Abstract
Background
Ghrelin in the gastric mucosa of rats can improve myocardial cell apoptosis and cardiac remodeling during myocardial ischemia and myocardial infarction, and it has a protective effect on the myocardium, but its mechanism is unclear.
Objectives
The rat myocardial ischemia/reperfusion (I/R) model was established to investigate Ghrelin’s role in the disease’s pathogenesis.
Materials and Methods
The study subjects were 80 male and female Sprague-Dawley (SD) rats: Sham operation group, I/R model control group, I/R + Ghrelin group, and I/R + Ghrelin + 5-hydroxydecanoic acid salt (5-HD) group. Before surgery, rats in the I/R + Ghrelin group were given Ghrelin 100 µg/kg intravenously, and those in the I/R + Ghrelin + 5-HD group were given 100 µg/kg and 5-HD 5 mg/kg. An I/R model was made, and a cardiac ultrasound index was detected after reperfusion. The venous blood of rats in each group was taken, with the values of the myocardial enzyme creatine kinase (CK), troponin I (cTnI), and so on. Malondialdehyde (MDA) and adenosine triphosphate (ATP) were measured from the myocardial tissues of rats. Mitochondrial protein was extracted from the ischemic myocardium cells of rats in each group, and the content of Cx43 protein in the mitochondria was detected.
Results
After reperfusion, it was found that left ventricular developed pressure (LVDP), left ventricular ejection fraction (LVEF), and left ventricular fractional shortening (LVFS) in the echocardiogram of the I/R model control group were significantly decreased, and the difference was statistically significant compared with the sham operation group (p < 0.001). LVDP, LVEF, and LVFS in the I/R + Ghrelin group were significantly higher than those in the I/R model control group, and the difference was statistically significant (p < 0.001). At the end of reperfusion, myocardial troponin and other data, as well as MDA content in the peripheral blood of the control group, were different from those in the sham operation group (p < 0.05). The former ATP value was lower than the latter (p < 0.05). The CK, CK-myocardial band (CK-MB), and cTnI values in the I/R + Ghrelin group were lower than those in the good group (p < 0.05). Mitochondria were isolated from rat myocardial cells in each group. After reperfusion, Cx43 data in the control group were lower than those in other groups. After pretreatment with Ghrelin, related expressions were unchanged. After 5-HD was added, the expression of Cx43 decreased slightly, but it was not obvious. The content of MDA in the I/R + Ghrelin + GHRP-6 group was significantly increased, which was not significantly different from that in the I/R + Ghrelin + 5-HD group. Still, the expression was reversed considerably compared with that in the I/R + Ghrelin group, and the difference was statistically significant (p < 0.001). After the intervention of GHRP-6, ATP content in the myocardial tissue of rats in the I/R + Ghrelin group was also reversed, and the difference was statistically significant (p < 0.05). By western blot detection, it was found that after the intervention of GHRP-6 in the rats pretreated with Ghrelin, the expression level of Cx43 in the myocardial mitochondria of the rats was significantly decreased. Still, the difference was insignificant compared to that in the I/R + Ghrelin + 5-HD group. Ghrelin protein reduces the mitochondrial ATP value, and reperfusion can maintain the stability of energy metabolism of myocardial cells and protect them. Ghrelin protein protects the myocardium by maintaining the expression of Cx43. Ghrelin protein can maintain the expression of Cx43 by increasing the activity of the mitochondrial ATP-sensitive potassium channel, thus playing a protective role in cardiomyocytes to a certain extent.
