Abstract
Background
It is estimated that the plant has been utilized in India’s traditional medical system for close to 2,500 years. Alkanin is a key active component in Boraginaceae medicinal plant roots. Many scientists are interested in this tiny molecule due to its biological potential.
Objectives
To examine the hypoglycemic effects of alkannin (a naphthoquinone pigment).
Materials and Methods
Initially, we determined the dosage of alkannin by conducting an acute oral toxicity study per Organisation for Economic Co-operation and Development guidelines. Subsequently, streptozocin was used to induce diabetes in mice, and we chose several parameters such as body weight, food and water intake, blood glucose level, and biochemical estimation for the in vivo evaluation. The enhanced pharmacokinetic features of alkannin, such as its better cytochromes P450 metabolism profiles and higher intestinal absorption as compared to glimepiride, were validated by in silico investigations utilizing Swiss ADME (Absorption, Distribution, Metabolism, and Excretion) and AutoDock Vina.
Results
In particular, blood glucose levels and body weight in the alkannin-treated group drastically decreased to 138 ± 1.45 mg/dL and 27.9±0.54 g, respectively, as compared with the diabetic-treated group. Moreover, alkaline phosphatase levels dropped to 57.26 ± 3.05 U/L in the alkannin group, suggesting enhanced liver and kidney functions. Creatinine levels also decreased from 1.44 ± 0.10 mg/dL to 0.92 ± 0.09 mg/dL, and serum glutamic oxaloacetic transaminase/aspartate aminotransferase levels decreased from 64.16 ± 3.14 U/L to 47.85 ± 2.01 U/L. Additionally, diabetic controls maintained cholesterol levels between 102.1 ± 11.46 mg/dL and 93.53 ± 2.61 mg/dL. With a considerable binding score of –9.2 kcal/mol for alkannin, molecular docking demonstrated a high degree of binding efficiency and robust interaction with biological targets. The docking results revealed that alkannin exhibits drug-like properties. In addition, it satisfies the lead likeness requirement and has a lower synthetic accessibility score compared to glimepiride.
Conclusion
The results revealed the promising characteristics and positive outcomes of alkannin, underscoring its potential as a viable candidate for further investigation and prospective advancement as a treatment drug for diabetes.
Introduction
Diabetes mellitus (DM), a significant global public health concern, has a significant negative economic and health impact. Approximately 463 million individuals worldwide had diabetes in the year 2019, and by the end of the year 2045, this number will increase to 700 million (Saeedi et al., 2019; Zhang et al., 2022). It is a broad category of disorders characterized by hyperglycemia and partial or complete inability to secrete or respond to insulin (Saeedi et al., 2019). According to the World Health Organization, diabetes kills over a million people each year and necessitates hospitalization for an increasing number of people (Shoaib et al., 2022). It is a chronic disorder characterized by slow degradation of pancreatic beta cells. Type 1 diabetes develops when the pancreas fails to produce sufficient insulin (El Malahi et al., 2022); type 2 diabetes mellitus (T2DM) develops when the body can no longer effectively use the insulin that it produces; or during pregnancy (known as gestational DM). The body consistently produces insulin in T2DM, a form of DM that is not insulin-dependent, but cells are not as receptive to it as they should be. Numerous environmental and genetic factors, including population growth, obesity, aging, inactivity, and urbanization, frequently result in T2DM (Khan et al., 2022). Because of this, T2DM is quite common and accounts for around 90% of all instances of diabetes (Motala et al., 2022). T2DM affects about 6% of the elderly population of developed nations. Hyperglycemia may subsequently result in catastrophic damage to the blood vessels and nerves in addition to discomfort and blurred eyesight. Untreated diabetes can also have several serious side effects, such as renal failure, erectile dysfunction, heart issues, diabetic neuropathy, eye damage, stroke, metabolic acidosis, and even death (Chamberlain et al., 2022).
By altering one’s lifestyle, staying away from cigarettes, and increasing exercise time, T2DM can be avoided or managed. Even after surgery, weight loss in some T2DM patients seems to be a successful therapy (Shoaib, Salem-Bekhit et al., 2020). Medication, including or omitting insulin, may be recommended if all other therapies are ineffective for certain patients (Nguyen et al., 2022). However, adopting healthy diets that include foods with high phytochemical content and strong antioxidant capabilities, as shown in several epidemiological studies, can minimize the likelihood of developing diabetes and its predisposing variables (Saeed et al., 2021).
