Abstract
Objectives
The primary aim of this study is to investigate the specific effects and mechanisms of ganoderic acid A (GAA) on castration-resistant prostate cancer (CRPC).
Materials and Methods
We successfully established an androgen-independent prostate cancer cell line (LNCaP-AI). The LNCaP-AI cells demonstrated accelerated growth, elevated levels of androgen receptor and prostate-specific antigen (PSA), and resistance to the drug enzalutamide. Then, we administered LNCaP-AI cells with GAA or a control and determined the cell growth rate. Next, LNCaP-AI cells with and without GAA treatment were subjected to RNA-seq for mRNA sequencing. The genes were subjected to a Kyoto Encyclopedia of Genes and Genomes analysis. We also checked the expression levels of the targeted genes. Finally, normal LNCaP or LNCaP-AI cells were subcutaneously injected into nude mice to further confirm the effect of GAA.
Results
Experiments showed that GAA effectively suppressed the growth rate, PSA secretion, migration, and invasion of LNCaP-AI cells. The results of a subsequent RNA-seq analysis identified the PI3K/AKT pathway as the key signaling pathway influenced by GAA. Notably, AKT and GSK-3β phosphorylation was decreased, and β-catenin protein levels were also lowered, as a result of GAA treatment. Moreover, we observed decreased activity in crucial genes, including c-Myc, Cyclin D1, and MMP-2, which play integral roles in CRPC development. Furthermore, GAA inhibited the tumorigenesis of LNCaP-AI cells in nude mice in vivo.
Conclusion
GAA effectively suppresses the growth of androgen-independent cells by inhibiting the AKT/GSK-3β/β-catenin signaling pathway. These findings demonstrate the potential of GAA as a therapeutic candidate for CRPC.
Keywords
Introduction
The fifth most common malignancy in the world is prostate cancer (PCa), with the United States ranking first in male cancer incidence and second in mortality (Sung et al., 2021). About 81% of new patients with PCa in the United States have clinically localized disease. The first-line treatment for these patients is radical surgery or radical radiotherapy, with a five-year survival rate of nearly 90% after radical treatment (Sung et al., 2021). In contrast, most new patients with PCa in China are locally advanced or have extensive metastases; unfortunately, patients with extensive metastases are not suitable for local radical treatment (Zhu et al., 2021).
PCa has androgen-dependent properties, and lowering androgen levels or blocking androgen receptors (ARs) can inhibit the progression of the disease. Androgen deprivation therapy (ADT) is the typical type of treatment for locally advanced and metastatic PCa (Rebello et al., 2021). While initial ADT is effective for most patients with the disease, after 18–20 months of remission, nearly every patient will progress to castration-resistant prostate cancer (CRPC) (Evans, 2018), which is clinically challenging to treat. Therefore, this study explored new pathways for use in treatment to prolong the survival of patients with intermediate to advanced PCa.
Ganoderma lucidum, also known as Reishi, is a medicinal edible fungus of the genus Ganoderma and is distributed worldwide, mostly in tropical and subtropical regions (Ahmad et al., 2021). G. lucidum has antitumor, immunomodulatory, and antiaging properties, with triterpenoids (important antitumor active components) as its main active ingredient, and polysaccharides (Cor et al., 2018; Lu et al., 2020). Most of the components of G. lucidum are related to PCa. For example, the commercial G. lucidum product Reishimax, which claims to contain 6% G. lucidum triterpenes, suppresses the growth of PC-3 cells by inhibiting the expression of vascular endothelial growth factor (VEGF) and transforming growth factor-1 (TGF-1) (Jiang et al., 2004; Stanley et al., 2005). Ganoderic acid A (GAA) is one of the most bioactive compounds from G. lucidum. GAA demonstrates inhibitory properties on the proliferation and invasion of hepatocellular carcinoma cells, which triggers apoptosis (Wang et al., 2017). Additionally, Wan et al. observed that GAA ameliorated lung damage brought on by lipopolysaccharide (Wan et al., 2019). Ganoderic acid DM suppresses PCa cell growth by activating multiple pathways of cell death and immune recognition (Johnson et al., 2019), and GAA functions via STAT3 signaling, which prevents PC-3 PCa cells from proliferating, remaining viable, and producing reactive oxygen species (ROS) (Gill et al., 2016). Our previous study found that G. lucidum triterpenes (provided by Nanjing Zhongke, 98% purity) suppressed the migration and inhibited the proliferation of highly aggressive DU-145 PCa cells by inhibiting matrix metalloproteinase (MMP)-2 and MMP-9 (Qu et al., 2017). Thus, G. lucidum triterpenes have antiprostate-cancer effects. However, whether other components of G. lucidum affect CRPC cell growth and the specific mechanism of that growth (i.e., by modulating the Wnt pathway) are unknown.
