Abstract
A naturally occurring xanthone, mangostanin (1,3,6-trihydroxy-7-methoxy-2,8-bis(3-methylbut-2-en-1-yl)-9H-xanthen-9-one), was isolated from Garcinia mangostana L. It demonstrates a range of pharmacological effects, such as antibacterial, antifungal, antiviral, anti-inflammatory, and anticancer activity. The α-mangostin’s isoprenyl and benzopyrone skeleton side chains are crucial to its antibacterial activity. In this review, we review the current approach for the isolation of α-mangostin from plant sources and the recent report on its biological activity. The structure–activity relationship is discussed to demonstrate the pharmacophore of α-mangostin responsible for its anti-infective properties, including antibacterial, antifungal, and antiviral activities. The potential approaches for developing α-mangostin for the treatment of infectious diseases are proposed as future perspectives.
Introduction
Microbes, encompassing bacteria, viruses, fungi, and parasites, serve as the origin of infectious ailments. Within our bodies, a diverse array of microorganisms coexist. While many of these organisms are harmless or even advantageous under normal circumstances, certain ones have the potential to induce illness in specific contexts. Certain communicable diseases can disseminate through direct human-to-human contact or via transmission facilitated by insects or other animal vectors (Mir, 2022). Additionally, swallowing tainted food or water or being exposed to environmental organisms are two other ways that infectious diseases can spread. Every stage of aging has a tendency to see an increase in infectious diseases. The leading infectious diseases in developed countries include cancer, acquired immunodeficiency syndrome (AIDS), diabetes, and other diseases caused by various bacterial and virus species (Coates et al., 2020). Infectious diseases pose a substantial burden on public health and global economies. Their occurrence and spread are primarily influenced by socioeconomic, environmental, and ecological factors. These interconnected elements play a crucial role in shaping the emergence and transmission of infectious diseases, highlighting the complex nature of their dynamics. Addressing these underlying factors is essential for effective prevention, control, and mitigation strategies to safeguard public health and mitigate the economic impact of infectious diseases on a global scale (Shaw-Taylor, 2020).
Globally, approximately half of the deaths are caused by infectious diseases yearly. Most of the pathogens involved in infectious diseases are bacteria and rickettsia. This is generally illustrated by the rise of multidrug-resistant bacterial strains, particularly vancomycin- and methicillin-resistant Staphylococcus aureus (MRSA) (Sagar et al., 2019). The challenge posed by antibiotic resistance has taken center stage in public healthcare in the twenty-first century. In 2013, the Centers for Disease Control and Prevention (CDC) reported that around 2 million Americans experienced severe infections caused by antibiotic-resistant bacteria, which were no longer susceptible to commonly used antibiotics. Tragically, out of those cases, approximately 23,000 individuals lost their lives due to these resistant infections (Kadri, 2020). Similarly, in 2007, Europe witnessed approximately 400,000 individuals being infected with multidrug-resistant bacteria, leading to the unfortunate deaths of 25,000 people (Rachid, 2019). These statistics highlight the alarming global impact of antibiotic resistance and the urgent need to address this growing public health concern (Kaur et al., 2020).
Herbal medicines have been used in the primary healthcare of humans and animals since ancient times. The type of microbe that causes an infectious disease will determine how it is treated. Infections that are caused by bacteria are typically treated with antibiotics, which eradicate the bacteria and end the infection. Viral infections, however, are often treated with supportive therapy like rest and increased fluid intake. However, there are situations when people can benefit from antiviral drugs like oseltamivir phosphate. Fluconazole and mebendazole are examples of antifungal and antiparasitic medications used to treat infections brought on by fungi and other parasites (Mequanent, 2022). However, due to resistance to these available medicines for infectious diseases, there is a promising situation across the world to find new ways to treat infectious diseases.
