Abstract
Background and Objectives
The effect and mechanism of the extract of
Materials and Methods
We detected mice’s brain damage after cerebral I/R and tested neuronal injury following oxygen-glucose deprivation/re-oxygenation (OGD/R) to evaluate the neuroprotection of ECJ. Besides, we tested the expressions of hydrogen sulfide (H2S) synthase cystathionine-β-synthase (CBS) and α subunit of large-conductance Ca2+-activated K+ channels (BKα) both in brain tissues and culture neurons. Importantly, the roles of iberiotoxin (IbTX), BKCa channel inhibitor, and CBS inhibitor aminooxyacetic acid (AOAA) on ECJ-mediated neuroprotection were assessed to explore the neuroprotective mechanism of ECJ.
Results
ECJ treatment could alleviate the mice’s brain injury following cerebral I/R and reduce the neuronal damage caused by OGD/R
Conclusion
ECJ can protect against mouse cerebral I/R injury, the mechanism of which is correlated with promoting the CBS/H2S-BKCa pathway.
Introduction
Ischemic stroke is known as a common cerebrovascular disease (Xue et al., 2017). Cerebral ischemia-induced brain damage is irreversible and can lead to disability and high morbidity in patients (Li et al., 2015). Until now, few ischemic stroke patients are eligible for tissue-type plasminogen activator (t-PA) treatment, which is the only FDA-approved treatment for ischemic stroke (Li et al., 2019). Hence, it is of great significance to demonstrate neuroprotective strategies and develop possible agents for the treatment of ischemic stroke.
Accumulating study has revealed that many plant extracts and/or constituents possess protective effects against cerebral ischemia injury (Dong et al., 2021; Zhu et al., 2021).
Hydrogen sulfide (H2S) is a novel gasotransmitter molecule and participates in modulating the neuronal biological function (Shan et al., 2019). H2S is involved in various physiological processes in the central nervous system (CNS); for instance, H2S modulates the brain microvascular diameter, regulates intracellular Ca2+ homeostasis, and modulates the release of neurotransmitters (Chen et al., 2022; Kimura, 2024). In addition, plenty of studies have reported the neuroprotection of H2S (Lobov et al., 2020) and revealed the protection of H2S against myocardial I/R injury (Li & Xiao, 2020). We previously have found that H2S could protect against mouse cerebral I/R injury, and reduce the oxygen-glucose deprivation/re-oxygenation (OGD/R)-induced neuronal injury (Yin et al., 2023). Endogenous H2S in the brain tissues is mainly catalyzed by cystathionine-β-synthase (CBS) (Powell et al., 2018) from
BKCa channels are widely distributed in excitable and non-excitable cells and are synergistically activated by intracellular Ca2+ and the depolarization of cellular membrane (Kopec et al., 2019). Activation of BKCa channels participates in regulating the release of neurotransmitters and the regulation of neuronal excitability (Liu et al., 2013). We have reported that activation of BKCa channels participated in the endogenous H2S-mediated neuroprotective effects (Wen et al., 2021). Therefore, we likewise demonstrate the effect of BKCa channel activation in the ECJ-mediated neuroprotection and explore the relationship between H2S and BKCa channels.
Materials and Methods
Reagents
ECJ was obtained from Xi’an Wan Fang Biotech Co., Ltd. (Xi’an, China). The procedures of the extraction of plants were provided by the supplier. Briefly, the flowers of
Animals
C57BL/6 mice (6–8 weeks old, 18–22 g) were housed in the Animal Center of Anhui Medical University under standard laboratory conditions. Briefly, the relative humidity was maintained at 54 ± 2%. The temperature was maintained at 22 ± 2°C. The experimental procedures of mice were approved and supervised by the Ethical Committee of Anhui Medical University (Certification No. LLSC 20210661).
