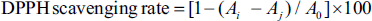Abstract
Background
Finding a new promising plant antioxidant to combat or prevent cardiovascular disease remains a major challenge for researchers. Lychee pericarp, which includes a variety of polyphenols, is a prospective source of botanical antioxidants.
Objectives
In response to this issue, we assessed the effect of lychee bark extract on oxygen radical scavenging capacity and separate vascular protective ability in vivo.
Materials and Methods
In the current study, the lychee pericarp enrichment product (RLP) was obtained by optimizing an extraction method and separated using modern separation methods. The structure of the isolated compounds was assessed using 1H-NMR, 13C-NMR, and electrospray ionization-mass spectrometry, and the content of polyphenols obtained from the extract was quantified using the area normalization method. In addition, the capacity of RLP to scavenge oxygen radicals in vitro was assessed using the DPPH (1,1-diphenyl-2-picrylhydrazyl) and T-AOC (total antioxidant capacity) techniques. Furthermore, an isoprenaline hydrochloride (ISO)-induced rat model of acute myocardial ischemia was established, and four biochemical indicators were used to assess the effect of lychee bark extract on oxygen radical scavenging capacity and cardiovascular protective ability in vivo.
Results
RLP extraction produced 11 chemicals, 6 of which were structurally assessed. Quantitative analysis revealed that epicatechin, PC-C(epicatechin-(4β↓8,2β↓O↓7)-epicatechin-(4β↓8)-epicatechin), type A proanthocyanidin trimer, proanthocyanidin A2, and type B proanthocyanidin dimer were the most abundant polyphenols. The in vitro DPPH and T-AOC findings revealed that RLP exhibited strong free radical scavenging activity, approximately 1.5-fold that of vitamin C. The four biochemical indicators confirmed the antioxidant activity and cardiovascular protective ability of RLP in vivo.
Conclusion
RLP has good in vivo and in vitro antioxidant activity, confirming lychee pericarp as a prospective source of botanical antioxidants.
Introduction
The global yearly output of lychee is 4.41 million tonnes, producing a substantial amount of lychee pericarp (approximately 30% of the total weight of lychee) during its deep processing (Jiang et al., 2013; Kilari & Putta 2016; Ruenroengklin et al., 2008; Zhang et al., 2013; Zhao et al., 2020). The ability to employ modern science and technology to utilize lychee trash and transform it into treasure has enormous social and economic potential. Polyphenols, found in the skin, roots, leaves, and fruits of plants, are a type of secondary metabolite that can protect lipids, proteins, enzymes, carbohydrates, and DNA against oxidative damage (Chu, 2022; Joseph et al., 2016; Kilari & Putta 2016; Majidinia et al., 2019; Rajha et al., 2022; Tresserra-Rimbau et al., 2018).
The rind of the lychee fruit contains many polyphenol compounds with hypoglycemic, hypolipidemic, antioxidant, anti-bacterial, anti-inflammatory, and other biological activities. Moreover, it has great potential to be utilized in the development of healthcare products, cosmetics, pharmaceutical products, and other effective products (Contreras-Castro et al., 2022; Ibrahim & Mohamed, 2015; Kumar et al., 2015; Queiroz et al., 2018; Wen et al., 2023; Zeng et al., 2019). Lychee is rich in polyphenols, with a level as high as 170 mg/100 g, which is greater than that detected in pineapple, banana, and other tropical fruits and second only to passion fruit (Gong et al., 2018).
Cardiovascular disease is one of the most serious diseases threatening human beings in today’s world, and according to statistics, its morbidity and mortality rates have exceeded those of oncological diseases and ranked first in the world, while myocardial ischemia is one of the diseases with the highest mortality rates among cardiovascular diseases. When myocardial ischemia occurs, some cardiac biomarkers with high specificity and sensitivity will diffuse into the blood, such as creatine kinase (CK), lactate dehydrogenase (LDH), cardiac troponins (cTns), and myoglobin (MYO) (Dreyfus et al., 1960; Rosalki et al., 2004) These cardiac biomarkers reflect heart disease and contribute to early diagnosis, clinical stratification, and prognosis evaluation of myocardial injury, especially acute myocardial infarction (AMI). In addition, studies on the etiology of myocardial ischemia have found that the excessive accumulation of reactive oxygen species (ROS) leads to the apoptosis of cardiomyocytes (Incalza et al., 2018). Therefore, in recent years, from the perspective of ROS, researchers have been looking for suitable treatments to reduce ROS production, block the further activation of oxidative stress, and provide new means and methods for the treatment of cardiovascular disease. Compounds such as proanthocyanidins and kaempferol in lychee pericarp are polyphenols widely present in plants, which can be used as natural antioxidants to clear the reactive oxygen radicals in the human body and play the role of antioxidants (de Pascual-Teresa, 2014).