The Wnt gene is the combined name of the int-l gene, which was discovered in murine breast cancer in 1982, and the Wingless gene, discovered in Drosophila in 1973 (Liu et al., 2022). The human genome has 19 Wnt genes that encode Wnt proteins, which initiate intracellular signaling pathways via the binding of Frizzled receptors on the surface of cell membranes (Zhou et al., 2022). In lower- to higher-level organisms, one of the signaling pathways that is most conserved at present is the Wnt signaling system, which plays an important role in cell proliferation, differentiation, and embryonic development (Rim et al., 2022). The Wnt/β-catenin signaling pathway is linked to CRPC, which is an important marker of PCa progression (Murillo-Garzon & Kypta, 2017). One study used early immunohistochemical techniques to detect the expression of β-catenin in 23% of radical prostatectomy specimens compared with 38.8% in specimens with advanced PCa (the CRPC stage) undergoing transurethral resection for urinary obstruction (de la Taille et al., 2003), with a significant difference between the two. In patients with PCa treated with ADT followed by radical prostatectomy, the expression level of β-catenin was considerably greater in the resected specimens of each patient than in comparison to the comparable prostate puncture biopsy specimens (Chen et al., 2019). In another study, the inhibition of the Wnt/β-catenin signaling pathway conferred resistance to enzalutamide in hormone-dependent PCa cells, and ICG001, an inhibitor of the Wnt/β-catenin signaling pathway, made hormone-resistant PCa cells sensitive to enzalutamide once again (Zhang et al., 2018). Thus, the Wnt/β-catenin signaling pathway plays a key role in the development of CRPC and can be used as a new target for the treatment of PCa.
This study aimed to clarify whether GAA, another chemically defined component of G. lucidum isolates, suppresses the growth of LNCaP-AI cells via the Wnt/β-catenin signaling pathway.
Methods
Cell Culture and Establishment of the LNCaP-AI Cell Line
Human prostate cancer cells (LNCaP) were obtained from the American Type Culture Collection (Manassas, VA, USA) and cultured as previously reported (Qu et al., 2017). Briefly, cells were cultured in RPMI 1640 medium containing 1% glutamine, 5% sodium bicarbonate, penicillin, and streptomycin (each of 50 U/mL), and 10% fetal bovine serum (FBS) (purchased from Gibco, Thermo Fisher Scientific, MA, USA) in a humidified incubator with 5% CO2 at 37°C. GAA was donated by Shanghai Yuanye Bio-Technology Co. Ltd. (Nanjing, China). The concentration of GAA used for the culture of the PCa cells was 20 µM (Radwan et al., 2015), with 20 mg/kg used for the mice (Cao et al., 2017).
To induce LNCaP-AI cells, cells were cultivated in RPMI 1640 without FBS for 2 days; then, they were cultured with a medium containing FBS treated with activated charcoal/dextran (for the filtration of androgens) and were subsequently cultivated for at least 4 months to screen stable LNCaP-AI cells. This method for inducing LNCaP-AI cells is widely used (Chen et al., 2022; Li et al., 2020). For the cell treatment, 200 µg/mL GAA was added for all the experiments (Lixin et al., 2019).
RT-qPCR
TRIzol™ (Invitrogen, Carlsbad, CA, USA) was used to extract total RNA from PCa cells, and RNA was then reverse transcribed to cDNA. For mRNA detection, the following primers were synthesized by GenePharm (Shanghai, China): AR1 (F, 5′-CCA GGGACCATGTTTTGCC-3′; R, 5′-CGAAGACGACAAGAT GGACAA-3′), AR2 (F, 5′-GACGACCAGATG GCTG TCATT-3′; R, 5′-GGGCGAAGTAGAGCA TCCT-3′), STEAP1 (F, 5′-ACTGGGCACAATACA CGCAT-3′; R, 5′-GGTGA CGTCTTCCCAACCAT-3′), FKBP5 (F, 5′-CTACACCTG CTGAAG GGACG-3′; R, 5′-CTCCAGCAAACCCTGGTACA-3′), and GAPDH (F, 5′-TGAAGGTCGGAGTCAACGG-3′; R, 5′-CCTGGAAGATGGTGATGGG-3′). We assessed the relative expression of mRNA using the 2−∆∆Ct method, with GAPDH serving as an internal reference.
Wound-healing Assay
LNCaP-AI cells were cultured and a wound-healing assay was performed using the previously reported methods (Qu et al., 2017). Briefly, cells were cultured in 6 cm dishes to 100% confluence, and the resulting monolayer was scratched with a 10 µL pipette tip to create a narrow wound. The cells were washed with a culture medium and cultured with or without GAA. Images were captured prior to or 24 hours after treatment by a Nikon phase-contrast microscope (Nikon Instruments, Florence, Italy). After 24 hours, the wounded area was normalized to 0 hours, and the final data were normalized to the control group.
