Abstract
Background
Preeclampsia is a pregnancy complication characterized by high blood pressure and signs of damage to another organ system, often the kidneys. Recent studies suggest that endothelial dysfunction and trophoblast apoptosis are involved in the pathogenesis of preeclampsia.
Objectives
To investigate the effects of tanshinone IIA (TIIA) on clinical symptoms, vascular endothelial function, and placental trophoblast apoptosis in preeclampsia rats.
Materials and Methods
Twenty-four pregnant Sprague-Dawley rats, between 8 and 10 weeks old, were randomly divided and allocated into three groups: Control, model, and treatment, with each group consisting of 8 rats. The control group received normal saline injections via the tail vein; the model group received NG-nitro-l-arginine methyl ester (l-NAME) to induce preeclampsia, followed by normal saline injections; and the treatment group received TIIA (10 mg/kg) via tail vein injection after preeclampsia induction with l-NAME. Various parameters were measured, including tail artery systolic pressure, 24-hour proteinuria, offspring and placental weight, levels of endothelial nitric oxide synthase (eNOS), endothelin-1 (ET-1), placental growth factor (PLGF), and soluble FMS-like tyrosine kinase 1 (sFlt-1) in serum, and expression of B-cell lymphoma-2 (Bcl-2) and Bcl-2-related X protein (Bax) in placental trophoblasts.
Results
TIIA treatment led to decreased tail artery systolic pressure and 24-hour urine protein levels, increased body and placental weights of offspring, and favorable changes in serum levels of ET-1, sFlt-1, eNOS, and PLGF. Moreover, TIIA treatment resulted in decreased Bax levels and apoptosis in placental trophoblasts, along with increased Bcl-2 levels.
Conclusion
TIIA can improve the clinical symptoms of preeclampsia in rats induced by l-NAME, alleviate the apoptosis of placental trophoblast cells, and improve vascular endothelial function.
Introduction
Preeclampsia typically develops after the 20th week of pregnancy and is identified by the sudden onset of high blood pressure and protein in the urine in expectant mothers. It may also involve complications affecting the heart, liver, brain, and other organs. It is a common pregnancy syndrome that contributes to maternal and perinatal mortality. Its incidence rate accounts for 3–5% of pregnant women (Chappell et al., 2021). The exact cause of preeclampsia is not completely understood. It is commonly acknowledged that abnormalities in the placenta and resulting maternal issues are important factors in the development and advancement of preeclampsia. In early pregnancy, impaired trophoblast differentiation and excessive apoptosis lead to the failure of maternal spiral artery reconstruction, placental ischemia, placental synthesis, and the release of numerous proinflammatory factors (Rana et al., 2019). In the third trimester of pregnancy, abnormal trophoblast invasion leads to repeated episodes of hypoxia/reperfusion, massive inflammation, and cytokine production (Phoswa & Khaliq, 2021). This process results in vascular endothelial cell dysfunction, characterized by a reduction in placental growth factor (PLGF) (Guerby et al., 2021) and an elevation in soluble FMS-like tyrosine kinase 1 (sFlt-1) (Veisani et al., 2019), ultimately leading to the clinical symptoms associated with preeclampsia. Preeclampsia is classified as a type of blood stasis syndrome in traditional Chinese medicine. Salvia miltiorrhiza, a traditional Chinese medicine, has various effects such as antihypertensive, sedative, lipid-lowering, and anticoagulant effects, as well as enhancing blood flow and eliminating stagnant blood to improve circulation (Li et al., 2018; Lobina et al., 2018; Zhang et al., 2016). Tanshinone IIA (TIIA), the most active lipophilic component from S. miltiorrhiza root extract, has received approval from the National Administration of Traditional Chinese Medicine for managing cardiovascular conditions, such as coronary heart disease and myocardial infarction. Studies have confirmed that TIIA can increase nitric oxide (NO) synthase activity in vascular smooth muscle and maintain NO release to reduce blood pressure. Inhibition of COX-2 expression and decreased activity in a variety of cells; scavenging lipid free radicals and tumor necrosis factor-α (TNF-α); increasing PLGF expression; ameliorating tissue ischemia-reperfusion injury; reducing low-density lipoprotein, antiatherosclerosis; it can inhibit the influx of Ca2+ ions in vascular smooth muscle cells, improve microcirculation, and reduce renal function damage (Zhong et al., 2021). Based on the aforementioned functions, we hypothesized that TIIA may have a specific therapeutic effect on preeclampsia. Therefore, we examined the therapeutic efficacy of TIIA on the clinical manifestations, vascular endothelial cell function, and placental trophoblast apoptosis in rats with preeclampsia by establishing an animal model.