Natural products are a significant source of chemical variety and have produced significant therapeutic medicines for many infectious illnesses caused by bacteria. It is generally known that various plant extracts have a variety of effects against bacteria that spread infectious diseases. They work together synergistically in terms of pharmacology because they each target a different spot and affect it in a different way to increase the response of the cell (Abdel-Razek et al., 2020; Chopra & Dhingra, 2021). Furthermore, certain plant extracts showcase a spectrum of pharmacological properties, including antimicrobial, anti-inflammatory, antiallergic, immunomodulatory, and antioxidant effects. These properties hold potential for the treatment of infectious diseases. Since antiquity, humans have used plants to treat common infectious diseases. The WHO reports that traditional medicine was used for primary healthcare by populations of more than 80% in various Asian and African nations. Ayurvedic, naturopathic, homeopathic, traditional oriental, and Native American Indian medicine are just a few examples of the complementary and alternative therapies that between 70% and 80% of people in many affluent nations rely on (Peltzer & Pengpid, 2015; Siddique et al., 2021). The most widely used traditional form of medicine, and the most lucrative on the global medical market, according to the WHO, is herbal medicine. In addition, the WHO Traditional Medicine Plan 2014–2023 emphasizes enhancing the quality, safety, and efficacy of traditional medicine by setting regulatory and quality assurance requirements (WHO, 2019). Natural products with their second metabolites, such as xanthones, flavonoids, and terpenoids, have been a source of the most active ingredients. Apart from antibiotics, several bacteria, fungi, and viruses have been reported to be sensitive to xanthones, which are found in some higher plant families, lichens, and fungi (Goga et al., 2020; Kandar, 2021). There are five xanthone groups: Prenylated xanthones, xanthonolignoids, xanthone glycosides, oxygenated xanthones, and miscellaneous xanthones (Aravind et al., 2016; Lee et al., 2023). The prenylated xanthones are isolated from pericarp, bark, leaves, and whole fruit of mangosteen, a tropical tree harvested in tropical rainforests of some Southeast Asia countries, e.g., Thailand, Malaysia, and Indonesia. The xanthones obtained from the mangosteen fruits, such as α-, β-, and γ-mangostin, gartanin, garcinone E, and 8-deoxygartanin, produce important biological activities (Ansori et al., 2020). Several xanthones from the pericarp of mangosteen are used as medicinal agents for treating skin infections, diarrhea, and wound healing (Rizaldy et al., 2021). In addition, they have inhibitory effects on the growth of S. aureus, Candida albicans, Pseudomonas aeruginosa, Salmonella typhimurium, Bacillus subtilis, HIV, and Propionibacterium acnes (Anand et al., 2020; Dikpınar & Süzgeç-Selçuk, 2020; Loureiro et al., 2019; Verma et al., 2021). α-Mangostin has been reported to show antibacterial, antifungal, and antiviral activities. The chromone (or 1,4-benzopyrone) and iso-phenyl structures could play a significant role in its action (Hart & Cock, 2016; Patil et al., 2021). In this review, the authors summarize information regarding the antibacterial potential of α-mangostin, which can be useful for developing antibacterial agents and treating infectious diseases.
Chemistry Extraction, Isolation, and Structure Elucidation
Mangostin, first discovered in 1855 from the mangosteen tree plant (Pimchan, 2017), is a specialized compound found in Garcinia mangostana. To obtain α-mangostin from the plant, various extraction methods have been employed, including Soxhlet extraction (Kumaran & Sukumaran, 2021; Muchtaridi et al., 2017; Tatiya-Aphiradee et al., 2021; Zakia et al., 2020), decoction extraction (Moussavi et al., 2024), microwave-assisted extraction (Fang et al., 2011; Hiew et al., 2021; Ihsanpuro et al., 2023; Kwansang et al., 2022; Meah & Panichayupakaranant, 2020; Mohammad et al., 2019; Tangpromphan et al., 2023), maceration (Al-Massarani et al., 2013; Eukun Sage et al., 2018; Moongkarndi et al., 2015), ultrasonic-assisted extraction (Asasutjarit et al., 2019; Plaza et al., 2021; Zhang et al., 2015), supercritical fluid extraction (Chhouk et al., 2016; Rahayu et al., 2023), supercritical carbon dioxide extraction (Farooq et al., 2023; Hamid et al., 2018; Lee et al., 2019), aqueous micellar biphasic system extraction (Kok et al., 2021; Tan et al., 2017), and eutectic solvents (Mulia et al., 2016, 2019; Yuvanatemiya et al., 2022). The choice of solvent extraction method is crucial for isolating α-mangostin effectively. Researchers have investigated various single solvent extraction methods, such as hexane (Van Quang, Minh, et al., 2022; Van Heuvel, Schatz, et al., 2022), chloroform (Mohamed et al., 2017; Paul et al., 2023), 70% ethanol or 85% ethanol (Al-Massarani et al., 2013; Tangpromphan et al., 2023), 95% ethanol (Suhandi et al., 2023; Upegu et al., 2015), and methanol (Naing et al., 2023; Mohamed et al., 2014). For quantitative and qualitative analysis of α-mangostin, researchers have employed various analytical techniques to accurately determine its presence and concentration in different samples, including high-performance liquid chromatography (HPLC) (Permana et al., 2023; Pothitirat & Gritsanapan, 2009), ultra-performance liquid chromatography (UPLC), liquid chromatography–mass spectrometry (LC–MS), and nuclear magnetic resonance (NMR) (Masullo et al., 2022; Pandey et al., 2015).
