Abstract
Background
Shilajit a natural phytomineral has a proven record of treating many human body ailments.
Objectives
This study aimed to explore the influence of Shilajit on cell proliferation and apoptosis induction in oral cancer cells (OCC) and to comprehend the molecular mechanism associated with OCC migration.
Materials and Methods
Human gingival fibroblast (hGF) and OCC (KB-1 (KERATIN-forming tumor cell line HeLa)) were exposed to Shilajit solution dilutions. Cell growth and apoptosis were measured by MTT and Annexin-V tests (control/test group). Cellular morphology using an inverted microscope, cellular apoptosis using acridine orange/ethidium bromide dual staining, reactive oxygen species production analysis using 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) staining, and gene protein expression using real-time polymerase chain reaction test were used to measure study outcomes. The data enumerated were the average of three trial experiments. Categorical and numerical data were expressed as frequency distribution and means, respectively. Differences in groups (control/test; zero/24 h/48 h) were determined using Student’s t-test and one way analysis of variance, with probability ‘p’ value considered significant at ≤0.05.
Results
The viability of OCC exhibited a concentration and time-dependent response to Shilajit. Notably, Shilajit demonstrated selectivity against cancer cells. Through an examination of the Annexin-V apoptosis assay, it was observed that Shilajit induces apoptosis by upregulating the proapoptotic gene expression (p ≤ 0.05) and downregulating antiapoptotic proteins (p ≤ 0.05). Furthermore, the impact of Shilajit on cell migration decreased significantly when compared to control cells through modulating the urokinase-type plasminogen activator (uPA) and its receptor (uPAR) and chemokines gene expression.
Conclusion
Shilajit exhibited greater cytotoxicity, decreased OCC proliferation and migration, and initiated OCC apoptosis as equivalent to normal cells. These promising outcomes indicate that Shilajit holds potential as a robust, promising option for oral cancer treatment, underscoring the need for further research in this domain.
Introduction
Oral cancer is a significant global health concern, characterized by its high incidence (Ghantous & Abu Elnaaj, 2017), aggressive nature (Ghizoni et al., 2020), and if not diagnosed, may often lead to devastating outcomes, including mortality (Cunha et al., 2019). Oral cancer, which comprises malignancies of the lip, several other oral tissues, and the oropharynx, is the 13th most prevalent type of cancer globally (Gupta et al., 2016). As per World Health Organization (WHO) estimates, there were 177,757 fatalities and 377,713 new cases of lip and oral cavity cancer worldwide in 2020 (World Health Organization, 2019). Men and older individuals are more frequently affected by oral cancer, with higher mortality rates in men than women (Cunha et al., 2019). Socio-economic status has been found to be related to the prevalence of oral cancer (Agarwal et al., 2007), as well as significantly impacting staging, survival, and diagnosis (Azimi et al., 1995). Carcinogens associated with oral cancer comprise alcohol, tobacco, and areca nut (betel quid), while other agents have been linked with the progress of different types of oral cancers (Rivera, 2015). Human papillomavirus infections are increasingly identified as the causative factor for mouth malignancies in children in North America and Europe (Stjernstrøm et al., 2019; Yete et al., 2018). The choice of treatment for oral cancer is dependent upon the stage of the cancer and is considered to be challenging due to the involvement of different contiguous oral structures (Borse et al., 2020). While in the early stages, maximum oral cancers can be treated by surgical excision, treating advanced cases still challenges physicians and surgeons, despite improvements in diagnostic and therapeutic techniques, thus highlighting the crucial need for novel and efficient therapies. In most cases, when cancer is present in either the maxilla or the mandible, the postsurgical case scenario becomes the rehabilitation of either a maxillectomy or a hemimandibulectomy, respectively (Kumar et al., 2023; Singh et al., 2015).
Approximately half (47%) of contemporary drug molecules utilized in cancer chemotherapy originate from or are derived from natural products (Newman & Cragg, 2007), making the exploration of natural substances for new drug development a keen interest for researchers. Shilajit (also called mummiyo, salajeet, mumijo, mumie, or moomiyo) is a phytomineral exudating from rock (sedimentary) layers, although it is believed to originate from plant decomposition (Windmann, 2023). With a semi-solid consistency that has a brown (pale to black) color, strong (sharp) odor, and powerful pungent taste, it has been found in various high-altitude (≥1,000 m) mountains/ranges (Himalayas, Altai, Caucasus) during the summer months (Agarwal et al., 2007). Shilajit is naturally designed by the prolonged microbial humification (long-term) process of various plants, chiefly bryophytes (Mishra et al., 2020). With an opulent history of use in traditional medicine systems like Ayurveda, Shilajit has a proven record of use in treating various body ailments (genitourinary, digestive, nervous, asthma, diabetes, and so on) (Agarwal et al., 2007; Stohs, 2014; Windmann, 2023). It serves as a natural medicinal food that has been highly competent in wound healing, bleeding (anemia), bone fractures, weakness, and anti-inflammatory treatments (Wilson et al., 2011). A very high frequency (80%) of developing countries’ population consumes plant-based drugs to maintain health care needs (Ara et al., 2020), with the WHO estimating the same percentage of the global population will rely on plant-based medicines in the near future (Rai et al., 2020). Chemically, Shilajit is a humic substance that contains humic acid (HA) and fulvic acid (60–80%), which contribute mainly to its biological properties. Other components include acids (fatty, ellagic, aromatic carboxylic, amino, and benzoic), ichthyol, resin, phenolic lipids, and microelements (Kloskowski et al., 2021). Wilson et al. (2011) reported its beneficence in reducing inflammation, rheumatism, arthritis, pain, ulcerations, stress, and anxiety. Early evidence suggests that the humic matter that is associated with Shilajit may impede cancer by slowing the growth of cancer cells and later inducing cancer cell death or apoptosis (Yang et al., 2004). Additionally, the cytotoxic characteristic of HA has been associated with the generation of nitric oxide (NO) and the formation of reactive oxygen species (ROS) (Ting et al., 2010). Studies have also unveiled its antiviral potential against the herpes simplex virus (HSV) (Cagno et al., 2015), which may hold potential against viral infections that cause cancer (human papillomavirus and hepatitis). Barouji et al. (2020) studied the anticancer properties of Shilajit in breast cancer cells (epithelial-mesenchymal transition (EMT) and MCF-7/MDA-MB-231 cancer cell invasiveness) besides its anti-inflammatory and antioxidant abilities. The authors concluded that Shilajit (mummy) triggered EMT and metastasis inhibition in cancer cells by downregulating of transforming growth factor-beta 1 (TGFβ1) activity. Pant et al. (2016) observed that Shilajit reduced proliferation in hepatocarcinoma cells by inducing the generation of ROS and promoting cell death by impeding miRNA-22 and miRNA-21. Kloskowski et al. (2021), in their urinary bladder cancer investigation, reported that Shilajit exhibits a time- and concentration-dependent influence on bladder cancer cell viability (T24 and 5637) as opposed to normal uroepithelial cell types (SV-HUC1). It hinders cell cycle advancement and triggers apoptosis. Notably, the results demonstrate that Shilajit exerts significantly higher cytotoxicity on urinary bladder cancer cells compared to normal cells. The earliest evidence of the anticancer activity potential of Shilajit was found in Ehrlich ascites, where Shilajit extract significantly inhibited proliferation by cancer cells (Kamgar et al., 2023). Its active component, HA, has been described to stop proliferation (antiproliferative) and growth inhibition in acute T lymphoblastic leukemia (acts on HL-60 cells by inducing apoptosis through activation of caspase-3/mitochondrial cytochrome-c cycle). Damage to DNA due to the formation of thymine dimer leads to mutations as a consequence of ultraviolet (UV) rays (radiation exposure). HA, having photo-protective ability, can thus prevent such cancers that result from radiation exposure (Meena et al., 2010). HA has the capability to deactivate and reduce the bioavailability of hazardous materials (radioactive materials, pesticides, heavy metals, and so on), which have carcinogenic or mutagenic compounds (Perminova & Hatfield, 2005). Processed HA inactivates various mutagens when tested on yeast (Marova et al., 2011).