Keywords
Introduction
Coronary atherosclerotic heart disease, also referred to as coronary atherosclerotic disease (CHD), refers to the sclerosis of the coronary arteries or the accumulation of lipids in the intima of the coronary arteries, resulting in vascular obstruction and stenosis, which can lead to the occurrence of myocardial deficiency or hypoxic disease (Lazdunski, 1994). Acute myocardial infarction is one of the serious types. It is the acute ischemic necrosis of the myocardium caused by coronary artery obstruction, which is the main cause of sudden cardiac death. Studies by scholars have found that this phenomenon is mostly caused by myocardial ischemia/reperfusion (I/R) injury (Lemos Caldas et al., 2015). Myocardial I/R injury means a blockage in a coronary artery blood flow and ischemia of the myocardium, which can damage a part of the myocardial tissue’s ultrastructure, function, and metabolism, causing electrophysiological further changes (Wang et al., 2014). The mechanism of its occurrence is relatively complicated. The relevant mechanism after reperfusion was studied to explore effective methods of myocardial protection to improve the malignant outcome of clinical patients. The mitochondrial adenosine triphosphate-sensitive potassium channel (mitoKATP) is a K+ inward-rectifying ion channel located on the mitochondrial membrane. It stabilizes mitochondrial energy metabolism, reduces Ca2+ overload in mitochondria, and inhibits the generation of oxygen free radicals (Zhou et al., 2013). Cx43 is a connexin, mainly in the myocardial tissue of the nonconducting system (Mühlfeld et al., 2010), and it is the main component of the gap connection. Recent studies have found that Cx43 is also one of the effector targets to protect the function of myocardial cells, and it has a protective effect on the myocardium during ischemic post-treatment (Chen et al., 2014). Several studies have shown that the Cx43 protein located on the mitochondrial membrane is involved in myocardial protection during ischemic preconditioning (IPC) and ischemic post-processing (IPOC) (Rankinen et al., 2015). The opening of the mitoKATP channel can maintain the production of ATP by mitochondria, which helps maintain the phosphorylation state of the Cx43 protein (Bainey & Armstrong, 2014). In recent years, the Ghrelin protein found in the gastric mucosa of rats has been associated with many diseases, which can improve myocardial cell apoptosis and cardiac remodeling during myocardial ischemia and myocardial infarction and has a protective effect on the myocardium, but its mechanism is not clear (Matsushima et al., 2014). Using the rat myocardial I/R model in the system, a preliminary study of Ghrelin in myocardial I/R exists in the process of protection, and through the experiment in the pretreatment for mitoKATP-specific inhibitors of 5-hydroxydecanoic acid salt (5-HD), whether the preliminary study shows that mitoKATP and Cx43 play a role.
Materials and Methods
Experimental Animals
The subjects were 80 Sprague-Dawley (SD) rats, half male and half female, purchased from the Shanghai Slack Experimental Animal Technology Service Department. Body weight (250 ± 20 g), kept at the Experimental Animal Center of our hospital, humidity 50 ± 10%, temperature 24°C, and 12-hour day and night rotation. Breeding conditions are specific pathogen free (SPF), with clean and disinfected drinking water, ordinary rat feed, and raised according to relevant regulations.
Animal Model Grouping and Administration
SD rats fed for 1 week were randomly divided into four groups according to the blind selection method: I/R model control group (n = 20), sham operation group (n = 20), myocardial I/R + Ghrelin group (I/R + Ghrelin group, n = 20), and myocardial I/R + Ghrelin + 5-HD group (I/R + Ghrelin + 5-HD, n = 20). The rats in the Ghrelin group were given 100 µg/kg of Ghrelin intravenously 10 minutes before surgery. The rats in the Ghrelin + 5-HD group were given 100 µg/kg + 5-HD 5 mg/kg of Ghrelin intravenously. The rats in the I/R model control group were given the same dose of normal saline. Rats in the myocardial I/R + Ghrelin group were further randomly divided into two groups: One group received normal Ghrelin intervention, and the other group was fed 30 mg/kg of Ghrelin receptor antagonist (GHRP)-6 (I/R + Ghrelin + GHRP-6 group). In the intervention process, the researchers and the animal handlers involved in the experiment agreed with the double-blind principle on the dose and intervention method to reduce the subjective bias of the research results.
Animal Model Construction
The incision was located in the third intercostal space, 0.5 cm from the left edge of the sternum, and the skin tissue, with a length of about 1.5 cm, was cut longitudinally. The subcutaneous fascial tissue was bluntly separated, and the small intercostal outer muscle blood vessels were clamped with vascular forceps. Use small, curved forceps to pass the double-strand ligature through the third intercostal space, bluntly separate the intercostal artery of the third intercostal space, and ligate it at both ends of the third rib. After the ligation, the third rib was cut in the middle of the ligature at both ends, and the pleura and pericardium were separated and opened in layers, one by one. After confirming the position, it was given to reduce expenditure. Use a 3/8-gauge suture needle to close the myocardium’s surface and observe the electrocardiogram (ECG) changes. After it has stabilized, continue to observe for 15 minutes. If there is no obvious change, place a cotton thread on the surface of the rat heart to be ligated, ligate the rat with live ligation, and then treat the descending branch. At this time, ECG monitoring showed that the QRS complex of lead II suddenly increased and widened, the ST segment showed a dorsal archup type, the color of myocardial tissue in the corresponding area of the heart surface became darker, and myocardial pulsation weakened, indicating successful ischemic modeling. After 30 minutes, the ST segment of lead II decreased by 1/2 to show myocardial ischemia, and the color gradually turned red after the resumption of blood perfusion.
