Abstract
Background
Potentilla discolor Bunge (PDB) is a plant of the Rosaceae family. A wide range of pharmacological activities, such as antibacterial, hypoglycemic, and others, can be achieved through its chemical components, which include terpenes, steroids, polyphenols, and flavonoids. However, the effects of the herb on the heart are unclear.
Objectives
To explore the effect of the main components of PDB on myocardial cell membranes.
Materials and Methods
We investigated the role of different concentrations of PDB in cardiomyocytes by evaluating myocardial electrical activity. We used high-performance liquid chromatography to analyze the components. Patch-clamp experiments were conducted to detect the drugs’ impact on the ion channels of cardiomyocytes.
Results
PDB regulated 14 potential metabolites in mice. Interestingly, quercetin exerts certain effects on the heart by acting on the inositol phosphate metabolism pathway. Quercetin is a monomer compound that can enhance the activity of IK1 on the myocardium membrane.
Conclusion
This study showed that monomeric compounds of PDB can significantly enhance IK1 activity and influence ion channels in a concentration-dependent manner.
Introduction
In traditional folk medicine, it is believed that the consumption of cooked pig lung food containing Potentilla discolor Bunge (PDB) roots can potentially alleviate cough symptoms. PDB can be used as a whole grass and boiled with sugar to create a remedy that effectively treats phlegm-induced asthma. The plant is referred to as “Potentilla discolor Bunge” due to its dark green upper leaf surface and gray–white lower surface. PDB is a perennial herbaceous plant in the field of rosacea, characterized by its robust and extensive root system measuring approximately 5–8 centimeters in length. The lower portion is frequently characterized by a thick, spindle-shaped, or cone-like structure and contains substantial starch. It encompasses an array of chemical constituents, predominantly terpenes, steroids, polyphenols, and other flavonoids. The principal chemical components comprise kaempferol, quercetin, luteolin, and corydalis (Zhang et al., 2010).
According to the records in the “Compendium of Materia Medica,” PDB exhibits a diverse range of pharmacological activities, including heat-clearing, detoxifying effects, dysentery alleviation, hemostasis, antibacterial properties, and so on. An escalating body of research has demonstrated that numerous Chinese herbal formulations incorporating the aforementioned herb exhibit advantageous properties in addressing metabolic disorders, inflammation, and related conditions. Studies have shown that PDB can reduce blood glucose, improve insulin resistance (IR), and protect pancreatic β-cells in type 2 diabetes (Wang et al., 2013). Luteolin, a flavonoid present in plants, exerts inhibitory effects on fat synthesis by targeting the LXR-SREBP-1c signaling pathway (Li et al., 2023). Additionally, it activates the Sirt1/AMPK pathway, thereby promoting oxidation and suppressing adipogenesis (Zhang et al., 2010). The natural form of glycosides contains quercetin, which is one of the most abundant flavonoids (Singh et al., 2021). Quercetin decreases fatty accumulation in the liver by controlling gene expression for lipid metabolism, lipid peroxidation triggered by cytochrome P450-2E1, and its associated lipid toxicity (Hosseini et al., 2021), thereby reducing IR and nonalcoholic fatty liver disease (NAFLD) activity scores (Ji et al., 2022). However, the role of flavonoids in heart disease remains unclear. Therefore, the objective of this investigation was to investigate the effects of chemical components in the PDB on the heart and to confirm the impacts of different flavonoid concentrations on ion channels in the cardiac membrane during isolated conditions.
The inward rectifying potassium channel (IK1) is a special potassium channel mainly found in cardiomyocytes and plays a role in establishing the resting potential of ventricular muscle (Hibino et al., 2010). Inward rectification of potassium channels refers to the phenomenon that the permeability of potassium channels decreases with the change of depolarization and increases with the hyperpolarization of the membrane. In other words, the more depolarized potassium ions in the membrane, the less potassium ions will flow out, and the more hyperpolarized potassium ions will flow out. This property is called inward rectification. The channel is a voltage-dependent mechanical channel whose permeability is only regulated by voltage rather than a voltage-gated channel (Algalarrondo & Nattel, 2016). Therefore, the primary focus of this study was to investigate the impact of PDB extraction on the inward-rectifying potassium channel of myocardial membranes.
Materials and Methods
Reagent
Na2ATP, collagenase II, HEPES, and KCl were all purchased from Sigma Company. Quercetin was provided by Chengdu Pufei De Biotech Co., Ltd. (Chengdu, China). Quercetin was mixed with normal saline and dimethyl sulfoxide (DMSO) (Sigma–Aldrich, MO, USA) and was dissolved for animal injections. The final medium contained less than 0.1% DMSO.