Materials and Methods
Extraction of Phenolic Compounds from Lychee Pericarp
The lychee pericarp was subjected to polyphenol extraction and enrichment using the technique of Zhu et al. (2019) and pre-experiment. Dried and crushed lychee fruit pericarp (cv. Black Leaf, collected in Guangdong Dianbai on 20 June, 2023) was placed in a 24 mesh and refluxed at 60°C twice for 93 min per cycle, before freeze-drying to obtain a polyphenol crude extract powder. The crude extract sample solution was pretreated on an AB-8 macroporous resin chromatography column (10 × 100 cm) at a concentration of 0.2 g mL−1; the flow rate of the sample solution was set at 6 BV h−1. The eluent was collected for freeze-drying to obtain the lychee pericarp enrichment product (RLP).
Composition Properties of Polyphenolic Compounds
The Folin–Ciocalteu phenol reagent was used to determine the total polyphenol content of RLP. In brief, 80 µg mL−1 gallic acid standard reserve solution was prepared and diluted to 8.06, 16.09, 24.06, 40.30, 48.20, and 80.09 µg mL−1 working solutions. The working solution (1 mL) was added to a measuring flask, followed by the addition of 1 mL distilled water and 0.5 mL Folin–Ciocalteu reagent. The mixture was vortexed for 8 min, which was followed by the addition of 1.5 mL saturated Na2CO3 and vortexing for 2 h. Using a spectrophotometer, the absorbance of the final solution was determined at 760 nm. The gallic acid content was used as the horizontal coordinate and the absorbance value was considered the vertical coordinate to set a linear regression equation. The absorbance data were entered into the linear regression equation to determine the concentration of RLP polyphenols in different batches using the abovementioned approach.
First, RLP was diluted in methanol to a working solution of 1 µg mL−1, which was then analyzed using ultra-performance liquid chromatography–mass spectrometry (UPLC–MS) under the following conditions: ACQUITY UPLC® HSS T3 (2.1 × 100 mm; 1.8 µm); column temperature, 35°C; mobile phase, 1% formic acid (A) and acetonitrile (B); gradient elution; detection wavelength, 280 nm; and flow rate: 0.3 µg mL−1.
Efficacy and Evaluation of Antioxidant Activity in vitro
The radical-scavenging activity of 1,1-diphenyl-2-picrylhydrazyl (DPPH) was determined using the technique of La et al. (Baliyan et al., 2022; Iordănescu et al., 2021). A stock solution of 240 mg L−1 RLP was prepared using anhydrous ethanol as the solvent. The stock solution was diluted to 4.80, 9.61, 19.23, 28.87, 38.46, and 48.07 mg L−1 working solutions. To 1 mL RLP test solution, 3 mL DPPH reagent was added, mixed, and incubated for 30 min in the dark, before measuring the absorbance at 517 nm (Ai). Ethanol was used to replace the DPPH reagent as the blank, which was recorded as Aj. Ethanol was used to replace the working solution, which was used as the control and was recorded as A0. The experiment was operated three times in parallel, and the average was taken. Furthermore, vitamin C (Vc) was considered the positive control in the same operation. The DPPH• residue in the samples was computed using a DPPH• linear regression equation, and the clearance rate was determined using the following equation:
The T-AOC radical-scavenging activity was determined using the technique of Roy et al. (2010). In this method, anhydrous ethanol was employed as a solvent to prepare a 3.02 mg L−1 RLP stock solution. The RLP stock solution was diluted to working solutions of 0.12, 0.15, 0.20, 0.30, and 0.60 mg L−1, followed by the determination of the absorbance of the different concentrations of the test solution at 520 nm according to the manufacturer’s recommendations. In addition, Vc was considered the positive control in the same operation.
Efficacy Evaluation on Myocardial Ischemia-Reperfusion Injury In Vivo
All animal experimental procedures were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and approved by the Laboratory Animal Management Committee of Guangdong Pharmaceutical University (Laboratory Animal No. SCXK (GD) 2022-0002). Male Sprague-Dawley (SD) rats (180–220 g) were obtained from the Center for Animal Experimentation and kept in plastic cages at 22 ± 2°C with free access to food and water. Sixty SD rats (males) were randomly divided into the following 6 groups of 10 rats each: control group (CG), model control group (MG), metoprolol tartrate group (PG), H-RLP group (200 mg kg−1), M-RLP group (100 mg kg−1), and L-RLP group (50 mg kg−1), the latter three being the dosage groups. Each dosage group was administered the solution once a day for 5 days via gavage.