Cell Counting Kit-8 (CCK-8) Assay
To detect cell viability and growth, LNCaP-AI cells were cultured in 96-well plates at a density of 1 × 105/cm2 with or without GAA for 1, 3, and 5 days. Then, 10 µL of CCK-8 reagent (Dojindo Molecular Technologies, Inc., Kumamoto, Japan) was added to the cells and incubated for 1 hour. Cells were collected and subjected to measure absorbance at 450 nm using a microplate spectrophotometer (Bio-Rad, Hercules, CA, USA). The cell growth rate was normalized to 0 days for the relative growth rate.
Transwell Assay
To detect cell migration and invasion, a Transwell system was used. LNCaP-AI cells were cultured in the upper chamber of the Transwell system in a serum-free state, and the lower chamber was filled with 20% FBS culture medium to induce cell migration and invasion. After a 24-hour period, the migrated and invaded cells were fixed and stained with 0.1% crystal violet (#C0121; Beyotime, Shanghai, China), images were taken, and the number of migrated/invaded cells was measured. The bottom of the upper chamber was a cell-penetrating membrane; for the assessment of invasion, the upper chamber was coated with Matrigel® matrix (#356234; Corning, MA, USA).
Colony Formation Assay
The LNCaP cells were obtained and re-counted, and 2,000 cells were seeded into six-well plates for culture at 37°C for 2 weeks until the appearance of visible clones. Then, the cell clones were washed, fixed with methanol, and stained with 1 mg/mL crystal violet (Beyotime) for 20 min. Images were obtained, and the number of obvious clones was measured.
Western Blotting
Samples were lysed and proteins were extracted with a RIPA buffer (Beyotime) at room temperature; then, proteins were loaded onto SDS-PAGE 8% gels and transferred to a polyvinylidene difluoride (PVDF) membrane as in our previous report (Qu et al., 2017). 5% skimmed milk was used to block the membranes for 40 min. After washing, the membranes were incubated with primary antibodies as follows: AR (#ab133273; Abcam, Cambridge, MA, USA; 1:1,000), prostate-specific antigen (PSA) (#ab76113, 1:500; Abcam), HK2 (#ab273721, 1:1,000; Abcam), phosphor-AKT (#ab38449, 1:500; Abcam), total AKT (#ab8805, 1:1,000; Abcam), phospho-GSK-3β (#ab75814, 1:500; Abcam), total GSK-3β (#ab93926, 1:1,000; Abcam), and GAPDH (#AF5009, 1:2,000; Beyotime). Then, the membranes were incubated with secondary HRP-conjugated antibodies (#A0208 or #A0216, Beyotime) for 1 hour at room temperature and visualized with an ECL kit (#P0018S, Beyotime). GAPDH was used as a loading control, with the expression levels normalized first to GADPH and then to the control group.
RNA-seq Analysis
LNCaP-AI cells with or without GAA treatment were subjected to an RNA-seq analysis. First, RNA quantity and quality were evaluated. Then, the samples were sent to Novogen Bioinformatics Technology Co., Ltd. (Beijing, China) for RNA sequencing on an Illumina ×10 sequencing instrument with an average sequencing depth of 100×. Next, the pretreated data of RPKM were analyzed using the R software package (Version 3.0) to identify differentially expressed genes (DEGs). The screening threshold was |logFC| > 1.58, with p < 0.05. A principal component analysis (PCA), Gene Ontology, and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses were conducted, and a DEG heatmap was created using R packages.
Top-Flash Assay
To detect canonical Wnt signaling activity, cells were transfected with pRL-renilla (Promega Corp., Madison, WI, USA) vectors or 2 µg of Top-Flash luciferase vectors with wild-type TCF binding sites and Top-Flash luciferase vectors with mutant TCF binding sites Fugene HD (Roche Applied Sciences, Indianapolis, IN, USA) according to the manufacturer’s guidelines. Four hours later, the cells were treated with GAA or the control for 48 hours. The cells were lysed, and the luciferase activities were measured using a Promega Dual-Luciferase Assay System. The relative activity was normalized to the control group.
Mouse Tumor Xenograft Model
The right flank of BALB/c nude mice (20 ± 2 g) were subcutaneously injected with LNCaP-AI cells. When the average tumor volume reached about 100 mm3, the mice were randomized into two groups, each containing five mice; one group received a vehicle control, and the other group was intravenously injected with GAA, 20 mg/kg (dissolved in DMSO in saline) (Cao et al., 2017) every other day. The mice’s body weight and tumor volume were measured and recorded. Subsequently, after 25 days, the mice were beheaded to end their lives, and the tumors were excised for final weight measurement.
Statistical Analysis
For each experimental group, the results were shown as the mean and standard deviation. Differences between two groups were analyzed using an unpaired Student’s t-test, and differences between multiple groups were analyzed by a two-way analysis of variance using GraphPad (Version 9.4.1; GraphPad Software, Inc., San Diego, CA, USA) software. To assess the differences in tumor volume, a nonparametric Mann–Whitney U test was performed. A value of p < 0.05 was considered statistically significant.