Naphthoquinones are common phenolic substances found in nature. They are byproducts of the secondary metabolism of plants as well as bacterial and fungal processes, with the three most common substances being juglone, lawsone, and plumbagin. Naphthoquinones have powerful antibacterial, antifungal, antiviral, insecticidal, anti-inflammatory, and antipyretic effects (Rahman et al., 2022). In addition, they exhibited substantial cytotoxicity. Naphthoquinones have historically been employed as dyes and are found in a variety of plant groups. However, many biological activities of substances such as lawsone, lapachone, and lapachol have recently been discovered (Cardoso et al., 2018). It has been shown that these natural naphthoquinones can heal wounds, which may be because of their anti-inflammatory and antioxidant capabilities. Additionally, their pharmacological effects on the reproductive and cardiovascular systems have been proven. They attach to deoxyribonucleic acid (DNA) and prevent DNA replication, interact with several proteins (enzymes), disrupt cell and mitochondrial membranes, and interfere with the electrons of the respiratory chain on mitochondrial membranes (de Sousa Portilho et al., 2022).
Many medicinal plants with an unusual multiplicity of phytochemicals have been identified for the treatment of diabetes. The phytochemical family of naphthoquinones has several promising medicinal uses, one of which has an antidiabetic effect. Among the many ways in which naphthoquinone works include blocking α-glucosidase and protein tyrosine phosphatase 1B, increasing glucose uptake in myocytes and adipocytes via glucose transporter type (GLUT) 4 and GLUT2 translocations, increasing peroxisome proliferator-activated receptor (PPAR) ligand activity, and restoring normal liver carbohydrate metabolizing enzyme levels (El-Najjar et al., 2006; Wang et al., 2020).
A naturally occurring naphthoquinone is alkannin. It is one of the primary active ingredients in the roots of some medicinal plants of the Boraginaceae family (Papageorgiou et al., 1999). Several research teams have been interested in this small compound because of its extraordinary biological potential (Arampatzis et al., 2022). More precisely, alkannin and its derivatives have been shown to possess several pharmacological characteristics over the years, with the most significant ones being anti-inflammatory, antibacterial, anticancer, antioxidant, wound healing, and regenerative actions (Guo et al., 2019). Its positive benefits for metabolic illnesses have been revealed in several investigations published in the last few years. To the best of our knowledge, there are no data available on the antidiabetic potential of alkannin. The purpose of the current study was to explore the antidiabetic potential of alkannin in in vivo and in silico models.
Materials and Methods
Chemicals
Alkannin (purity ≥98%) and glimepiride (pharmaceutical secondary standard grade) were obtained from Cayman Chemicals, Michigan, USA. Streptozocin (STZ, purity ≥98% high-performance liquid chromatography (HPLC) grade) and all other chemicals used were of analytical grade and procured from Sigma-Aldrich (St. Louis, MO, USA).
In vivo Antidiabetic Study
Animals
Female mice (10 weeks old; 20–25 g) were housed under standard laboratory conditions of 23°C ± 1°C, relative humidity of 55%, and 12-h/12-h light/dark cycle. The mice were provided standard pellets and drinking water ad libitum. Every possible endeavor was undertaken to minimize the distress experienced by the animals and to decrease the number of animals utilized. The experimental animal protocol was approved by the Research Ethics Committee (REC) School of Pharmacy & Technology Management, NMIMS, Shirpur, Maharashtra (SPTM/2022/IAEC/06).
Acute Oral Toxicity Investigation in Mice
The acute toxicity of alkannin was assessed according to the Organisation for Economic Co-operation and Development guideline no. 423 (OECD-423) (OECD Guideline for Testing of Chemicals, 2001). At each time point, three nonpregnant mice were used. An alkannin suspension was prepared using a 0.5% carboxymethylcellulose (CMC) solution. This suspension was administered via intragastrically at specific doses of 5, 50, and 300 mg/kg of body weight (BW). Animal behavior was evaluated at distinct time intervals: 30 min after administration, with particular focus during the first 4 hours, and then continuously for 2 weeks. The toxicity profile was assessed based on mortality and expressed as the lethal dose (LD50).
STZ-induced Diabetic Model
Diabetic mice were produced by intraperitoneal injection of freshly prepared STZ (St. Louis, MO, USA) dissolved in citrate buffer (0.1 M, pH 4.5) at 40 mg/kg BW after overnight fasting. Blood glucose levels were monitored 72 hours post-STZ injection by using an Accu-Chek glucometer. Diabetic mice were identified by fasting glucose levels above 245 ± 5 mg/dL for further research. Three days after the STZ injection, test and reference drug treatments were performed (Shoaib, Salem-Bekhit et al., 2020).