Despite such preventive and therapeutic evidence, many of the potential applications of Shilajit including its prospective role in oral cancer treatment, remain unexplored, although its potential applications in medical sciences have been observed earlier (Flammang et al., 2022). Presently, the specific effects and cellular signaling pathways linked to Shilajit’s anticancer activity remain unclear. Therefore, this study was undertaken with the primary aim of exploring the influence of Shilajit on the regulation of cell viability, proliferation, migration, and induction of apoptosis in KB-1 (KERATIN-forming tumor cell line HeLa) cancerous cells. Additionally, the primary objective of the study was to unravel the molecular mechanism associated with oral cancer cell (OCC) migration, with a focus on assessing the anticancer potential of Shilajit in OCCs. Shilajit, recognized for its diverse therapeutic benefits in traditional remedies, holds significant demand as a crucial ethno-medicinal food. Considering the widespread consumption of Shilajit in the customs of South Asian countries, this research bears potential implications for comprehending therapeutic benefits in the prevention and management of oral cancer.
Materials and Methods
Study Design, Sampling, and Grouping
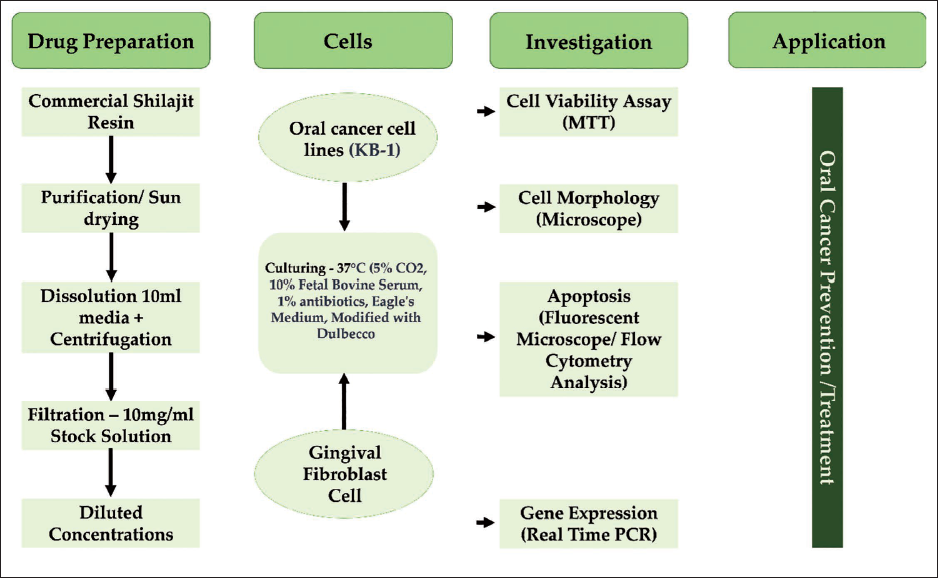
This study was designed as an in vitro experimental approach that was conducted in three different phases. The first phase was primarily dedicated to cell line procurement/maintenance and Shilajit solution preparation, including its various dilutions. The second phase principally focused on the intervention, with cell lines receiving no treatment serving as cell controls (control group) and those exposed to Shilajit solution serving as treated or experimental groups. Sample groups for assessing cell morphology were based on the exposure time (control, 24 hours, 48 hours). The final phase predominantly measured the outcome through cell viability assay, flow cytometry analysis, and real-time polymerase chain reaction (PCR). Each experiment was repeated three times to impart the test-retest reliability for individual outcome measurement as guided by other similar studies (Kloskowski et al., 2021). The experimental sequence that was performed on two different cell lines is depicted in Figure 1. The experimental armamentarium, including materials, measuring tools, and analyzing software, is summarized in Table 1.
Armamentarium (Materials, Measuring Devices and Kits, Software).

Cell Line Procurement and Maintenance
The OCC lines (KB-1) were provided by the National Centre for Cell Science (NCCS), Pune, and human gingival fibroblast (hGF) cells were obtained from Saveetha Dental College, Chennai, India. These cells were cultured in a culture media (Dulbecco’s Modified Eagle Medium (DMEM), fetal bovine serum (FBS) (10%), 1% antibiotics (penicillin, streptomycin, and amphotericin)). The growth of cell lines was accomplished in a tissue culturing flask (T25-flask, 75 cm2, Corning, USA) maintained at a temperature of 37°C in a humidified environment with 5% carbon dioxide (CO2).