Materials and Methods
Source of Materials
Sprague–Dawley rats aged 8–10 weeks (Beijing Vitong Lihua Laboratory Animal Technology Co., Ltd., Beijing, China) were kept in an environment with a temperature ranging from 18℃ to 28℃ and a relative humidity between 40% and 70%. The female and male rats were housed together in the same cage at a 1:1 ratio. Examining vaginal secretions every morning, the day of sperm discovery was considered the first day of pregnancy. All studies were authorized by the Ethics Committee at Tianjin Central Hospital of Obstetrics and Gynecology (2020KY122).
Methods
Establishment of Animal Models
The control group rats were administered 1 mL of saline via the tail vein on gestation days 10, 15, and 20. The rats in the model group were injected with NG-nitro-
Measured the systolic blood pressure in the caudal artery and quantified the 24-hour urine protein.
On the 20th gestational day, the BP2000 small animal noninvasive blood pressure analysis system (Visitech Systems, USA) was used to measure the systolic pressure of the tail artery. Simultaneously, the rats were kept in standard metabolic cages, and the 24-hour urine from each group was collected.
Collection of Specimens
On the 21st day of pregnancy, 3–4% isoflurane was used to induce anesthesia, 1–2% isoflurane was used to maintain anesthesia, the flow rate was 0.2–0.3 L/min, and a cesarean section was performed. The offspring rats and placental tissues were removed for further analysis. Upon completion of the experiment, the rats were euthanized by injecting potassium chloride into their hearts, and then their bodies were collected and incinerated.
Detected the Function of Vascular Endothelial Cells in Serum Using an Enzyme-linked Immunosorbent Assay
Utilized enzyme-linked immunosorbent assay to detect the levels of endothelin-1 (ET-1), endothelial nitric oxide synthase (eNOS), PLGF, and sFlt-1 in the serum of rats. The kit was obtained from Wuhan Cypei Biotechnology Co., Ltd. (Wuhan, China). The operation was performed following the guidelines outlined in the provided kit, and the experiment was repeated three times.
Isolation of Placental Trophoblast Cells
Primary placental trophoblasts were isolated using modified density gradient centrifugation. The placental tissue was cut into pieces using sterile surgical scissors and was digested with 0.25% trypsin and an equal volume of deoxyribonuclease (DNase) in a water bath at 37°C three times for 10 minutes each time. The cell suspension was obtained and centrifuged for 5 minutes at 1,500 rpm. After discarding the supernatant, the DMEM/F12 medium was resuspended. The Percoll separation solution was added, and the mixture was centrifuged for 30 minutes at 1,500 rpm. The cloudy cell layer appeared at the interface of different concentrations of Percoll solution. The cell layer was sucked, and then it was washed twice with culture medium and antibiotic saline.
The expression levels of Bax/Bcl-2 messenger ribonucleic acid (mRNA) in placental tissues of pregnant rats in the three groups were detected using real-time fluorescence quantitative polymerase chain reaction (PCR).
Total RNA was extracted from placental trophoblast cells using TRIzol reagent, and the NanoDrop 2000 (Thermo Fisher, Waltham, MA, USA) was used to determine the concentration and purity of total RNA. Each sample was taken with 500 ng of RNA and processed utilizing the high-capacity cDNA reverse transcription kit (Thermo Fisher, Waltham, MA, USA) to reverse-transcribe RNA into cDNA. The CFX96 fluorescence quantitative PCR instrument (BioRad, Hercules, CA, USA) was used for detection. The reaction was carried out with the following conditions: Predenaturation at 95°C for 1 minute, denaturation at 95°C for 5 seconds, followed by annealing at 60°C for 20 seconds. A total of 40 cycles were performed, and the primer designs are presented in Table 1. Using the 2–∇∇Ct method with GAPDH as a loading control to determine the mRNA expression levels of Bax/Bcl-2, we repeated the experiment three times.
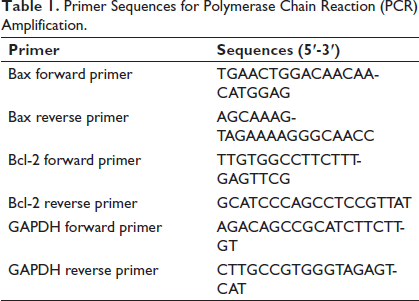
Primer Sequences for Polymerase Chain Reaction (PCR) Amplification.
The expression levels of Bax/Bcl-2 proteins in placental tissues of pregnant rats in each group were detected using Western blot analysis.
