Abstract
Background
Hypertension is a prevalent chronic disorder that affects approximately 1.28 billion adults worldwide. In the use of amlodipine, though it is a cost-effective antihypertensive agent, it causes side effect that may lead to discontinuation of the medication.
Objectives
To examine the effect of the combination of Elettaria cardamomum plus a low dose of amlodipine on Nω-nitro-l-arginine methyl ester (L-NAME)-induced hypertensive rat model.
Materials and Methods
Thirty adult male Wistar rats were randomized into five groups of six rats, respectively. The standard control group received only normal saline (first group). The other four groups received L-NAME (40 mg/kg/day, orally) for 21 days as follows: Group 2 served as L-NAME group only; Group 3 was given E. cardamomum (400 mg/kg/day, orally); Group 4 received amlodipine (5 mg/kg/day, orally); Group 5 received E. cardamomum (400 mg/kg/day, orally) + a low dose of amlodipine (2.5 mg/kg/day, orally) and served as the combination treatment group. Twenty-four hours after the last treatment, blood pressure was measured, then blood samples were collected, rats were sacrificed, and hearts and kidneys were harvested. Serum levels of lipids, liver enzymes, urea, and creatinine were determined.
Results
L-NAME administration induced a notable increase in systolic, diastolic, and mean arterial blood pressure, with significant changes in biochemical parameters. However, administration of E. cardamomum alone or in combination with a low dose of amlodipine showed a better reduction in systolic and diastolic blood pressure than amlodipine alone. Consequently, effects on liver enzymes, lipid profile, and cardiorenal pathology were significantly improved with combination treatment compared with amlodipine alone.
Conclusion
The present study has shown that the E. cardamomum–low-dose amlodipine combination could be beneficial in lowering blood pressure without the attendant adverse effects associated with the normal dose of amlodipine alone. This could be due to covasorelaxant effects.
Introduction
Hypertension is among the most prevalent chronic disorders that contribute to serious health problems, such as cardiovascular diseases (CVDs) and diabetes mellitus (DM) (Félétou & Vanhoutte, 2006; Kamide et al., 1996). Recently, it was reported that hypertension can cause premature deaths and affect approximately 1.28 billion adults worldwide (World Health Organization, 2023). Several studies have shown that hypertension results in target organ damage (TOD) (Li et al., 2017), which causes complications in the functions of the vital organs, including the brain, heart, eye, kidney, and liver. Additionally, a large body of evidence has documented that lipid metabolic disorders can be induced by hypertension (Lyu et al., 2016; Zhu et al., 2016).
At early stages, hypertension is managed with lifestyle modifications, and if not controlled, anti-hypertensive medications such as thiazide diuretics, angiotensin-converting enzyme inhibitors (ACEIs), angiotensin receptor blockers (ARBs), and calcium channel blockers (CCBs) will be prescribed (Unger et al., 2020).
In the case of amlodipine use, though it is a cost-effective antihypertensive agent, its side effect profile may cause discontinuation of the medication. Clinical evidence has shown that amlodipine could cause side effects like palpitations, dizziness, and flushing (Wang et al., 2023). Reports have also shown amlodipine-induced hyponatremia among patients taking 10 mg daily, and it could potentially lead to brainstem herniation, affect respiration, and cause death (Tun et al., 2022). In addition, amlodipine-induced pedal edema is a recognized adverse effect, where patients are usually advised to use half of the maximum dose recommended (Skovbjerg et al., 2023). Furthermore, it is also documented that gingival hypertrophy is induced by amlodipine among patients taking the maximum dose (Yolcu & Aydogdu, 2020). Therefore, a reduced dose of amlodipine might see a decrease in these aforementioned side effects.
Therefore, identifying effective antihypertensive treatments with few side effects, either as a reduced single therapy or in combination with existing ones at lower doses, will be a valuable add-on to the current therapeutical management.
Numerous studies have reported the efforts of scientists in exploring alternatives for managing hypertensive patients with agents that are tolerable and have fewer side effects. In this regard, components of natural products have been reported to be ideal candidates that can be combined with other therapeutic agents to enhance drug efficacy by increasing drug sensitivity, achieving therapeutic synergy, and decreasing adverse drug reactions (Li et al., 2019; Sun et al., 2022). Therefore, natural products such as Elettaria cardamomum have been documented to potentiate the pharmacological effects of synthetic drugs such as CCBs (Clement et al., 2007). E. cardamomum is a herbaceous plant that is cultivated in Central Asian countries, specifically in India (Abu-Taweel, 2018). E. cardamomum has been shown to pose different pharmacological effects, such as anti-inflammatory, antioxidant, and fibrinolytic properties (Rajan et al., 2017). These effects are attributed to the presence of phenols, flavonoids, tannins, and alkaloids (Souissi et al., 2020).
Evidence has shown that E. cardamomum contains bioactive components that have been shown to be effective in treating various types of infectious diseases, such as gum and teeth infections, in traditional medicine (Ashokkumar et al., 2020; Das et al., 2012). It is also reported to be effective in the treatment of cardiac diseases and pulmonary congestion and has anti-inflammatory properties (Moulai-Hacene et al., 2020). In addition, Vaidya and Rathod (2014) indicated that the extract of cardamom showed potential in modulating macrophages; hence, its use in lung infections and tuberculosis (Vaidya & Rathod, 2014). According to a recent study, E. cardamomum contains volatile oils identified as α- and β-pinene, methyl eugenol and trans-nerolidol, alpha-phellandrene, limonene, myrcene, p-cymene, linalool, sabinene, terpinolene, terpinene, and cineole (Cornejal et al., 2023).