Preparation of the Shilajit Stock Solution
Shilajit paste was custom prepared using a commercially available natural resin (200 mg) (Lajit Gold Shilajit Resin, Lajit Gold, USA) derived from plants and sourced from high altitude Himalayan mountains while manufactured in the United States of America (USA) (Lajit, 2022). This resin underwent a validated purification process based on traditional Ayurvedic techniques
3-[4,5-Dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide (MTT) cell viability assay: To test the vitality of Shilajit-treated hGF cells and an OCC line, the MTT assay was utilized (Kloskowski et al., 2021). This assay involved the alteration of the yellow salt (tetrazolium) into insoluble formazan crystals with a purple color. For the assay, 5 × 103 trypsinized cells of the cell line were plated per well in 96-well plates. After plating, they were cultured in serum-free media for 3 hours at 37°C. The cells were then starved, followed by two washes with 100 µL of serum-free medium. Control cells were exposed to a serum-free medium initially. Subsequently, both cell lines were exposed to Shilajit at various concentrations (ranging from 0.25 to 1.5 mg/mL) for 24 and 48 hours. Following treatment, 100 µL of MTT-containing DMEM (0.5 mg/mL) was further added to each well containing control and Shilajit-treated cells. Later, the cell culturing for four hours at 37°C in a CO2 incubator was initiated. Following the removal of the solution containing MTT, 1 × phosphate-buffered saline (PBS) was used to wash the cells. Dimethyl sulfoxide (100 µL) was used to dilute the formazan crystals that were generated. At 570 nm in wavelength, the generated color’s intensity was determined by a micro enzyme-linked immunosorbent assay (ELISA) plate reader. Utilizing the following formula, the fibroblast viable cell frequency (percentage) was established: [A570 nm-treated/A570 nm – control] × 100.
Intervention, Measurement, and Analysis
Cell morphological/cytotoxicity characteristics: The optimal dose, half-maximal inhibitory concentration (IC50), determined as 1 mg/mL through the MTT assay for the OCC line, was selected for further analysis. In six-well plates, cells were plated to a density of 2 × 105 and subjected to Shilajit for 24 hours. After the Shilajit treatment, they were washed once in PBS (pH 7.4) and examined using an inverted microscope to assess cell morphology. The cytotoxic potential of Shilajit was also evaluated with the MTT assay. KB-1 (5 × 103) cells per well were plated in the 96-well plates, and after reaching 80% confluence, the cells were treated with various concentrations of Shilajit for 24 and 48 hours. Data obtained were analyzed using recommended software (FlowJo v10.3).
Apoptosis Recognition and Analysis
Double labeling using the acridine orange (AO) dye and a substance called ethidium bromide (EtBr) determined the manner of cell death. As mentioned in earlier studies (Kloskowski et al., 2021), the acridine orange/ethidium bromide (AO/EtBr) dual staining method was utilized to analyze the impact of Shilajit on OCC death. Following 24 hours of Shilajit (0.5 and 1 mg/mL) treatment, the cells were gathered and rinsed with PBS. Subsequently, the pellets were incubated with 5 µL of AO (1 mg/mL) and 5 µL of EtBr (1 mg/mL). The Shilajit-induced apoptotic changes in the labeled cells were visualized with the help of an inverted fluorescence microscope. The data obtained were further assessed using computer software (BD FACSDiva v. 8.0.1, BD Biosciences, USA).
Apoptosis Assay (Annexin-V Flow Cytometry Analysis)
The KB-1 cells were seeded in a 60-mm dish. IC50 concentration (1 mg/mL) of Shilajit was treated and incubated for 24 hours. The Shilajit-induced apoptosis in OCC was assessed with an Annexin V/fluorescein isothiocyanate (FITC) Apoptosis Detection kit (BD Biosciences, USA). The Shilajit-treated KB-1 cells were harvested, and the cells were labeled with propidium iodide (PI) and Annexin-V-FITC and their examination was made by using a fluorescence-activated cell sorting (FACS) Calibur Flow Cytometer (BD Biosciences).
Scratch Wound Healing Assay
An in vitro scratch wound healing assay was performed to assess the antimigrative potential of Shilajit in OCC (Elumalai et al., 2014). Human OCC (2 × 105 cells/well) were seeded onto six-well culture plates. The cell monolayer was scratched using a 200 µL tip to create a wound, washed with PBS, and photographed with an inverted microscope. Shilajit (0.5 and 1 mg/mL) was treated for 24 hours, and control cells were received with serum-free culture medium. After the treatment period, the wounded area was photographed using the same microscope. The experiments were performed in triplicates.
Real-time PCR
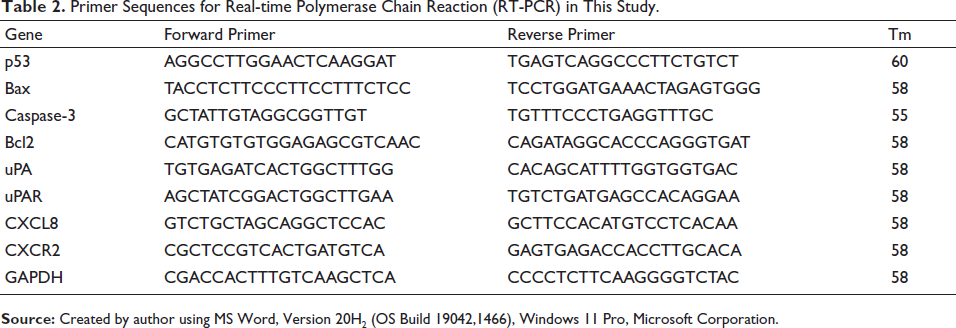
The gene expression of apoptosis signaling molecules was analyzed using real-time PCR. The Trizol from Sigma was used to isolate the total ribose nucleic acid (RNA) using a standard protocol. 2 µg of RNA is used for copy DNA (cDNA) synthesis using Prime Script, 1st strand cDNA synthesis kit (TakaRa, Japan) by the reverse transcription process. The targeted genes were amplified using specific primers. The primer sequences used in the study are tabulated and presented in Table 2. The PCR reaction was performed with GoTaq, qPCR Master Mix (Promega), which contains N,N-dimethyl-N′-[4-[(E)-(3-methyl-1,3-benzothiazol-2-ylidene) methyl]-1-phenylquinolin-1-ium-2-yl]-N′-propylpropane-1,3-diamine (SYBR green I) dye and all the PCR components. Real-time PCR was completed in a CFX96 PCR system (Bio-Rad). The raw data, cycle threshold (CT value) from the real-time PCR machine were analyzed by comparative CT method, and to generate fold change graph 2-(–delta-delta-CT) method was used as described by Schmittgen and Livak (2008).
Primer Sequences for Real-time Polymerase Chain Reaction (RT-PCR) in This Study.