Treatment Schedule
A total of 24 mice (18 diabetic and 6 normal) were separated into four groups (n = 6) for the experiments. For gastric intubation, the treated group received alkannin and glimepiride (standard) every morning, after fasting, for 28 days. Group I (control group) was a normal control group that was served with a normal diet, Group II was a diabetic control group treated with STZ (40 mg/kg BW), Group III was the diabetic treated group with alkannin (10 mg/kg BW p.o.), and Group IV was diabetic group treated with standard glimepiride (Glamp, 10 mg/kg BW, p.o.; positive control group). Three days after STZ administration, all treatments (except the normal control) were administered orally for 28 days. BW and blood glucose was recorded on the first and last day of treatment. On day 28, cardiac puncture was used to collect blood from the heart, which was allowed to clot at room temperature and centrifuged at 4°C at 3,000 rpm to separate serum for further research.
Estimation of BW
The animal’s weight was determined using a digital balance on both the first and last days of treatment (Khalid et al., 2022).
Food and Water Intake
Food and water consumption were observed in all groups during the 28 days of treatment on the initial and final days (Shoaib, Siddiqui et al., 2020).
Estimation of Blood Glucose Level
On the first and last day of the treatment, a glucometer was used to check the blood glucose level from a tail prick (Shoaib, Salem-Bekhit et al., 2020).
Biochemical Analysis
The Erba CHEM-5 Plus V2 auto-analyzer (Transasia Bio-Medicals Ltd.) was used to analyze biochemical parameters after the serum was separated from nonheparinized blood. These parameters included the estimation of serum aspartate aminotransferase (AST), serum alanine aminotransferase (ALT), and alkaline phosphatase (ALP), urea, creatinine, cholesterol, and triglycerides (Khalid et al., 2022).
Pharmacokinetics Prediction via Swiss ADME (Absorption, Distribution, Metabolism, and Excretion) and admetSAR
We used the Swiss ADME web tool to determine the physicochemical qualities of alkannin and glimepiride. We started by putting the chemical structures of alkannin and glimepiride into the Swiss ADME tool. Different factors such as molecular weight, lipophilicity, and amount of rotatable bonds were looked at to see how drug-like they were examined to determine how well they might be absorbed. In addition, admetSAR, a computer program for guessing ADMET qualities, was used to examine how alkannin and glimepiride are absorbed, distributed, broken down, and flushed out of the body. The last step was to contrast the results from Swiss ADME and admetSAR to find out how alkannin and glimepiride work in the body.
Molecular Docking Analysis
AutoDock Vina tools were used to determine the interactions between aleglitaar and alkannin (Pratap Singh & Pattnaik, 2024; Singh & Pattnaik, 2024). Before starting molecular docking, we retrieved alkannin ligand (PubChem ID: 72521) of interest from PubChem and the three-dimensional structure of aleglitaar (3G9E) from the Protein Data Bank (PDB) database. The target was cleaned by removing water molecules, co-crystallized ligands, and any other heteroatoms that were not directly involved in the binding site. The protein structure was prepared for docking by adding hydrogen atoms and giving each atom the correct charge. The grid box parameters for the ligand-binding site of aleglitaar were adjusted as X = 60, Y = 60, and Z = 60. The center grid box coordinates were X = 7.384, Y = 22.009, and Z = 18.269, with a spacing of 0.35 Å. The most favorable docking position was chosen from the nine poses based on the interacting residues that produced hydrogen bonds with a strong binding affinity (kcal/mol). Protein–ligand interactions of the docked complexes were created and visualized using Discovery Studio 4.0. client.
Statistical Analysis
Software called GraphPad Prism V5.01 was used to perform statistical analysis on the data. One-way analysis of variance (ANOVA) was used to determine the significant difference from the corresponding controls for each experimental test condition. All the values were expressed as mean ± SEM; where * denotes p < 0.05, ** denotes p < 0.01, and *** denotes p < 0.001.
Results
Acute Oral Toxicity Study in Mice
The LD50 of alkannin in Swiss mice was determined to be 100 mg/kg of BW. There were no notable alterations in overall behavior and body mass. Nevertheless, there was a noticeable rise in mortality that was directly proportional to the dosage.
