Abstract
Background and Purpose
Cantharidin can or cannot reverse the drug resistance of non-small cell lung cancer to cisplatin (DDP) by reducing the expression of programmed cell death ligand 1 (PD-L1).
Materials and Methods
The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide method was used to screen for sublethal doses of cantharidin and determine its sensitizing effect on DDP. Detect the apoptosis rate of A549/DDP cells treated with cantharidin, DDP, or the combination of cantharidin and DDP. The effects of cantharidin on the expressions of p53 and PD-L1 in the cell line were investigated. Furthermore, pifithrin-α (PFT-α) pretreatment was performed to inhibit the function of p53 and verify whether cantharidin can affect the expression of PD-L1 through p53.
Results
Cantharidin significantly enhanced the sensitivity of cells to DDP and increased the apoptosis of cells induced by DDP. Cantharidin upregulated the expression of p53 in A549/DDP cells with wild-type p53 and downregulated the expression of PD-L1. Pretreatment with PFT-α inhibited the apoptosis of cells induced by the combination treatment of cantharidin and DDP.
Conclusion
Cantharidin can regulate the expression of p53 and downregulate the PD-L1 in non-small cell lung cancer with wild-type p53. It may enhance the sensitivity of non-small cell lung cancer to DDP by further altering the levels of PD-L1 through the regulation of p53 expression.
Introduction
Most lung cancer patients are diagnosed at an advanced stage, thus causing a low 5-year survival rate (Sung et al., 2021). Targeted therapy is preferred for patients with unresectable advanced lung adenocarcinoma who have sensitive gene mutations. However, for patients without sensitive gene mutations, chemotherapy, radiation therapy, and immunotherapy are the main treatment options, among which chemotherapy is indispensable (Fouad et al., 2021). Platinum-based combination chemotherapy, especially cisplatin (DDP), has been proven to be an effective treatment for advanced lung adenocarcinoma. However, chemotherapy resistance is a major barrier. It is important to know DDP resistance in lung adenocarcinoma treatment and find new targets and drugs to prevent and reverse drug resistance (Burki, 2021).
Programmed cell death ligand 1 (PD-L1) plays an important role in cancer cells evading immune surveillance. After binding with programmed cell death receptor 1 (PD-1), PD-L1 causes T cell exhaustion, which will further inhibit immune function (Zhang et al., 2021; Zheng et al., 2021).
Cantharidin is an effective ingredient of the insect, great cantharides. It has the medical effects of relieving itching, improving local neural nutrition, stimulating hair follicles, and promoting hair growth. It is mainly used for the treatment of cancer (Wang et al., 2018). Our research aims to investigate whether cantharidin can reverse the DDP resistance of non-small cell lung cancer (NSCLC) by reducing the expression of PD-L1. Currently, there are no related studies, making our research innovative.
Materials and Methods
Materials
Cantharidin: No. M19851, Meryer.
A549/DDP: No. 3111C0001CCC000519, Cell Resources Center, Institute of Basic Medicine, Chinese Academy of Medical Sciences.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT): No. 12-4657, Biyuntian Biotechnology Co., Ltd.
Glyceraldehyde 3-phosphate dehydrogenase (GAPDH): 70R-17422, Fitzgerald.
PD-L1: BES-T71-41, Shanghai Bolsen Biotechnology Co., Ltd.
p53: BES207608RP, Shanghai Bolsen Biotechnology Co., Ltd.
Methods
MTT assay to evaluate the effect of cantharidin on the proliferation capability of A549/DDP cells.
Cells were obtained and divided into a control group and a cantharidin group. The control group was added with 10 µl of culture medium, while the cantharidin group was treated with 10 µl of prepared culture medium containing cantharidin at 5, 10, 15, 20, 25, and 30 µM concentrations. The final concentrations of cells in each well were set at 0.5, 1, 1.5, 2, 2.5, and 3 µM after a 48-hour incubation. Cell viability was then determined according to the outlined steps of the MTT assay.
Flow Cytometry to Detect the Effect of Cantharidin on Apoptosis of A549/DDP Cells
Cells were obtained and cultured until they adhered to the wall, then 1.5 µM cantharidin, 8 µM DDP, or a combination of 1.5 µM cantharidin and 8 µM DDP were added to each group. Analysis using flow cytometry.
Cells were divided into the control group, the 30 M pifithrin-α (PFT-α) group, the 8 µM DDP group, the 30 M PFT-α combined with 8 µM DDP group, the 1.5 µM cantharidin combined with 8 µM DDP group, and the combination of 30 pM PFT-α, 1.5 µM cantharidin and 8 µM DDP group. Flow cytometry was employed for analysis.