Physicochemical Properties and Chemical Constituents of α-Mangostin
α-Mangostin, depicted in Figure 1, is a prominent xanthone compound sourced from the mangosteen tree, known for its yellow color and as a major constituent among the 50 xanthones isolated from the fruit’s pericarp (Aizat et al., 2019). With a molecular formula of C24H26O6, α-mangostin features a 9H-xanthene framework with hydroxy groups at positions 1, 3, and 6, a methoxy group at position 7, an oxo group at position 9, and prenyl groups at positions 2 and 8. While insoluble in water, it demonstrates solubility in various organic solvents. Initially identified as mangostin in 1855, it was later renamed α-mangostin, with its structural attributes elucidated by Dragendorff (1930) and Murakami (1932). The molecular formula was subsequently determined by Yates and Stout in 1958, with Dragendorff accomplishing its isolation and characterization in 1968 (Yates & Bhat, 1968). Predominantly, xanthone derivatives like α- and β-mangostins act as the primary bioactive secondary metabolites in G. mangostana, drawing significant interest for their pharmacological properties (Pimchan, 2017). While over 60 additional xanthones have been isolated from various parts of the G. mangostana plant, research has chiefly concentrated on extracting and elucidating their structures (Ong et al., 2020). α-Mangostin exhibits a diverse range of biological activities, including antiviral, antibacterial, anti-inflammatory, antioxidant, antiobesity, antidiabetic, neuroprotective, and antimalarial properties, with its isoprenyl group notably recognized for destabilizing MRSA membranes, enhancing permeability, and optimizing antibacterial activity (Chen et al., 2018; Phitaktim et al., 2016).
Structure of α-Mangostin.
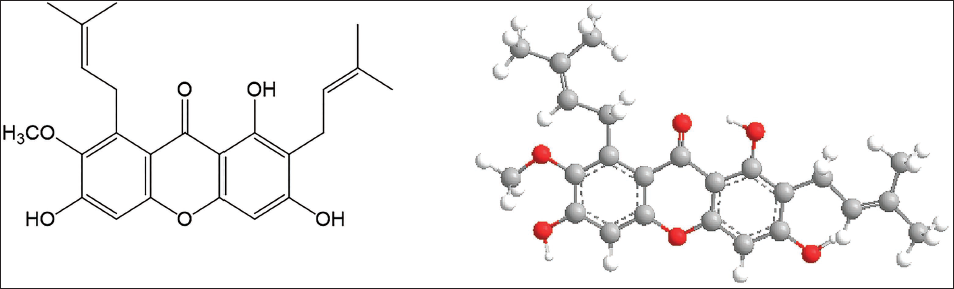
Uses of α-Mangostin in Infectious Diseases
Bacterial Infections
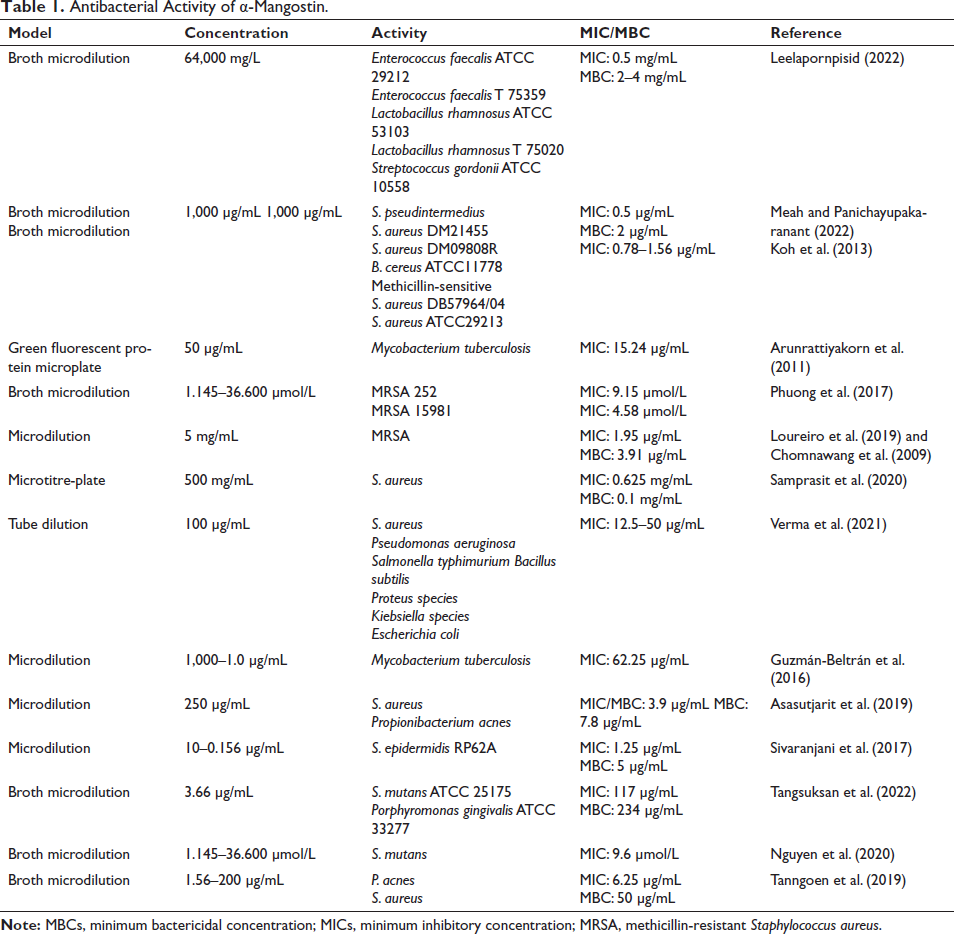
The increasing prevalence of vancomycin-resistant Enterococci (VRE) and MRSA infections poses a significant global health threat, with rising rates of mortality and morbidity (Mancuso et al., 2021). α-Mangostin has demonstrated potent antibacterial properties against MRSA and VRE, with minimum inhibitory concentration (MICs) and minimum bactericidal concentration (MBCs) ranging from 0.78 to 6.25 µg/mL (Koh et al., 2013; Meah et al., 2020; Sakagami et al., 2005; Sivaranjani et al., 2017; Sultan et al., 2022). Its effectiveness extends to other pathogens such as Staphylococcus epidermidis, S. aureus, Streptococcus pyogenes, and P. acnes, with MIC values ranging from 0.78 to 6.25 µg/mL (Ahmad et al., 2019). α-Mangostin exhibits synergistic effects with various antibiotics, enhancing their efficacy against resistant strains. However, caution is warranted concerning its consumption, as it may disrupt the gut microbiome (Gutierrez-Orozco et al., 2015). Furthermore, α-mangostin shows promise as an antituberculosis agent and in inhibiting HIV-1 protease. The broad spectrum of antibacterial activities exhibited by α-mangostin underscores its potential as a valuable therapeutic agent against diverse microbial pathogens (Nchiozem-Ngnitedem et al., 2023; Saleem, 2018). The review of literature highlighted numerous antibacterial activities of α-mangostin against various microorganisms (Table 1).