Cardiac Ultrasound Index Detection
Rats were given peritoneal anesthesia with 1 mL of 10% chloral hydrate. At the same time, echocardiography of the papillary muscle section was obtained with an X5 VET small animal ultrasound instrument. The probe frequency was controlled at 40 MHz, and the indexes of left ventricular developed pressure (LVDP), left ventricular ejection fraction (LVEF), and left ventricular fractional shortening (LVFS) were recorded.
Creatine Kinase (CK), CK-Myocardial Band (CK-MB), and Troponin I (cTnI) Detection
The biochemical kit was used to prepare samples according to the instructions. Rat venous blood was measured before and after reperfusion, and an automatic biochemical analyzer (Roche Cobas C501) detected the content of CK-MB, CK, and cTnI in whole plasma.
Determination of Malondialdehyde (MDA) Content in Myocardial Tissue
Prechilled normal saline; remove the heart, absorb the filter paper, weigh it with water, and homogenize it at a low temperature. Mass volume of 1:9; centrifuge at 1,000 rpm for 15 minutes at 4°C. After the sample preparation is completed, the protein concentration is measured by the bicinchoninic acid (BCA) method. A standard curve is plotted on the ordinate to represent the absorbance value; the abscissa is the concentration of the standard, and the concentration calculation equation is calculated.
Determination of ATP Content in Myocardial Tissue
The heart was quickly cutoff. After the model was built, it was quickly weighed. A full layer of myocardial tissue was cut from the target site. 100–200 µL of lysate was added to every 20 mg of myocardial tissue and homogenized in a glass homogenizer. The supernatant was centrifuged at 12,000 g at 4°C for 5 minutes, and the ice was cleaved for 10 minutes. The supernatant was collected and placed at 636 nm on the micrometer to measure the absorbance.
Extraction of Mitochondria from Myocardial Tissue and Detection of Cx43 Protein Expression in Mitochondria by Western Blotting
Add an equal volume of sample buffer to the obtained mitochondrial solution to lyse the mitochondria, prepare a protein sample, unwind the protein, add protein markers and samples in a predetermined order, and separate gels and concentrated gels with corresponding concentrations. Install the electrophoresis device. Then start electrophoresis, transfer the membrane ↓ block ↓ incubate the primary antibody ↓ incubate the secondary antibody ↓ develop color ↓ imaging treatment.
Observation of Mitochondrial Changes in Rat Myocardial Cells by Transmission Electron Microscope
Heart tissue blocks were fixed overnight in 2.5% glutaraldehyde phosphate buffer ↓ rinsed with 0.1 m phosphate-buffered saline (PBS) for 15 minutes × 2 times ↓ added 1% osmium acid and fixed for 1 hour ↓ stained with 2% uranium acetate solution for 30 minutes ↓ dehydrated with alcohol, in turn, ↓ dehydrated with acetone ↓ permeated with pure embedding agent ↓ embedded with pure embedding agent ↓ modified blocks and ultrathin sections ↓ stained ↓ observed and photographed under TECNAI 10 transmission electron microscope.
Statistical Methods
Stata 23.0 statistical software was used for processing, expressed as mean ± standard deviation (x ± s). Classification data were tested by chi-square test, measurement data were compared by paired t-test, and relative expression of quantitative reverse transcription polymerase chain reaction (qRT-PCR) results was measured using 2–∆∆Ct means. p < 0.05 was considered statistically significant.
Results
Construction of a Rat Model of Myocardial I/R Injury
An SD rat model was established. A total of 80 rats were randomly divided into four groups: Sham operation group, I/R model control group, I/R + Ghrelin group, and I/R + Ghrelin + 5-HD group. In the sham operation group, anesthesia, tracheal intubation, and thoracotomy were performed according to the same surgical methods, but the anterior descending coronary artery was ligation with threading only, not ligation. The I/R + Ghrelin group was treated with 100 µg/kg of Ghrelin 10 minutes before the myocardial I/R model was constructed. The I/R + Ghrelin + 5-HD group was treated with Ghrelin and 5-HD at concentrations of 100 µg/kg and 5 mg/kg, respectively, before model construction. The I/R model control group was given the same dose of normal saline before modeling (Figure 1).