Western Blot (WB) Method to Detect the Effects of Cantharidin on Various Proteins
A549/DDP cells were obtained and cultured until reaching wall adherence. Fresh culture medium containing cantharidin was added, with a final concentration of 1.5 µM. The cells were then cultured for 0, 12, 24, and 48 hours, respectively. WB method was used to detect the proteins according to standard procedures.
A fresh culture medium containing different substances was added. The control group did not receive PFT-α (final concentration of 30 µM) or cantharidin (final concentration of 1.5 µM). The PFT-α combined with the cantharidin group received a final concentration of 30 µM PFT-α and 1.5 µM cantharidin. The cells were then cultured for 48 hours in the incubator, and the WB method was used to detect the proteins accordingly.
Statistical Analysis
Three repeated experiments were conducted, using SPSS 19.0 as the statistical software. Between-group comparisons were made using a t-test, with p < 0.05 indicating significant statistical differences.
Results
The effect of cantharidin on the proliferation of A549/DDP cells.
Statistical analysis showed that 1.5 µM cantharidin began to inhibit the proliferation of A549/DDP cells. Therefore, we selected 1.5 µM cantharidin as the drug concentration for subsequent A549/DDP cell sensitization tests. The results are shown in Table 1.
The Effect of Cantharidin on the Survival Rate of A549/Cisplatin (DDP) cells.

The Effect of Cantharidin on the Apoptosis of A549/DDP Cells
Cantharidin enhanced the apoptotic induction effect of DDP on A549/DDP cells. There is a difference in the apoptotic rate of cells between the DDP group and the DDP + cantharidin group, as shown in Figure 1.
The administration of PFT-α alone does not alter the apoptotic status of A549/DDP cells, nor does it change the apoptosis caused by DDP. However, PFT-α can partially reverse the sensitizing effect of cantharidin-induced DDP. There is a statistical significance in the apoptotic rate of cells between the cantharidin + DDP group and the cantharidin + DDP + PFT-α group, as shown in Table 2.
The Effect of Cantharidin on the Apoptosis of A549/Cisplatin (DDP) Cells.
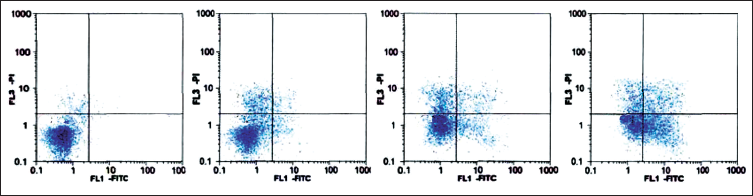
The Effect of Cantharidin on the Apoptosis of A549/Cisplatin (DDP) Cells.

The Effects of Cantharidin on Various Proteins
We found that 1.5 µM cantharidin upregulated the p53 protein. Significant differences in the expression of p53 protein were observed after 12 hours of cultivation, as shown in Figure 2.