Cerebral I/R Model of Mice
Mice cerebral I/R model was undertaken using bilateral common carotid arteries (BCCAs) occlusion (2-VO) (Wen et al., 2019; Yan et al., 2015). Animals were first anesthetized using 5% isoflurane, after that, BCCAs were isolated gently through a midline incision in the mice’s neck. The BCCAs were ligated with threads, and 20 min later, the blood flow was restored by loosening the vessel occlusion. BCCAs of sham mice were only exposed without vascular ligation. During the operation of mice 2-VO, the temperature was consistently maintained at 37°C using a warm table. Mice were randomly divided into the following groups (male to female: 1:1,
Primary Culture and OGD/R Model of Neurons
The procedures of primary culture of hippocampal neurons were described in our present study (Wen et al., 2021). Briefly, hippocampal tissues were isolated from neonatal mice after anesthesia with isoflurane. Then, hippocampal tissues in phosphate buffer saline (PBS, pH 7.45) were cut into small pieces, which were then digested with 0.125% trypsin solution. Finally, cells were obtained after centrifugation and resuspended in DMEM/F12 (1:1). The medium contained penicillin-streptomycin (1%) and 20% fetal bovine serum (FBS). Subsequently, cells seeded in culture plates were incubated in an incubator. The medium of culture cells was replaced with fresh Neurobasal™ medium (Gibco Invitrogen). Neurons were identified using immunofluorescence with neuronal marker microtubule-associated protein 2 (MAP2) and with Hoechst 33258, the cell nuclei dye.
Cultured neurons were divided into normal, OGD/R, NaHS (200 µmol/L), NaHS + IbTX (100 nmol/l), ECJ (90, 270, 810 mg/L), ECJ (810 mg/L) + IbTX (100 nmol/L), and NaHS (200 µmol/L) + IBTX (100 nmol/L) groups (
Morris Water Maze (MWM) Test of Mice
MWM test of mice was undertaken in a circular pool of the maze filled with water to a depth of 21 cm, which is divided into four quadrants, namely northeast (NE), southeast (SE), southwest (SW), and northwest (NW). In the SW quadrant, a platform is placed 1 cm below the water surface (Zhu et al., 2014). During the MWM test, the water was maintained at 22 ± 1°C, and the mouse was gently released into the water at one of four quadrants by the experimenter. The time the animal required to find and stay on the escape platform was recorded and defined as escape latency. Importantly, if an animal failed to find the escape platform, it would be guided to find and stay on the platform for 15 seconds, and the escape latency would be recorded as 60 seconds. In the following three days of MWM test, the starting points of mice released into the water were undertaken in a clockwise direction.
In the spatial probe phase of MWM on the fifth day, the mice were allowed to swim freely in the pool for 60 s after the platform was removed. We recorded the number of mice entering the SW quadrant, as well as noted the time and distance of the mouse crossing the SW quadrant. After the MWM test, the animals were sacrificed under anesthesia, and then we collected the brain and sera of mice for future use.
Western Blot
Total protein was extracted from mouse brain tissues and culture neurons (Lu & Wen, 2020). Proteins (30 µg) were separated by using sodium dodecyl-sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). After that, the proteins in gels were transferred by electrophoresis onto a polyvinylidene difluoride (PVDF) membrane. Proteins on PVDF membranes were blocked with skim milk (5%) in tris-buffered saline for 2 h at room temperature. Afterward, the PVDF membranes were respectively incubated with monoclonal antibodies, such as anti-BKα, anti-CBS, and anti-β-actin, overnight at 4°C. PVDF membranes were then incubated with HRP-labeled secondary antibody at 37°C for 1 h. Finally, the immunoreactive bands on the PVDF membranes were presented using an enhanced chemiluminescence kit (Thermo, USA). The densitometry of the target protein determined the relative intensity of the band compared with β-actin, an internal control.
Biochemical Measurements
The activities of NSE and LDH in mouse sera and in cell culture supernatants and the MDA content in brain tissues were assessed by using biochemical assay kits in accordance with the manufacturer’s manual. The neuronal viability was tested using a CCK-8 assay kit.