The Expression of Cardiac Ultrasound Indexes in Each Group of Rats
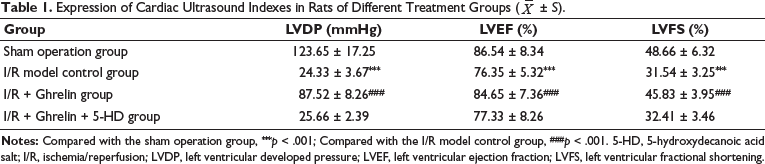
It can be seen from the experiment that LVDP, LVEF, and LVFS in the I/R model control group, after myocardial I/R treatment, were significantly decreased in echocardiography, and the difference was statistically significant compared with the sham operation group (p < 0.001). LVDP, LVEF, and LVFS treated with Ghrelin protein before I/R showed a significant increasing trend compared with the I/R model control group but did not exceed the sham operation group; the difference was statistically significant (p < 0.001). In the I/R + Ghrelin + 5-HD group, the expression of LVDP, LVEF, and LVFS was also significantly decreased, which was close to the level of the I/R model control group, and there was no statistical significance between the two groups (Table 1).
Expression of Cardiac Ultrasound Indexes in Rats of Different Treatment Groups (
± S).
CTnI Value and Other Expressions of Rats in Each Group
In this study, it can be seen that the concentration of cTnI in peripheral blood of the I/R model control group treated with myocardial I/R was significantly upregulated, which was different from that of the sham group (p < 0.001). The concentration of cTnI in the peripheral blood of rats treated with Ghrelin protein before I/R treatment was higher than that in the sham operation group (p > 0.05), but the related concentration data, such as the cTnI value, decreased compared with the previous (p < 0.05). In the Ghrelin + 5-HD group, the concentration of cTnI in peripheral blood also significantly increased, which was close to that of the I/R model control group (p > 0.05) (Table 2).
Contents of Creatine Kinase (CK), CK-Myocardial Band (CK-MB), and Troponin I (cTnI) in Peripheral Blood of Rats in Different Treatment Groups (
± S).

Expression of MDA and ATP in Myocardial Tissue of Each Group of Rats
In the sham-operated group without I/R, the MDA content was lower, at 1.86 ± 0.32 nmol/mgprot. The content of ATP was higher, at 238.59 ± 77.25 mol/gprot, and the branch was ligated, which caused ischemia to restore the blood supply for 30 minutes. It could be found that the content of MDA increased significantly to 2.37 ± 0.23 nmol/mgprot. The content of ATP decreased significantly to 56.68 ± 17.17 mol/gprot, which was different from the data in the sham operation group (p < 0.05). In the rats pretreated with Ghrelin before I/R treatment, the MDA content did not increase significantly compared with the sham operation group (p > 0.05); the content was 1.74 ± 0.18 nmol/mgprot. The MDA content was lower than that of the control group (p < 0.05). The data related to the ATP content of I/R + Ghrelin increased compared with the control group (p < 0.05) and decreased compared with the sham operation group (p > 0.05). After pretreatment with ion channel-specific inhibitors, it was found that after blocking the opening of ATP-sensitive K+ channels on the mitochondrial membrane, the related MDA content was higher than that of the sham group (p < 0.05) but was less obvious than that of the control group (p > 0.05) (Table 3).
Contents of Malondialdehyde (MDA) and Adenosine Triphosphate (ATP) in the Myocardium of Rats in Different Treatment Groups (
± S).

The Expression of Cx43 in the Mitochondrial Protein of Rat Myocardium in Different Treatment Groups
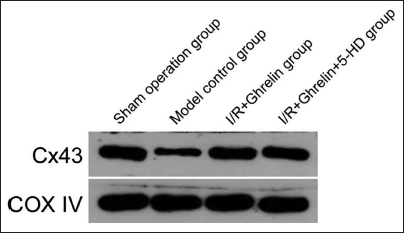
Cx43 expression was present in the myocardial mitochondrial proteins of rats in the sham-operated group that did not undergo I/R treatment. The expression of Cx43 in myocardial mitochondrial proteins of the I/R model control group rats after I/R treatment was significantly down. Before the I/R treatment, the expression level of Cx43 in the myocardial mitochondria of rats pretreated with Ghrelin protein was restored. The expression in the model treatment group was significantly increased compared with the sham operation group (p > 0.05). In rats given the Ghrelin protein and the mitochondrial ATP-sensitive K+ channel blocker 5-HD group, the expression of Cx43 in myocardial mitochondria protein was not significantly downregulated (Figure 2).