Due to the fact that many locals use E. cardamomum as a herbal remedy alongside their antihypertensive medications, many studies have investigated the effect of E. cardamomum on hypertension (Bhaswant et al., 2015). In addition, it has been reported that α-terpineol, an active component of E. cardamomum, was able to reduce mean arterial pressure (MAP) by decreasing vascular resistance and restoring enzymatic antioxidants in hypertensive rats (Sabino et al., 2013). This could be in part due to its calcium channel-blocking property (Gilani et al., 2008).
The mechanism of action of E. cardamomum has been reported to be via calcium channel blockade (Khan et al., 2011; Yahyazadeh et al., 2021). Additionally, the reports indicated that it has nitric oxide synthase (NOS)-inducing properties. These effects may be implicated in the antihypertensive and cardioprotective effects of E. cardamomum.
Previous studies have described the essential role of nitric oxide (NO) in attenuating glomerular hypertension that can be caused by NOS inhibition, which helps to maintain the homeostasis of vascular tone (Chen et al., 2019; Seth et al., 2016), to prevent NO biogenesis. Specifically, it occurs because the continuous overload of systemic blood pressure gradually exceeds autoregulatory mechanisms for maintaining adequate pressure of glomerular filtration. As a result, glomerular hypertrophy, inflammation, and albuminuria may progressively develop (Schlondorff, 2008; Stanko et al., 2020). Therefore, it is envisaged that patients with chronic hypertension are at high risk of renal dysfunction as well.
The pharmacological activity of E. cardamomum on hypertension has been assessed in several animal models. One of these models is hypertension induced by Nω-nitro-l-arginine methyl ester (L-NAME), an inhibitor to NO biogenesis through inhibition of NOS. This model has shown that L-NAME-induced hypertension may lead to the disruption of liver function, resulting in elevated liver enzymes, lipid profile changes, and renal injury (Angele et al., 2000; Bilanda et al., 2017; Clemens, 1999).
However, the effect of using E. cardamomum concomitantly with a low dose of amlodipine in L-NAME hypertensive rats has not been investigated, since both appear to be vasorelaxant. Hence, the objective of the current research is to evaluate the interaction between E. cardamomum (cardamom) and a low dose of amlodipine in L-NAME-induced hypertension in rats.
Materials and Methods
Chemicals
L-NAME and amlodipine besylate were obtained from Sigma–Aldrich (Darmstadt, Germany), while lipid profile analysis kits were purchased from Labtest Diagnostica (Minas Gerais, Brazil). Other chemicals of analytical grade were procured from standard commercial suppliers.
Plant Material and Crude Extraction
E. cardamomum was purchased from a local store in Al-Ahsa, Eastern Region, Saudi Arabia. The whole plant material was expressed to air-drying according to universal standard herbarium procedures. The identification of the plant was kindly done by Dr. Hany Ezzat Khalil, Professor of Pharmacognosy in the Department of Pharmaceutical Sciences, College of Clinical Pharmacy, King Faisal University, Al Ahsa, Eastern Region, Saudi Arabia. A voucher specimen was preserved in the Department of Pharmaceutical Sciences, College of Clinical Pharmacy, King Faisal University. The whole seeds of E. cardamomum were ground to a fine powder, then 220 g of powder was solubilized in 500 mL of distilled water and left to stand for 1–2 hours. Later, it was filtered using a sieve cloth. The resultant solution was connected to an evaporator pump. A yield of 0.277% was obtained, culminating in 55.4 mg extract/g weight.
Experimental Animals
Thirty male Wistar rats weighing between 170 and 220 g (8–10 weeks) were included (six rats/group). All rats were kept at room temperature and had a 12-hour light–dark cycle. They had free access to a regular chow diet and water. Animal care and experimental procedures in this study design were carried out in accordance with the approved guidelines of the Research Ethics Committee (protocol ID: KFU-REC-2023-FEB-ETHICS554) at King Faisal University, Saudi Arabia.
Experimental Design
One week after acclimatization, rats were randomized into two main groups. Six animals in the normal control group received distilled water (vehicle); however, 24 rats in the other group were administered L-NAME (40 mg/kg/day, orally) for 3 weeks (Abdelrahman et al., 2023; Adaramoye et al., 2012; Adedapo et al., 2020; Bin Jardan et al., 2021). After three weeks, the rats in the latter group were randomly divided into four subgroups: (a) The L-NAME group, the hypertension control group; (b) the E. cardamomum group, which received E. cardamomum (400 mg/kg/day, orally) according to modified methods of Kanthlal et al. (2020) and Shahidullah et al. (2017); (c) the CCB group, which administered a regular dose of amlodipine (5 mg/kg, orally); and (d) the combination group, which received a low dose of amlodipine (2.5 mg/day, orally) and E. cardamomum (400 mg/kg/day, orally). Treatment duration was 3 weeks after the induction of hypertension in rats by L-NAME.