The H2S level in mouse brain tissues and in cell culture supernatants was detected using methylene blue method (Wen et al., 2021), which was undertaken according to the procedure provided by the assay kit. Briefly, 0.1 mL of supernatant fluid of brain homogenate or culture supernatant was mixed with 1% acetic acid zinc (0.5 mL) and distilled water (2.5 mL). 10 minutes later, the above mixture was added with 1 mL of 10% trichloroacetic acid. Subsequently, the supernatant of the mixture was collected after centrifugation. Then, the supernatant was added with 30 mmol/L FeCl3 in 1.2 mol/L HCl (0.4 mL) and 20 mmol/L
The content of H2S in mouse brain tissues (µmol/g protein) = 0.727 × A670 ÷ weight of sample, and the content of H2S in culture supernatants (µmol/L) = 13.0625 × A670.
Determination of Brain Water Content
The brain water content was tested using a wet-dry approach (Wen et al., 2018). The brains were cut into slices, and the weight of fresh brain slices was recorded as the wet weight. Then, the dry weight of brain slices was obtained by weighing them after drying them at 105℃ for 48 h. The following formula was used to calculate the brain water content of mice:
(wet weight – dry weight)/wet weight × 100% = brain water content (%).
Data Analysis
The present study was designed to generate groups of equal size. Besides, the research was undertaken using randomization and blinded analysis. All data in the study were presented as mean ± standard deviation (SD). SPSS 21.0 (RRID: SCR_002865) and GraphPad Prism 8.0 (GraphPad Software Inc., USA, RRID: SCR_002798) were used to perform data analysis. The normal distribution of data was analyzed using Shapiro–Wilk test. The homogeneity of variance was assessed by Levene’ test. The results of the analysis showed that all data conformed to normal distribution and homogeneity of variance. Finally, the difference between groups was analyzed and determined using one-way analysis of variance (ANOVA) followed by Duncan.
Results
ECJ Protects against the Impairment of Place Navigation and Spatial Probe Ability of Mice