One hour after the last injection, the rats in each group were anesthetized, placed in the supine position, and attached to an electrocardiograph to record normal electrocardiograms. Then, the rats in the model group and each dosage group received several subcutaneous injections of isoprenaline hydrochloride (ISO, 2 mg kg−1), whereas the normal control group received an equal quantity of saline. Rat electrocardiograms were recorded 1, 3, 5, 8, 10, 15, 20, 25, and 30 min after ISO treatment, and the downward shift of the J-point in the electrocardiograms at the above time points was considered ∇J (the J-point is the point where the QRS wave cluster meets the ST segment).
Blood was collected from the abdominal aorta 24 h after the last ISO dose, and serum was separated via centrifugation at 4°C and 3,000 rpm for 10 min. The serum was evaluated for CK, LDH, nitric oxide synthase (NOS) activity, and total antioxidant capacity (T-AOC) according to kit instructions.
To prepare 10% cardiac tissue homogenate, a piece of myocardium was incised, cleaned, and crushed in cold saline. Then, centrifugation was performed to obtain the homogenate supernatant. A kit was used to determine the levels of protein, superoxide dismutase (SOD), malonaldehyde (MDA), and glutathione peroxidase (GSH-Px) in the supernatant. After the rat was sacrificed, the heart was collected. After draining the blood, the apical section of the myocardial tissue was excised, fixed in neutral formalin, embedded in regular paraffin, sectioned, and stained with hematoxylin and eosin (H&E). Pathological alterations in the myocardial tissue were observed under a microscope.
Data Analysis
All results are presented as the mean ± standard deviation for the triplicate determinations of each experiment. Data were analyzed using a one-way analysis of variance, followed by the Student–Newman–Keuls test. Statistical significance was defined as p < 0.05. All statistical analyses were performed using SPSS statistical package version 13.0 (SPSS Inc., Chicago, IL, USA).
Results
Lychee Pericarp Structure Identification and Content Determination
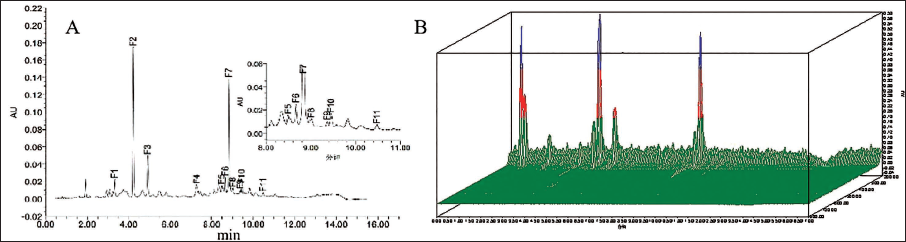
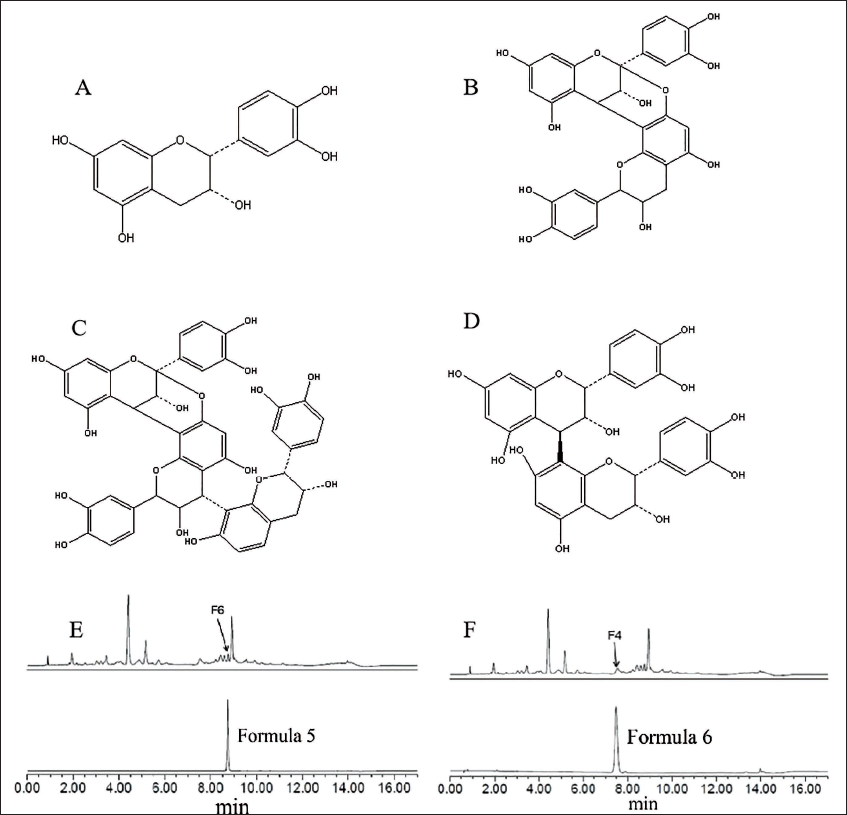