Statistical Analysis
Individual experiments were conducted thrice, and depending upon the distribution, either mean or median was considered as the final measurement value. The average viable cell was determined as a percentage that was relative to the control. All categorical variables (morphology) were expressed in frequency distribution (percentage and number) while numerical data were expressed as mean ± standard deviations (SDs). Student’s t-test assessed the differences between the control and test (cellular apoptosis) and at 0- and 24-hour (cellular migration) groups. A one-way analysis of variance (ANOVA) with Tukey’s post hoc test determined multiple group comparisons for cellular viability (control, 24 hours, 48 hours). Differences between groups were considered significant for the probability ‘p’ value to be either equal or less than 0.05 (p ≤ 0.05). All statistical tests were performed using Statistical Package of Social Sciences (SPSS 25.0) (IBM, Armonk, NY, USA) software.
Results
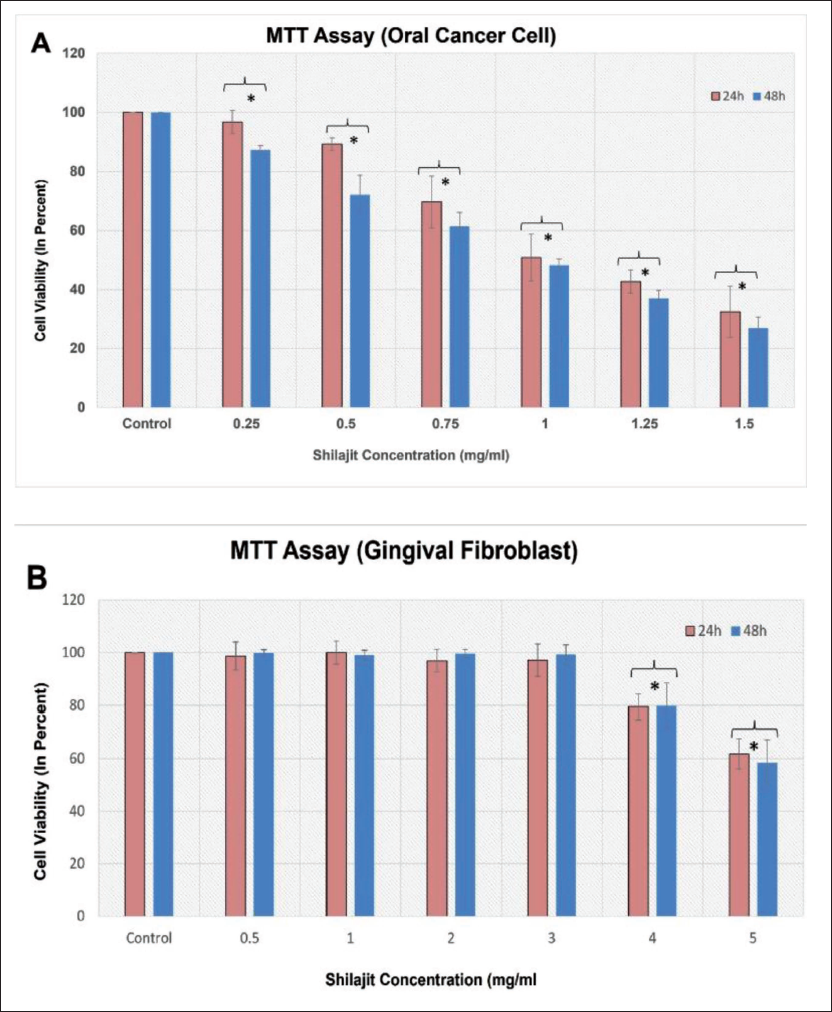
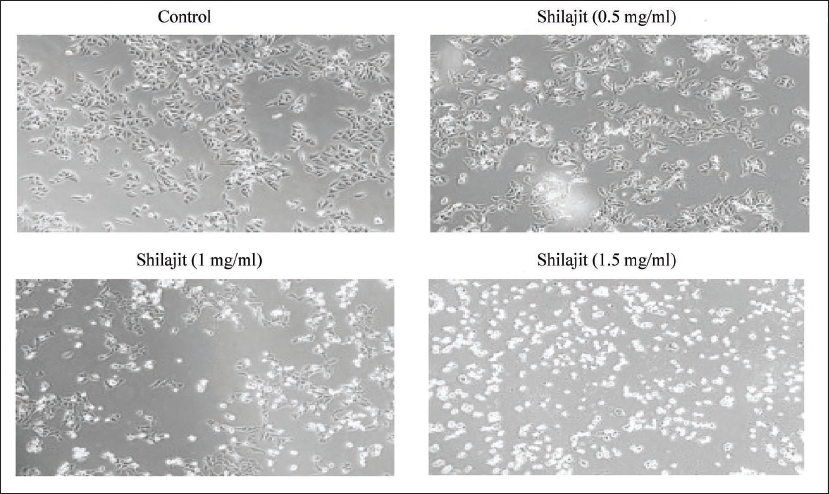
Cell viability and cellular morphology: The results revealed a significant reduction (p ≤ 0.05) in cell proliferation, with percentages of 96.71%, 89.20%, 69.65%, 50.84%, 42.66%, and 32.44% in 24 hours and 87.24%, 72.08%, 61.33%, 48.24%, 36.90%, and 26.93% in 48 hours time points at concentrations of 0.5, 0.75, 1, 1.25, and 1.5 mg/mL of Shilajit, respectively, in KB-1 cells (Figure 2A). Shilajit significantly reduced the viability of OCCs compared to the control (untreated) group at both 24- and 48-hour time intervals (p ≤ 0.05). The percentage of cell viability gradually decreased significantly (p ≤ 0.05) with an increase in the concentration of Shilajit, reaching 50% growth inhibition at a dose of 1.049 ± 0.122 (24 hours) 0.965 ± 0.026 (48 hours) (Figure 2A). Notably, all calculated IC50 values for Shilajit were lower in cancer cells (KB-1) than for normal cells (hGFs). The cell viability percentage of gingival fibroblast cells followed 98.79%, 99.90%, 96.86%, 97.09%, 79.49%, and 61.76% for 24 hours and 99.91%, 99.07%, 99.68%, 99.33%, 79.48%, and 58.53% for 48 hours time points at concentrations of 0.5, 1, 2, 3, 4, and 5 mg/mL of Shilajit, respectively (p ≤ 0.05) (Figure 2B). Therefore, the aforementioned dose was chosen for subsequent experiments. Lower concentrations of Shilajit did not exhibit significant effects. The OCC line was exposed to Shilajit (0.5, 1, and 1 mg/mL) for 24 hours. In comparison to the control cells, the treated cells exhibited notable morphological alterations, including cell shrinkage and reduced cell density. Characteristic features of apoptotic cells were evident in the Shilajit-treated cells. Additionally, cells undergoing apoptosis displayed other morphological changes, such as rounding up, shrinkage, and loss of contact with neighboring cells. Some sensitive cells were also observed to detach from the surface of the plates (Figure 3).