The changes of place navigation and spatial probe ability of mice tested using MWM were used to assess brain injury in animal experiments. The change of escape latency is a common indicator of spatial navigation ability (Zhang et al., 2019). The results revealed that there were no differences in average escape latency among all groups on days 1 and 2 of training (Figure 1A and B). However, the escape latency was gradually reduced from day 3 to day 4 of training. The results showed (Figure 1C and D) that cerebral I/R could induce significant impairment of mouse spatial navigation ability, evidenced by higher escape latency in the cerebral I/R mice (compared with the sham group mice,
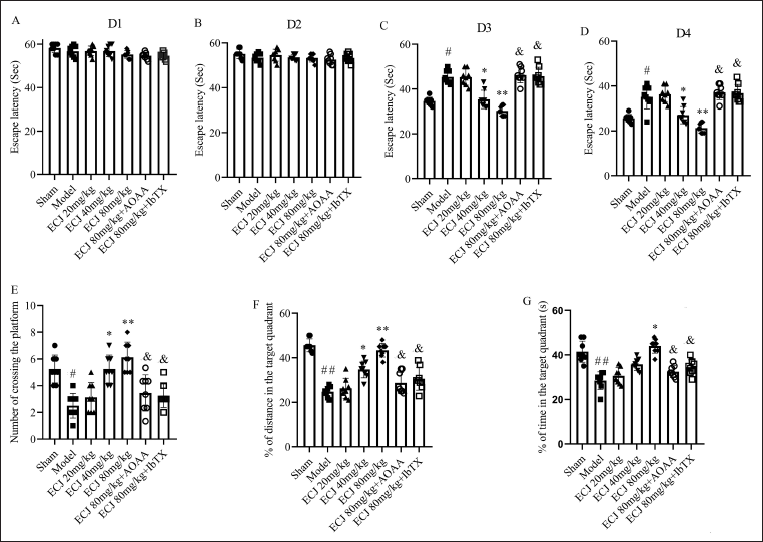
ECJ Reduces the Cerebral I/R-induced Brain Injury of Mice
For the sake of confirming the role of AOAA and IbTX on the ECJ-mediated neuroprotective effects against mouse cerebral I/R injury, we likewise demonstrated the change of brain water content, an important index of brain edema (Wen et al., 2018). The results were shown in Figure 2A that cerebral I/R led to marked enhancement of brain water content of mice (compared with sham group mice,
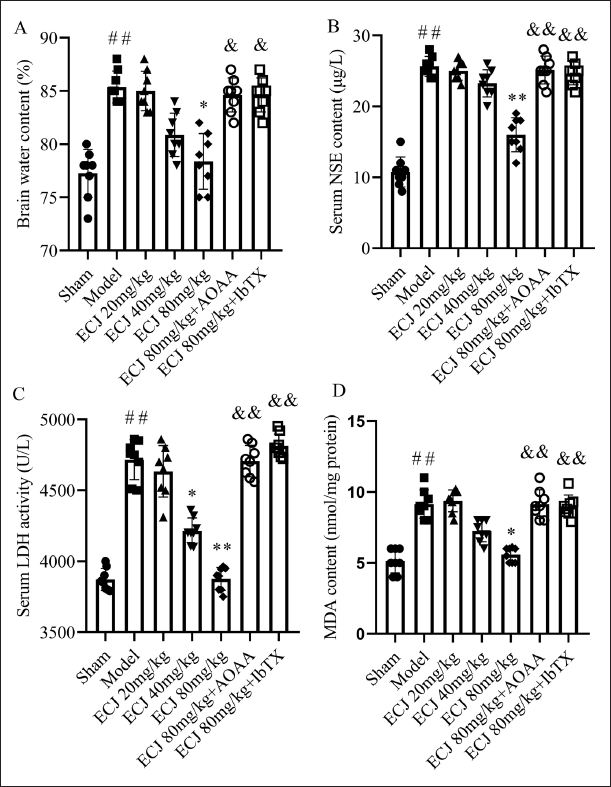
Besides, serum LDH level and NSE activity leaked from injured neurons are well known as major indexes of brain injury. MDA in brain tissues, a product of lipid peroxidation, also can be used as an indicator of brain injury (Wen et al., 2018). Therefore, we tested LDH level and NSE activity in mouse sera and assessed the changes of MDA content in mice brain tissue to further confirm the neuroprotective effect of ECJ and explore the relationship between the ECJ-mediated neuroprotection and IbTX and AOAA. Not surprisingly, cerebral I/R resulted in an obvious increment of LDH level and NSE activity in mouse sera and induced increased MDA content in mouse brain tissues (Figure 2B–D), suggesting brain injury (compared with sham group mice,
ECJ Promotes the Expressions of CBS and Improves the CBS-derived H2S Production
For the sake of confirming the relationship between ECJ-mediated neuroprotection and CBS-produced H2S, we tested the H2S content in mouse brain tissues. As shown in Figure 3A, 40 or 80 mg/kg ECJ could upregulate the H2S content in brain tissues of cerebral I/R mice (compared with the model group mice,
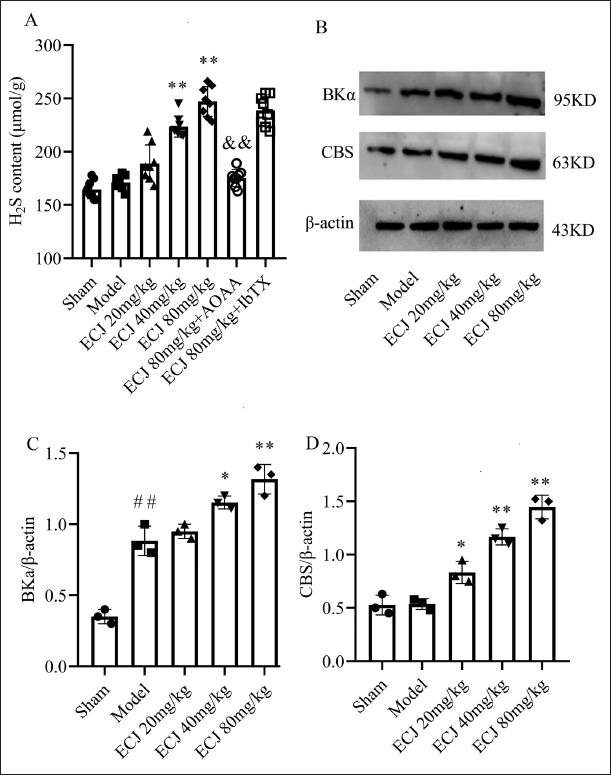
ECJ Improves the Expression of α Subunit of BKCa Channels (BKα)