Primary Culture of Cardiomyocytes
Kunming mice (1–3 days old) were purchased from the Experimental Animal Center of the Second Affiliated Hospital of Harbin Medical University (Harbin, China). In the super clean platform, the chest of suckling mice was opened to expose the heart. The heart was removed with curved tweezers and placed in precooled DMEM. Next, the heart tissue was cut up, transferred to a 15 mL centrifuge tube, and treated with the preheated trypsin digestion solution of two times the volume of the heart. Then, under the condition of a constant temperature 37℃, the centrifuge tube was vibrated at an appropriate speed. When the trypsin digestion solution was slightly turbid, the supernatant was transferred to high-glucose DMEM containing 10% serum to terminate the digestion. The trypsin digestion solution was added to the remaining tissue blocks, and the above operations were repeated until most of the heart tissue blocks were digested. The digested cell suspension was filtered through a 200-mesh copper filter screen. The filtrate was collected and centrifuged at 1,500 rpm for 5 minutes. The supernatant was discarded, high-glucose DMEM with 10% serum was used to precipitate the cells at the bottom of the centrifuge tube, and 100 U/mL penicillin and streptomycin were added. Subsequently, the cells were uniformly distributed throughout the culture bottle and cultivated in an incubator with 5% CO2 at a consistent 37°C temperature and humidity level. Following a 1.5-hour incubation period in which cardiomyocytes were hung in the culture media, cardiac fibroblasts were attached to the bottom of the culture bottle. The culture plate was equally distributed after the culture bottle was carefully taken out and its bottom cautiously aspirated. The supernatant suspension was then collected, diluted, and distributed with new culture solution. Following 48 hours, the cardiomyocytes attached themselves to the wall, allowing for the microscopic observation of the cell shape and density, as well as the conduct of additional tests.
Quadrupole Ion Trap (Q TRAP) LC/MS/MS
When coupled to the UHPLC via the ESI interface (Applied Biosystems, Foster City, CA, USA), the AB SCIEX 4000 Q TRAP™ composite triple quadrupole/linear ion trap tandem mass spectrometer (SCIEX, Framingham, MA, USA) was used in the positive ion mode. The column effluent was separated using a zero dead. About half of the flow is fed into the mass spectrometer through the volume “T” link. The mass spectrometer had the following interface and parameters: Spray capillary voltage of 5.5 kW, dry gas pressure of 40 psi, curtain gas pressure of 10 psi, DP of 70 V, EP of 0 V, and CE of 45 V; dry gas temperature of 600°C; and nebulizer pressure of 40 psi. The Analyst (SCIEX) was used for the acquisition and processing of all data. Using O-TRAPLC/MS/MS, the mass spectrum fragment ions of MS and MS3 could be obtained.
Electrode Control
On a drawing instrument, a two-step Faraday microelectrode was created and polished (Narishige company, Japan). K-glutamate 130, KCl 15, NaCl 5, MgATP 5, MgCl2 1, EGTA 5, CaCl2 1, and HEPES 10 (mmol/L) were all present in the pipette solution, which had been pH-adjusted to 7.2 with KOH.
Experimental Instruments
Patch-clamp instrument: MultiClamp P700B, Axon; digital-to-analog converter, DigiData 1440A, Axon; inverted microscope, IX51, Olympus; 3D manipulator MP-225, Axon; microelectrode drawing Instrument P97, Sutter Instrument.
Patch-Clamp Technology
One single cell’s ion current was recorded using the whole-cell patch-clamp method. The microelectrode was made by a two-step drawing instrument. After filling the electrode’s interior with solution, the microelectrode’s resistance ranged from 3 to 8 mΩ. DigiData 1440A digital-to-analog converter and a MultiClamp 700B patch-clamp amplifier are used to gather, store, and analyze the current signal. The cell suspension was placed on the inverted microscope platform in a 1 mL bath. After cell attachment, the extracellular sodium channel current solution was perfused at a flow rate of 1 mL/min. Rod cells with similar sizes and clear textures were chosen for the experiments. After the high-resistance seal is formed, the membrane capacitor, series resistance, and leakage current are compensated. After equilibration at 5 MN, the membrane current was recorded after the exchange between the intracellular solution and the extracellular solution. The magnitude of the membrane current is expressed by the current density, that is, the membrane current per unit membrane capacitance.