Partial restoration of the upregulation of p53 expression induced by cantharidin in A549/DDP cells was achieved with PFT-α treatment. Comparative analysis between the cantharidin group and the PFT-α + cantharidin group revealed an increase in the expression of p53 protein, as shown in Figure 3.
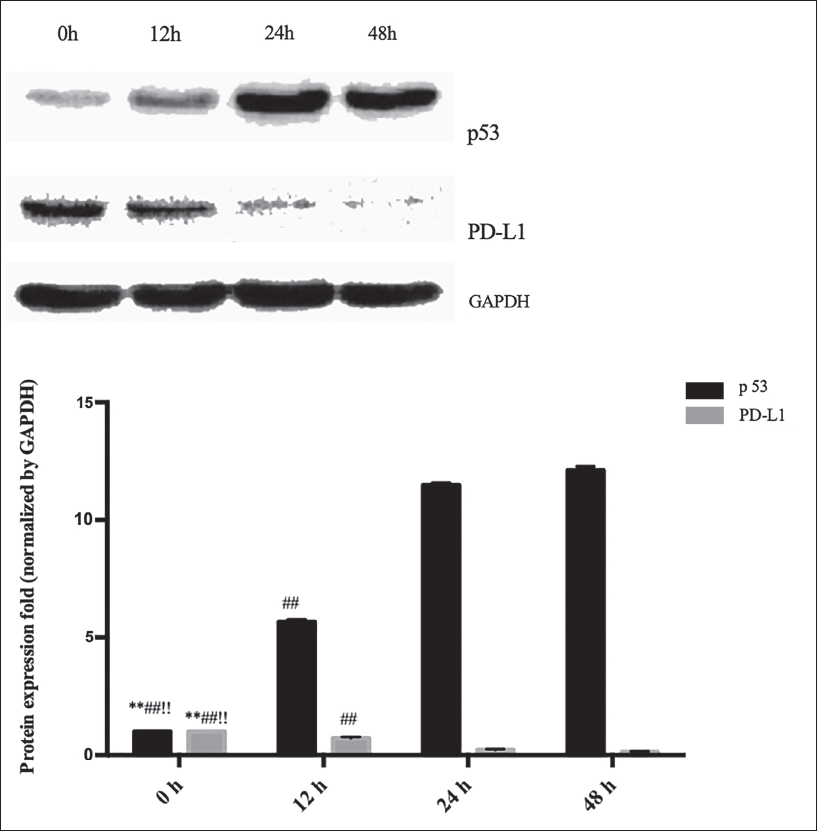
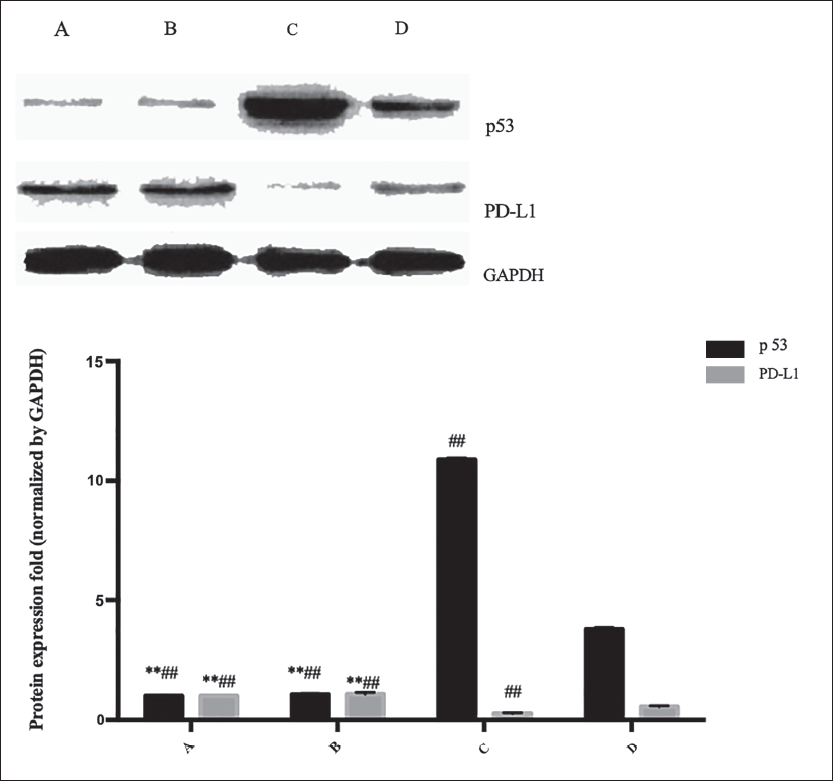
Discussion
PD-L1 in tumor cells has special functions other than an immune checkpoint ligand, including stimulating cancer progression, promoting epithelial–mesenchymal transition, and resistance to chemotherapy drugs. Studies have also suggested that it may be related to the resistance of lung adenocarcinoma cells to DDP, and it indicates strong resistance to DDP (Naba et al., 2020). Another study examined the expression of PD-L1 in tumor tissue and adjacent tissue, evaluated the correlation between PD-L1 expression and tumor histological type, and lymph node metastasis of tumor tissue, and assessed the overall survival rate. PD-L1 is associated with histological type and overall survival rate. The PD-L1 was higher in adenocarcinoma or patients with a postoperative survival time of less than 3 years (Zhu et al., 2021). Cox model analysis also suggested that PD-L1 is a poor prognostic factor (Kang et al., 2020; Pecere et al., 2003).
Another study showed that a variety of chemotherapy drugs can increase the PD-L1 in tumor cells, such as paclitaxel, fluorouracil, oxaliplatin, and irinotecan. Other studies suggested that DDP-containing regimens in neoadjuvant therapy for lung cancer can increase. High expression of PD-L1 protein has also been observed in DDP-resistant cell lines (Rawangkan et al., 2018). However, there is currently limited clinical literature concerning the effects of DDP on PD-L1 expression in lung tumor tissue, making it difficult to conclude whether DDP increases PD-L1 expression (Wherry, 2011). Therefore, we are currently collecting tissue specimens from advanced lung adenocarcinoma patients before and after chemotherapy containing DDP. These samples were obtained through lung puncture and will be analyzed in small groups to observe relevant results (Melosky et al., 2020).
p53 can regulate cellular stress responses, participate in DNA damage repair, inhibit abnormal mitosis processes, and mediate various antiproliferative processes. It is important in preventing tumor formation, and mutations or dysfunctions of the p53 gene play an important role in the occurrence of most human tumors. MiRNA has various regulatory effects in animal and plant cells, mainly participating in posttranscriptional gene expression regulation. miRNA has been proven to regulate epigenetic mechanisms by guiding target mRNA cleavage or translation inhibition to regulate gene expression (Raver-Shapira et al., 2007).