Electron Microscopy of Myocardial Mitochondria in Rats of Different Treatment Groups
Table 4 and Figure 3 show the electron microscopy performance of myocardial mitochondria in different treatment groups. In the model group, many mitochondrial ridge structures were broken and missing. After Ghrelin and combination treatment, mitochondrial crest structure breakage and loss were reduced.
The Results of Ultrastructure Observation of Different Groups of Rat Heart Tissue under Electron Microscope.


The Content of MDA and ATP in Myocardial Tissue and the Expression of Cx43 in Myocardial Mitochondrial Protein in Different Ghrelin Expression Intervention Groups
To further understand the role of Cx43 and mitoKATP in the Ghrelin effect, GHRP-6 intervention was further performed in rats with the myocardial I/R + Ghrelin intervention group, and it was found that MDA content in the I/R + Ghrelin + GHRP-6 group was significantly increased. There was no significant difference between the I/R + Ghrelin + 5-HD group and the I/R + Ghrelin + 5-HD group, but the expression was significantly reversed compared with the I/R + Ghrelin group, and the difference was statistically significant (p < 0.001). At the same time, ATP content in myocardial tissue of rats in the I/R + Ghrelin group was also reversed after GHRP-6 intervention; the difference was statistically significant (p < 0.05), and it was close to that in I/R + Ghrelin + 5-HD group; the difference was not significant (Table 5). At the same time, protein western blotting showed that after the intervention of GHRP-6 in the rats pretreated with Ghrelin, the expression level of Cx43 in the myocardial mitochondria of the rats was significantly decreased, but the difference was not significant compared with that in the I/R + Ghrelin + 5-HD group (Figure 4).

Malondialdehyde (MDA) and Adenosine Triphosphate (ATP) Contents in Myocardial Tissue of Rats in Different Ghrelin Expression Intervention Groups.

Discussion
Myocardial I/R injury refers to the process that occurs after myocardial ischemia, when the obstructed coronary arteries are recanalized. After the myocardial blood supply is restored, due to the massive generation of oxygen free radicals, the imbalance of energy metabolism, and the destruction of the balance of vasoactive substances, the myocardial ultrastructure is changed, and irreversible damage is formed (Wu et al., 2015). It is an urgent problem to study the mechanism of the injury and explore the feasible treatment of myocardial protection.
CK and similar enzymes belong to the myocardial enzyme spectrum. CK is widely present in the cytoplasm and mitochondria of muscle cells, such as skeletal muscle, cardiac muscle, brain tissue, prostate tissue, and the uterus, and is related to ATP formation (Hwang et al., 2015). In this experiment, CTnI in the peripheral blood of rats was used as a lateral indicator to evaluate the area and severity of myocardial infarction. The body produces free radicals, which can attack fatty acids through lipid peroxidation. MDA is a more important substance in the cell and is used as the final product of lipid peroxidation (Wang et al., 2015). ATP is the body’s most important ability to supply molecules. It has a greater role in cells and is related to developing the patient’s condition (Heusch et al., 2014). In this study, MDA content in the myocardial tissue of rat models from different treatment groups was determined as an assessment of the oxidative stress response level in rats, and ATP content in myocardial tissues was determined as a way to understand changes in mitochondrial function and energy metabolism (Liu et al., 2014). Gap junctions are intercellular channels that mediate intercellular communication and communicate the cytoplasmic interactions of adjacent cells. They affect the regulation of intracellular Ca2+, ATP, and NAD+ levels (Chouchani et al., 2014). Ca2+ overload, acidosis, and other phenomena during myocardial I/R lead to decreased expression of phosphorylated Cx43, increased levels of dephosphorylated Cx43, and decreased electrical coupling between cells, which are also some of the causes of severe ventricular arrhythmias (Rodrigo et al., 2013).