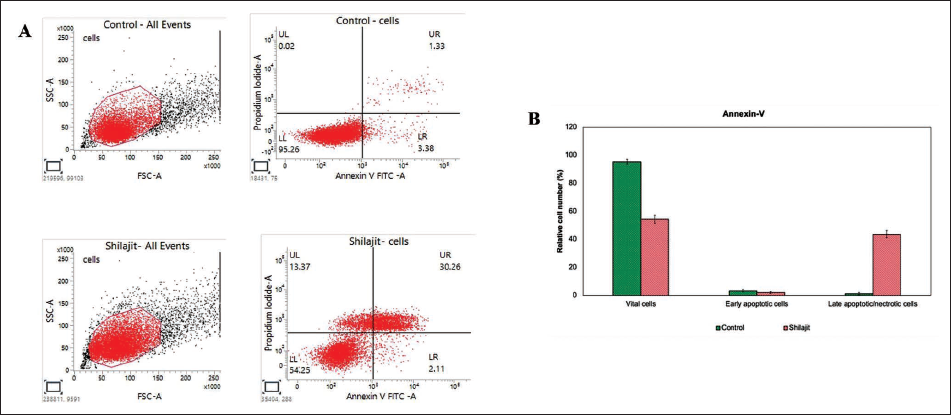
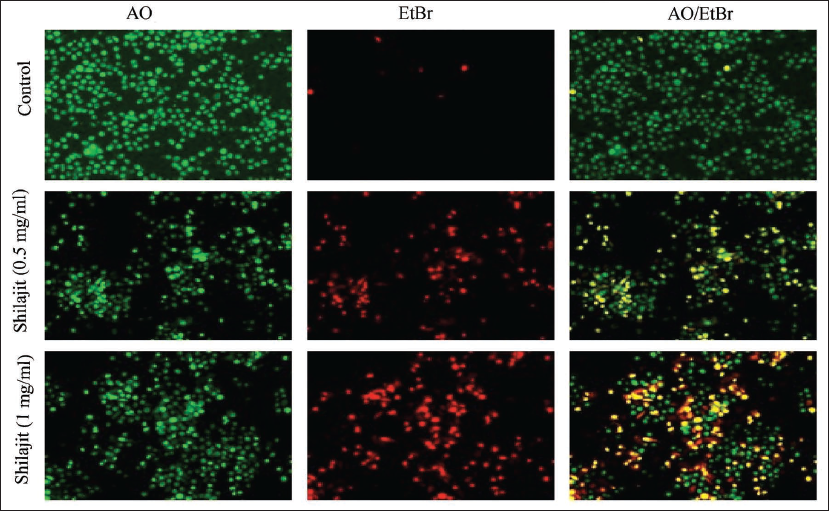
Cellular apoptosis by activating the p53-mediated intrinsic apoptotic pathway: To ascertain whether cultured OCCs undergo apoptosis, we analyzed untreated and Shilajit-treated KB-1 cells using Annexin-V and PI. Shilajit-treated KB-1 cells exhibited a higher percentage of apoptotic cells when compared to the untreated group. Overall, the percentage of apoptotic cells increased in Shilajit-treated KB-1 cells (32.37%, n = 3), (p ≤ 0.05) (Figure 4) compared to untreated cells, with less than 13.37% of the cells showing necrotic signs in Shilajit-treated cell lines and control cells showing only 0.02%. In the group of hGFs treated with Shilajit (1 mg/mL), there was only a slight decrease in cell viability from 100% to 99.00%. This finding underscores the inhibitory efficiency of Shilajit on the growth of OCCs, demonstrating minimal cytotoxicity to the normal hGF cells. To further understand cell death mechanisms, specific apoptosis, or necrosis, we utilized the AO/EtBr dual staining procedure to examine the nucleus morphology in both control and Shilajit-treated cells (Figure 5). The AO/EtBr dual staining results aligned with our quantitative apoptosis analysis, providing confirmation that the highest rate of apoptosis, characterized by nuclear deformation and loss of cell wall integrity, was observed in OCCs treated with Shilajit.
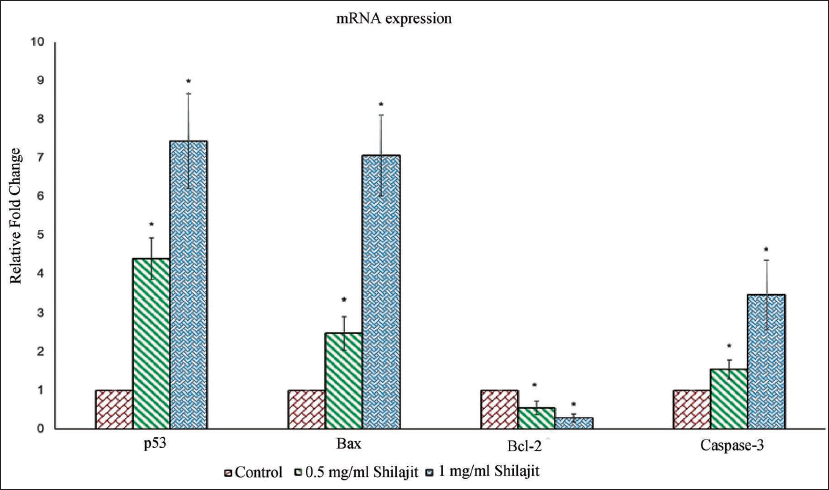
Intracellular signaling molecules governing proapoptotic activity in KB-1 cells: The B-cell lymphoma 2 (Bcl-2) protein family consists of both proapoptotic proteins (p53, Bcl-2-associated X protein (Bax), and caspase-3) and antiapoptotic protein (Bcl-2), which play crucial roles in regulating mitochondrial outer membrane integrity, the release of cytochrome c into the cytosol, and activation of caspase. Consequently, we analyzed the mRNA expression of Bcl-2 family proteins through real-time PCR. Shilajit demonstrated a substantial rise in the expression of proapoptotic proteins, including p53, Bax, and caspase-3, in OCC lines (Figure 6). Furthermore, it downregulated the expression of the antiapoptotic protein (Bcl-2) in the KB-1 cell line. These findings suggest that Bcl-2 family members play a pivotal role in Shilajit-mediated apoptosis induction in OCCs.