In addition, we likewise assessed the expression of BKα in mice brain tissues to explore the relationship between the role of ECJ and BKCa channels. The results (Figure 3B and C) showed that ECJ can markedly and dose-dependently increase the expression of BKα in brain tissues of cerebral I/R mice (compared with model group mice,
ECJ Protects against the Neuronal Injury Induced by OGD/R via CBS/H2S-BKCa Pathway
Primary culture of mouse hippocampal neurons was used in the present study to confirm the roles of CBS/H2S and BKCa channels in ECJ-mediated neuroprotection. As shown in Figure 4A–C, ECJ (270, 810 mg/L) significantly promotes the expressions of BKα and CBS in OGD/R-treated hippocampal neurons (compared with OGD/R neurons,
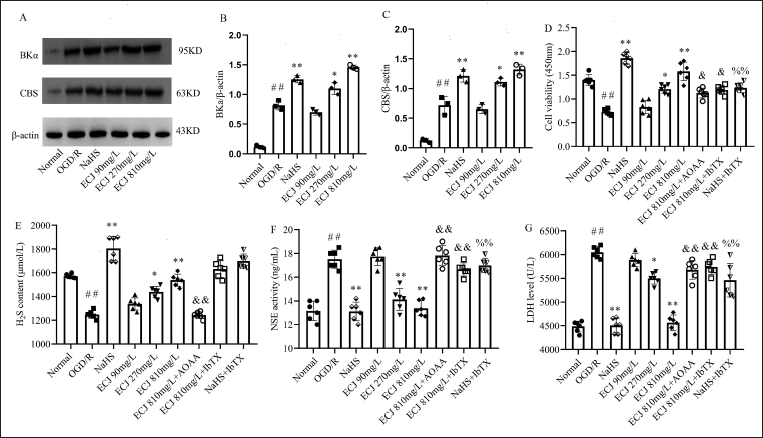
Subsequently, we detected the neuronal viability, NSE activity, and LDH level in the culture medium of neurons to evaluate the effect of ECJ and exogenous H2S donor NaHS on the OGD/R-induced neuronal injury. The results are shown in Figure 4F and G that both ECJ (270, 810 mg/L) and NaHS could promote cell viability, reduce NSE activity, and decrease LDH level in cell culture medium (compared with OGD/R neurons,
Discussion
Ischemic stroke, accompanied by a high rate of disability and mortality, is one of the common diseases. However, few numbers of ischemic stroke patients are eligible for t-PA treatment due to the therapeutic time window and secondary impairments (Wang et al., 2016). Thus, further study must be undertaken to reveal the internal neuroprotective strategies and explore the potential agents for ischemic stroke treatment.
As one of the endogenous gasotransmitters, H2S is discovered following nitric oxide (NO) and carbon monoxide (CO) (Chen et al., 2016). Endogenous H2S in brain tissues is mainly produced by CBS. H2S can promote neural functional recovery after ischemic injury (Lee et al., 2009). Although exogenous H2S donors such as Na2S and NaHS are widely used in animal research, they cannot be used in clinics. Considering that promoting the release of endogenous H2S is used to explore the protective mechanism of traditional Chinese medicines against various diseases (Guo et al., 2020; Jiao et al., 2019), we aimed to explore the therapeutic potential of ECJ on brain injury following cerebral I/R and investigate whether CBS-produced H2S is involved in the role of ECJ.