Antibacterial Activity of α-Mangostin.
Acne Infection
Acne, stemming from factors like skin inflammation, hyperproliferation of ductal epidermis, excessive sebum, and P. acnes colonization, finds potential treatment in α- and β-mangostins due to their antibacterial properties. Research utilizing CCK-8 indicates their effectiveness in hindering P. acnes growth and keratinocyte proliferation, while also inhibiting mitogen-activated protein kinase (MAPK) and nuclear factor-kappa B (NF-κB) pathways, reducing proinflammatory cytokines. This mechanism mitigates the inflammatory response tied to P. acnes infection, offering promise for acne treatment (Araviiskaia et al., 2022; Mendoza et al., 2013; Xu et al., 2018). With rising resistance to conventional antibiotics, α-mangostin’s synergistic effects with erythromycin, tetracycline, and clindamycin highlight its potential as a cutting-edge acne medication (Ahmad et al., 2019).
Multidrug-resistant Bacteria
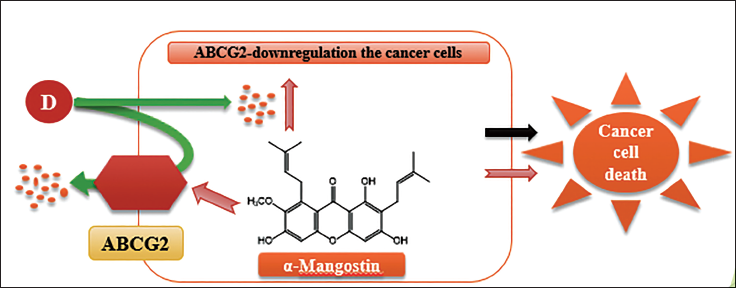
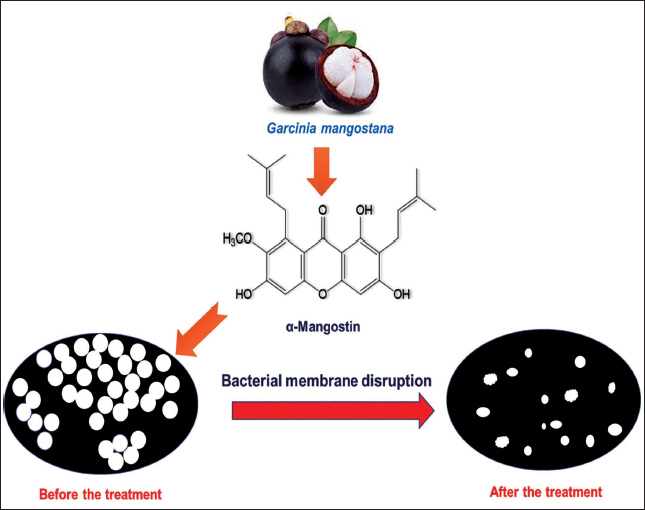
Antibiotic resistance presents a substantial challenge in healthcare, driven by three primary mechanisms: Alterations in the active site of the target, enzymatic degradation or modification of antibiotics, and mutations in the target site. Enzymatic processes like hydrolysis and group transfer contribute to antibiotic inactivation, while reduced outer membrane permeability and increased antibiotic efflux decrease drug accumulation, collectively fostering resistance (Hemaiswarya et al., 2008; Martínez et al., 2020). Multidrug therapy, incorporating natural antibacterial compounds like xanthones, emerges as a promising strategy to combat resistance (Panichayupakaranant et al., 2019). Furthermore, multidrug resistance (MDR) in cancer treatment poses additional hurdles, often associated with upregulated ATP-binding cassette (ABC) drug transporter expression (Kukal et al., 2021; Wu et al., 2017). Natural compounds like α-mangostin offer potential solutions, inhibiting drug transport in ABCG2-overexpressing MDR cancer cells and reversing MDR (Figure 2). Transcriptome and proteome analyses reveal α-mangostin’s diverse effects on cellular processes, including membrane integrity, biofilm formation, and stress responses, shedding light on its antibacterial mechanisms against pathogens like S. epidermidis. Notably, α-mangostin exhibits synergistic effects with conventional antibiotics and demonstrates rapid bactericidal activity by disrupting bacterial membranes, suggesting its potential as a therapeutic agent against antibiotic-resistant infections and MDR cancers (Sivaranjani et al., 2019) (Figure 3).