The UPLC chromatogram and 3D chromatogram of the lychee chinensis extract were obtained at 280 and 200–800 nm (Figure 1A and B), respectively, using UPLC–MS, which preliminarily identified 11 components. Under the negative ion detection mode, the comparison of the obtained selected ion chromatograms with the control peaks revealed that component F2 was epigallocatechin; components F7 and F11 were both A-type procyanidin dimers; F1 was procyanidin B2; the five fractions F3, F4, F8, F9, and F10 were all A-type procyanidin triplexes; and F5 was rutin. Silica gel chromatography and high-performance liquid chromatography (HPLC) were used for separating RLP, yielding six formulas. The structures of the six formulas were characterized using UPLC–MS, electrospray ionization-mass spectrometry (ESI-MS), and nuclear magnetic resonance (NMR) (Figure 2).
Ultra-performance Liquid Chromatography (UPLC) Chromatogram (280 nm) and 3D Chromatogram (200–800 nm) of Lychee Pericarp Enrichment Product (RLP).
Structure of Formulas 1–6. (A) (−)-Epicatechin; (B) Procyanidin A2; (C) Procyanidin B2; (D) Epicatechin-(4β↓8,2β↓O↓7)-epicatechin-(4β↓8)-epicatechin; (E) B-type Procyanidin Dimer; (F) A-type Procyanidin Trimer.
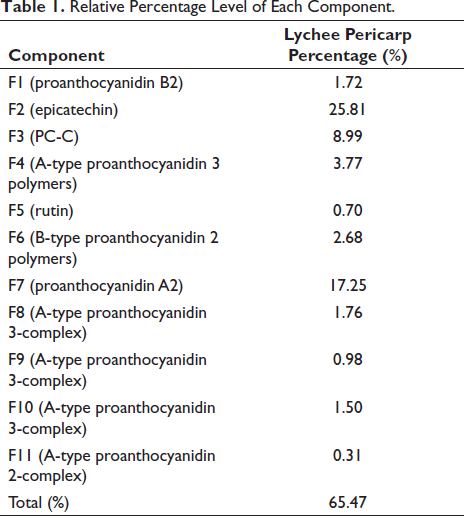
The area normalization method was used to calculate the relative percentages of the identified fractions with respect to all fractions detected at 280 nm (UPLC chromatography), and the results are shown in Table 1. The results revealed that the proanthocyanidin monomers in the lychee pericarp extract were mainly dominated by epicatechin. Oligomeric proanthocyanidins were primarily dominated by A-type proanthocyanidins, with a significant proportion of proanthocyanidin A2 and proanthocyanidin (PC) and relatively low content of B-type proanthocyanidins.
Relative Percentage Level of Each Component.
Qualitative and Quantitative Analysis of RLP
At gallic acid mass concentrations ranging from 8 to 80 mg L−1, the linear regression equation y = 0.0099x + 0.0246 (R2 = 0.9995) demonstrated satisfactory linearity. The polyphenol concentration in the three distinct batches of the lychee bark extract samples was 82.6 ± 4.12%, 81.0 ± 3.68%, and 80.4 ± 4.56% (Table 2). These findings demonstrated that the extraction and purification process completely extracted polyphenols from the lychee pericarp efficiently, separated polysaccharides and other contaminants, and produced a satisfactory purifying effect.
Polyphenols Content in Different Batches of Lychee Pericarp Extract.