Results
Construction of Androgen-independent PCa Cell Line
To construct the LNCaP-AI cell line, LNCaP cells were cultivated in a medium filtered by activated carbon/dextran material. Fifteen days later, the LNCaP cells gradually began to protrude pseudopods, and after 2 months, the outwards had connected to form a network (Figure 1A), and the cells were in an autocrine state. After 3 months, the cells began to get rid of the autocrine state, and the protrusions gradually decreased. After 4 months of subculture, the cell growth changed to spread-like growth, with decreased apoptosis. Culture continued for 6 months to form a hormone-independent LNCaP cell sub-line (LNCaP-AI) (Figure 1A).
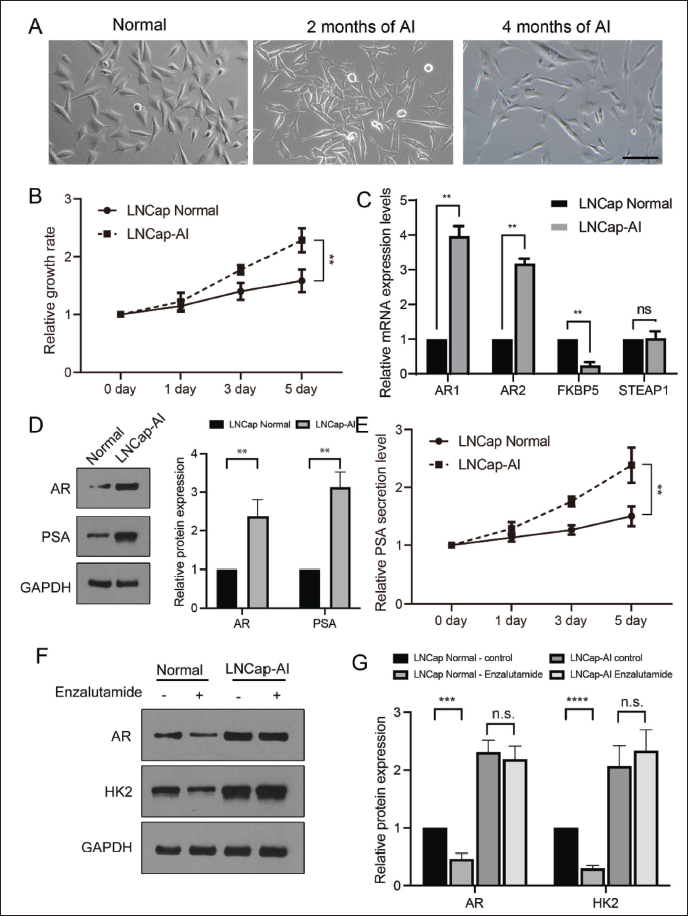
To identify the gene profiles of the established LNCaP-AI cell line, the mRNA level of AR, FKBP5, and STEAP1 (AR-related signal pathway) were detected by RT-qPCR. The growth rate of LNCaP-AI was much faster than that of normal LNCaP cells. As shown in Figure 1C, the mRNA levels of AR1, AR2, and FKBP5 in the LNCaP-AI cells were substantially greater than those in normal LNCaP cells, while STEAP1 showed no changes (Figure 1B). In terms of the protein levels revealed by Western blotting, both AR and PSA from the LNCaP-AI cells were also much higher than those from the normal LNCaP cells.
High levels of PSA secretion in blood usually suggest larger tumor burden. Thus, secreted PSA levels are another marker for the identification of the androgen-independent phenomenon. As shown in Figure 1E, the secreted level of PSA in the LNCaP-AI cells was markedly upregulated compared with the case in normal LNCaP cells, as determined by enzyme linked immunosorbent assay (ELISA). Furthermore, the protein expression of AR and HK2 was downregulated by the administration of enzalutamide (AR signaling inhibitor (Campanelli et al., 2023)), HK2 as the positive marker for the response to enzalutamide (Mansinho et al., 2018) in normal LNCaP cells but not in LNCaP-AI cells (Figure 1E and F). These data indicated the successful establishment of the androgen-independent cell line LNCaP-AI.