Displaying ABCG2-overexpressing Multidrug Resistance (MDR) Cancer Cells Blocked by α-Mangostin.
α-Mangostin Disrupts the Bacterial Cell Membrane.
Used in Fungal Infections
Oral Candidiasis
Oral thrush, a form of candidiasis affecting the mouth’s mucous membranes, has become increasingly prevalent, particularly among individuals with compromised immune systems, endocrine disorders, cancer, advanced age, or undergoing radiotherapy (Lu, 2021). Existing antifungal medications, primarily polyenes and azoles, remain effective treatments, although limited options persist due to escalating antifungal resistance (Lakhani et al., 2019). α-Mangostin demonstrates promising antifungal activity against C. albicans, the primary causative agent of oral candidiasis, outperforming clotrimazole and nystatin. Its minimal fungicidal concentrations (MIC and MFC) against C. albicans are 1,000 and 2,000 µg/mL, respectively, with minimal cytotoxicity to human gingival fibroblasts at 4,000 µg/mL (Kaomongkolgit & Jamdee, 2015). Furthermore, microbial metabolism studies reveal α-mangostin’s potent antifungal effects and identify metabolites generated during its interaction with fungi, highlighting its potential as a therapeutic option for oral candidiasis (Arunrattiyakorn et al., 2011). Different antifungal actions of α-mangostin against various pathogens are identified in this review (Table 2).
Antifungal Activity of α-Mangostin.
Used in Viral Infections
Dengue Fever
Dengue virus (DENV), transmitted by mosquitoes, poses a significant global health threat, with up to 50–100 million infections and 20,000 fatalities annually in over 100 endemic countries (Dhiman et al., 2022). While the recently approved dengue vaccine, Dengvaxia, shows limited efficacy, effective antiviral medications remain unavailable (Khetarpal & Khanna, 2016; Tully & Griffiths, 2021). α-Mangostin emerges as a promising candidate for treating severe DENV infection, reducing virus production and cytokine expression in HepG2 and Huh-7 cells. It significantly reduces infection rates of all four DENV serotypes by 47–55% and suppresses DENV1-4 production by 100-fold (Tarasuk et al., 2017). α-Mangostin’s potency surpasses conventional antiviral medications and painkillers, presenting it as a potential treatment for DENV infection by inhibiting viral replication and alleviating inflammation (Sugiyanto et al., 2019).
HIV/AIDS
In the fight against HIV-1, the primary cause of AIDS, combination therapy, known as highly active antiretroviral therapy (HAART), targets essential viral enzymes like HIV-1 reverse transcriptase and HIV-1 protease. These enzymes are pivotal in the replication process of the virus (Van Heuvel et al., 2022; Vethakanraj et al., 2015). From the activity-guided fractionation of G. mangostana, α-mangostin and β-mangostin were identified as active compounds, displaying notable inhibition of HIV-1 protease with IC50 values of 5.1 and 4.8 M, respectively. However, in experiments evaluating their impact on recombinant HIV-1 reverse transcriptase, epicatechin and α-mangostin exhibited modest inhibitory effects, suggesting their potential in combating HIV-1 protease activity (Chang et al., 2015).
Used in Inflammation
In research conducted by Pinto et al. (2005) and Tangsuksun et al. (2022), a crude extract of mangosteen fruit pericarp demonstrated the ability to inhibit contractions induced by histamine and serotonin in isolated thoracic rabbit aorta, suggesting a potential role as histaminergic and serotonergic receptor blockers for α- and β-mangostin, respectively. Subsequent investigations by the same team revealed that α-mangostin effectively attenuated histamine-induced contractions in rabbit thoracic aorta and guinea-pig trachea in a dose-dependent manner, irrespective of cimetidine presence, demonstrating inhibition of histamine H1 receptor activity (Richard et al., 2017). Furthermore, evaluation of α-mangostin’s anti-inflammatory properties using carrageenan-induced mouse paw edema indicated significant inhibition, with α-mangostin exhibiting faster action than sulindac. Although α-mangostin did not markedly reduce paw edema in mice, it showed considerable inhibitory activity against 12-human lipoxygenase (12-LOX) with an IC50 of 0.58 M (Ibrahim et al., 2016). Additionally, in studies by Liu et al. (2012), α-mangostin demonstrated anti-inflammatory effects by reducing tumor necrosis factor (TNF)-α and interleukin (IL)-4 expression in lipopolysaccharide (LPS)-stimulated U937 and EL4 cells, suggesting its potential as a therapeutic agent against inflammation and allergies.