Evaluation of in vitro Antioxidant Capacity
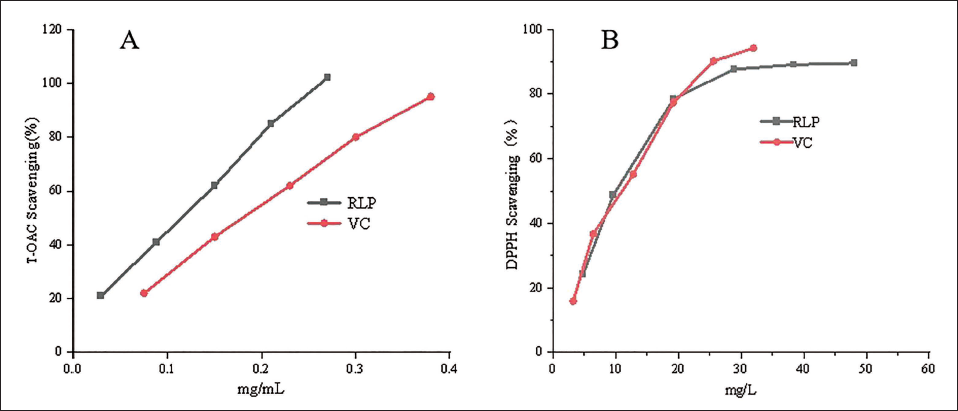
The results of the total polyphenol scavenging ability of the lychee pericarp in comparison with Vc are shown in Figure 3A. The lychee pericarp extract had a strong scavenging effect on DPPH•, and the scavenging rate of DPPH• reached approximately 90% when the concentration was about 30 mg L−1, which was not significantly different from the antioxidant capacity of Vc. A kit was used to measure the T-AOC of the lychee bark extract, as shown in Figure 3B. The T-AOC of 0.76 mg L−1 lychee bark extract was equivalent to that of 0.23 mg L−1 Vc. Thus, the lychee pericarp extract had a higher T-AOC, approximately 1.5-fold that of Vc.
Rate of 1,1-Diphenyl-2-picrylhydrazyl (DPPH) and Total Antioxidant Capacity (T-AOC) Elimination Using RLP.
Evaluation of In Vivo Antioxidant Capacity
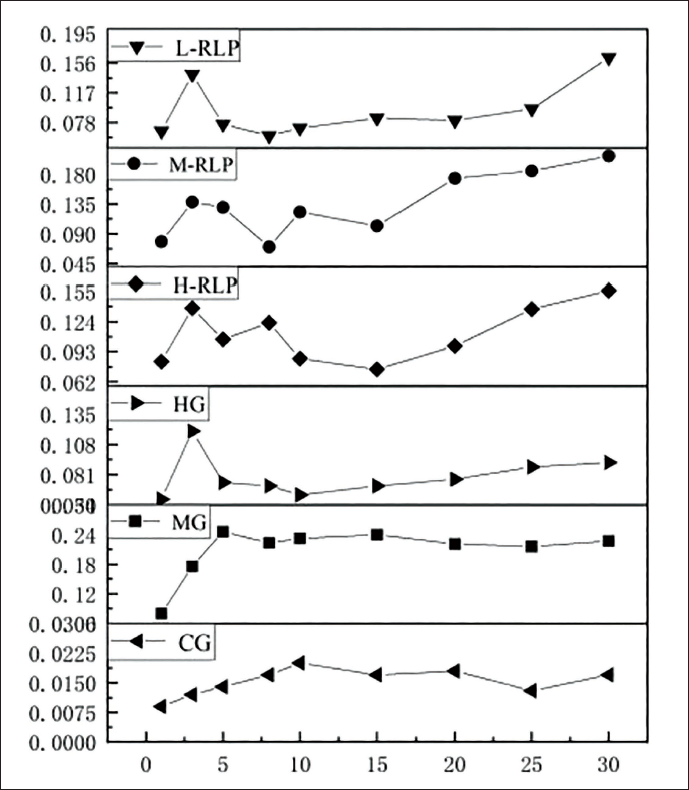
To varying degrees, the high-, medium-, and low-dosage groups of the lychee bark extract reversed the J-point downshift caused by ISO-induced cardiac damage in rats (Figure 4). At 5–20, 8–15, and 5–15 min following ISO injection, the high-, medium-, and low-dosage groups of the lychee bark extract significantly counteracted the J-point downshift generated by ISO-induced cardiac damage in rats (p < 0.05). Compared with the model group, the metoprolol tartrate group showed the highest suppression of the J-point downshift 30 min after the ISO injection (p < 0.05).