Data Processing
The least significant difference (LSD) test and one-way analysis of variance (ANOVA) were performed using the experimental data, which are represented by the software X ± SPSS.
Results
Screening for Potential Biomarkers
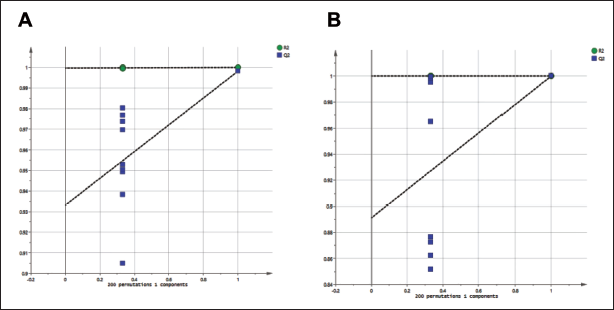
To explain the regulatory effect of PDB extracts on the metabolites of mice, principal component analysis (PCA) was used, and orthogonal partial least squares discriminant analysis (OPLS-DA) methods were used to analyze the differences in metabolism between the samples. The similarity of the data was classified. To verify that there were variations in sample segments between the administration group and the control group, PCA was employed. This indicates that metabolic changes took place in the mice. Following curing, the treatment group’s distribution of sunshine products showed a pattern that was essentially different from the control groups, but it also showed signs of convergence with the control group’s distribution (Figure 1). Using OPLS-DA, significant variations in the sample distribution across the three groups were found. Additionally, a substantial separation between the control and PDB delivery groups was found along the axis by the orthogonal component score value in the orthogonal signal correction treatment (Figure 2). This implies that the mice’s regular physiological metabolic process was disturbed and that the PDB extract greatly affected the mice’s ability to regulate their metabolism. In addition, paired array tests were performed on three groups to validate the effectiveness of orthogonal partial least squares with orthogonalized attributes (OPLS-OA). Furthermore, we found that there were no fitting phenomena, good differential stability, and high predictive reliability in the OPLS-DA model developed in this experiment. The result shows that the intercept in R2 was smaller than the initial value, and the intercept in Q2 was likewise negative on the vertical axis (Figure 3).
Mouse Metabolites Controlled by Potentilla discolor Bunge (PDB) Extract. Mice Stool Sample Principal Component Analysis (PCA) Score Plots: (A) Control vs +PDB, Positive Ion Flow. (B) Negative Ion Flow, Control vs +PDB, n = 10.
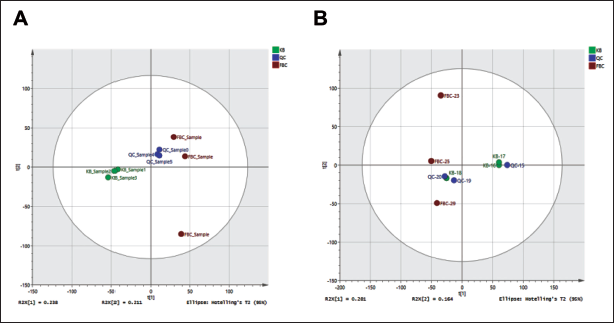
Potentilla discolor Bunge (PDB) Extract Controlled the Mice’s Metabolite Levels. Score Plots of Mice Stool Samples using Orthogonal Partial Least Squares Discriminant Analysis (OPLS-DA): (A) KB vs. +PDB, Positive Ion Flow. (B) Negative Ion Flow, KB against +PDB, n = 10.
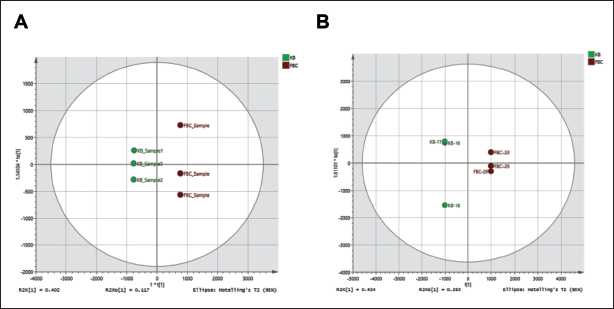
The Number of Divergent Metabolites in Mice was Induced by Potentilla discolor Bunge (PDB) Extract. Diagram showing the Displacement Test Result for the Orthogonal Partial Least Squares Discriminant Analysis (OPLS-DA) Model, (A) Control vs +PDB, Positive Ion Flow. (B) Negative Ion Flow, Control vs +PDB, n = 10.