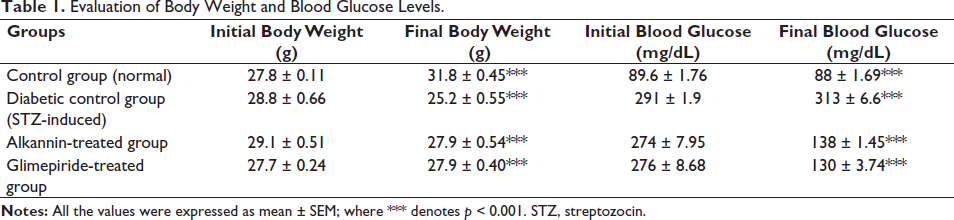
Determination of Body Mass and Evaluation of Blood Glucose Concentration
The study examined the disparity in BWs between the beginning and end of the study, and it was determined to be statistically significant for Group I (31.8 ± 0.45***) (Table 1). The glucose levels in the blood remained relatively stable at 88 ± 1.69*** in the normal control group. During the study, there was a notable decrease in final BW (25.2 ± 0.55***) and a significant increase in blood glucose levels (313 ± 6.6***), indicating the potential development of diabetes. The results of Group III showed a notable decrease of 138 ± 1.45*** in blood glucose levels and a significant reduction in final BW of 27.9 ± 0.54*** among diabetics treated with alkannin. When comparing the results, it is evident that in Group IV, the diabetics treated with the standard drug, the animal experienced a significant increase in their final BW of 27.9 ± 0.40***, while their blood glucose levels (130 ± 3.74***) showed a substantial decrease.
Evaluation of Body Weight and Blood Glucose Levels.
Evaluation of Consumption of Food and Water
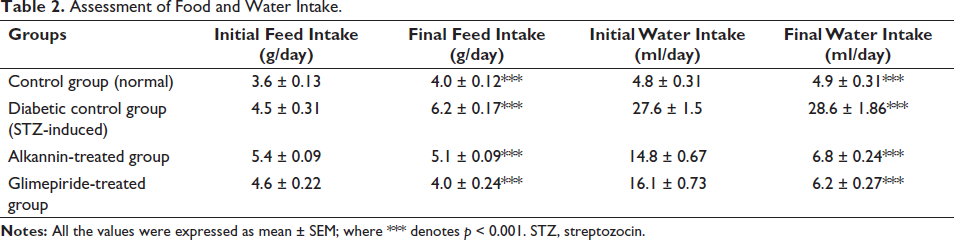
The findings of the food and water intake demonstrate that the diabetic mice (Group II) consumed more food (6.2 ± 0.17***) and water (28.6 ± 1.86***) than the control group, which is probably because of their diabetes (Table 2). Groups III and IV, treated with alkannin and glimepiride, respectively, showed substantial changes in food and water consumption, suggesting that these medications may have an impact on hunger and glucose regulation.
Assessment of Food and Water Intake.
Determination of Serum Glutamic Oxaloacetic Transaminase (SGOT)/AST
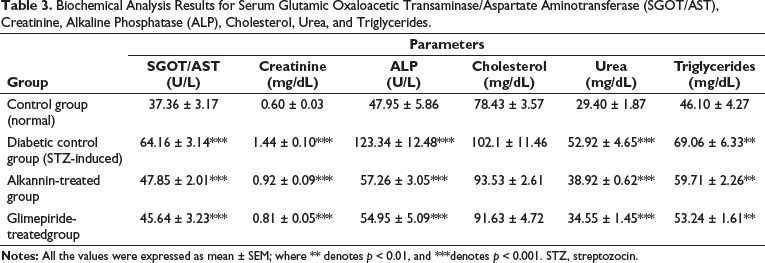
According to the findings, all three experimental groups showed significantly higher SGOT/AST levels than the control group. Group II (64.16 ± 3.14***) and Group III (47.85 ± 2.01***) have the highest SGOT/AST values, while Group IV (45.64 ± 3.23***) has the lowest (Table 3).
Biochemical Analysis Results for Serum Glutamic Oxaloacetic Transaminase/Aspartate Aminotransferase (SGOT/AST), Creatinine, Alkaline Phosphatase (ALP), Cholesterol, Urea, and Triglycerides.
Determination of Creatinine
Group II showed a significant elevation in creatinine levels (1.44 ± 0.10***), indicating kidney dysfunction associated with diabetes. Group III significantly reduces creatinine levels (0.92 ± 0.09***), suggesting a potential influence on renal function in diabetic mice. Group IV had the lowest creatinine level, indicating an impact on kidney function (Table 3).
Evaluation of ALP
The ALP level in mice with STZ-induced diabetes (Group II) was markedly higher than that of the control group. The ALP level in mice treated with alkannin (Group III) was considerably lower (57.26 ± 3.05***) than that in the diabetic group (Group II) (123.34 ± 12.48***). The reduction in ALP levels can be seen as a favorable outcome, indicating potential liver improvement in response to alkannin treatment. Interestingly, Group IV mice had a significantly lower ALP level (54.95 ± 5.09***) compared to Group II and Group III. The diminished ALP levels found in the group treated with alkannin and the standard drugs may indicate enhanced liver function and reduced liver inflammation (Table 3).