Effect of RLP on the J Spot Down Value of Electrocardiogram (ECG) in Rats with Isoprenaline Hydrochloride (ISO)-induced Acute Myocardial Infarction (AMI). ∇J = J-point Value at the Corresponding Time Point after ISO Injection − J-point Value Before Injection (Data represented mean ± SD; ***p < 0.001, **p < 0.01, and *p < 0.05).
The serum antioxidant capacity effect revealed that compared with the normal control group, the serum LDH level of rats in the model group was significantly increased (p < 0.05). The CK level also increased, but there was no statistically significant difference, indicating that a high dose of ISO caused myocardial ischemic injury in rats. The serum LDH level of rats in the high- and medium-dosage groups of the lychee bark extract decreased significantly (p < 0.05) compared with the model group (Figure 5A), with the CK level also decreasing (Figure 5B). These results indicated that the lychee bark extract had an alleviating effect on the ISO-induced myocardial ischemic injury in rats. Figures 5A and 5B showed that, compared with the model group, the metoprolol tartrate group had a propensity to lower blood LDH and CK levels.
Effect of RLP on Biochemical Indicators in Rats with Isoprenaline Hydrochloride (ISO)-induced Acute Myocardial Infarction (AMI) (x ± s, n = 10).
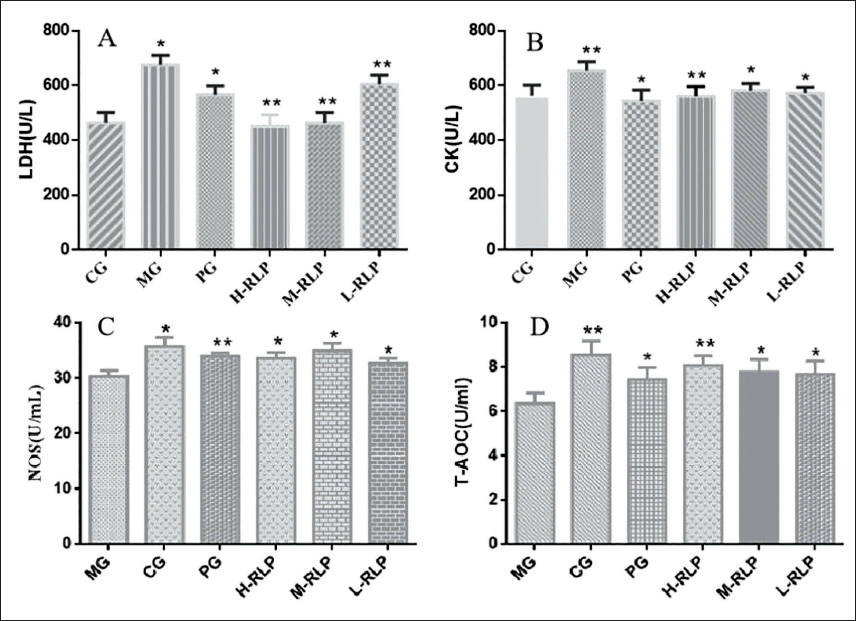
Moreover, compared with the normal control group, the serum level of NOS and T-AOC in the model group rats was significantly reduced (p < 0.05), indicating that the antioxidant capacity of serum in the rat myocardium was decreased during myocardial ischemia. The level of NOS and T-AOC activity in the high-, medium-, and low-dosage groups of the lychee bark extract was significantly higher (p < 0.05) than that in the model group, indicating that the lychee bark extract could effectively improve the antioxidant capacity of rat serum and alleviate the ISO-induced oxidative damage in rat cardiomyocytes (Figure 5C and D). Finally, NOS and T-AOC levels were significantly higher in the metoprolol tartrate group than in the model group (p < 0.05).