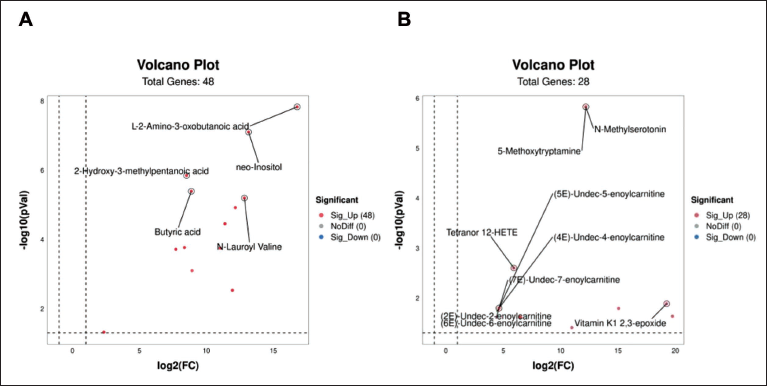
The differences in the endogenous metabolite content in mice before and after PDB therapy were then graphically depicted using heat maps, with the aim of identifying the metabolites that have specific effects on mice (Figures 4 and 5). Furthermore, biomarkers for controlling physiological reactions in vivo in mice were found using S-dot plots. Every dot on the chart denotes a variable. The further the variable is from the origin, the larger the VIP value, which denotes a more significant contribution to the difference between feet (Figure 6A and B). From mouse blood samples, five distinct compounds were found in the positive ion stream: l-2-amino-3-oxobutanoic acid, 2-hydroxy-3-methylpentanoic acid, neo-inositol, butyric acid, and N-lauroyl-valine (Figure 4A). In the negative ion stream, nine different metabolites were detected: 5-methoxytryptamine, N-methylserotonin, (5E)-undec-5-enoylcarnitine, tetranor 12-HETE, (4E)-undec-4-enoylcarnitine, (7E)-undec-enoylcarnitine, (2E)-undec-2-enoylcarnitine, (6E)-undec-6-enoylcarnitine, and vitamin K1 2,3-epoxide (Figure 4B). According to the findings, as opposed to the control group, the blood levels of
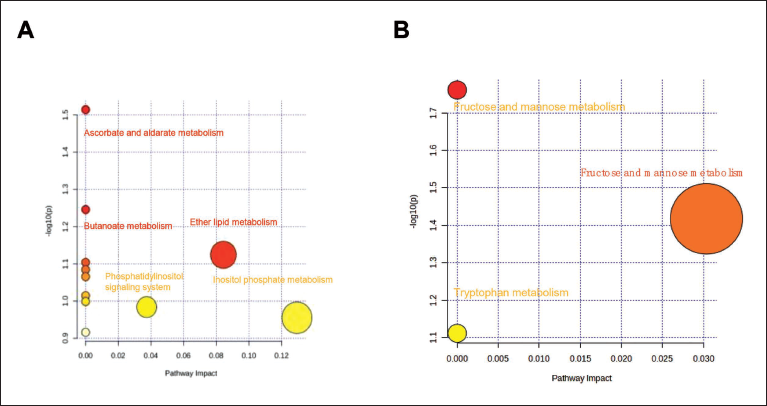
Regulated Metabolic Pathways Extracted from the Potentilla discolor Bunge (PDB). (A) The Positive Ion Flow’s Metabolic Route. (B) The Negative Ion Flow’s Metabolic Route.
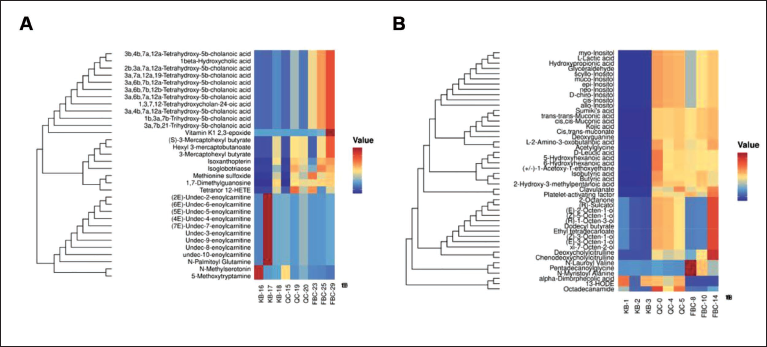
Potentilla discolor Bunge (PDB) Controls the Mice’s Endogenous Metabolites. Heat Map of the Positive Ion Flow’s Metabolites (A). (B) Metabolites’ Heat Map in the Flow of Negative Ions.