Evaluation of Cholesterol Level
The cholesterol levels in mice with STZ-induced diabetes (Group II) were significantly elevated at 102.1 ± 11.46 compared to the control group (Group I), suggesting that the development of diabetes with STZ resulted in an augmentation of cholesterol levels. When analyzing the results, it was observed that during treatment with alkannin (Group III), there was a slight decrease in cholesterol levels (93.53 ± 2.61) in comparison to diabetic mice (Group II). After analyzing the results, it seems alkannin can potentially enhance cholesterol regulation in mice with diabetes. On the other hand, Group IV showed the lowest cholesterol levels (91.63) compared to the other groups. Cholesterol levels in the glimepiride-treated group showed a significant decrease, that is, 91.63 ± 4.72, compared to the diabetic group (Group II) and the alkannin-treated group (Group III). These findings indicate that alkannin could be beneficial in regulating cholesterol levels in diabetic mice (Table 3).
Estimation of Urea Level
The urea level in mice with STZ-induced diabetes (Group II) was found to be significantly higher (52.92 ± 4.65***) than in the control group. Based on these results, it appears that the induction of diabetes with STZ causes changes in urea metabolism, indicating potential kidney dysfunction. The mice treated with glimepiride (Group IV) exhibit a significantly lower urea level of 34.55 ± 1.45***, in contrast to both the diabetic mice (Group II) with a urea level of 52.92 ± 4.65*** and the alkannin-treated mice (Group III) with a urea level of 38.92 ± 0.62***. It appears that the use of alkannin in the experiment positively impacted urea metabolism, which could lead to improved kidney function or prevent the rise in urea levels associated with diabetes (Table 3).
Determination of Triglycerides
The triglyceride level in Group II was significantly higher than in the control group (Group I), that is, 69.06 ± 6.33**. This suggests that the diabetic condition in Group II led to an increase in triglyceride levels. Group III included mice that were administered alkannin. The triglyceride level in this group was 59.71 ± 2.26, indicating that alkannin treatment decreased triglyceride levels. Group IV consisted of mice that were treated with glimepiride. The triglyceride level in these mice was 53.24 ± 1.61, indicating a significant reduction due to glimepiride treatment. These results suggest that both glimepiride and alkannin could effectively manage triglyceride levels, with alkannin showing promising results (Table 3).
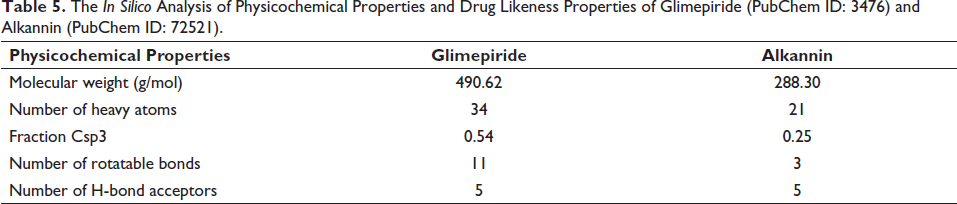
Analysis of Physicochemical Properties with admetSAR
Compared to glimepiride, alkannins possess several advantageous drug-like features. These properties include lower molecular weight, lower proportion of sp3 hybridization, fewer rotatable bonds, and lower lipophilicity. One possible explanation for the potential appropriateness of alkannin as a drug candidate with enhanced pharmacological properties is that these features may contribute to its potential applicability.
Analysis of ADMET Properties via SwissADME
Both alkannin and glimepiride have strong human intestinal absorption properties; however, alkannin has a slightly greater projected probability, indicating a possible improved absorption profile. Alkannin has superior Caco-2 permeability compared to glimepiride, suggesting enhanced cell permeability, which is a desirable characteristic of potential therapeutic candidates. Alkannin exhibits a more advantageous cytochromes P450 (CYP450) profile due to its non-substrate nature for certain CYP450 enzymes and its inhibitory effect on certain enzymes. In contrast, glimepiride is a substrate for CYP450 2C9. The subcellular localization of alkannin in mitochondria is more advantageous than the localization of glimepiride in lysosomes. The FHMT results indicated that alkannin and glimepiride were likely to have a high level of toxicity to fish. Alkannin and glimepiride are considered P-glycoprotein substrates, implying that they may interact with this efflux transporter similarly. Furthermore, both are anticipated to be noninhibitors of renal organic cation transporters, implying no significant inhibition of these transporters, and non-AMES are hazardous, implying a lower risk of generating mutagenic effects. Lipinski’s rule of five is satisfied by both alkannin and glimepiride, which indicates that these compounds possess characteristics often linked with high oral bioavailability and comparable drug-likeness. Alkannin can pass through Ghose’s filter, whereas glimepiride has only two violations. According to Ghose’s filter, alkannin’s molecular weight and molar refractivity are within the permitted range. The Veber rule is satisfied by alkannin, which indicates that it possesses qualities that are beneficial for oral bioavailability. On the other hand, glimepiride has one violation that is connected to the number of rotatable bonds. Alkannin is in accordance with Egan’s rule. However, glimepiride has one infraction that is connected to the polar surface area (TPSA). In contrast, glimepiride has three violations about molecular weight, rotatable bonds, and XLOGP3, while alkannin satisfies the criterion for lead likeness. While glimepiride has a higher synthetic accessibility score, alkannin has a lower value, which indicates that it may be simpler to produce using synthetic methods. The results are presented in Tables 4 and 5.