Cellular Migration Potential
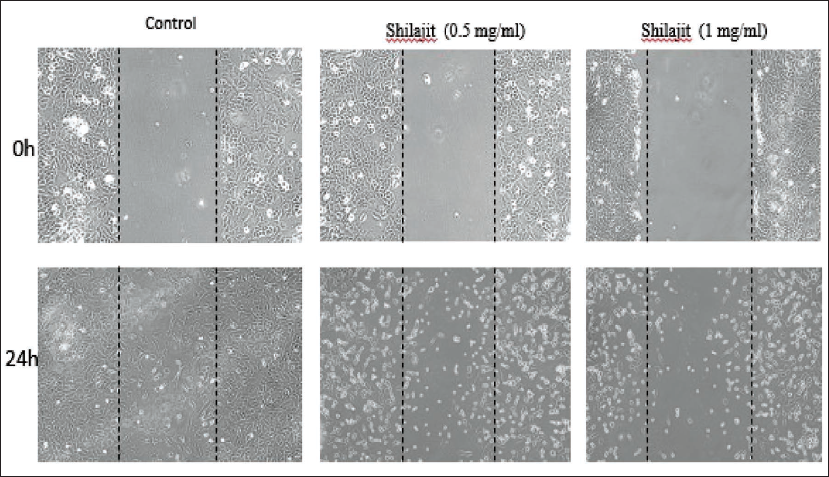
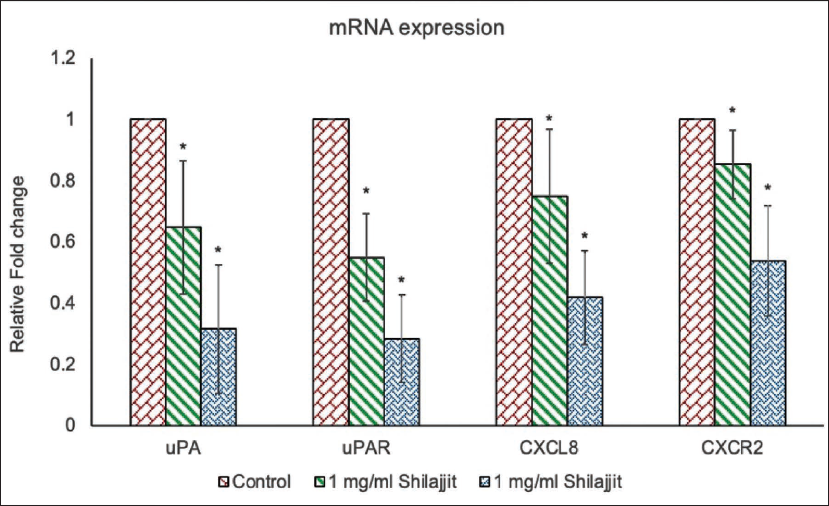
Shilajit inhibits the migratory potential of OCCs by inhibiting the urokinase-type plasminogen activator (uPA) and its receptor (uPAR) and the chemokine signaling pathway. A scratch test was performed to study the impact of Shilajit on the migration of OCCs. The results showed that Shilajit inhibits the cell migration rate when compared to control cells. The following observations were made: In the control group, the cells migrated to almost half of the scratched area after 24-hour time points. Treatment with Shilajit at a concentration of 1 mg/mL significantly inhibited the migration of OCCs compared to the control group (p ≤ 0.05). The migration distance of cells in the Shilajit group decreased compared with that observed within the control group (Figure 7). Next, we analyzed the mRNA expression of the uPA and uPAR and the chemokine signaling proteins in OCCs. Substantial clinical and experimental evidence indicates that uPA and uPAR interaction on the cell surface facilitates both extravasation and intravasation of cancer cells, playing a crucial role in the proteolysis process and the extracellular matrix attachment of the cell. We assessed the messenger ribonucleic acid (mRNA) expression of uPA and uPAR in KB-1 cell lines using real-time PCR. Shilajit demonstrated a significant reduction in the mRNA expression of both uPA and uPAR in both cell lines (Figure 8) (p ≤ 0.05). Chemokines constitute a group of cytokines secreted by cells, primarily functioning as a chemoattractant to promote the cell migration process. The mRNA expressions of CXCL8 and CCR2 were evaluated using semi-quantitative PCR (Figure 8). C-X-C motif chemokine ligand 8 (CXCL8) chemokines bind with C-C chemokine receptor type 2 (CCR2) and promote the inflammatory response. Shilajit treatment markedly decreased CXCL8 and CXCR2 mRNA expression in OCCs.
Discussion
The current in vitro experimental research was aimed primarily at discovering the potential application of Shilajit as a prospective anticancer therapeutic drug in the treatment of oral cancer by studying its influence on cancer cell viability, morphology, proliferation, migration, and apoptosis. The study also aimed to reveal the mechanism of action of Shilajit at the microscopic cellular level. The main findings of the study showed that Shilajit had a greater cytotoxic effect on OCCs than normal cells (same organ) without harming healthy oral fibroblast cells, had a time-dependent effect on KB-1 cell viability, significantly increased apoptotic cancer cells and activity (verified through Annexin-V and PI exclusion assays), and prevented cancer cell migration. Shilajit use has been investigated in the medical field for various purposes, showing potential applications as an anti-inflammatory, antiallergic, and antifungal agent (Carrasco-Gallardo et al., 2012; Kim et al., 2021; Shalini & Srivastav, 2008). Its applications for cancer prevention and treatment are based on its antiulcerogenic (Agarwal et al., 2007), antioxidant (Kamgar et al., 2023), and immunomodulator (Carrasco-Gallardo et al., 2012) properties. Shilajit (mumio) constitutes a major and popular form of traditional medical treatment (Liao et al., 2018). Its use in modern medicine came to light a few decades back when it was shown to improve urinary bladder blockage due to benign prostate enlargement (Andriukhova et al., 1997). Its therapeutic potential in cancer prevention (radioprotective) (Marova et al., 2011) and treatment (anticarcinogenic) (Perminova & Hatfield, 2005), besides antimicrobial properties, has been chiefly attributed to its chemical constituents fulvic and benzoic acid. Additionally, mumio polyphenolic antioxidants like allergies and tannic acids limit superoxide anion radical generation and phospholipase A2 activity (Kloskowski et al., 2021). Investigations on mumio’s effects on liver cancer cells have identified suppression of well-differentiated hepatocyte-derived carcinoma cell line (Huh-7), inhibition of cancer cell multiplication, and, at the same time, apoptosis (Pant et al., 2016). The cytotoxic effects on human cancer cell lines and Medical Research Council cell strain 5 (MRC-5) lung fibroblast cells were more effective than normal lung fibroblast line (Barouji et al., 2020), except for breast cancer (Elumalai et al., 2012).