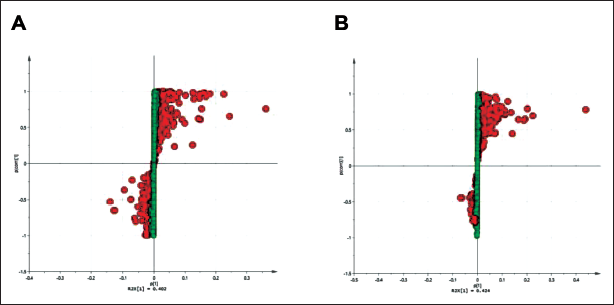
Potentilla discolor Bunge (PDB) Extract Increased the Number of Distinct Metabolites in Mice. (A) Control vs +PDB S-plot in the Positive Ion Flow. (B) Control vs +PDB S-plot in the Negative Ion Flow.
Metabolic Pathway Analysis
To reveal the modulatory effects of the PDB extract on metabolic pathways during cardiac function regulation, the pathways of various metabolites in all samples were evaluated using MetaboAnalyst 5.0.
We found that PDB can regulate the metabolism of ascorbic acid, aldose, butyric acid, lipid, β-alanine, pyruvate, propionic acid, fructose, mannose, galactose, Inositol phosphate, glycine, serine, threonine, and ubiquinone, and regulate glycolysis/gluconeogenesis, the phosphatidylinositol signaling system, and other terpenoid quinone biosynthesis (Figure 7A and B). Among the above 14 metabolic pathways, the inositol phosphate metabolism pathway is related to the regulation of cardiac function. Inositol phosphate can affect a number of physiological processes, including cell division, proliferation, and death, and is an important cell signaling molecule. Inositol phosphate metabolism is an important metabolic process in the human body, and it plays a variety of physiological functions in the cell. The prevention and treatment of numerous diseases require a comprehensive understanding of the mechanism and physiological function of inositol phosphate metabolism. Therefore, we speculate that the PDB regulates cardiac function by acting on the inositol phosphate metabolism pathway, which provides new evidence for clinical research on the new mechanism of regulating cardiac function and improving metabolism.
(A) Metabolite Volcano Map in the Positive Ion Flow. (B) Metabolite Volcano Map in the Negative Ion Flux.
Effects of Quercetin on IK1 Channel of Mouse Cardiomyocytes
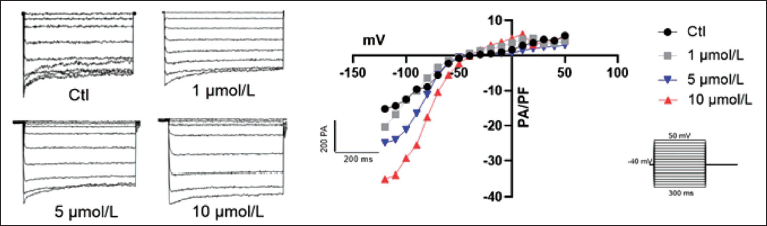
First, we studied the effect of quercetin on the IK1 channel of the myocardium. Quercetin was used to treat primary cardiomyocytes at concentrations of 1 µM/L, 5 µM/L, and 10 µM/L in order to see how it affected the cardiac potassium channel. The findings indicated that quercetin did not significantly affect the myocardial IK1 channel at 1 µM/L, but it might raise the channel’s current at 10 µM/L, which could result in normal atrial and ventricular fibrillation and electrocardiographic interference (Figure 8). In conclusion, the influence of quercetin on ion channels of the myocardial membrane is mainly concentrated on the potassium ions channel.
Quercetin’s Impact on IK1 Channels in the Heart.
