Abstract
Background
Huanglian Jiedu decoction (HLJDD) is a classical herbal formula for treating inflammatory diseases with Traditional Chinese Medicine (TCM). However, the impacts of HLJDD on neuroinflammation and the precise mechanisms involved remain poorly understood.
Purpose
In the present study, HLJDD was found to inhibit lipopolysaccharide (LPS) induced neuroinflammatory responses in BV2 microglia via regulating NLRP3 inflammasome activation.
Materials and Methods
Anti-inflammatory activity was evaluated with ELISA assay and the polarization changes were observed using immunofluorescent staining. The levels of NLRP3 inflammasome activation were determined using qPCR and Western blotting.
Results
The administration of HLJDD successfully countered LPS-induced neuroinflammatory responses by attenuating microglial activation and fostering a phenotypic conversion from the pro-inflammatory M1 to the anti-inflammatory M2. Additionally, HLJDD treatment demonstrated inhibition of NLRP3 inflammasome activation.
Conclusion
The findings of the present study may provide insights into how HLJDD could be a therapeutic drug for neuroinflammatory diseases associated with regulating NLRP3 inflammasome and microglial activation.
Introduction
Neuroinflammation encompasses diverse immune responses occurring within the central nervous system (CNS) triggered by diverse factors and stimulations, such as disease, injury, infection, or stress. This condition results in the secretion of proinflammatory mediators, leading to synaptic impairments and neuronal degradation (Leng & Edison, 2021). Such inflammatory processes play a crucial role in the onset and advancement of various neurodegenerative disorders, including ischemic stroke, multiple sclerosis, Alzheimer’s, and Parkinson’s disease (Mishra et al., 2021).
Microglia, the CNS’s innate immune cells, provide a vital role in the maintenance of tissue homeostasis by monitoring the environment, performing physiological housekeeping, and defending against modified-self and non–self-injurious agents (Hickman et al., 2018). During the activation, they can rapidly transform morphology, phenotype, and function when responding to neuropathological conditions. Similar to macrophages, microglia exhibit functional plasticity across a spectrum from classically-activated M1 (classical activation) to alternatively-activated M2 (alternative activation) phenotypes (Wolf et al., 2017). M1 microglial cells secrete several proinflammatory agents, such as IL-1β, IL-18, tumor necrosis factor-alpha, and inducible nitric oxide synthase (iNOS), contributing to increased blood-brain barrier permeability and the facilitation of neuronal apoptosis. Conversely, M2 microglia are characterized by the production of anti-inflammatory compounds and neurotrophic factors, including IL-10, arginase-1 (Arg-1), and insulin-like growth factor 1, which are crucial for neuroprotection (Lee et al., 2016; Tang & Le, 2016). A meta-analysis showed that a few M1 inhibitors, such as cyclooxygenase (COX) -2 inhibitors, are not as effective against neurodegenerative diseases (Miguel-Álvarez et al., 2015). Therefore, suppression of microglial activation and shifts toward an M2 phenotype are becoming crucial research areas to find new therapeutic targets for neuroinflammatory-related diseases (Orihuela et al., 2016).
It is a recent discovery that the nucleotide-binding oligomerization domain-like receptor (NLR) family pyrin domain-containing 3 (NLRP3) inflammasome, containing NLRP3, ASC, and procaspase-1, is the best-characterized inflammasome (Mangan et al., 2018). Within microglia, the NLRP3 inflammasome acts as a critical mechanism for the maturation and release of IL-1β and IL-18, triggering significant inflammatory responses when encountering immune stimuli, including damage-associated molecular patterns (DAMPs), pathogen-associated molecular patterns, as well as various external threats or environmental stressors (Cunha et al., 2016). Lipopolysaccharide (LPS), widely used as an in vitro model for inflammation, is demonstrated to trigger both microglial activation and NLRP3 inflammasome initiation through the TLR4/NF-κB signaling pathway, functioning as DAMPs (Liu et al., 2020; Yang et al., 2020).
In traditional Chinese medicine (TCM), Huanglian Jiedu decoction (HLJDD) is one of the well-known prescriptions and has been widely used to clear heat and detoxification for several years. It was first recorded in Ge Hong’s Elbow Backup Emergency, which consists of Gardenia jasminoides J. Ellis, Phellodendron amurense Rupr., Scutellaria baicalensis Georgi and Coptis chinensis Franch (The plant’s scientific name has been confirmed using the International Plant Names Index database.
This research is designed to explore HLJDD’s impact on microglial activation and neuroinflammation in vitro and to evaluate how the NLRP3 inflammasome contributes to the anti-inflammatory effects of HLJDD.
Materials and Methods
Reagents
Materials for this study were sourced as follows: BV2 mouse microglial cells and LPS from Procell (Wuhan, China) and Sigma, respectively. IL-1β, IL-18, and TNF-α ELISA kits from Elabscience, with the nitric oxide (NO) assay kit from Nanjing Jiancheng Bioengineering Institute (NJJCBIO, Nanjing, China). Antibodies for NLRP3 and iNOS were provided by Huabio and Wuhan Sanying, respectively. Cell Signaling Technology supplied the Arginase-1 antibody, and the anti-Iba1 antibody was from Abcam. RT and PCR reagents, including HiScript® II Q select RT SuperMix and SYBR Green Master Mix, were from Vazyme Biotech. Boster provided all secondary antibodies, including fluorescein isothiocyanate-conjugated goat anti-rabbit IgG, Cy3-conjugated goat anti-mouse IgG, and horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG. HLJDD was prepared by Wuhan NO.1 Hospital following the Chinese Pharmacopeia (2010 Edition).
