription and first-strand synthesis from each sample was carried out using the PrimeScript RT-PCR System kit (TaKaRa Dalian Biotechnology, Dalian, China). The resulting complementary DNAs were used as templates for real-time quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) in the ABI 7500 Real-Time PCR system (Applied Biosystems, California, USA) using SYBR® Premix Ex Taq™ Mix (TaKaRa Dalian). The primers were synthesized and purified by TaKaRa Dalian with the following sequences: GABAA receptors, forward: 5′-AAATGACGCTGTGGATTCTGCTC-3′, reverse: 5′-TCCGATGTCAGGTCGAAGTTTG-3′ (194 bp product); GABAB receptors, forward: 5′-GAACCCTGCTTGGAGCTATTCAC-3′, reverse: 5′-TGCTGCTGAATGACAGGAGTTG-3′ (110 bp product); β-actin, forward: 5′-CATCCGTAAAGACCTCTATGCCAAC-3′, reverse: 5′-ATGGAGCCACCGATCCACA-3′ (171 bp product). The PCR reaction was amplified in the program including an initial denaturation step at 95°C for 30 s, followed by 40 cycles for denaturation at 95°C for 5 s, annealing at 60°C for 34 s, subsequent melting curves for 15 s at 95°C, 1 min at 60°C and 15 s at 95°C to ensure that only a single product was amplified. Absolute values from each sample were normalized to β-actin (constitutive gene) mRNA as a reference standard.
Statistical analysis
The continuous data are expressed as the mean ± SD. The experimental data were analyzed using one-way analysis of variance with post hoc analysis. All statistical analyses were performed using SPSS software for Windows (version 13.0, SPSS, Chicago, Illinois, USA). The least significant difference multiple comparisons test was used to compare differences between the control group and dosed groups. The confidence level for accepting the hypothesis was 5%.
Results
Signs of acute toxicity induced by β-cypermethrin
The onset of toxic symptoms such as apparent licking, anxiety and scratching were observed in mice at 1 h, followed by tremors, hyperthermia, convulsions, hunched posture, CS, unsteady or crawling gait with splaying of the hindlimbs and ataxia from 2 to 4 h after administration with 80 mg/kg β-cypermethrin. Slight licking and scratch symptoms were observed in some mice after administration with 40 mg/kg β-cypermethrin, and the peak effect of these clinical signs was at 2 h. No significant sign of toxicity was seen in mice at the 20 mg/kg dose during the experiment. No animals died, and no differences were seen between the sexes during the experiment.
Effect of β-cypermethrin on GABA
At the 80 mg/kg dose, β-cypermethrin caused significant GABA increases in the cerebral cortex of mice at 2 h (p < 0.05) compared with the control group and at 4 h (p < 0.01) compared with the other three groups after treatment. In addition, no significant differences (p > 0.05) in GABA level were found at both 2 and 4 h between the lower dose (20 or 40 mg/kg) β-cypermethrin and the control group (Figure 1).

Altered GABA levels in the cerebral cortex of mice treated with β-cypermethrin. The GABA levels in the cerebral cortex were detected using HPLC-UV method at 2 and 4 h after β-cypermethrin administration. The control mice were treated with the same volume of vehicle. Each value represents the mean ± SD of each group (n = 10). *p < 0.05, compared with the control group. ## p < 0.01, compared with the other three groups.
GABA immunohistochemistry results suggested that the number of positive granules was increased in the cerebral cortex of mice 4 h after exposure to 80 mg/kg β-cypermethrinin compared with the control (Figure 2).