A study found that when screening miRNAs regulated by p53, the abundance of miR-34a increased most significantly after p53 activation. Both p53 activation and DNA damage can induce its primary transcripts in a p53-dependent manner. Changes in miR-34a can cause cells to stagnate, thereby inhibiting tumor cell proliferation. p53 can transplant tumor mouse models by binding-to-binding sites within the miR-34a gene. Through transfection experiments, it has been demonstrated that silencing miR-34a can weaken p53-mediated tumor cell apoptosis (Prajapati et al., 2011).
p53 protein increase in miR-34a, miR-34b, and miR-34c levels, as well as a decrease in PD-L1 expression levels, indicating a negative correlation. The negative correlation between miR-34a and PD-L1 in vivo, the researchers compared tumor tissue samples. The negative relationship between them in NSCLC cells and tumor samples suggests that p53 may inhibit PD-L1 expression through miR-34 (Gong et al., 2017).
Cantharidin is an effective ingredient of the insect, great cantharides. It has the medical effects of relieving itching, improving local neural nutrition, stimulating hair follicles, and promoting hair growth. The Chinese alias is hexahydro-3a, 7a-dimethyl-4,7-epoxidisobenzofuran-1,3-dione, which is an organic compound with the chemical formula C10H12O4. It is insoluble in cold water, but soluble in hot water, acetone, chloroform, ethyl acetate, and oil. It is slightly soluble in ethanol and has a certain inhibitory effect on various experimental animal tumors. Especially when the number of tumor cells inoculated with ascitic tumors is small, it can be seen that the survival period of mice is prolonged. Its mechanism of action may be to inhibit the synthesis of proteins and nucleic acids. Low doses do not affect the body’s immune function, while high doses lead to a decrease in immune function. Cantharidin is present in the body of cantharides and dried beetles and can be used as a skin redness, foaming, or hair growth agent. N-hydroxy cantharidin prepared from cantharidin has been tested for liver cancer and has shown certain therapeutic effects. At present, it has been found in clinical practice that taking too much may lead to symptoms such as vomiting and diarrhea, and in more severe cases, heart and kidney failure may occur. Banjiao Su has a strong irritant effect. After swallowing it, it takes its way through the bladder and is excreted with urine. When it flows through, it creates a burning sensation in the bladder and urethra, which stimulates the sexual organs due to reflex effects. When using, pay attention to the fact that cantharidin is a highly toxic drug. If it is abused, overused, used together with alcohol garlic, used raw, with a large external area, accumulated, liver and kidney dysfunction, or taken orally, it can cause poisoning. Therefore, the dosage should be very accurate in order to be effectively applied to the treatment of diseases (Wang et al., 2018).
Tumor cells expressing wild-type p53, an increase in p53 protein levels leads to a decrease in PD-L1 expression. To further confirm the negative correlation of PD-L1 in vivo, the researchers compared tumor tissue samples with wild-type p53 and mutant p53. The results showed that compared with wild-type p53 tumors, tissues with mutant p53 had higher PD-L1 expression. The negative relationship between NSCLC cells and PD-L1 in tumor samples suggests that p53 may inhibit PD-L1. Its research confirms that p53 can regulate the expression of PD-L1 (Cortez et al., 2016).
Conclusion
Our study is the first to investigate the mechanism by which cantharidin reverses DDP resistance in lung cancer. Cell apoptosis is the main way DDP induces cell death. Our research further confirmed the correlation between high PD-L1 expression and DDP resistance in lung cancer cells and emphasized that the combination of cantharidin and DDP can promote the apoptosis of lung cancer cells. This study found that cantharidin increases the chemotherapy sensitivity of lung cancer cells and enriched the theoretical basis and its potential as a sensitizing agent for DDP resistance, thus laying the foundation for further clinical applications.
Footnotes
Abbreviations
NSCLC: Non-small cell lung cancer; DDP: Cisplatin; PD-L1: Programmed cell death ligand 1; WB: Western blot; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide.
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Research experiments conducted in this article with animals or humans were approved by the Ethical Committee and responsible authorities of our research organization(s) following all guidelines, regulations, and legal, and ethical standards as required for humans or animals. Our research is a cell experiment.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