Measurements of Blood Pressure
Measurements of rats’ blood pressure were obtained weekly by CODA, a High Throughput Noninvasive Blood Pressure System (Kent Scientific, Torrington, CT, USA). This system records tail blood pressure by means of volume pressure. Each rat was kept in a clear holder on a warming pad with the tail extended out, and both cuffs (an occlusion and a volume pressure-recording) were put close to the base of the tail. This system automatically measures the values for the systolic blood pressure (SBP), diastolic blood pressure (DBP), and MAP. Readings for 15 cycles were taken from each rat with the exclusion of extreme values. All measurements were acquired under similar circumstances at the same time of day to reduce stress-induced variations in blood pressure.
Blood and Tissue Sampling
On the last day of the experiment, animals were sacrificed by cervical dislocation after they had been anesthetized by urethane for blood sampling and tissue collection. Blood was withdrawn via retroorbital puncture, and samples of blood were immediately centrifuged at 3,600 rpm to separate the serum. The serum was stored at –80οC for further investigation (Valo et al., 2022).
Measurement of Liver and Kidney Biomarkers
The serum levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), and the estimation of levels of total proteins were done by using an automated chemistry analyzer (Merck, Wiesbaden, Germany) according to the manufacturer’s specifications.
Measurement of Serum Lipid Profile
Labtest kits, including cholesterol liquiform, triglycerides (TRG) liquiform, high-density lipoprotein (HDL) liquiform, and low-density lipoprotein (LDL) liquiform from Labtest Diagnostica (Minas Gerais, Brazil) and an automated chemistry analyzer (Merck, Wiesbaden, Germany) were utilized to measure lipid profiles.
Urine Collection
Experimental animals were kept in metabolic cages, from where their urine samples were collected and kept in urinary bottles at –85°C until assayed. However, an automated chemistry analyzer (Merck, Wiesbaden, Germany) was utilized to measure the markers of kidney damage, such as creatinine, urea, and uric acid, according to the manufacturer’s specifications.
Histopathological Preparation and Analysis
Hearts and kidney tissues of all experimental groups were collected and kept in a 10% buffered formaldehyde solution. After alcohol dehydration, 4 µm-thick sections were cut and stained with hematoxylin and eosin (H&E) (Merck, Darmstadt, Germany), and histopathological analysis of the prepared slides was performed as previously described (Yeşilada et al., 1997). The stained slides were evaluated using a light microscope (Olympus, Tokyo, Japan). Using a high-resolution, color digital camera (Olympus, Tokyo, Japan) adapted to the light microscope and connected to a computer, photomicrographs for the respective tissues were digitally obtained.
Statistical Analysis
Data are presented as mean ± standard deviation (SD). Statistical analysis was carried out with GraphPad Prism version 10.2 (San Diego, CA, USA). Data analysis was carried out using two-way analysis of variance (ANOVA), comparing statistical differences between control and treatment groups, whereas Tukey’s multiple comparisons test was used to measure statistical significance between intergroup treatments. The level of significance was set at <5%. Overall, statistical differences between the control were denoted with *, while the differences between the untreated hypertensive group and treatment groups were denoted with #.
Results
Effect on Blood Pressure
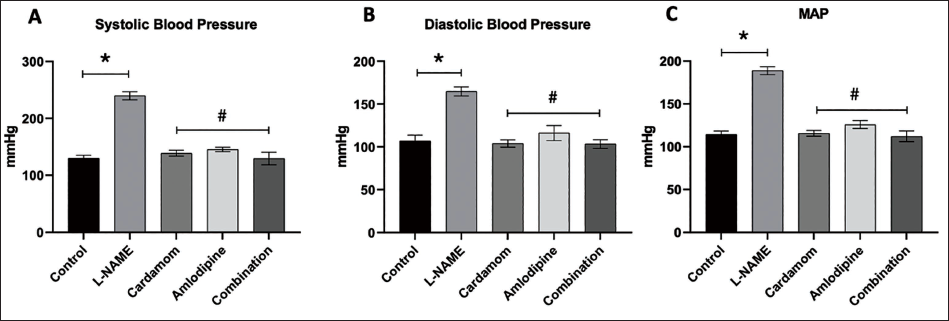
Figure 1 shows changes in blood pressure measurement in normal control, L-NAME, E. cardamomum, amlodipine, and combination (E. cardamomum and amlodipine) groups. L-NAME administration progressively increased SBP, DBP, and MAP by 84, 54, and 65%, respectively. However, in the E. cardamomum-treated animals, all three blood pressure parameters were significantly decreased compared to the L-NAME group (p < 0.05). Similar results were obtained with the amlodipine and combination groups. Thus, all treatment groups produced a lowering effect on SBP, DBP, and MAP in L-NAME-induced hypertensive animals. However, there were remarkable differences in the lowering effects between the treatment groups.
Changes in Systolic Blood Pressure (SBP) and Diastolic Blood Pressure (DBP) and Mean Arterial Pressure (MAP) in Nω-Nitro-l-Arginine Methyl Ester (L-NAME) Treated Rats Treated with Elettaria cardamomum and/or Amlodipine. (A) SBP; (B) DBP; (C) MAP. Data are Expressed as the Mean ± Standard Deviation (SD), n = 6. *Represents a Significant Statistical Difference between the Control and the Hypertensive Control Groups, Whereas # Represents a Significant Statistical Difference between the Hypertensive Control and All Treatment Groups.