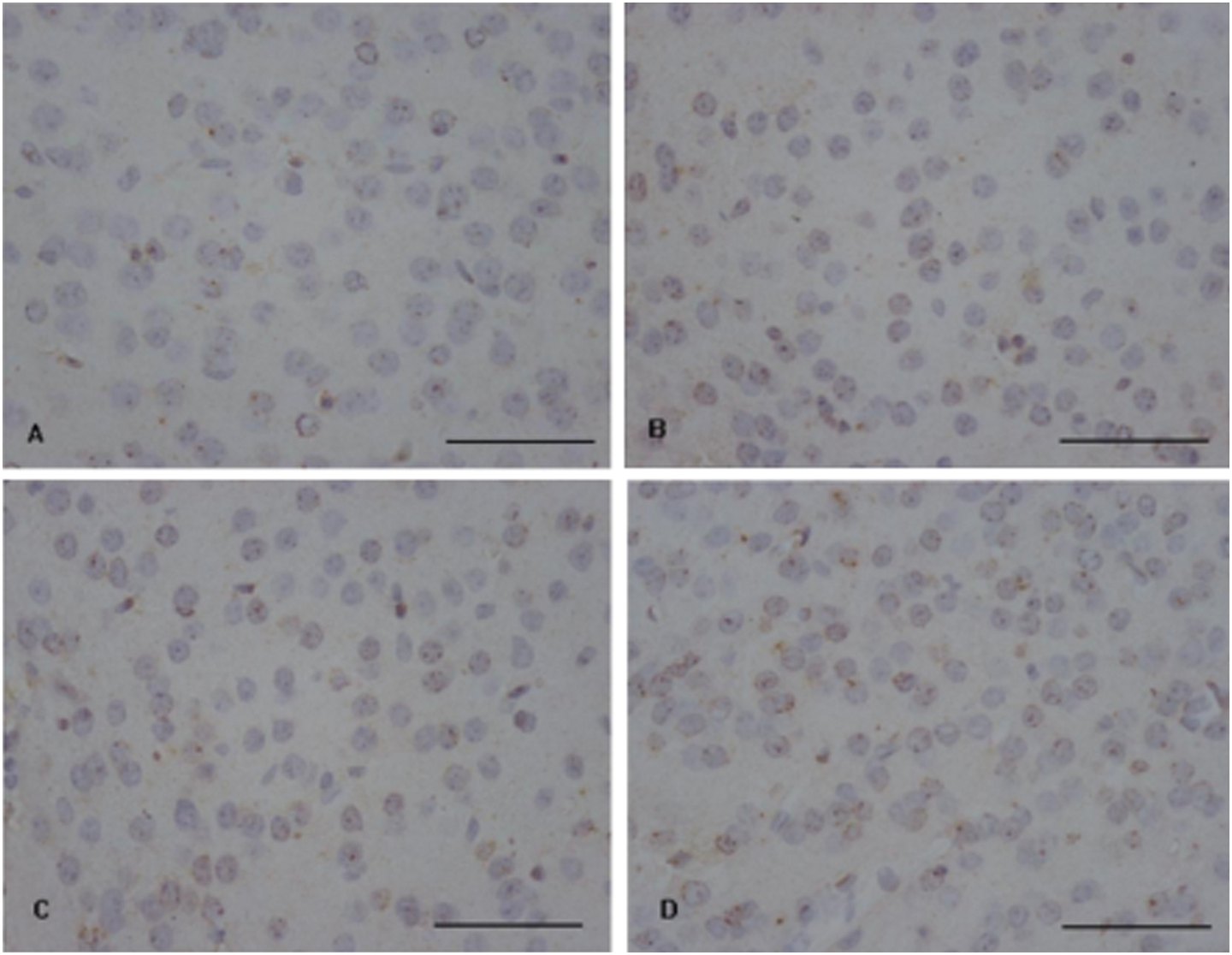
Immunohistrochemical assays for GABA in the mouse cerebral cortex. Brown granules imply the localization of GABA in the cerebral cortex of mice. The number of positive granules was increased in the cerebral cortex of mice treated with 80 mg/kg β-cypermethrinin compared with the control group. Bar = 50μm.
Effects of β-cypermethrin on GABAT and GAD activities
Compared with the control, significant decreases in GABAT activity were found at 2 h in the cerebral cortex of mice after β-cypermethrin administration with both 40 mg/kg (p < 0.05) and 80 mg/kg (p < 0.01). However, no significant differences (p > 0.05) were detected in GABAT activity of the cerebral cortex in mice 2 h after treatment with 20 mg/kg β-cypermethrin, as well at 4 h in each treatment group (Figure 3(a)). Furthermore, there were no significant differences (p > 0.05) in GAD activities of the cerebral cortex in mice among β-cypermethrin-treated groups and the control (Figure 3(b)).

GABAT and GAD activity in the cerebral cortex of mice exposed to β-cypermethrin. Enzyme (GABAT (a) and GAD (b)) activities in the cerebral cortex of mice were measured using colormetric method at 2 and 4 h after β-cypermethrin administration. The control mice were treated with the same volume of vehicle. Each value represents the mean ± SD of each group (n = 10). *p < 0.05 and **p < 0.01, compared with the control group.
Effect of β-cypermethrin on GABAT protein and GABA receptors mRNA expression
As shown in Figure 4, 2 h after administration, there were no significant differences (p > 0.05) in GABAT protein expression in the cerebral cortex of mice among the treatment groups. The GABAA and GABAB receptors mRNA expression in the cerebral cortex of mice are shown in Figure 5. At 2 h after administration, there was no significant difference (p > 0.05) in GABAA receptors mRNA expression between the groups. At 80 mg/kg, however, β-cypermethrin caused a significant decrease (p < 0.01) in GABAA receptors mRNA expression at 4 h compared with the other three groups. In addition, no significant differences (p > 0.05) in GABAA receptors mRNA expression were found between the lower doses (20 or 40 mg/kg) β-cypermethrin and the control group. The level of GABAB receptors mRNA expression did not show any significant changes (p > 0.05) at 2 or 4 h compared with the control group.

The effect of β-cypermethrin on GABAT protein expression in the mouse cerebral cortex. The expression of GABAT protein was determined by Western blot as described in the Materials and Methods section. The control mice were treated with the same volume of vehicle. Each value represents the mean ± SD of each group (n = 10). There were no significant differences (p > 0.05) in the expression of GABAT protein among the groups.