This study found a greater cytotoxic impact in one OCC line than in normal cells from the same organ. Jafari et al. (2019) found that mumio cytotoxically affected dose-dependent breast and lung cancer cell lines. Our research precisely examined the effects of Shilajit on normal and malignant cells from the same organ, unlike earlier studies. This method allows for more detailed pharmacological efficacy and safety assessments in early research. Our investigation found greater IC50 values for normal cells than OCCs. The cell viability of Shilajit-treated hGFs reduced from 95.1% to 82.53%, demonstrating that it inhibits OCC proliferation without harming normal oral fibroblast cells. Barouji et al. (2020), while studying the influence of Iranian pure Shilajit found dose-dependent inhibition of breast cancer cell growth. They also found that MD Anderson Metastasis Breast cancer (MDA-MB-231) cell lines were more susceptible to anticancer characteristics of Shilajit than Michigan Cancer Foundation-7 (MCF-7) cell lines. Interestingly, they also reported that Iranian Shilajit did not harm human normal cells (MCF-10A). In another study, Borek (2004) suggested that antioxidants generally protect cells from being injured by free radicals by a mechanism that involves neutralizing the electric charges present on the injurious free radical (Borek, 2004). Peña-Méndez et al. (2005) termed this property of Shilajit as free radical scavenging ability, and they found it to be more effective against the NO (nitrous) and OH (hydroxyl) free radicals. He also attributed such activity to the concentration level of the humic compounds in Shilajit. Our study aimed to determine if Shilajit had cytotoxic and proapoptotic properties against an OCC line. Our results showed that Shilajit has a time-dependent effect on KB-1 cell viability, with a substantial and dose-dependent decrease in cell viability. Shilajit significantly stimulated cell death at 1 mg/mL doses, suggesting a possible cytotoxic activity. Apoptosis, also known as programmed cell death, is distinguished by several distinct features. These include DNA fragmentation, cell shrinkage, chromatin condensation, and enzyme activation, also called caspases (Mohammad Mirzapour et al., 2023). Apoptosis induction in tumor cells is a well-recognized and effective anticancer strategy used in various cancer treatments (Barouji et al., 2020; Elumalai et al., 2012). Our findings indicate a substantial increase in the percentage of apoptotic cells after administering Shilajit treatment, suggesting that Shilajit has a proapoptotic effect. The verification of apoptosis induction was conducted through Annexin-V and PI exclusion assays. The IC50 concentration of Shilajit (1 mg/mL) was established, and flow cytometry was used to quantify the apoptosis induction. The percentage of apoptotic cells was significantly higher in the group treated with Shilajit (late apoptotic cells: 43.63 ± 2.54, n = 3, p ≤ 0.05, Figure 4). The KB-1 cells were compared to untreated KB-1 cells. Our findings are supported by a previous study, which found that Shilajit caused apoptosis in Huh-7 cells. This was confirmed through the annexin-V/PI assay. Shilajit inhibits cell proliferation by regulating microRNA (miRNA)-22 and suppressing miRNA-21. AO/EtBr staining was used to verify and quantify apoptosis in Shilajit-treated cells in our study. Shilajit-treated and untreated cells were examined for nuclei morphology using this approach. Live cells were green, early apoptotic cells were yellow, and late apoptotic cells had contracted and shattered nuclei with a red ethidium bromide stain. AO/EtBr staining confirmed our quantitative apoptosis analysis findings, showing that Shilajit-treated KB-1 cancer cells had the greatest rate of cell death, characterized by nuclear deformation and cell wall integrity loss (Figure 5).
Several signaling molecules govern apoptosis, a highly regulated cell death process. Cell cycle regulation and cancer prevention depend on p53 (mutant protein). Apoptosis of p53 controls cancer progression (Timofeev, 2021). This regulates the Bcl-2 family, pro and antiapoptotic proteins. Several kinds of cancer are connected to p53 gene alterations. Such changes may disrupt normal apoptotic pathways, enabling injured cells to avoid scheduled cell death. Damaged cells’ abnormal survival and proliferation contribute to cancer growth and progression (Sluyser, 2002). The structurally linked Bcl-2 family regulates programmed cell death by promoting or inhibiting apoptosis. Cellular homeostasis depends on the delicate balance between Bcl-2 family members such as Bax, Bad, Bcl-2, Bcl-xL, and Mcl-1 (Kashyap et al., 2021). Death stimuli cause the proapoptotic monomeric Bax to alter the shape in the cytoplasm, causing mitochondrial permeability transition (Qian et al., 2022). Antiapoptotic proteins including Bcl-2, Bcl-xL, and Mcl-1 inhibit intrinsic apoptotic signaling. This is done by suppressing Bax and Bak oligomerization. Dysregulation of apoptosis contributes to cancer development. In KB-1 cells, Shilajit treatment significantly boosted Bax mRNA expression and decreased Bcl-2 gene expression. Thus, Shilajit may promote intrinsic KB-1 cell death.