Colon Inflammation
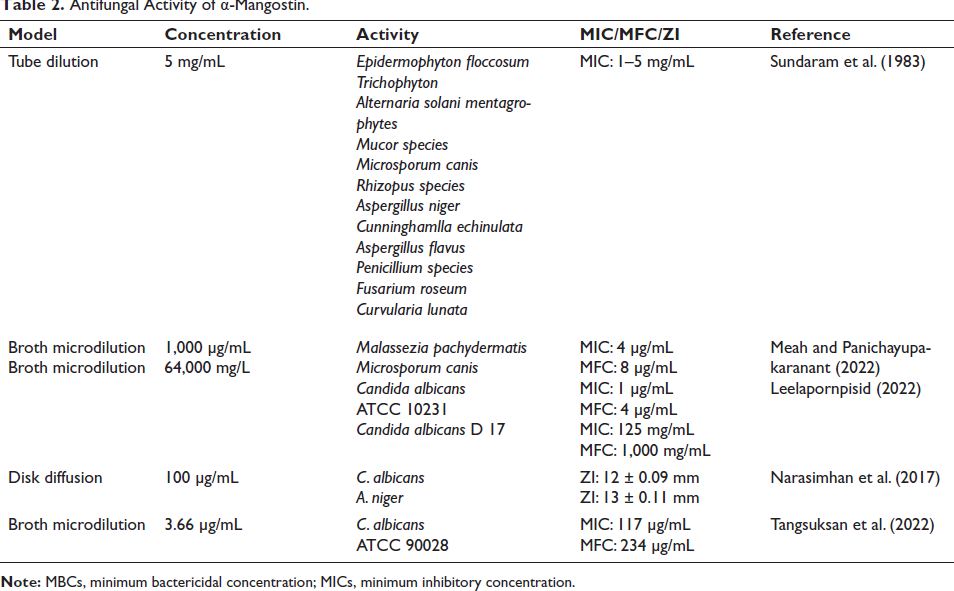
In investigations concerning the effects of α-mangostin on ulcerative colitis (UC), the predominant xanthone in mangosteen fruit, known for its anti-inflammatory and antibacterial properties, was examined. Surprisingly, experiments in mice with experimental colitis induced by dextran sulfate sodium (DSS) revealed that α-mangostin exacerbated colonic inflammation and damage. Mice fed with α-mangostin displayed increased colonic infiltration of CD3 (+) and F4/80 (+) cells, elevated levels of colonic myeloperoxidase, and higher serum concentrations of inflammatory markers compared to controls (Fan et al., 2023; Gutierrez-Orozco et al., 2015). Analysis of the gut microbiota in healthy mice given α-mangostin showed changes resembling those observed in human UC, suggesting a potential risk associated with α-mangostin supplements for individuals with UC. However, previous research indicated that α-mangostin’s high concentration in the colon inhibited the MAPK and NF-κB pathways, mitigating the severity of DSS-induced colitis. Additionally, α-mangostin inhibited inflammatory mediators in LPS-triggered cells, suggesting its potential therapeutic benefits in UC treatment (Banerjee et al., 2018; You et al., 2017). Furthermore, α-mangostin significantly inhibited nitric oxide (NO), TNF-α, prostaglandin E2 (PGE2), and inducible nitric oxide synthase (iNOS) synthesis in LPS-triggered RAW 264.7 cells (Figure 4) (Ibrahim et al., 2016; Liu et al., 2012; Pan, Chen, et al., 2017; Pan, Wu, et al., 2017).
Joint Infections
Researchers investigated the impact of α-mangostin on osteoarthritis (OA), a joint disorder characterized by inflammation and cartilage loss. They utilized rat chondrocytes and an OA rat model induced by destabilizing the medial meniscus (DMM). Rats received saline or α-mangostin pretreatments every other day. The severity of cartilage lesions was assessed up to 8 weeks post-surgery using staining techniques. Results showed that α-mangostin reduced IL-1-induced phosphorylation of the NF-κB signaling pathway, as well as the production of NO and PGE2 (Mandal et al., 2023). Additionally, compared to the control group, cartilage treated with α-mangostin exhibited reduced degradation. These findings highlight the potential therapeutic benefits of α-mangostin in managing OA (Pan, Chen, et al., 2017; Pan, Wu, et al., 2017).