Comparative ADME Profile of Alkannin and Glimepiride.
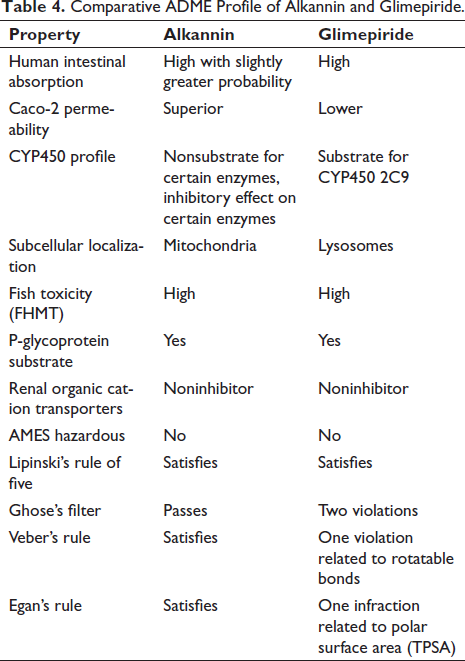
The in silico Analysis of Physicochemical Properties and Drug Likeness Properties of Glimepiride (PubChem ID: 3476) and Alkannin (PubChem ID: 72521).
Molecular Docking Analysis Result
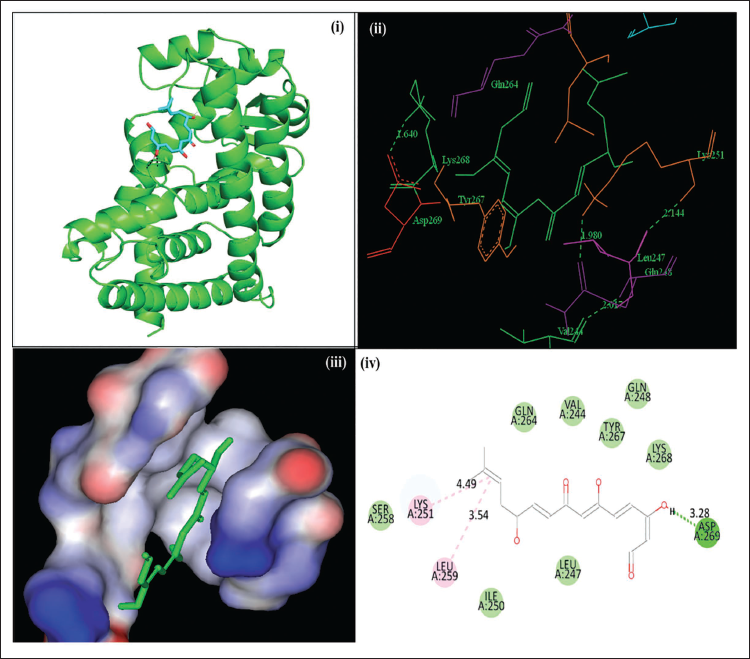
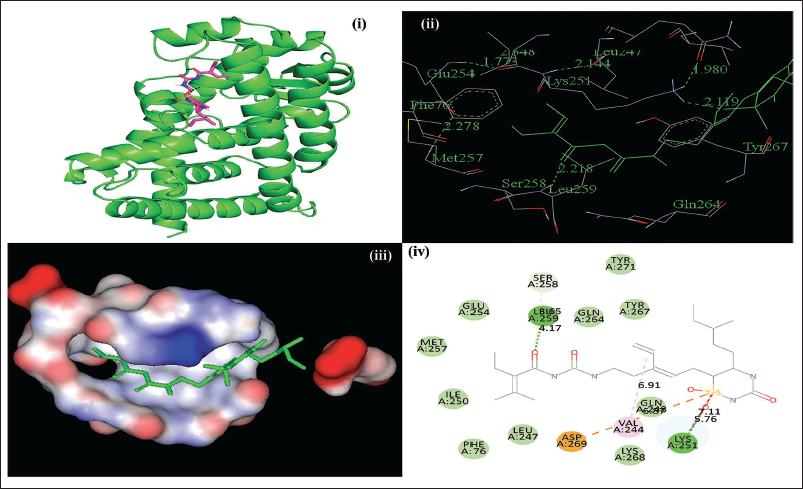
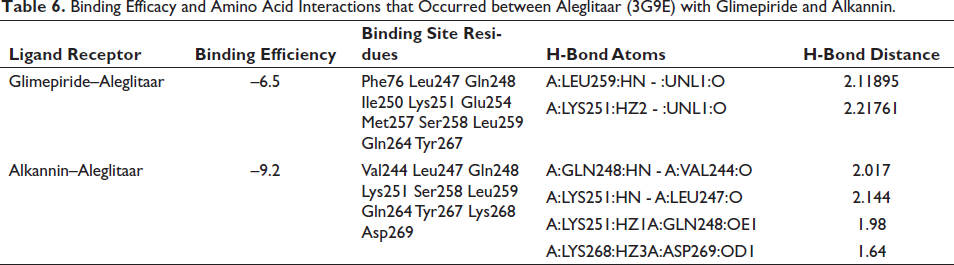
Alkannin–Aleglitaar demonstrates superior binding efficiency with a more pronounced negative value –9.2 kcal/mol, engages a larger quantity of amino acid residues at the binding site, and establishes a greater number of hydrogen bonds (4) in comparison to glimepiride–aleglitaar, which has a binding efficiency of –6.5 kcal/mol, involves a smaller number of residues, and forms a lesser number of hydrogen bonds (2). In Figures 1 and 2, we have shown the surface model, ribbon model, 2D and 3D binding patterns of alkannin (PubChem ID: 72521) and glimepiride (PubChem ID: 3476) with aleglitaar (3G9E). The results are presented in Table 6.
The Binding Patterns of Alkannin with Aleglitaar. (i) 3D Presentation of Alkannin with Aleglitaar in Ribbon Representation; (ii) 3D Stick Model Presentation of Alkannin with Aleglitaar with Labeled Three-letter Amino Acid Names; (iii) Surface Presentation of Alkannin Surrounded by Aleglitaar; (iv) 2D Presentation of Alkannin with Aleglitaar.
The Binding Patterns of Glimepiride with Aleglitaar. (i) 3D Presentation of Glimepiride with Aleglitaar in Ribbon Representation; (ii) 3D Stick Model Presentation of Glimepiride with Aleglitaar with Labeled Three-letter Amino Acid Names; (iii) Surface Presentation of Alkannin Surrounded by Aleglitaar; (iv) 2D Presentation of Glimepiride with Aleglitaar.
Binding Efficacy and Amino Acid Interactions that Occurred between Aleglitaar (3G9E) with Glimepiride and Alkannin.
Discussion
In this work, toxicological evaluation was conducted on rodents using normal procedures, administering alkannin at a single dose over a set length of time (Shoaib, Siddiqui et al., 2020). Similarly, in the present, during acute toxicity tests, female mice showed no mortality or behavioral changes from alkannin up to a maximum dose of 100 mg/kg BW orally. The initial symptoms of poisoning in acute toxicity studies include mortality, clinical signs, and behavioral abnormalities.