Effect on Liver Function
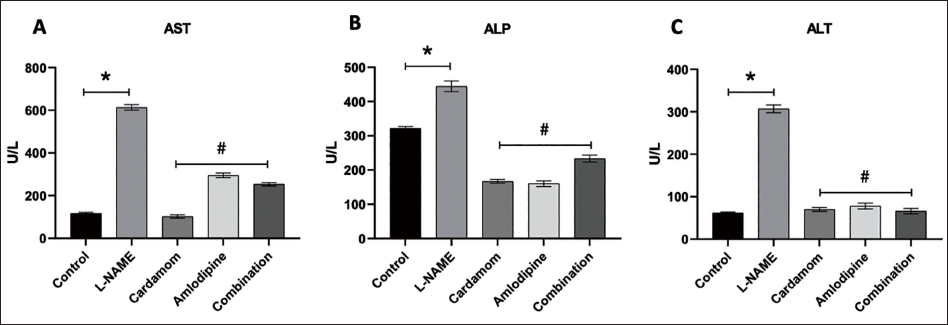
The effects of L-NAME, E. cardamomum, amlodipine, and combination (E. cardamomum + amlodipine) on the levels of serum AST, ALP, and ALT are illustrated in Figure 2. Figure 2A shows that the treatment groups had significantly (p < 0.05) lower AST levels compared to the hypertensive control group (L-NAME treatment). Moreover, a single treatment with E. cardamomum had the highest lowering effect compared to other treatment groups. Regarding the ALP enzyme, a highly significant decrease was noted in rats treated with E. cardamomum or amlodipine compared with the hypertensive control group (p < 0.05) as shown in Figure 2B. Surprisingly, both E. cardamomum and amlodipine-treated groups had a significant decrease in the ALP enzyme level compared with the combination group (p < 0.05). No significant difference was found in ALP and ALT levels between E. cardamomum and amlodipine. In Figure 2C, all treatment groups produced a significant (p < 0.05) decrease in the ALT levels compared to the L-NAME group. However, the combination group had the highest decrease compared to all the other treatment groups.
Effect of Elettaria cardamomum and/or Amlodipine on the Hepatic Levels of Aspartate Aminotransferase (AST), Alkaline Phosphatase (ALP), and Alanine Aminotransferase (ALT) in Nω-Nitro-l-Arginine Methyl Ester (L-NAME)-induced Hypertension in Rats. Data are Expressed as the Mean ± Standard Deviation (SD), n = 6. *Represents a Significant Difference between the Control and the Hypertensive Control Groups, Whereas # Represents a Significant Difference between the Hypertensive Control and All the Treatment Groups.
Effect on Lipid Profile
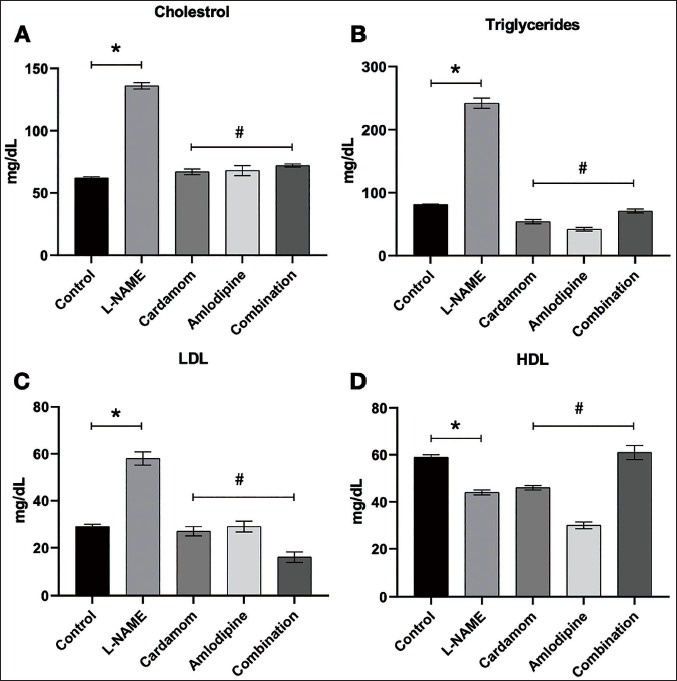
In Figure 3A, the results indicate that the treatment groups had significantly lower serum cholesterol levels (p < 0.05). However, the single treatment of E. cardamomum or amlodipine showed a superior lowering effect on cholesterol levels than the combination treatment. In Figure 3B, data showed that all the treatment groups had effectively reduced the triglyceride levels to a point that was better than both the control and L-NAME groups (p < 0.05), with superiority to the amlodipine in reducing the triglyceride levels. However, a different trend was observed in the LDL levels, where the combination group produced a better reduction compared to E. cardamomum and amlodipine-only groups, as shown in Figure 3C (p < 0.05). The HDL results, as presented in Figure 3D, showed that the combination group had a significant (p < 0.05) increase in HDL levels compared to other treatment groups and controls.