Liver Fibrosis
Chronic liver damage, characterized by excessive extracellular matrix buildup, is a hallmark of liver fibrosis, often leading to cirrhosis. Alcohol-induced fibrosis is a significant contributor to liver disease mortality (Parker et al., 2019; Kim et al., 2011). Hepatic stellate cells (HSCs) play a pivotal role in this process, transitioning into myofibroblasts upon activation triggered by paracrine signaling. Active HSCs boost endogenous transforming growth factor (TGF)-β production, further enhanced by alcohol metabolism, exacerbating oxidative stress and liver damage (Garbuzenko, 2022). In vitro studies by Lestari et al. showcased α-mangostin’s dose-dependent suppression of HSC activation, reducing TGF-β expression and hindering downstream signaling (Lestari et al., 2018). α-Mangostin also inhibited proliferation and activation of LX-2 cells induced by acetaldehyde, offering potential therapeutic benefits against alcohol-induced liver fibrosis (John et al., 2022).
Cancer
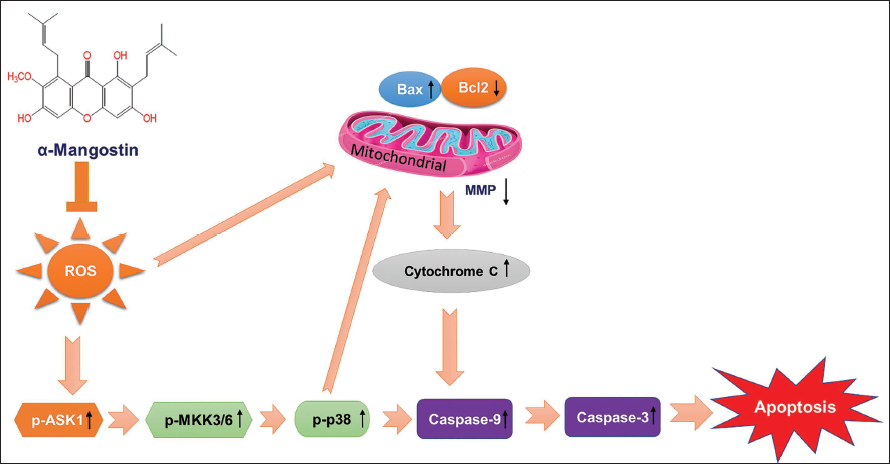
Scientists investigated the impact of six xanthones extracted from mangosteen fruit pericarp on inhibiting cell growth in the HL60 human leukemia cell line (Matsumoto et al., 2003). Assessing cytotoxic effects after 72 hours of exposure to xanthones at 5 or 40 µM concentrations, α-, β-, and γ-mangostins demonstrated notable potency starting from 10 µM. Among them, α-mangostin exhibited the strongest inhibitory action, with an IC50 of 7.5–10 µM, and its effect was confirmed in three additional leukemia cell lines (Kim et al., 2020; Nauman & Johnson, 2022). Further research by Alam et al. revealed that α-mangostin induces apoptosis in HL60 cells primarily through mitochondrial abnormalities (Alam et al., 2023), activating caspases 9 and 3, reducing mitochondrial membrane potential, generating reactive oxygen species (ROS), and releasing cytochrome C (Rojas-Ochoa et al., 2021). Additionally, the efficacy of α-mangostin against cholangiocarcinoma (CCA) was explored, showing inhibition of CCA cell growth, induction of apoptosis via the mitochondrial pathway, G1 cell cycle arrest, and suppression of metastasis. These findings position α-mangostin as a promising agent for cancer treatment and prevention, particularly in tropical regions where mangosteen is abundant (Aukkanimart et al., 2017; Benatrehina et al., 2018; Wahyuni et al., 2023).
Cervical Cancer
α-Mangostin exhibits promising anticancer properties in cervical cancer by inducing cell death pathways, including MMP loss, cytochrome C release, and caspase activation. Inhibition of p38 mitigates α-mangostin-induced cell death, indicating its involvement in the process (Figure 5) (Lee et al., 2017; Tripathi et al., 2024). Combined treatment with α-mangostin and CDDP enhances cytotoxic effects, apoptosis, and ROS production in cervical cancer cells, suggesting a potential neoadjuvant therapy (Díaz et al., 2023; Pérez-Rojas et al., 2016). Additionally, α-mangostin inhibits hepatocellular carcinoma (HCC) proliferation by inducing cell cycle arrest and reducing stemness markers, presenting potential for HCC prevention and treatment (Cai et al., 2016; Kim et al., 2020).
The Application of siRNAs Targeting ASK1, MKK3/6, or p38 Notably Reduced the Cell Death and Apoptotic Responses Triggered by α-Mangostin.
Used as Antimalarial
Research has shown that α-mangostin and β-mangostin inhibit the growth of Plasmodium falciparum clone D6, with IC50 values of 5.1 and 7 M, respectively (Tjahjani, 2017). Further studies demonstrated that α-mangostin is active against both chloroquine-resistant FcM29-Cameroon and chloroquine-sensitive F32 strains at concentrations ranging from 1.7 to 3.2 g/mL. Modified derivatives of α-mangostin with alkylamine groups exhibited the most potent inhibitory activity against P. falciparum in vitro (Ansori et al., 2020; Dutta et al., 2022). Isolated from the husk of mangosteen, α- and β-mangostin were evaluated for their in vitro antiplasmodial and cytotoxic activities. α-Mangostin showed higher susceptibility against the chloroquine-resistant P. falciparum strain (FCR3) compared to β-mangostin, with IC50 values of 0.2 ± 0.01 M and 121.2 ± 1.0 M, respectively. In a Plasmodium berghei malarial murine model, intraperitoneal delivery of α-mangostin proved most effective, resulting in an approximately 80% reduction in parasitemia with a daily dose of 100 mg/kg given twice daily for 7 days. However, oral administration of both substances was ineffective, and no toxicological side effects were observed (Mamede et al., 2020; Saraswathy et al., 2022; Tjahjani, 2017).