According to the findings of our research, diabetic mice that were given alkannin therapy had a substantial decrease in their blood glucose levels when compared to diabetic mice that were not given any treatment. In the STZ-induced diabetes rat model (60 mg/kg, i.p.), impaired insulin responsiveness to glucose leads to alterations in blood glucose levels and BW due to damaged pancreatic islet cells (Shoaib, Badruddeen, Dixit et al., 2019). The hypoglycemic effects of natural chemicals, particularly naphthoquinones, have been the subject of prior research, and our findings are consistent with these observations. Razaque et al. (2022) examined the hypoglycemic effects of derivatives of 2-phenylamino-1,4-naphthoquinone to develop new medications that can treat diabetes. Our research proves that alkannin has a hypoglycemic effect, which may be achieved by mechanisms such as increased insulin sensitivity or higher glucose uptake by tissues. Additionally, Yang et al. (2022) examined alkannin, produced from Alkanna tinctoria, for its anti-inflammatory activities. The study focuses on alkannin’s regulation of mitogen-activated protein kinase (MAPK) and nuclear factor kappa B (NF-κB) signaling pathways. This work highlights the possible pharmacological effects of alkannin on inflammatory responses, which are often linked to diabetes. Although it is unrelated to diabetes, this study sheds light on these effects.
The harmful effects of medications or chemicals may cause changes in BW; however, these changes may be due to fat formation and physiological adaptive responses (Kifayatullah et al., 2015; Shoaib, Siddiqui et al., 2020). This study found differences in BW between the beginning and end of the trial, which was statistically significant for the different groups. Additionally, the results of our research indicate that alkannin may enhance insulin sensitivity, which is an essential factor in improving glucose management in patients with diabetes. Furthermore, alkannin has shown the potential to protect against additional metabolic abnormalities associated with diabetes.