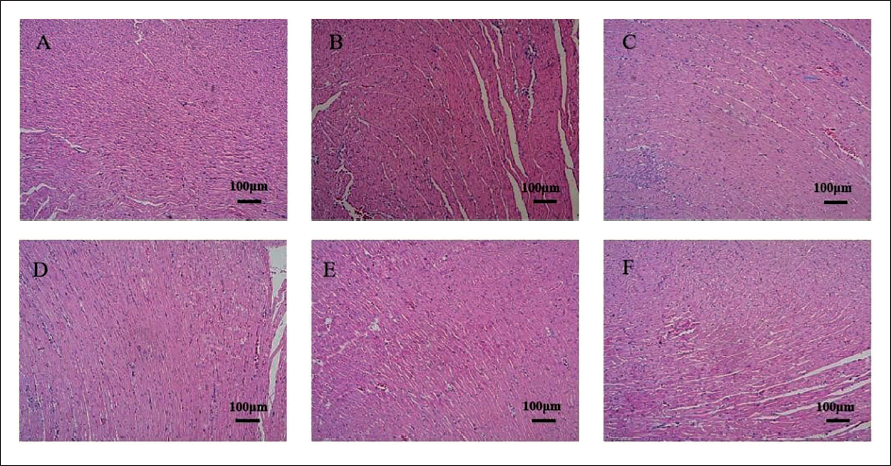
The H&E staining results revealed that the cardiac fibers of the normal control rats were neatly aligned, the cell membrane was intact, the cytoplasm was consistently stained, and nuclei were visible (Figure 6). The model group had disorganized cardiac fibers, and deteriorated and necrotic cardiomyocytes, with many inflammatory cells invading the interstitium (Figure 6B). Under the microscope, point-flake necrosis and degeneration were observed in each treatment group; however, they were greatly reduced in contrast to the model group and some of the cardiac tissues were similar to those in normal control rats, especially in H-RLP, M-RLP, and PG (Figure 6C–E), but the PG group had disorganized cardiac fibers and deterioration (Figure 6D).
Myocardial Tissue Morphology of Rats in Each Group (Hematoxylin and Eosin (H&E) Staining: 100×).
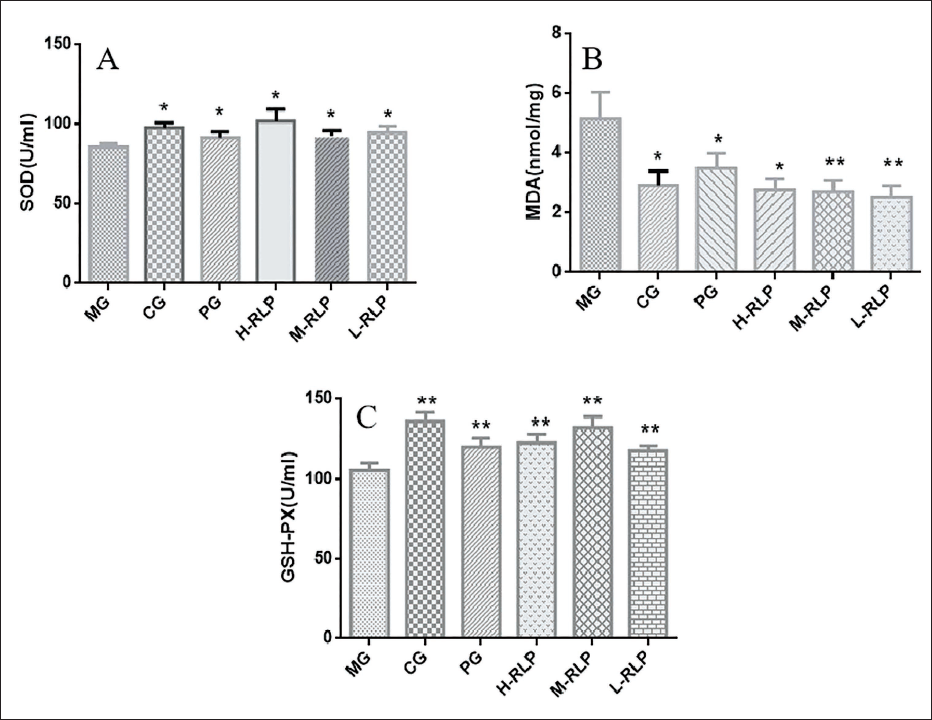
The results showed that the level of MDA in the myocardial tissue of rats in the model group was significantly increased (p < 0.05), the level of SOD was significantly decreased (p < 0.05), and the activity of GSH-Px was significantly decreased (p < 0.01), indicating that the oxidative stress response in rats with acute myocardial ischemia was enhanced and the products of lipid peroxidation were increased. In the high-, medium-, and low-dosage groups of the lychee bark extracts, the level of MDA significantly decreased (p < 0.05), the level of SOD significantly increased (p < 0.05), and the activity of GSH-Px significantly enhanced (p < 0.01) in the myocardial tissues of rats, indicating that the lychee bark extracts significantly improved the ability of the myocardium in resisting lipid peroxidation in ISO-induced myocardial injury in rats (Figure 7).
Effect of RLP on Antioxidant Capacity of Myocardial Tissue in Isoprenaline Hydrochloride (ISO)-induced Rats with Acute Myocardial Infarction (AMI) (x ± s, n = 10).