The placental trophoblast cells were treated with 100 µL of cell lysates, lysed on ice, and then the supernatant was centrifuged. A diquinoline formic acid kit was utilized to measure the protein concentration. Protein samples (30 µg) were subjected to polyacrylamide gel electrophoresis and then transferred to polyvinylidene fluoride membranes. The membranes with rat Bcl-2 and Bax antibodies (1:500, Abcam, Cambridge, MA, USA) and GAPDH antibody (1:1,000, Abcam, Cambridge, MA, USA) were incubated overnight at 4°C. Horseradish peroxidase-conjugated sheep antirabbit secondary antibody (1:2,000, Abcam, Cambridge, MA, USA) at room temperature was incubated for 2 hours. Subsequently, the luminescent solution was added to the polyvinylidene fluoride film for development, and ImageJ software was utilized to analyze the gray values.
Flow Cytometry was used for Detection
The placental trophoblast cells were digested with trypsin without ethylenediaminetetraacetic acid (EDTA). After the digestion was terminated, centrifuge the cells for 3 minutes at 1,500 rpm. The supernatant was aspirated, and the cells were washed once with phosphate-buffered saline (PBS). Subsequently, resuspend the cells in PBS after centrifugation for cell counting. After removing a suspension of 1E+5 cell volume, centrifuge it, discard the supernatant, and add 500 µL of binding buffer to resuspend the cells. Add 5 µL of Annexin V-APC, mix well, then add another 5 µL of 7-AAD staining solution, mix again, and keep the reaction in the dark at room temperature for 5–15 minutes before transferring it to ice. Flow cytometry was performed within 1 hour.
Statistical Analysis
Statistic Package for Social Science (SPSS) 22.0 software (IBM, Armonk, NY, USA) was utilized to analyze the data. Measurement data that followed a normal distribution were expressed as
Results
Compared Systolic Pressure, 24-hour Urine Protein Levels, Body Weight of Offspring, and Placental Weight in Each Group
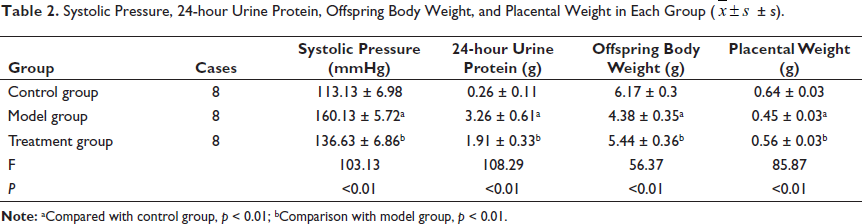
On the 20th day of pregnancy, the model group showed significantly higher levels of tail artery systolic pressure and 24-hour urine protein than the control group. On the other hand, the model group exhibited significantly lower body weight and placental weight in offspring rats compared to the control group. These observed differences reached statistical significance (p < 0.01). On the 20th day of pregnancy, the treatment group showed significantly lower levels of tail artery systolic pressure and 24-hour urine protein than the model group. On the contrary, the treatment group exhibited significantly higher body weight in offspring rats and placental weight than the model group. These observed differences reached statistical significance (p < 0.01), as demonstrated in Table 2.
Systolic Pressure, 24-hour Urine Protein, Offspring Body Weight, and Placental Weight in Each Group
.
The Expression Levels of Functional Factors in Vascular Endothelial Cells of Rats in Each Group
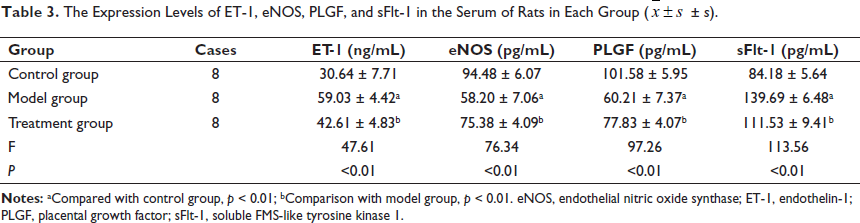
In the model group, there was a notable increase in the levels of ET-1 and sFlt-1 in the serum compared to the control group. In contrast, the levels of eNOS and PLGF were significantly reduced in the model group. These observed differences reached statistical significance (p < 0.01). In the treatment group, there was a notable decrease in the levels of ET-1 and sFlt-1 in the serum compared to the model group. In contrast, the levels of eNOS and PLGF were significantly increased in the treatment group. These observed differences reached statistical significance (p < 0.01), as demonstrated in Table 3.
The Expression Levels of ET-1, eNOS, PLGF, and sFlt-1 in the Serum of Rats in Each Group
.