The term “cancer migration” describes the movement of cells to different sites, which is important for tumor invasion and metastasis (Seyfried, 2012). Contiguous and intertwined structures (muscles, mucosa, ligaments) around the oral cavity affect adjacent structures immediately compared to an organ’s cancer. Even if the location of the cancer is external, the local spread of the cancer can be abrupt and fast, involving immediate adjacent structures (Mattoo et al., 2014). Our results showed that Shilajit can prevent cancer cells from migrating and that the amount given impacts this capacity. This finding is consistent with other studies that highlighted Shilajit’s ability to inhibit cancer cells from migrating (Barouji et al., 2020; Pant et al., 2016). Cancer cells have the potential to separate from the primary tumor, alter their cytoskeleton, and exhibit alterations in adhesion properties (Yilmaz & Christofori, 2009). They are able to enter adjacent organs as a result of their migratory capacity. It is essential to investigate migration and metastasis in cell culture to find possible treatment targets and develop anticancer tactics. It is also essential to add research using animal models and clinical studies to cell culture studies to completely understand cancer migration’s complexities. To comprehend how Shilajit affects cancer cell migration toward the scratched region, we used a scratch test in our study. Our findings suggest that Shilajit may be useful in treating oral cancer by successfully preventing cancer cells from migrating. Protease expression is commonly elevated in cancer cells to help them migrate via extracellular matrix breakdown (Wolf & Friedl, 2011). This mechanism is crucial to tumor spread across different malignant cancers (Li et al., 2023). The protease uPA is crucial to this process, as well as cell migration and metastases (Zeitlmayr et al., 2023). uPA receptor binding favors plasminogen-to-plasmin transformation. Plasmin activates metalloproteinases to break down extracellular matrix protein, increasing cancer cell invasion. High uPA levels and its inhibitor (PAI-1) are linked to oral cancer aggressiveness and poor outcomes (Yu et al., 2021). In the current study, Shilajit-treated OCC lines had considerably lower uPA and uPAR mRNA expression. This implies that Shilajit may modulate cancer cell invasion proteases, explaining its antimetastatic characteristics. Chemokines, a class of chemoattractant cytokines, are important in tumor creation and metastasis, making them prospective targets for anticancer therapy (Hundsdorfer et al., 2005). Primary tumor and stromal cells generate these chemicals in local and distant metastatic microenvironments. Chemokines, along with their respective receptors, affect breast cancer development, angiogenesis, invasion, and metastasis due to their extensive distribution (Najafiyan et al., 2024). The upregulation of chemokines relative to normal cells in tumor biology implies that targeting them might be “magic bullets.” This method may specifically target cancer cells and metastases while preserving nonmalignant cells. CXCL8, a proangiogenic chemokine, stimulates the proliferation and migration of endothelial cells via CXCR2 and C-X-C motif chemokine receptor 1 (CXCR1) (de Miguel-Gómez et al., 2020). In vitro growth, motility, invasion, matrix metalloproteinase (MMP)-9, and vascular endothelial growth factor (VEGF) production are increased by CXCL8. Oral cancer commonly expresses high CXCL8 and VEGF. Our investigation found that Shilajit administration dramatically decreased CXCL8 mRNA and CXCR2 expression in treated cells. Shilajit targets several angiogenic factors to inhibit OCC migration, as seen by this drop in CXCL8 and CXCR2 mRNA expression. Shilajit also boosts the immune system to produce more cytokines through the activation of immunological cells, which maintain the cell integrity of normal cells (Verma et al., 2016). Based on this study’s result, Shilajit has the potential to decrease the migration of OCCs. Other well-known mechanisms of Shilajit to cause cancer cell apoptosis are its antioxidant and anti-inflammatory properties (Barouji et al., 2020). Moreover, based on these properties, it could be interesting to test its experimental combinations with other preventive therapies in treating inflammatory diseases of the mouth, like periodontitis.
Strength and limits of the study: Based on our study findings, it can be concluded that Shilajit represents a promising molecular targeted therapy approach for challenging oral cancers. To strengthen our findings, randomized controlled clinical trials are required to ascertain the therapeutic potential of Shilajit. Shilajit comprises a diverse array of both organic and inorganic constituents. The limitation of this study is the inability to isolate the bioactive molecule that is responsible for the observed effect. Several chemical constituents in Shilajit pose challenges in determining its specific anticancer mechanisms. While the comprehension of cellular pathways may be achieved by in vitro experimentation, the human body’s complexity presents additional challenges, which is a further limitation of this study. Exercising care is necessary when attempting to extrapolate human in vivo reactions from in vitro studies. An in vitro study may demonstrate the presence of anticancer properties; nevertheless, the underlying processes responsible for these effects may remain ambiguous. The development of customized pharmaceuticals requires a comprehensive comprehension of the underlying systems involved. In order to assess the efficacy of Shilajit, it is necessary to do a comparative analysis with conventional chemotherapeutic medications as well as other naturally occurring compounds.
Conclusion
Shilajit, a commonly utilized remedy in Asia for various health concerns, has exhibited promising proapoptotic and antiproliferative properties in our investigation. Our study reveals that Shilajit may impede the migration of OCCs by controlling the expression levels of both proapoptotic and antiapoptotic genes. Specifically, our findings indicate that Shilajit targets uPA/uPAR and chemokines gene expression, suggesting a probable mechanism for inhibiting OCC migration. This study is the first to provide evidence that Shilajit regulates proliferation, apoptosis, and cell migration in OCCs, as observed in our in vitro experiments. However, it is crucial to note that clinical applications of Shilajit require further exploration through in vivo research to elucidate its specific anticancer mechanisms. Such investigations could pave the way for developing novel Shilajit-related drugs with potential therapeutic benefits in the prevention and treatment of oral cancer.
Abbreviations
AO/EtBr: Acridine orange/ethidium bromide; Bcl-2: B-cell lymphoma 2; CCR2: C-C chemokine receptor type 2; CXCL8: C-X-C motif chemokine ligand 8; CXCR2: CXC chemokine receptor 2; DCFH-DA: 2′,7′-dichlorodihydrofluorescein diacetate; DMEM: Dulbecco’s Modified Eagle Medium; FITC: Fluorescein isothiocyanate; hGFs: Human gingival fibroblasts; IC50: Half-maximal inhibitory concentration; MTT: 3-[4,5-Dimethyl-2-thiazolyl]-2,5 diphenyl-2H-tetrazolium bromide; PBS: Phosphate-buffered saline; ROS: Reactive oxygen species; uPA/uPAR: Urokinase-type plasminogen activator (uPA) and its receptor (uPAR); PCR: Polymerase chain reaction.
Footnotes
Acknowledgments
The authors would like to acknowledge the Department of Pharmacology, Saveetha Dental College, Hospitals, and Administration for their assistance with providing the necessary infrastructure and laboratory facilities. The authors would like to express their gratitude to Lajit Gold, USA for generously providing a valuable source of Shilajit that greatly contributed to the successful completion of our research.
Authors’ Contribution
Conceptualization, A.A, J.H., and H.Z.; methodology, J.H., H.Z., I.K., and E.P.; investigation, R.M., A.A.M.A, S.A.A., N.Z.A.H., and E.P.; data curation, A.A, H.Z., I.K., T.A.H., and K.M.; visualization, A.A, T.A.H., J.H., and A.A.M.A; writing—original draft, A.A, J.H., T.A.H., E.P., K.M., I.K., A.A.M.A, S.A.A., and R.M.; writing—review and editing, J.H., R.M., K.M., N.Z.A.H. All authors have read and given their approval for the final version of the article.
Data Availability Statement
All analytical data have been included in the manuscript. The raw datasets generated during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The study was duly approved by the Research Ethics Committee, College of Dentistry, King Khalid University (Vide Reg No.: IRB/KKUCOD/ETH-W/2023-24/002).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