The diabetic group consumed more food and water than the normal control and treated groups (Shoaib, Badruddeen, Siddiqui et al., 2019). Similar results were also observed in the present study. Biochemical indicators reveal the health of major organ systems such as the liver, renal, hematopoietic, and immunological systems (Schilter et al., 2003). Enzymes are commonly utilized in toxicology to identify and assess cellular damage. Early indications for toxicity include ALT and AST enzymes (Dzoyem et al., 2014). Our study’s findings showed that the levels of SGOT and AST in all three experimental groups were considerably higher than those in the control group.
A lipoprotein metabolic disorder may be caused by lipoprotein metabolism. Thus, serum total cholesterol, low-density lipoprotein (LDL), and triglyceride levels will rise, while high-density lipoprotein (HDL) levels will decrease. The gastrointestinal tract absorbs lipids as chylomicrons, triglycerides, phospholipids, and cholesterol (Khalid et al., 2020). Alkannin stimulates AMP-activated protein kinase (AMPK), which, in turn, enhances fat metabolism and restricts fat production. In addition, alkannin suppresses the transcription factors involved in fat production. The results of our study support the hypothesis that both glimepiride and alkannin might help to control triglyceride levels, but alkannin showed the most promising effects.
In addition, in silico studies that make use of Swiss ADME anticipate that alkannin will have desirable drug-like qualities, which indicates that it may have the potential to be a candidate for treatment. Alkannin showed superior pharmacokinetic qualities when compared to glimepiride, which is a medicine that is often used to treat diabetes. These properties include intestinal absorption, cell permeability, subcellular localization, and metabolism by CYP450 enzymes. According to the findings of molecular docking experiments, alkannin–aleglitaar exhibits a higher binding efficiency (–9.2 kcal/mol) and generates a greater number of hydrogen bonds at the binding site than glimepiride–aleglitaar. In a study published by Noboru Motohashi, the lowest binding energy of alkannin (–9.0 kcal/mol) and the same pharmacokinetic prediction was demonstrated (Motohashi et al., 2017). These characteristics may probably contribute to the prospective applicability of alkannin, which is one of the possible explanations for its potential appropriateness as a therapeutic candidate with enhanced pharmacological capabilities.
Therefore, our research contributes to the expanding body of evidence substantiating the hypoglycemic effects of naphthoquinones, phytochemicals such as alkannin.
Conclusion
The present study is a comprehensive evaluation for the determination of toxicity of alkannin in mice. The compound did not induce significant toxicity or pathological changes in the experimental animals in the present study. From the in vivo study, we can conclude that alkannin reduces insulin resistance and increases hyperglycemia in a type 2 diabetes animal model. This study offers several interesting prospects for developing innovative treatments for diabetes. From docking studies, it was found that alkannin has a smaller molecular weight, fewer rotatable bonds, lower molar refractivity, and higher lipophilicity than glimepiride. However, further research is needed to determine how alkannin affects insulin resistance and hyperglycemia. Therefore, we conclude that alkannin’s intriguing properties and findings in our investigation suggest that it could be used as a novel compound for managing diabetes.
Footnotes
Abbreviations
ADME: Absorption, Distribution, Metabolism, and Excretion; ALP: Alkaline phosphatase; ALT: Alanine aminotransferase; ANOVA: Analysis of variance; AST: Aspartate aminotransferase; CMC: Carboxymethylcellulose; CYP450: Cytochromes P450; DM: Diabetes mellitus; DNA: Deoxyribonucleic acid; GLUT: Glucose transporter type; HPLC: High-performance liquid chromatography; LD50: Lethal dose; OECD: Organisation for Economic Co-operation and Development; PDB: Protein Data Bank; SGOT: Serum glutamic-oxaloacetic transaminase; STZ: Streptozotocin; T2DM: Type 2 diabetes mellitus.
Authors’ Contribution
MS, AS, and LA contributed to the analysis, literature surveying, writing, visualization, and draft preparation. MS contributed to data curation. AS and MAK helped in data collection. VB, CA, and AM handled animals. MS and IA contributed to the manuscript’s conceptualization, validation, data curation, and editing. MJA and SAG performed in silico studies. MS helped in validation and interpretation.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The experimental animal protocol was approved by the Research Ethics Committee (REC), School of Pharmacy & Technology Management, NMIMS, Shirpur, Maharashtra (SPTM/2022/IAEC/06).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been funded by Scientific Research Deanship at University of Ha’il - Saudi Arabia through project number RG-21 122.