GAA Suppresses the Development of LNCaP-AI Cells
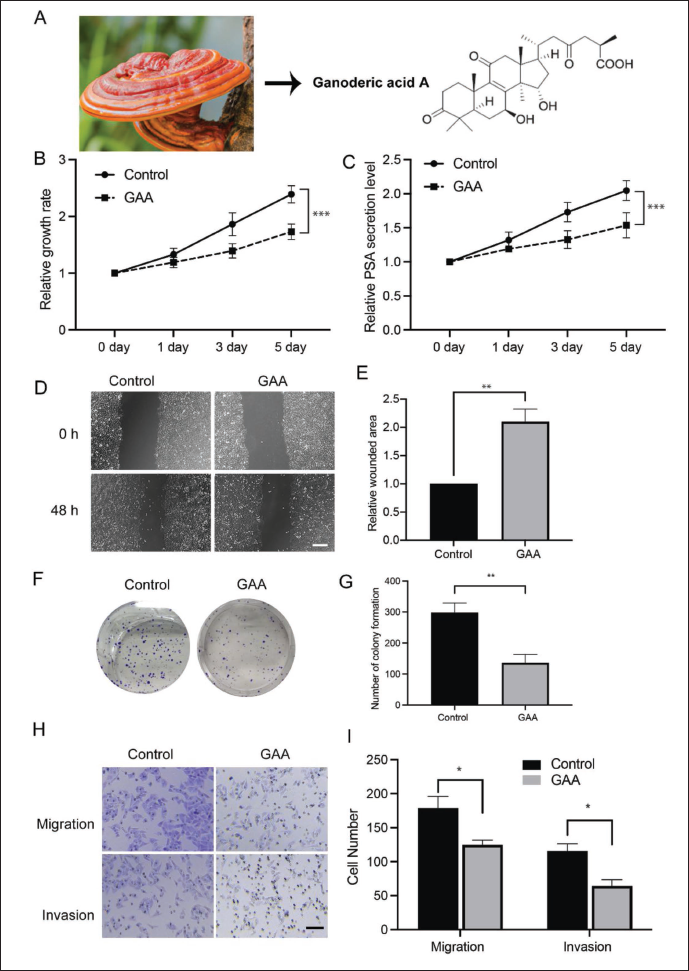
Next, we tried to determine whether GAA would impact the development of LNCaP-AI cells. GAA is a monomer with a clear structure and is derived from the extract of G. lucidum (Figure 2A). We administered LNCaP-AI cells with GAA or control and determined the cell growth rate. As shown in Figure 2B, the relative growth rate revealed by CCK-8 assay was significantly suppressed in the GAA group. According to ELISA, PSA secretion was also markedly inhibited by the administration of GAA (Figure 2C). A wound-healing assay showed that GAA increased the size of the wounded area compared with that of the control group (Figure 2D and E). Colony formation exhibited the same trend, and GAA significantly inhibited the number of clones (Figure 2F and G). Additionally, a Transwell experiment demonstrated that administration suppressed the migration and invasion of LNCaP-AI cells (Figure 2H and I). All these data indicate that the administration of GAA suppressed the development of androgen-independent cells.
GAA Functions via the AKT/GSK-3β/β-catenin Pathway
To explore the detailed mechanism underlying GAA-regulated CRPC cell development, LNCaP-AI cells with and without GAA treatment were subjected to RNA-seq for mRNA sequencing. The score plots of the PCA showed that the three samples from the control and GAA groups were well clustered, indicating good repeatability, with no strong deviation between the groups (Figure 3A). A total of 15,759 genes were detected, and 618 upregulated genes and 206 downregulated genes were identified as DEGs; these were plotted on a volcano plot (Figure 3B), and the relative expression in each sample was illustrated on a heatmap plot (Figure 3C).
RNA-seq Assay of Androgen-independent Prostate Cancer Cells (LNCaP-AI) Treated with and without GAA. LNCaP-AI Cells Treated with and without GAA were Subjected to RNA-seq. (A) Principal Component Analysis (PCA) Plot, (B) Volcano Plot, (C) Heatmap Plot, (D) Cnet Plot of Gene Ontology Analysis, and (E) Kyoto Encyclopedia of Genes and Genomes Analysis.