Ghrelin is produced by the glands that secrete gastric acid at the bottom of the stomach and has an important protective effect on the cardiovascular system. It is mainly reflected in protecting the function of vascular endothelial cells, inhibiting the inflammatory response of the vascular wall, and inhibiting the formation of lipid plaques in the arterial wall. Reducing I/R injury, inhibiting the occurrence of ventricular arrhythmias after ventricular myocardial infarction, ventricular remodeling, and so on, but the specific molecular mechanism of their roles is still not very clear (Ma et al., 2015; Morel et al., 2014). In the model, we observed that the blood supply was restored when the coronary artery was ligated 30 minutes later, which could cause significant myocardial damage. It is manifested as myocardial ischemia on the ECG and arrhythmia with ventricular premature contraction. The indexes of myocardial enzymes in peripheral blood increased, and the level of troponin cTnI increased, suggesting that myocardial infarction persisted. However, the MDA level in myocardial tissue was significantly upregulated, suggesting that myocardial cells had a serious oxidative stress reaction, lipid peroxides were increased, and the energy metabolism of myocardial cells was impaired. ATP content significantly decreased. The process of I/R causes the generation of a large number of oxygen free radicals and the energy barrier of cardiomyocytes, resulting in damage to cardiomyocytes (Guan et al., 2015). However, in the rat model pretreated with Ghrelin before, the MDA content and ATP level in the myocardial tissue were significantly lower than those in the I/R model control group. The content of ATP was significantly higher than that of the I/R model control group, indicating that the pretreatment of Ghrelin protein reduced the oxidative stress response of myocardial tissue and the production of reactive oxygen species, reduced the cell’s energy metabolism disorder, and maintained the normal ATP level. Therefore, in the peripheral blood of rats in this group, we also observed that CK, CK-MB, and cTnI levels were significantly lower, suggesting that myocardial damage was significantly reduced and that Ghrelin protein had a significant protective effect on cardiomyocytes.
To preliminarily understand the mechanism by which Ghrelin protein protects cardiac myocytes, we conducted a preliminary study on the effect of Ghrelin protein on Cx43 and whether it plays a role through the mitoKATP pathway (Brennan et al., 2015). We used the blocker 5-hydroxydecanoate (5-HD) of the mitoKATP channel to block the mitoKATP channel and pretreated rats with Ghrelin protein, which can cause I/R. MDA content increased, and ATP content decreased in rat myocardial tissue. Peripheral blood cTnI was elevated. This result suggests that after blocking the mitoKATP channel, the protective effect of the Ghrelin protein on the myocardium is reversed. Meanwhile, mitochondrial proteins of rat cardiomyocytes in different treatment groups were isolated and extracted, and the expression of Cx43 protein was detected by western blot. The results showed that the expression of Cx43 in the myocardial mitochondria of rats treated with I/R was significantly decreased, indicating that the I/R process caused damage to myocardial cells by downregulating the expression of Cx43. However, after pretreatment with Ghrelin protein and then receiving I/R treatment, the expression of Cx43 did not change significantly, suggesting that Ghrelin protein has the effect of stabilizing Cx43 expression and exerts a protective effect on myocardial cells through this pathway. To further understand the role of Cx43 and mitoKATP in the Ghrelin effect, GHRP-6 was added to the rats pretreated with Ghrelin protein for intervention, and the results showed that MDA and ATP contents in myocardial tissue of rats were effectively reversed. It is also close to the expression of mitoKATP after blocking the channel. At the same time, we also found the same effect when detecting Cx43 protein expression by western blot, suggesting that Ghrelin protein can maintain the expression of Cx43 by improving the activity of the mitochondrial ATP-sensitive potassium channel, thus playing a protective role in cardiomyocytes to a certain extent.
Conclusion
Ghrelin protein reduces the generation of oxygen free radicals by increasing the activity of mitochondrial ATP-sensitive potassium channels, maintains the stability of energy metabolism of myocardial cells, and has a protective role in myocardial I/R. Ghrelin protein protects the myocardium during myocardial I/R by maintaining the expression of Cx43. Ghrelin protein can maintain the expression of Cx43 by increasing the activity of the mitochondrial ATP-sensitive potassium channel, thus playing a protective role in cardiomyocytes to a certain extent.
Footnotes
Abbreviations
5-HD: 5-hydroxy decanoicacid salt; ATP: Adenosine triphosphate; CK: Creatine kinase; cTnI: Troponin I; cx43: Connexin 43; ECG: Electrocardiogram; I/R: Ischemia/reperfusion; IPC: Ischemic preconditioning; IPOC: Ischemic post-processing; GHRP: Ghrelin receptor antagonist; LVDP: Left ventricular developed pressure; LVEF: Left ventricular ejection fraction; LVFS: Left ventricular fractional shortening; MDA: Malondialdehyde.
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the ethics committee of No. 906 Hospital of the Joint Logistic Support Force of PLA and the ethics committee of Wenzhou Yining Geriatric Hospital.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