The mRNA and Protein Expression Levels of Bax and Bcl-2 in the Placenta of Each Group of Rats
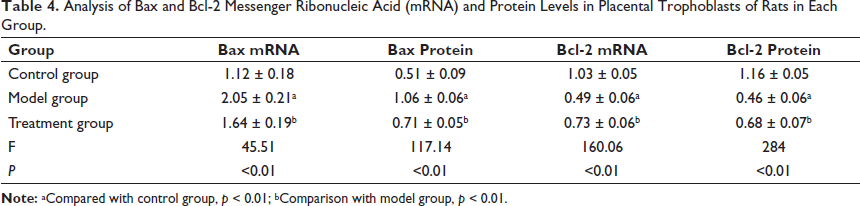
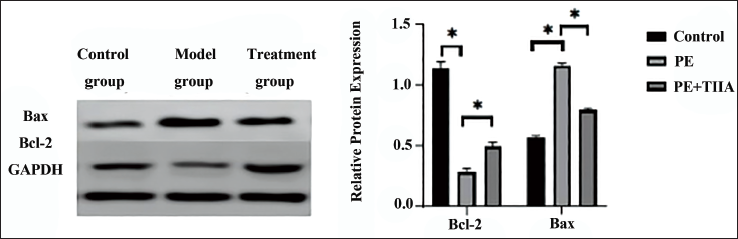
The model group showed a notable increase in the levels of Bax mRNA and protein in placental trophoblasts compared to the control group. Conversely, the levels of Bcl-2 mRNA and protein were significantly reduced in the model group. These observed differences reached statistical significance (p < 0.01). The treatment group showed a notable decrease in the levels of Bax mRNA and protein in placental trophoblasts compared to the control group. Conversely, there was a significant increase in the levels of Bcl-2 mRNA and protein in the treatment group. These observed differences reached statistical significance (p < 0.01), as shown in Figure 1 and outlined in Table 4.
Analysis of Bax and Bcl-2 Messenger Ribonucleic Acid (mRNA) and Protein Levels in Placental Trophoblasts of Rats in Each Group.
Analysis of Bax and Bcl-2 Protein Levels in the Placental Trophoblasts of Rats in Each Group.
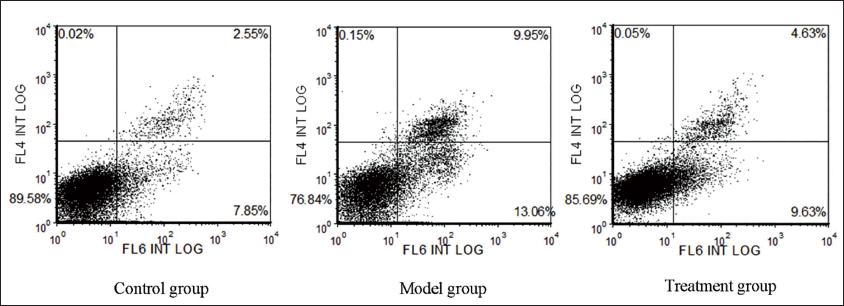
The model group had a significantly higher percentage of apoptotic cells (21.18 ± 1.45%) than the control group (10.92 ± 1.71%). These differences reached statistical significance (t = 13.07, p < 0.01). The treatment group had a significantly lower percentage of apoptotic cells (15.25 ± 1.05%) compared to the model group (21.18 ± 1.45%). These observed differences reached statistical significance (t = 9.51, p < 0.01) (Figure 2).
Used Flow Cytometry to Detect Apoptosis in Placental Trophoblast Cells.
Discussion
Research Status of S. miltiorrhiza Drugs for Treating Preeclampsia
There is a lack of effective treatment for preeclampsia. At present, symptomatic treatment measures, such as antihypertensive, spasmolytic, and sedation, are mainly used in clinical practice. The only effective method mentioned is the termination of pregnancy (Jung et al., 2022). S. miltiorrhiza has a long history of use in treating preeclampsia and has demonstrated favorable outcomes. It has the effects of dilating placental blood vessels, improving placental perfusion, and enhancing fetal development. Compound S. miltiorrhiza injection can promote blood circulation, prevent platelet aggregation, protect vascular endothelial cells, remove free radicals, and improve cellular resistance to hypoxia (Yang et al., 2018; Zhao et al., 2021). He et al. (2018) discovered that when S. miltiorrhiza injection is used in conjunction with magnesium sulfate to treat severe preeclampsia, it can effectively decrease patients’ blood pressure and reduce the likelihood of eclampsia. Mizutani and Mizutani (2015) demonstrated that a combination of S. miltiorrhiza injection and magnesium sulfate could effectively improve the renal and coagulation function of patients with preeclampsia.