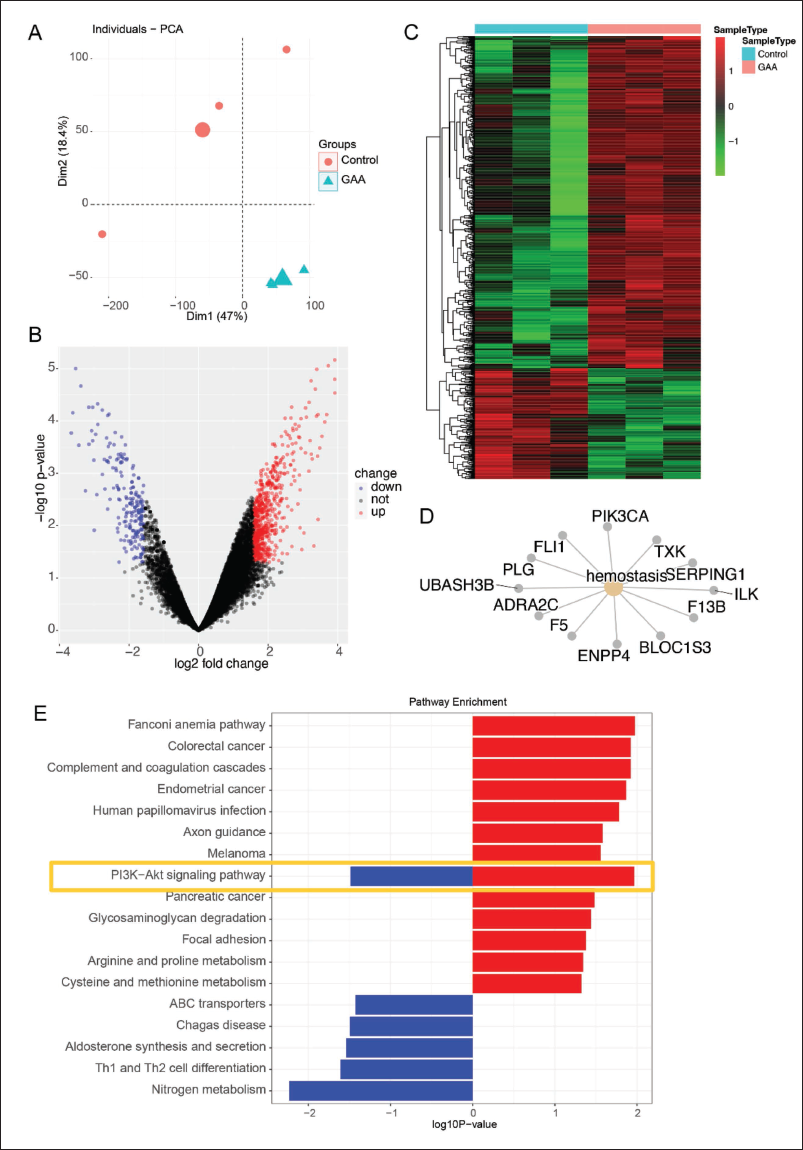
Next, the genes were subjected to a KEGG analysis to identify the most important signal transduction pathway. As shown in Figure 3D and E, the analysis revealed the involvement of the PI3K/AKT signaling pathway. However, since GSK-3β/β-catenin regulation is associated with CRPC (Arjsri et al., 2023), we tried to determine whether AKT/GSK-3β signaling can regulate β-catenin levels to regulate CRPC. The LNCaP-AI cells with and without GAA treatment were subjected to Western blotting. As shown in Figure 4A and B, the phosphorylated levels of AKT and GSK-3β were significantly downregulated with GAA treatment, and the β-catenin level was suppressed. Furthermore, the Top-Flash luciferase assay revealed that the induced β-catenin activity could be inhibited by the administration of GAA (Figure 4C).
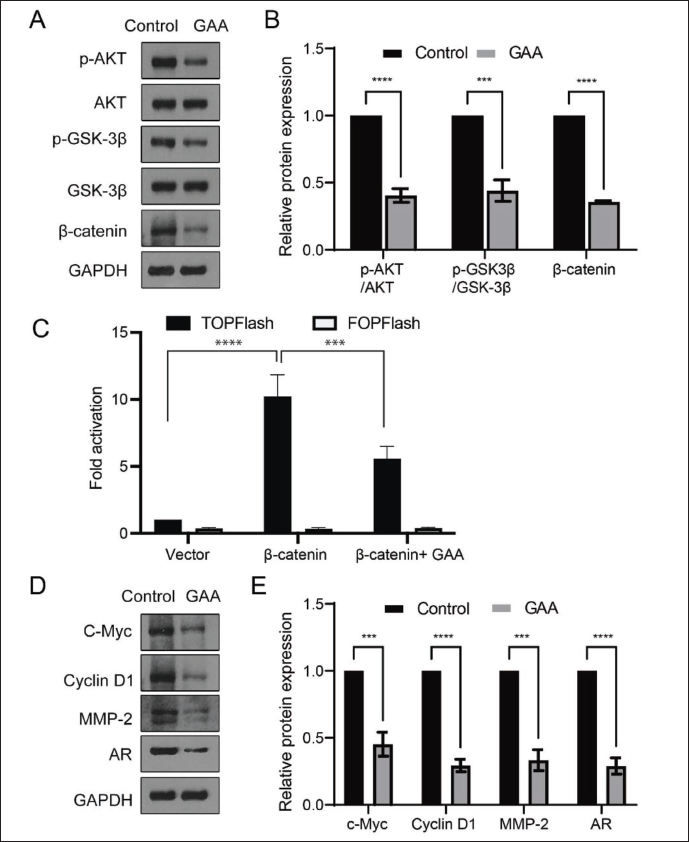
We also examined the expression levels of the targeted genes in the AKT/GSK-3β/β-catenin signaling pathway, such as c-Myc, Cyclin D1, and MMP-2. The data showed that all these levels were suppressed by the administration of GAA, along with the downregulation of AR. These data were consistent with the RNA-seq data, indicating that GAA may function via the AKT/GSK-3β/β-catenin signaling pathway to regulate cell growth under the condition of CRPC.