The 2-VO (Wen et al., 2021) was undertaken to build the mouse cerebral I/R model. The mice brain injury was evaluated via testing the impairment of spatial learning memory ability and brain edema (Huang et al., 2018). In addition, the MDA content in brain tissues, as well as serum LDH level and NSE activity leaked from injured neuron, have also been widely applied to evaluate brain injury (Jiang et al., 2017). The results revealed that cerebral I/R led to obvious brain injury, as evidenced by brain edema, impairment of mouse learning memory ability, and the increment of LDH and NSE levels in mouse sera and the increased content of MDA in brain tissues. Not surprisingly, the mouse brain injury induced by cerebral I/R could be remarkably ameliorated by ECJ treatment (40 or 80 mg/kg).
AOAA, the CBS inhibitor, was widely used to inhibit the release of CBS-produced H2S
BKCa channel is widely expressed in the nerve cell membrane. BKCa channel activation limits neuronal depolarization following ischemia injury, lowers intracellular Ca2+, and ameliorates neuronal disorders (Contet et al., 2016; N’Gouemo, 2014). We previously found that BKCa channel activation participates in the H2S-mediated neuroprotection (Wen et al., 2021). Therefore, we likewise sought to further demonstrate whether BKCa channel activation is related to the neuroprotection of ECJ by using IbTX.
IbTX, BKCa channel inhibitor, can be used to inhibit the activation of BKCa channels (Bao et al., 2023). We previously found that IbTX obviously inhibits the activity of BKCa channels in neurons, but does not affect both the cerebral I/R injury of mice and neuronal injury following OGD/R (Wen et al., 2021). Thus, in the present study, IbTX was used to assess the effect of BKCa channels in ECJ-mediated neuroprotection. We found that IbTX blocked the neuroprotection of ECJ against mouse brain injury following cerebral I/R. Besides, ECJ increases the expression of BKα in brain tissues. These data indicated that BKCa channel activation is also related to ECJ-mediated neuroprotection.
In order to further demonstrate the neuroprotective role of ECJ and explore the relationship between H2S and BKCa channels in ECJ-mediated neuroprotection, we likewise assessed the roles of exogenous H2S donor NaHS and ECJ in OGD/R-mediated neuronal injury. The results revealed that ECJ could protect against OGD/R-induced neuronal injury, as evidenced by ECJ-mediated increased cell viability, decreased LDH level, and reduced NSE activity in culture supernatant released from injured neurons. Exogenous H2S donor NaHS has a similar neuroprotective role against OGD/R-induced neuronal injury. Furthermore, both ECJ and NaHS obviously promote the expressions of CBS and BKα in culture neurons following OGD/R and improve the H2S production from OGD/R-treated neurons. Moreover, the beneficial effects of ECJ and exogenous H2S donor NaHS on the neuronal injury following OGD/R could be inhibited by IbTX. Thus, we proposed that the ECJ-mediated neuroprotection against OGD/R-induced neuronal injury is related to the CBS/H2S-BKCa pathway.
Conclusion
In summary, we found that (a) ECJ has eminent protection against the cerebral I/R injury of mice and protects against OGD/R-induced neuronal injury
Footnotes
Abbreviations
AOAA: Aminooxyacetic acid; CBS: Cystathionine-β-synthase; CCAs: Common carotid arteries; H2S: Hydrogen sulfide; I/R: Ischemia-reperfusion; LDH: Lactate dehydrogenase; MDA: Malondialdehyde; MWM: Morris water maze; NSE: Neuron-specific enolase; IbTX: Iberiotoxin.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The protocol of animal experiment was reviewed and approved by the Ethics Review Committee of Hefei Technical College, approval number is LLSC 20210661, which abided by the rules outlined in the Guide for the Care and Use of Laboratory. Animals published by the US National Institutes of Health (NIH publication number 86-23, revised 2011).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science Foundation of Colleges and Universities of Anhui Province in 2023 (No. 2023AH052541) and the Natural Science Foundation of Hefei Technology College (No. 2023KJA04),