Effect on Kidney Function
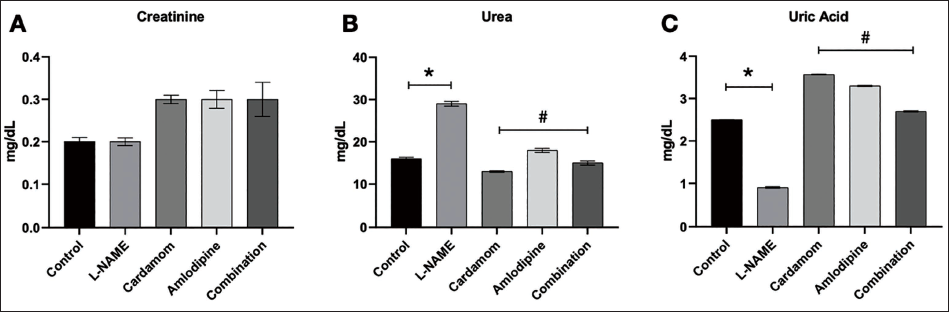
The effects of L-NAME, E. cardamomum, amlodipine, and the combination (amlodipine + E. cardamomum) on the creatinine, urea, and uric acid levels are illustrated in Figure 4. In Figure 4A, results for creatinine levels showed that the treatment and combination groups appeared to increase compared to control and L-NAME groups. This observation could not be explained from our point of view. In Figure 4B, the treatment groups showed significantly (p < 0.05) lowered urea levels. Moreover, the E. cardamomum and combination (amlodipine + E. cardamomum) groups produced a better-lowering effect compared to a single amlodipine treatment. In Figure 4C, L-NAME was found to reduce uric acid excretion. However, E. cardamomum and amlodipine only produced the highest uric acid excretion.
Effect of Elettaria cardamomum and/or Amlodipine on Kidney Function in Nω-Nitro-l-Arginine Methyl Ester (L-NAME)-induced Hypertension in Rats. Data are expressed as the mean ± Standard Deviation (SD), n = 6. *Represents a Significant Difference between the Control Group and the Hypertensive Control Group, Whereas # Represents a Significant Difference between the Hypertensive Control Group and all the Treatment Groups.
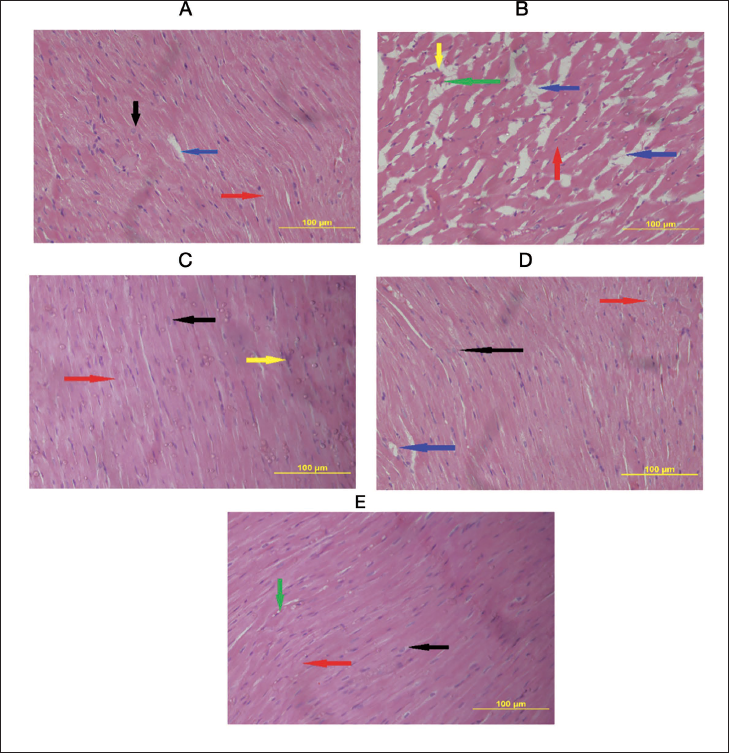
Histopathological Analysis of the Cardiac Tissue
Figure 5 displays photomicrographs of the experimental rats’ heart tissues from the normal control and the different treatment groups with L-NAME, E. cardamomum, amlodipine, and a combination of E. cardamomum plus amlodipine. Rat cardiac sections from the control group (Figure 5A) show a normal histological appearance, typical cardiac myocytes with oval nuclei, and a normal pattern of cross-striation. In contrast, the L-NAME treated group (Figure 5B) showed distorted intercalated discs having enlarged intercellular spaces, filled with extravascular red blood cells. In addition, blood vessels were congested, and cellular infiltrations of neutrophils were observed as well. These results indicated that L-NAME administration produced cardiac injury in the experimental rat heart tissue. Furthermore, treatment with E. cardamomum (Figure 5C) attenuated the cardiac injury seen with L-NAME alone. However, the administration with amlodipine (Figure 5D) showed mild tubular congestion with blood and recovering intercalated discs, although it was better than the L-NAME group. Lastly, the combination of E. cardamomum plus amlodipine (Figure 5E) also attenuated the injury in cardiac cells seen with L-NAME alone. Cardiac tissue (Figure 5C and 5E) showed near-normal cardiac appearance.