GAA Suppresses Tumor Formation in Nude Mice
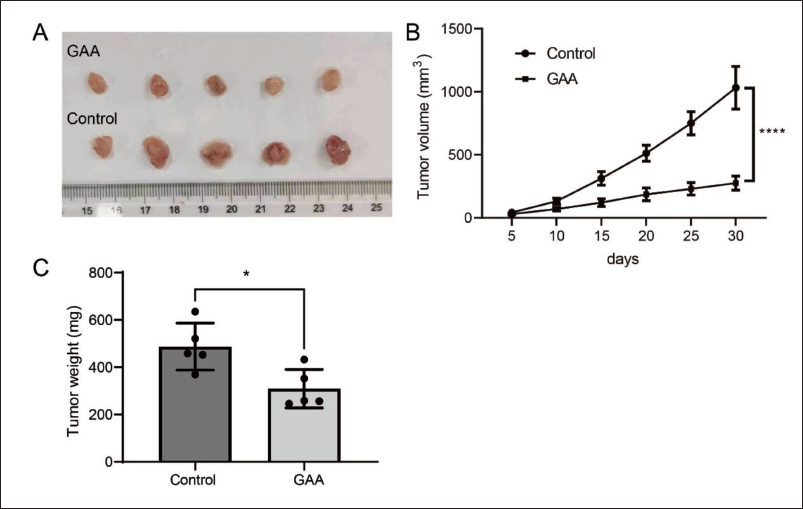
To further confirm the suppressive effect of GAA on cell growth in CRPC, in vivo tumorigenesis experiments were performed. Normal LNCaP-AI cells were subcutaneously injected into nude mice. GAA was injected (20 mg/kg) every other day, and the tumor volume was recorded over a period of 25 days. Then, the mice were euthanized, and the tumor weight was measured. As Figure 5A illustrates, the tumor in the control group was substantially larger than in the GAA-treated group, and the tumor volume and weight were much lower in the GAA group than in the control group (Figure 5B). These data were consistent with the in vitro data and suggest that GAA suppresses the proliferation of PCa cells in CRPC in vivo.
Discussion
The impact of GAA on CRPC and the detailed underlying mechanism remains unclear. Herein, we constructed an androgen-independent cell line (LNCaP-AI) to mimic the CRPC phenomenon and determined the character of LNCaP-AI cells. Then, these cells were exposed to GAA, which considerably inhibited the growth, colony formation, wound healing, migration, and invasion ability of the LNCaP-AI cells. An RNA-seq analysis identified a large portion of DEGs. Subsequently, the participation of the AKT/GSK-3β/β-catenin pathway was confirmed to be associated with GAA administration. Finally, we found that GAA treatment significantly suppressed the in vivo tumorigenesis of LNCaP-AI cells. These findings suggest that GAA may suppress CRPC in PCa via the AKT/GSK-3β/β-catenin pathway.
The mechanism of CRPC is the problematic focus of current research, and a variety of possibilities have been proposed, mainly including the following formation mechanism: (a) androgen-receptor-related mechanism. Activation of the AR signaling pathway plays an important role (Dai et al., 2017). (b) Mechanism of stem cell formation. Stem cell growth is independent of AR and can progress to CRPC after ADT treatment (Bora-Singhal et al., 2015). (c) Neuroendocrine transformation mechanism. ADT can induce neuroendocrine differentiation (NED) in PCa cells (Niu et al., 2018). PCa NED cells no longer depend on the AR signaling pathway and stimulate the proliferation of PCa cells via paracrine and autocrine effects (Li et al., 2016). However, the exact mechanism of CRPC remains unknown.
At present, drugs such as docetaxel, cabazitaxel, abiraterone, and enzalutamide, as well as anti-bone-metastasis therapy and immunotherapy, are the main treatment modalities for CRPC (Schaeffer et al., 2023). However, the above treatments can only control tumors for a limited period, and they have different side effects. These treatments cannot prolong the survival time of patients with CRPC, and the survival prognosis is not ideal, with a median survival period of no more than 12 months, with most patients eventually dying of PCa (Moussa et al., 2020). Therefore, how to delay the appearance of CRPC or suppress CRPC after ADT treatment, thereby prolonging the survival of patients, is worth investigating.