At present, most studies on the application of S. miltiorrhiza in preeclampsia are primarily focused on clinical practice, with limited exploration of the mechanism of action and small sample sizes. The conclusions drawn may be biased and need to be further verified. Therefore, in this study, we used
TIIA Alleviates Vascular Endothelial Cell Dysfunction in Preeclamptic Rats
Research has indicated that TIIA can regulate the function of immune cells, lower the concentrations of interleukin-1β (IL-1β) and TNF-α in the bloodstream, diminish the generation of inflammatory agents, and modulate signaling cascades like MAPK/NF-κB and TLR/NF-κB pathways to achieve anti-inflammatory, antioxidant, and protective effects on vascular endothelial cells (Guo et al., 2020). Numerous basic experiments and clinical investigations have demonstrated that TIIA plays an anti-inflammatory role in a variety of diseases, such as coronary heart disease, nervous system diseases, bronchitis, and sepsis (Li et al., 2020). In addition, TIIA can directly reduce blood pressure through its protective effect on the endothelial eNOS-NO pathway (Li et al., 2015). In terms of preeclampsia, basic studies have shown that TIIA can inhibit the release of high-mobility group protein B1 induced by hypoxia, thereby protecting the function of endothelial cells (Zhao et al., 2017). In our study, TIIA partially reversed the
TIIA Alleviates Placental Trophoblast Apoptosis in Preeclamptic Rats
Studies have shown that in the placental trophoblast cells of preeclampsia patients, the balance of the Bcl-2 family is disrupted, shifting toward apoptosis (Hu & Zhang, 2022; Tomas et al., 2011). Increased apoptosis in the placental villous trophoblast can lead to shallow placental implantation and failure of maternal spiral artery reconstruction, resulting in decreased placental perfusion (Kasture et al., 2021). Reduced placental perfusion induces oxidative stress, which, in a positive feedback loop, exacerbates trophoblast apoptosis. This leads to further oxidative stress and the release of cytokines into the maternal circulation, such as reactive oxygen species (ROS) (San Juan-Reyes et al., 2020), malondialdehyde (MDA) (Ferreira et al., 2020), IL-1β (Wang et al., 2022), and TNF-α (Cunningham et al., 2020). These cytokines act on the maternal vascular endothelium, causing dysfunction in the endothelial cells throughout the body. At the same time, cytokines inhibit eNOS production in endothelial cells and reduce the level of circulating nitric oxide, causing clinical symptoms of preeclampsia, such as increased blood pressure and organ function damage (Opichka et al., 2021). Therefore, excessive trophoblast apoptosis is considered to be one of the pathogenic causes of preeclampsia. In this study, TIIA was used to treat the preeclampsia rat model, and placental trophoblast cells were isolated. Flow cytometry, as well as Bcl-2/Bax protein and mRNA expression analysis, demonstrated that TIIA could effectively reduce excessive apoptosis in placental trophoblast cells in preeclampsia rats. This action helps prevent dysfunction of endothelial cells and alleviates the clinical manifestations of preeclampsia.
At present, there is no direct evidence supporting the use of TIIA in the clinical treatment of preeclampsia. However, based on the various pharmacological effects of TIIA, this study speculates that TIIA may have a certain therapeutic effect on preeclampsia. To investigate this, a rat model was established for further study and to explore its possible mechanisms. It should be noted that this study has several limitations. The study investigated the therapeutic effect of TIIA on preeclampsia in rats. However, there are species differences between rats and humans, and further research in other animal models of preeclampsia or even human clinical trials is necessary in the future. At the same time, further studies are needed to explore its role in the treatment of preeclampsia, including its mechanisms, indications, dosage, safety, and effectiveness. Due to the multiple effects of TIIA, its ability to improve early symptoms of preeclampsia through other mechanisms has not been thoroughly researched.
Conclusion
The present study demonstrates that TIIA can reduce placental trophoblast apoptosis and alleviate vascular endothelial cell dysfunction in preeclampsia. This intervention effectively alleviates the clinical symptoms of
Footnotes
Abbreviations
Bax: Bcl-2-related X protein; Bcl-2: B-cell lymphoma-2; eNOS: Endothelial nitric oxide synthase; ET-1: Endothelin-1; IL-1β: Interleukin-1β;
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All studies were authorized by the Ethics Committee at Tianjin Central Hospital of Obstetrics and Gynecology (2020KY122).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Tianjin Health Science and Technology Project (KJ20127) and the Tianjin Central Hospital of Gynecology and Obstetrics/Tianjin Key Laboratory of Human Development and Reproductive Regulation (2021XH03).