Photomicrographs of Cardiac Tissues of Controls and Treated Experimental Animals [Sections Stained with Hematoxylin and Eosin (H&E)]. (A) A Photomicrograph of Control Rats, Showing Normal Cardiac Structure with Oval Nucleus (Black Arrow), Normal Intercalated Disc (Red Arrow), and Perinuclear Vacuolization (Blue Arrow). (B) Histopathological Changes Induced by Nω-Nitro-l-Arginine Methyl Ester (L-NAME) Showing Disordered Intercalated Discs with Fragmented Muscle Fiber (Red Arrow) and Enlarged Intercellular Space (Blue Arrow). Also Observed were Blood-Filled Intercellular Spaces (Indicated with a Green Arrow). Furthermore, Cellular Infiltration of Neutrophils was seen in this Group (Yellow Arrow). (C) Showing Group Treated with Elettaria cardamomum with Near-to-Normal Cardiac Architecture having Normal Nuclei (Black Arrow) and Intercalated Disc (Red Arrow). However, a Few Infiltrated Neutrophils were Observed (Yellow Arrow). (D) Represents the Group Treated with Amlodipine, Showing Recovery in the Intercalated Disc (Red Arrow), Normal Oval Nucleus (Black Arrow), and with (Blue Arrow) Showing Vacuolation. (E) A Photomicrograph of the Group Treated with the Combination of Elettaria cardamomum Plus Amlodipine Displaying Near-to-Normal Cardiac Architecture of Nuclei and Intercalated Muscle Discs (Black and Red Arrow). Also, a Mild Congested Intercellular Space (Green Arrow) (H&E, 400×).
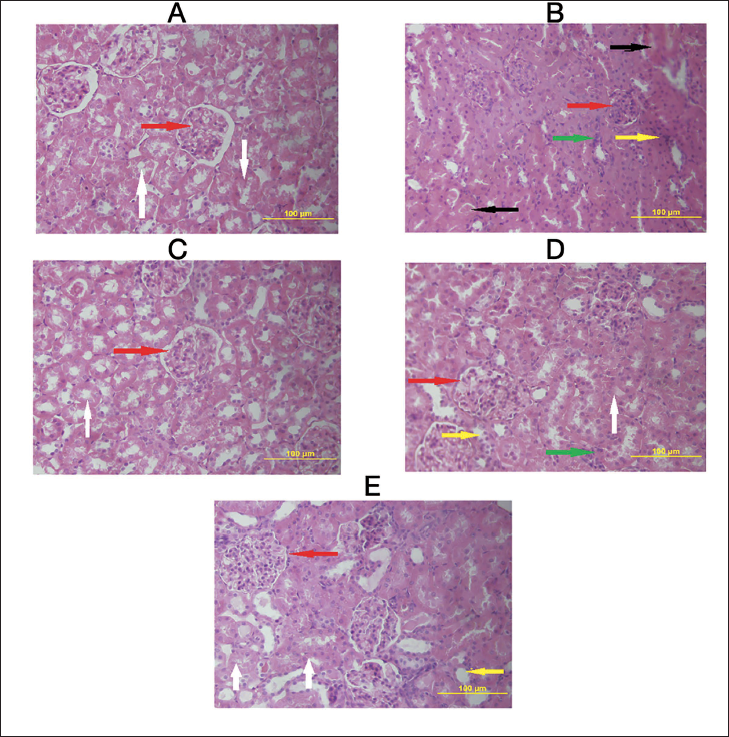
Histopathological Analysis of the Renal Tissue
Figure 6 shows observed histopathological changes in renal tissues due to treatments with L-NAME, E. cardamomum, amlodipine, and a combination of E. cardamomum plus amlodipine. Results showed that the untreated group (Figure 6A) showed no interstitial inflammations and distorted tubulus proximalis, as seen in the group treated with L-NAME (Figure 6B), which had distorted tubulus proximalis that were reduced in numbers. Interstitial inflammations and glomeruli found in this group were significantly reduced in size and number compared with the control. In addition, interstitial hemorrhage with tissue damage alongside leukocyte infiltration was prominent, indicating renal tissue injury. However, the group treated with E. cardamomum (Figure 6C) showed recovery with mild interstitial hemorrhage, few leukocyte infiltrations, and near-to-normal glomeruli and tubules. Treatment with amlodipine (Figure 6D), although it had a near-normal structure compared to the L-NAME group, still had interstitial hemorrhage and tubules congested with blood, indicating some form of damage but not to the extent seen with L-NAME. Lastly, the group treated with the combination of E. cardamomum plus amlodipine (Figure 6E) also showed recovery with mild interstitial hemorrhage, few leukocyte infiltration, and near-to-normal glomeruli and tubules. This indicates that treatment with either E. cardamomum or a combination with amlodipine significantly improved the injury caused by L-NAME.
Photomicrographs of Renal Tissues of Controls and Treated Experimental Animals (Sections Stained with H&E). (A) An Image of Kidney from a Control Rat Showing Normal Architectural Features with Normal Glomeruli and Tubulus Proximalis (Red and White Arrows). (B) Morphological Structure of Rats’ Kidney Tissues Treated With L-NAME Shows Glomerular Atrophy Appearing Diminished in Size (Red Arrow), Tubules with Congested Blood (Black Arrow), Interstitial Congestion (Yellow Arrow), and Leukocyte Infiltration of Cells (Green Arrow). (C) A Photomicrograph shows Near-Normal Glomerulus and Nephrons in the Group Treated with the Elettaria cardamomum. (D) Showing Recovery with Amlodipine Treatment However with Few Infiltrated Neutrophils (Yellow Arrow) and Recovering Tubulus Proximalis (White Arrow). (E) Represents a Photomicrograph of a Rat’s Kidney Treated with a Combination of Elettaria cardamomum Plus Amlodipine, Showing also Near-to-Normal Renal Architecture with Normal Glomeruli and Tubulus Proximalis (Red and White Arrow) However having Few Infiltrated Neutrophils (H&E, 400×).