The Wnt/β-catenin pathway is important for many organs to grow normally (Polakis, 2012). The abnormal activation of this pathway is always related to human cancers, including liver, colorectal, and PCa (Liu et al., 2022; Wang et al., 2022). The Wnt/β-catenin pathway interacts with AR signaling to regulate CRPC. There is crosstalk between the two pathways, and β-catenin also directly interacts with AR to activate downstream transcription (Khurana & Sikka, 2019). By sequencing CRPC samples using a next-generation technique, significant changes were found in multiple components of the Wnt/β-catenin pathway (Grasso et al., 2012; Rajan et al., 2014). Furthermore, abnormal paracrine Wnt ligands and the loss of secreted Wnt antagonists from prostatic stroma are involved in ADT resistance (Sun et al., 2012). Additionally, overexpressed β-catenin leads to the stem cell transformation of PCa cells, making them resistant to ADT treatment (Yeh et al., 2019). After 22 weeks of ADT treatment in patients with advanced PCa, the patients had 774 protein-coding genes with increased expression and 24 signaling pathways enriched for genes with increased expression, with the Wnt/β-catenin signaling pathway containing the highest number of genes with increased expression (Rajan et al., 2014). A comparative analysis of the gene expression data of patients with advanced PCa with enzalutamide (a second-generation antiandrogen drug) resistance or sensitivity showed that the Wnt/β-catenin signaling pathway was significantly activated in patients with CRPC (Zhang et al., 2018). A gene set enrichment analysis was performed on 101 patients with metastatic CRPC, and it was again discovered that the Wnt/β-catenin signaling pathway was significantly activated in patients with enzalutamide resistance (Chen et al., 2019). Thus, the Wnt signaling pathway plays a complex role in CRPC.
Herein, mRNA sequencing revealed the regulation of the PI3K/AKT pathway in CRPC LNCaP cells, indicating the relationship between GAA and the downstream canonical Wnt pathway (Figure 3). Next, we revealed the activation (increased level) of AKT and GSK-3 in LNCaP cells, which was inhibited by GAA treatment. Using a Top-Flash luciferase assay, we determined that activated β-catenin could also be suppressed (Figure 4). Our data showed the involvement of β-catenin in CRPC, which is consistent with previous reports, as summarized in this review (Khurana & Sikka, 2019).
Although certain immunotherapeutic treatments have shown effectiveness in PCa, the effective long-term treatment of the disease, especially CRPC, continues. G. lucidum, which contains many anticancer components, has gradually become more widely used (Cor et al., 2018). Our previous reports demonstrated that ganoderic triterpenes suppressed the growth and multiplicity of PCa cells (Qu et al., 2017). G. lucidum is related to the regulation of β-catenin in many cancers. Das et al. found that in the human meningioma cell line (IOMM-Lee), GAA and ganoderic acid DM (two triterpene acids with similar structures) from G. lucidum significantly inhibited the expression of the Wnt-2 protein, while in a nude mouse model, the injection of GAA and ganoderic acid DM inhibited tumor cell proliferation and reduced tumor volume (Das et al., 2020). Gill et al. first used computer software for bioinformatics analyses and found that the Lys180 and Asn220 residues of GAA were stably docked with the β-catenin protein molecule (Gill et al., 2018). The pharmacodynamic parameters are suitable as drug-like molecules, and it has been confirmed that GAA acts on β-catenin and can inhibit the proliferation and cell activity of the pancreatic cancer cell line RIN-5F (Gill et al., 2018). Zhang et al. found that in the human breast cancer cell line MDA-MB-231 and the mouse breast cancer cell line 4T1, G. lucidum significantly downregulated the phosphorylation level of the Wnt co-receptor LRP6, thereby suppressing the transcription of the Wnt target gene Axin2 and finally suppressing the proliferation and migration of breast cancer cells by blocking the Wnt/β-catenin signaling pathway (Zhang, 2017). G. lucidum significantly inhibited the transcription and protein expression of Wnt co-receptors LRP5, β-catenin, c-Myc, Cyclin D1, MMP-2, and MMP-9 in osteosarcoma cell lines (Zhang et al., 2019).
The above reports suggest that G. lucidum/G. lucidum triterpenes can exert antitumor effects through the Wnt/β-catenin signaling pathway. However, the remaining role of G. lucidum extract in CRPC remains unknown. By establishing a model of the androgen-independent LNCaP cell line to mimic CRPC, herein, we demonstrated that GAA suppressed the secretion of PSA and the growth, wound healing, clone creation, migration, and invasion abilities of LNCaP-AI cells in vitro (Figure 2) and tumorigenesis in vivo (Figure 5) by suppressing the activation of the AKT/GSK-3β/β-catenin pathway (Figure 4).
In conclusion, this study identified a novel role of GAA in the regulation of CRPC via the AKT/GSK-3β/β-catenin pathway, which provides a rationale for the future clinical exploration of CRPC treatment in patients with PCa.
Animals and Ethics Statement
BALB/c immunodeficient nude mice were obtained from Southern Medical University (animal approval: SCXK-2020-0056). All the procedures involving animals were approved by the Laboratory Animal Center of Jinan University (approval no. 20200316-07).
Author Contributions
ZC and LQ contributed to conception and design of the study. LQ organized the database. YQ and QL performed the statistical analysis. QL wrote the first draft of the manuscript. QL, YQ, GC, and JC conducted the experiments and wrote sections of the manuscript. All authors contributed to manuscript revision, read, and approved the submitted version.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Projects in Guangzhou (#202201020051) and the Pilot Program of the First Affiliated Hospital of Jinan University.