Discussion
Modern research shows that free radicals are constantly generated during the metabolic process of human life activities, and the damage caused by the accumulation of free radicals is the result of oxidation in the body. However, when the balance is broken and the level of free radicals in the body increases, free radicals will destroy the structure and function of cells through a series of oxidation reactions, thus inducing a variety of diseases, such as atherosclerosis, cardiovascular diseases, and tumors. Litchi skin is the rind of Litchi chinensis (Litchi chinensis Sonn.), a plant of the Sapindaceae family, which is rich in polyphenols, flavonoids, anthocyanosides, and other active ingredients, with strong synergistic antioxidant properties, significant therapeutic effects, and small side effects, making it a natural antioxidant with great prospects for development. At present, there are more studies on lychee skin, lychee kernel, and fruit pulp components, but there are fewer studies on the cardiovascular efficacy of lychee skin. Therefore, this experiment explored the protective effect of litchi pericarp on acute myocardial ischemia through previous literature research and team accumulation of Litchi Research.
During pretesting, the concentration of proanthocyanidins in fresh and dried lychee pericarps was compared using an established technique. Because the loss of proanthocyanidins in lychee pericarps after drying was significant, which affected the quality of the extract, fresh lychee pericarps were selected as the raw material to optimize the extraction procedure. Moreover, different amounts of methanol, propanol, and acetone were examined as extraction solvents for selecting the appropriate extraction solution. The results showed that acetone led to a greater yield than methanol and ethanol. However, both acetone and methanol are toxic, and residual acetone or methanol in the extract is more difficult to remove, making it a risk to the human body. Thus, ethanol was chosen as the extraction solvent. The technique of lychee pericarp extraction was finalized using the proanthocyanidin yield as the indicator via a four-factor, three-level response surface optimization test. The polyphenol content of three distinct RLP batches was also tested, and the findings demonstrated that the optimized extraction process could effectively extract polyphenols from the lychee pericarp.
UPLC–MS was used to perform the preliminary identification of 11 components in L. chinensis bark extract. Six formulas were isolated from the purified extract of L. chinensis bark via silica gel column chromatography, Sephadex LH-20 dextran gel column chromatography, and reversed-phase HPLC semipreparative chromatography. The formulas were structurally identified using NMR and ESI-MS. The preliminary compositional analysis of the lychee bark extract helped guide the subsequent activity assessments. The scavenging rate of DPPH• reached approximately 90%, not significantly different from the antioxidant capacity of Vc, and the T-AOC findings indicated that 0.76 mg L−1 RLP and 0.23 mg L−1 Vc had comparable T-AOCs. Next, an acute myocardial ischemia model in rats was established to assess the protective efficacy of RLP against myocardial ischemic damage. The results suggested that the lychee bark extract has a protective effect on myocardial ischemic injury in rats. With regard to its mechanism of action, RLP may attenuate the lipid peroxidation response of the myocardial cell membrane and block the drop in NO level during myocardial ischemia. Finally, the current study indicated that RLP had good antioxidant activity and cardiovascular protective effects, thereby laying a theoretical foundation for the application of lychee bark extract in cardiovascular disorder treatment.
Conclusion
Using black-leaved lychee pericarp as the study object, the extraction procedure was optimized and the contents of the lychee pericarp extract, along with its polyphenol content and free radical scavenging activity, were evaluated. The findings of in vitro and in vivo investigations revealed that the lychee pericarp extract had strong antioxidant activity with a cardioprotective effect, offering a theoretical and experimental framework for the production and use of the lychee pericarp.
Footnotes
Abbreviations
RLP: Lychee pericarp enrichment product; DPPH: 1,1-Diphenyl-2-picrylhydrazyl; T-AOC: Total antioxidant capacity; ISO: Isoprenaline hydrochloride; CK: Creatine kinase; LDH: Lactate dehydrogenase; NOS: Nitric oxide synthase; SOD: Superoxide dismutase; MDA: Malonaldehyde; GSH-Px: Glutathione peroxidase; UPLC–MS: Ultra-performance liquid chromatography–mass spectrometry; ESI-MS: Electrospray ionization-mass spectrometry; NMR: Nuclear magnetic resonance; AMI: Acute myocardial infarction; cTns: Cardiac troponins; MYO: Myoglobin; ROS: Reactive oxygen species; Vc: Vitamin C; HPLC: High-performance liquid chromatography; PC: Proanthocyanidin.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has received financial support from the Guangdong Provincial Forestry Science and Technology Innovation Project (2022KJCX012) and the Guangdong Provincial Drug Administration Science and Technology Innovation Section Project (2021TDB40).