Discussion
There is the availability of data showing the close relationship between hypertension, liver, heart, and kidney injury (Lyu et al., 2016; Schlondorff, 2008; Stanko et al., 2020; Zhu et al., 2016). It is known that L-NAME-induced NO deficiency in hypertensive rats could induce liver injury and disturbances in serum lipid profiles (Abdel-Rahman et al., 2017; Bilanda et al., 2017; Rajeshwari et al., 2014). In addition, it could also cause renal damage due to the development of glomerulosclerosis, tubulointerstitial fibrosis, and tubular atrophy, which are associated with a deteriorated glomerular filtration rate and increased urinary protein excretion (Hropot et al., 1994; Ikeda et al., 2009; Ndisang & Chibbar, 2015).
There is a wide range of antihypertensive medications that are approved for the management of hypertension. Due to their side effect profile, studies have recently been directed toward developing safer agents from natural products (Pasdaran et al., 2023). For this reason, the effects of different natural products on the treatment of hypertension are being investigated. Agents with proven evidence of antihypertensive benefits, such as coenzyme Q10, fish oil, garlic, vitamin C, and l-arginine, are undergoing further clinical tests (Houston, 2013; Wilburn et al., 2004). Additionally, several studies have reported the beneficial effects of the key components of the essential oil in E. cardamomum, as they revealed its anti-inflammatory, antioxidant, antiangiogenic, and antiapoptotic effects. Also, previous reports have documented the beneficial effect of E. cardamomum (cardamom) in lowering blood pressure in a hypertensive animal model (Silambarasan et al., 2016; Verma et al., 2009). E. cardamomum has been documented to possess smooth muscle relaxant properties as well as the induction of NOS and endothelial NO in tissues (Yahyazadeh et al., 2021).
So, it is expected that the administration of E. cardamomum with another synthetic antihypertensive medication may provide a better effect, such as acting in an additive fashion or synergistically. The current study focused on investigating the efficacy of the combination therapy in comparison with single treatment with amlodipine. It was found that the oral administration of L-NAME at a dose of 40 mg/kg/day for 3 weeks effectively induced a marked elevation of the blood pressure in rats. Furthermore, L-NAME significantly diminished liver function, leading to significant increases in the liver enzymes AST, ALP, and ALT, along with a distorted lipid profile in rats (Abdel-Rahman et al., 2017; Bilanda et al., 2017; Rajeshwari et al., 2014; Seth et al., 2016). These results were expected, as our data are in strong agreement with the previous studies that documented the negative effect of the NO inhibitor L-NAME on liver health (Seth et al., 2016; Silambarasan et al., 2016).
With respect to the L-NAME effect on kidney function, our results revealed an increase in urea production, a reduction in creatinine, and uric acid excretion by L-NAME treatment. These results did not agree with previous reports on L-NAME and renal function alteration (Amann et al., 2001; Dhaun et al., 2006). Besides, it was reported that L-NAME increased the serum uric acid levels, which may be related to the reduction in NO production (Kumar et al., 2000). These reported findings are not consistence with the findings of the present study on our observations of L-NAME effects on renal function. L-NAME showed a significant reduction in serum uric acid levels with no effect on the serum creatinine level. However, L-NAME increased only the serum urea level.
Comparing the single treatments of E. cardamomum or amlodipine, the combination treatment appeared to produce a significant lowering effect on the systolic, diastolic, and mean arterial blood pressure. The study showed that E. cardamomum had a better SBP-lowering effect than amlodipine. This result is consistent with the report of Verma et al., which documented that treatment with E. cardamomum (3 g) significantly decreased the SBP and DBP by 19 and 12 mmHg, respectively (Verma et al., 2009). The effect of the E. cardamomum and amlodipine combination, according to the results obtained, appeared to be either additive or synergistic. As can be seen, the systolic pressure was below the normal control. Moreover, Gilani et al. (2008) noted that E. cardamomum may have a calcium channel-blocking effect, which may describe its mechanism of lowering blood pressure.
Furthermore, it was observed that either single or combination treatment of E. cardamomum significantly lowered the serum levels of the liver enzymes, particularly AST and ALT. However, the combination treatment produced a better modulating effect on ALT levels compared to amlodipine or E. cardamomum alone. In agreement with these data, several studies have reported similar protective effects of E. cardamomum on liver damage, consistent with our findings (Alkhalifah et al., 2022; Kanthlal et al., 2020).
On the effect of E. cardamomum alone and combination treatment on lipid profile, our results showed that serum cholesterol levels and TRG were also reduced, but amlodipine showed a better decreasing effect. However, the combination treatment significantly decreased LDL and significantly increased HDL as well. LDL and HDL have become therapeutic targets, as their interaction could lead to the development of atherosclerosis (Barter, 2005). HDL prevents the oxidation of LDL, which is responsible for forming atheroma. Therefore, the significant increase in HDL caused by the combination of E. cardamomum and amlodipine in this study could be beneficial in hypertension management. However, the effect of E. cardamomum on LDL and HDL was also similar to the effect of the combination, showing a better lowering than amlodipine. These results are consistent with the report of Kanthlal and colleagues, in which administration of the aqueous extract of E. cardamomum significantly lowered the lipid profile serum levels (Bhaswant et al., 2015; Kanthlal et al., 2020).