Toxicity Assessment of α-Mangostin
In addition to 16 known xanthones, three entirely novel prenylated xanthones named mangostenones C, D, and E were obtained from the immature fruit of G. mangostana. Among these, mangostenone C demonstrated significant cytotoxicity against epidermoid carcinoma of the mouth (KB), breast cancer (BC-1), and small cell lung cancer (NCI-H187) cells, with IC50 values of 2.8, 3.53, and 3.72 g/mL, respectively. Notably, mangostenone C exhibited remarkable potency against BC-1 cells, surpassing that of ellipticine, a well-known drug (IC50 = 1.46 g/mL). Additionally, gartanin and mangostin displayed significant activity against KB and NCI-H187 cells, respectively, with IC50 values of 1.08 g/mL and 2.08 g/mL, respectively (Murthy et al., 2018; Suksamrarn et al., 2006).
In a separate investigation, the effects of eight xanthones from the fruit peel of G. mangostana on cell death were examined using PC12 rat pheochromocytoma cells. Among these xanthones, α-mangostin exhibited the highest efficacy, with an EC50 value of 4 M (Parekh et al., 2022; Sato et al., 2004). α-Mangostin displayed characteristics of the mitochondrial apoptotic pathway, including cytochrome C release and mitochondrial membrane depolarization (Reyes-Fermín et al., 2019). Furthermore, α-mangostin notably reduced the activity of sarco(endo)plasmic reticulum Ca2+-ATPase (SERCA), and the inhibitory effects of xanthone derivatives on SERCA activity were associated with their apoptotic effects.
Regarding toxicity, administration of a high dose of α-mangostin (200 mg/kg body weight) intraperitoneally in rats led to a significant increase in the activity of serum glutamic oxaloacetic transaminase (SGOT) and serum glutamic pyruvic transaminase (SGPT) enzymes, peaking after 12 hours. However, compared to paracetamol, α-mangostin administration did not cause a significant reduction in total liver protein in rats. This indicates that while paracetamol elevates SGOT and SGPT activity, α-mangostin does not have a similar effect on liver protein levels (Zonouz et al., 2023).
Conclusion and Future Direction
According to the current literature assessment, several viral disorders that affect the body’s important organs may be affected by α-mangostin and its constituents, particularly the xanthones. The current review also suggests that the populace has to be informed that α-mangostin also fulfills their dietary needs while having positive effects on their health. The compound’s isoprenyl and benzopyrone skeleton side chains are crucial for its antibacterial properties, making it a promising candidate for combating infectious diseases. Research on isolation techniques, structure–activity relationships, and potential synergistic effects with other drugs highlights its therapeutic potential. However, further exploration, including safety, bioavailability, pharmacokinetics, and clinical trials, is needed to fully validate its efficacy and potential as a new treatment option for infectious diseases. By investing in research and development efforts and exploring novel delivery systems and combination therapies, α-mangostin could become a valuable addition to the range of available treatments for infectious diseases in the future. In vitro and in vivo studies, however, have produced the most current scientific findings. Therefore, additional clinical research is necessary before it may be developed and used as a novel medicine.
Footnotes
Abbreviations
CDC: Centers for Disease Control and Prevention; AIDS: Acquired immunodeficiency syndrome; HAART: Highly active antiretroviral therapy; iNOS: Nitric oxide synthase; NO: Nitric oxide; PGE2: Prostaglandin E2; TNF-α: Tumor necrosis factor-α; NF-κB: Nuclear factor-kappa B; MAPKs: Mitogen-activated protein kinases; MPO: Myeloperoxidase; OA: Osteoarthritis; DMM: Destabilizing medial meniscus; CAA: Cholangiocarcinoma cell; ROS: Reactive oxygen species; DSS: Dextran sulfate sodium; UC: Ulcerative colitis; HSCs: Hepatic stellate cells.
Acknowledgments
The authors are thankful to the Phytomedicine and Pharmaceutical Biotechnology Excellence Center, PSU, and Dr. Pharkphoom Panichayupakaranant for providing all support.
Authors’ Contributions
All authors contributed to the conception and design of the study. M. S. Meah and P. Panichayupakaranant conceptualized the review topic and conducted extensive literature searches. M. A. Sayeed and M. G. Kim provided support in manuscript preparation, editing, and formatting, ensuring clarity and coherence throughout the document. All authors approved the final submitted version.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (Grant No. NRF-2020R1I1A2073610).