Analysis of GABAA and GABAB receptors mRNA expression in the cerebral cortex of mice. GABAA (a) and GABAB (b) mRNA expression was analyzed by qRT-PCR as described in the Materials and Methods section. Each sample was normalized to β-actin mRNA as a reference standard. Each value represents the mean ± SD of each group (n = 10). ## p < 0.01, compared with the other three groups.
Discussion
Generally, the normal function of the nervous system relies on the balance between excitability and inhibitory. As an important inhibitory neurotransmitter in the CNS, GABA exists in almost all the neurons in brain, mediates inhibitory synaptic transmission and prevents neuronal hyperexcitability. Hossain et al. 14 found that cyhalothrin increased the extracellular GABA in a dose-dependent manner, but deltamethrin decreased GABA levels in the hippocampus of rats using microdialysis following systemic administration. In addition, allethrin had an interesting dual effect: increasing GABA release at low doses (10 or 20 mg/kg) and decreasing GABA release at a high dose (60 mg/kg). The decreased GABA levels were also reported by Manna et al. 23 in the rat brain after a single oral dose of 150 mg/kg deltamethrin.
In this study, increased GABA levels were detected in the cerebral cortex of mice exposed to 80 mg/kg β-cypermethrin, and the result was supported by ours and another immunohistochemical study. 12 Moreover, we observed that the mice had a rapid onset of CS-syndrome in the current study, and some mice developed symptoms such as ataxia and splayed hindlimbs after administration of 80 mg/kg β-cypermethrin. Concerning the onset of toxic signs and their severity, no differences were attributed to sex. The relative balance of glutamate and GABA plays an important role in maintaining normal neural physiological functions. It has been reported that pyrethroids affected glutamate levels, an excitability neurotransmitter. 12,14 We also found that the level of glutamate decreased in the cerebral cortex of mice at 4 h, but not 2 h, after administration with 80 mg/kg β-cypermethrin (data not shown). These data suggest that pyrethroids induced the metabolic disturbance in glutamate and GABA.
In the nervous system, glutamate can be catalyzed to GABA by GAD. This study did not find a significant difference in GAD activity in the cerebral cortex of mice after treatment with different doses of β-cypermethrin. GABAT is another key enzyme in the metabolism of GABA, which is distributed widely in the CNS and exists mainly in the mitochondrial matrix. A significant decrease was found in GABAT activity after a single oral administration of 37.5 mg/kg deltamethrin. 18 In this study, significant decreases were detected in GABAT activity at 2 h, but not 4 h, in the cerebral cortex of mice after administration with 40 or 80 mg/kg β-cypermethrin. However, no significant changes were found in GABAT protein expression. Since there was no change in GABAT activity at 4 h, we only determined the expression of GABAT protein at 2 h. Based on comprehensive consideration of the alterations of GABA level and GABAT activity, in this study, increased GABA levels are supposed to result from the attenuation of GABAT activity in the cerebral cortex of mice treated with β-cypermethrinin.
Excessive GABA transmitters may trigger a cascade of biochemical events by binding to their receptors. There are three classes of GABA receptors: GABAA, GABAB and GABAC. GABAA receptors are ligand-gated ion channels (also known as ionotropic receptors), which mainly mediate fast synaptic transmission. 24 GABAB receptors are G protein-coupled receptors (also known as metabotropic receptors) that participate in slow synaptic transmission, which not only can affect the open activity of ion channels indirectly but also can activate a series of metabolic reactions in the postsynaptic neuron. 25 Like GABAA, GABAC receptors are ionotropic receptors, mainly found in the retina, spinal cord, superior colliculus, pituitary and gastrointestinal tube. 26 Some scholars believe that the GABAC receptors belong to the same family as GABAA receptors. Except for binding of GABA, these GABA receptors differ completely in their drug specificities. In the present study, 80 mg/kg β-cypermethrin caused a significant decrease in GABAA receptors mRNA expression, but not GABAB, in the cerebral cortex of mice 4 h after administration. According to the alteration of studied parameters at different doses and time points, we found that reduced GABAT activity was detected the earliest. Then, increased GABA, and then decreased GABAA receptors mRNA level were detected. The results suggest that the downregulated GABAA receptors mRNA expression could be a downstream event, which requires further study. GABAA receptors, which operate a Cl− channel, are the major inhibitory receptors of the vertebrate brain. The current study along with the previous studies 15 –17 raised the possibility that pyrethroids might directly or indirectly act on the GABA receptor–chloride ionophore complex. These data provide an important clue for further study on acute neurotoxicity caused by pyrethroids.
This study suggested that attenuation of GABAT activity induced by β-cypermethrin contributed to increased GABA levels in the cerebral cortex of mice and that the downregulated GABAA receptors mRNA expression is a downstream event. Our findings have implications for providing important insights into the neurotoxic potential of the pyrethroid insecticides.
Footnotes
Authors’ Note
Y HAN and D CAO contributed equally to this work.
Conflict of interest
The authors declared no conflicts of interest.
Funding
The work was financially supported by the National Natural Science Foundation of China (grant number 30872144).