The high levels of serum urea due to L-NAME treatment significantly decreased after the administration of E. cardamomum or amlodipine, either as single treatment or as a combination. Also, combination treatment was better compared to amlodipine alone. However, the serum creatinine did not display any significant changes in any of the treatment groups; however, uric acid excretions showed that the combination treatment gave the highest excretion compared to all treatment groups. It, therefore, indicates that cardamom can reduce uric acid levels, as reported by Andreucci et al. (2017), and is different from previous reports (Elkomy et al., 2015; Sabu & Kuttan, 2002). However, the present study shows the necessity for further study to explore its use in gout.
In addition, histopathological results for cardiac and renal tissues displayed various stages of tissue damage by L-NAME, which were attenuated by the administration of either E. cardamomum alone or in combination with amlodipine. These data came on the same line as previous studies, which reported that L-NAME administration (40 mg/kg) for 3 weeks induced cardiac and renal hypertrophy in a hypertensive rat model (Bachmann & Mundel, 1994; Eriguchi et al., 2015; Ribeiro et al., 1992; Sung et al., 2013). Treatment with E. cardamomum and amlodipine combination and E. cardamomum alone restored the damaged tissues to near-normal architecture, an indication of the protective effect potential of E. cardamomum.
However, treatment with amlodipine alone reduced cardiac injury but not as much as in the E. cardamomum combination. Also, on the renal tissue, amlodipine only did not completely restore the damage done by L-NAME administration, but it did in combination with E. cardamomum. Consistent with these results, the report of previous studies that supported the effect of E. cardamomum indicated that it has a potential renoprotective effect in addition to its blood pressure-lowering effect in rat hypertensive models (Gilani et al., 2008; Sabino et al., 2013; Verma et al., 2009; Yahyazadeh et al., 2021). This is evidenced by other studies that documented the beneficial antioxidant effect of E. cardamomum on the protection of kidney tissues in different rat models (Al-Johani & Al-Sowayan, 2023; Babaeenezhad et al., 2021; Elkomy et al., 2015).
Conclusion
The present study has confirmed the role of L-NAME in hypertension induction and the attendant consequences of high blood pressure. The findings of this study showed that E. cardamomum mitigated the effects of L-NAME-induced hypertension. However, the use of cardamom plus amlodipine is a novel study based on its use by locals and is indicated here to be additive or synergistic in action as reported here. Accordingly, this combination provided better SBP, DBP, and MAP lowering effects compared to amlodipine or E. cardamomum alone. Regarding its effect on the lipid profile, the combination significantly elevated HDL, which consequently reduced LDL levels in L-NAME-induced hypertension. Furthermore, kidney and cardiac histology were better improved by the administration of the combination of E. cardamomum and amlodipine compared to amlodipine alone. Although the present study could not determine the precise mode of action of the combination, further studies are needed to establish this, given the potential of this combination for drastically lowering L-NAME-induced hypertension in rats.
Study Limitations
To this study, there are some limitations that could elucidate the detailed mechanism of the combination of E. cardamomum extract and amlodipine. According to the literature, both are vascular relaxants. This study did not examine the expression of endothelial nitric oxide synthase (eNOS), which is believed to be responsible for decreased vascular smooth muscle and heart rate. In addition, electrocardiogram (ECG) was not measured to observe the extent of negative inotropic and chronotropic effects on the heart.
Footnotes
Abbreviations
ACEIs: Angiotensin-converting enzyme inhibitors; ALP: Alkaline phosphatase; ALT: Alanine aminotransferase; ANOVA: Analysis of variance; ARBs: Angiotensin receptor blockers; AST: Aspartate aminotransferase; CCBs: Calcium channel blockers; CVDs: Cardiovascular diseases; DBP: Diastolic blood pressure; DM: Diabetes mellitus; ECG: Electrocardiogram; H&E: Hematoxylin and eosin; HDL: High-density lipoprotein; LDL: Low-density lipoprotein; L-NAME: Nω-nitro-l-arginine methyl ester; MAP: Mean arterial pressure; NO: Nitric oxide; NOS: Nitric oxide synthase; SBP: Systolic blood pressure; TOD: Target organ damage; TRG: Triglycerides.
Acknowledgments
This article is derived from a research grant funded by the Research, Development, and Innovation Authority (RDIA)—Kingdom of Saudi Arabia—with Grant number (12884-KFU-2023-KFU-R-2-1-HW-).
Author Contributions
AKA conceived and designed the study, provided research materials, analyzed and interpreted data, and wrote the final draft of the article. DIA conducted research, collected and organized data, and wrote the initial draft of the article. SMA conducted research, collected and organized data, and wrote the initial draft of the article. ZAA conducted research, collected and organized data, and wrote the initial draft of the article. FJA conducted research, collected and organized data, and wrote the initial draft of the article. PME conceived and designed the study, provided research materials, provided logistic support, analyzed and interpreted data, and wrote the final draft of the article. ESG provided research materials, conducted research, provided logistic support, analyzed and interpreted data, and wrote the final draft of the article. All authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Animal care and experimental procedures in this study were carried out in accordance with the approved guidelines of the Research Ethics Committee (protocol ID: KFU-REC-2023-FEB-ETHICS554) at King Faisal University, Saudi Arabia.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia (Grant no. 5856).