Discussion
Up to now, the biological activity of PDB has been fully studied; PDB contains a variety of active compounds, including flavonoids, terpenoids, organic acids, and more. Flavonoids are a key active compound in PDB, with distinct chemical structure subtypes and diverse pharmacological and physiological functions (Chatree et al., 2020). About 20% of the flavonoid content is present in PDB. Conjugated glycosides and free forms are the main forms of flavonoids (Li et al., 2013). More and more studies have shown that PDB has anti-inflammatory, antioxidation (Yang et al., 2019), and reduces oxidative stress, leading to the enhancement of lipid metabolism and IR (Zang et al., 2015). Zhu et al. found that the PDB improves nonalcoholic steatohepatitis by inhibiting lipid deposition through Sirt1/AMPK signaling pathway (Sagawa et al., 2015). Interestingly, studies have shown that quercetin, the PDB extract, is found in glycosides, such as quercetin-3-O-β-rutin or quercetin-3-O-β-glucoside, in greater amounts than any other flavonoids naturally. Quercetin therapy can lower hepatic lipid accumulation by altering the expression of lipid metabolism genes, cytochrome P450-2E1-dependent lipid peroxidation, and result in a decrease in lipotoxicity and NAFLD activity scores (Phuwamongkolwiwat et al., 2014). Quercetin-3-O-β-glucoside has the ability to stimulate AKT activation in the gastrocnemius muscle, which is the key muscle that affects overall insulin sensitivity. Akt-induced insulin signaling pathway activation may result in a drop in plasma glucose concentration and IR. Uncertainty around the extract of PDB’s potential impact on cardiotoxicity belies its apparent anti-inflammatory properties. So, in clinical applications, it is very important to understand its harm to the heart in order to avoid untreatable side effects, especially cardiovascular disease. The metabolomics data, prospective biomarkers, and metabolic pathways were all analyzed and processed in this study using SIMCA software. The activity of potassium channels in the myocardial cell membrane was studied using the patch-clamp technique, which provided effective evidence for the cardiotoxicity of the main component of PDB extract.
In general, arrhythmia is usually a complication of spontaneous vascular disease, such as myocardial infarction, heart failure, diabetes, atherosclerosis, and so on. Abnormal electrophysiological activity of the myocardial membrane is the main factor leading to arrhythmia. Ion channels on the cell membrane are crucial for preserving cardiomyocytes’ regular electrical activity. However, when the external environment changes, the ion channel structure on the cell membrane changes, resulting in abnormal function and inducing various cardiovascular diseases. Several investigations have demonstrated that IK1 is primarily responsible for cardiac excitation. The upregulation of the IK1 current can lead to arrhythmia by shortening the duration of the action potential due to the accelerated inflow of potassium current. It is well known that when we use anti-inflammatory or antitumor drug treatment, it hurts the electrophysiological activity of the heart, which can cause arrhythmia. Thus, the patch-clamp technique is employed to investigate how drugs affect the activity of membrane ion channels, which provides a theoretical foundation for the practical application of drugs.
Our main focus was on studying the influence of PDB on the cardiac ion channel IK1. Patch-clamp results showed that increasing IK1 current under the treatment condition of 10 µm/L indicated that PDB exhibited the ability to dose-dependently induce aberrant cardiac cell electrophysiological activity.
In summary, our work proved that PDB has certain cardiotoxicity, and the toxic effect is dose-dependent. Although efforts have been made to confirm the cardiotoxic effects of quercetin monomer compounds, the exact mechanism remains unclear.
Conclusion
This study demonstrated the regulating impact of PDB on mouse metabolic function. Metabolomics studies have shown that PDB can affect the phosphatidylinositol metabolic pathway by affecting the content of metabolites such as
Abbreviations
DMSO: Dimethyl sulfoxide; IR: Insulin resistance; NAFLD: Nonalcoholic fatty liver disease; PCA: Principal component analysis; PDB: Potentilla discolor Bunge.
Author Contributions
G.L. and Q.W. had the conceptualization and resources. W.Z. and Y.C. did the project administration of the article. M.X., Z.C., and S.G. extracted the total flavonoids of PDB. Y.C., M.X., Z.C., and J.L. were responsible for feeding, gavage, and orbital blood sampling of SD rats. W.Z. and J.L. characterized the metabolites of PDB. The patch-clamp experiment of the quercetin monomer compound on the potassium channel of primary cardiomyocytes was conducted by X.L. and W.Z., and Y.C., W.Z., and L.L. did the formal analysis, methodology, and wrote the full article. G.L. gave critical supervision on the article.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
The use of animals in this study and the experimental process is strictly in accordance with the National Institutes of Health laboratory animal care and use guidelines and has been approved by the Ethics Committee of Harbin Medical University.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by financial support from the Heilongjiang Provincial Health Commission 2020 under grant no. 2020-299 and the National Natural Science Foundation of Heilongjiang Province-Outstanding Youth Project under grant no. YQ2020H008.