Drug Treatment
A total of 20 adult male S.D. rats, sourced from Hunan Slack Jingda Experimental Animal Co., Ltd., China, were randomly assigned to one of four groups: control, low-dose (LD) HLJDD, medium-dose (MD) HLJDD, and high-dose (HD) HLJDD. The LD, MD, and HD groups were given 1.339 g/(kg·d), 2.678 g/(kg·d), and 5.356 g/(kg·d) HLJDD by intragastric administration based on body surface area ratio between experimental animals and humans respectively, while the control group was given equal volume normal saline by intragastric administration at 1 mL/100 g for seven consecutive days. After the last intragastric dose, rats received an intraperitoneal injection of chloral hydrate for anesthesia. Subsequently, whole blood was collected through abdominal aortic puncture, with serum subsequently isolated to treat BV2 cells. In a sterile environment, LPS was prepared into 1 g/L LPS mother liquor, divided into 1.5 mL centrifuge tubes, stored at −20℃, and diluted to the required concentration during use.
Cell Culture
The BV2 mouse microglial cell line, an immortalized strain, was kept in a humidified incubator at 37°C with 5% CO2. Culture conditions included Dulbecco’s Modified Eagle’s Medium, enriched with 10% fetal bovine serum, 1% L-glutamine, and 1% penicillin/streptomycin. On reaching 90% confluency, cells were then subcultured using 0.25% trypsin for subsequent passages. The cells were passaged or used in experiments according to the ratio of 1:3. A series of concentrations of HLJDD and/or LPS (1 µg/mL) were applied to the cell for 24 h.
Real-time Polymerase Chain Reaction
TRIzol reagent was used to isolate the cell’s total RNA. HiScript II Q Select RT SuperMix for quantitative polymerase chain reaction (q-PCR) was used to synthesize cDNA and analyze samples using SYBR Green Master Mix. Table 1 shows the sequence information of the primers for q-PCR. Using glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as the internal reference, the relative mRNA expressions of NLRP3 (NM_001359638.1) were calculated by the 2-△△Ct method.
Primer Sequences for qPCR Analysis.

Western Bolt
Sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) separated the proteins after different stimuli and transferred them to nitrocellulose membranes. The membrane was re-probed with an antibody specific to GAPDH (1:1000) as an internal control. NLRP3 was detected using rabbit anti NLRP3 (1:800) followed by HRP-labeled, goat-anti-rabbit IgG antibodies. Finally, the X-ray films were fixed in a dark room after it was developed.
Determinations of IL-18, IL-1β, TNF-α, IL-4, IL-10, and NO
ELISAs were used to measure IL-18, IL-1β, TNF-α, IL-4, and IL-10 levels of cell supernatant after treatment, while the Griess reaction as described in the manufacturer’s instructions was used to determine NO concentration.
Immunofluorescence Double Staining
Cells were stimulated and then washed three times with PBS, followed by a 20-min permeabilization in PBS containing Triton X-100 and another three PBS washes for 3 min each. After blocking with 0.5% bovine serum albumin in PBS for 30 min at room temperature, the cells were incubated overnight at 4°C with primary antibodies against Iba-1, Arginase-1, and iNOS, all diluted to 1:100. Following a washing step, the cells were exposed to either fluorescein isothiocyanate- or Cy3-conjugated secondary antibodies for 1 h at room temperature and subsequently counterstained with 4’,6-diamidino-2-phenylindole (DAPI). Fluorescence microscopy was used for imaging.
Statistical Analysis
Experiments were conducted in triplicate, with results expressed as mean ± standard deviation. The unpaired t-test was applied to assess differences between two groups, while comparisons across multiple groups were made using one-way ANOVA and Tukey’s post hoc test, employing SPSS 25.0 for analysis. A significance level of p < 0.05 was established.
Results
Quality Control of HLJDD
The HPLC analysis was performed to evaluate the quality of HLJDD in this study, eight bioactive ingredients of HLJDD were identified, including baicalin, baicalein, wogonoside, wogonin, geniposide, berberine, jatrorrhizine, and palmatine hydrochloride. Then, we examined 10 batches of HLJDD, and their HPLC chromatogram showed similarity coefficients of more than 0.99 (Figure 1).
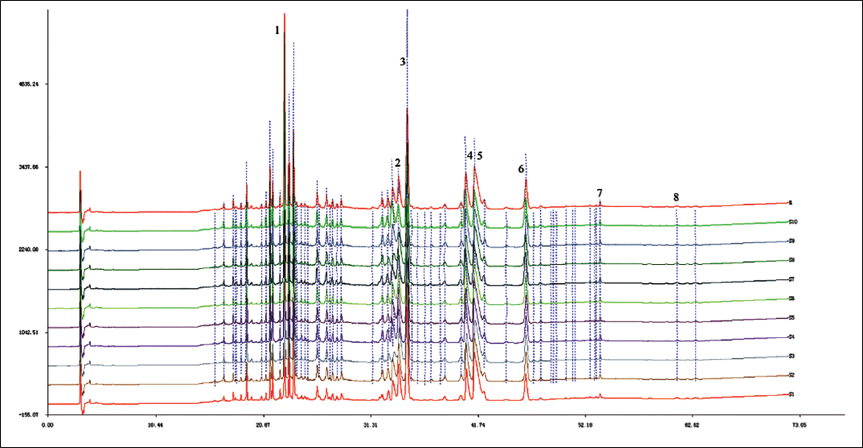
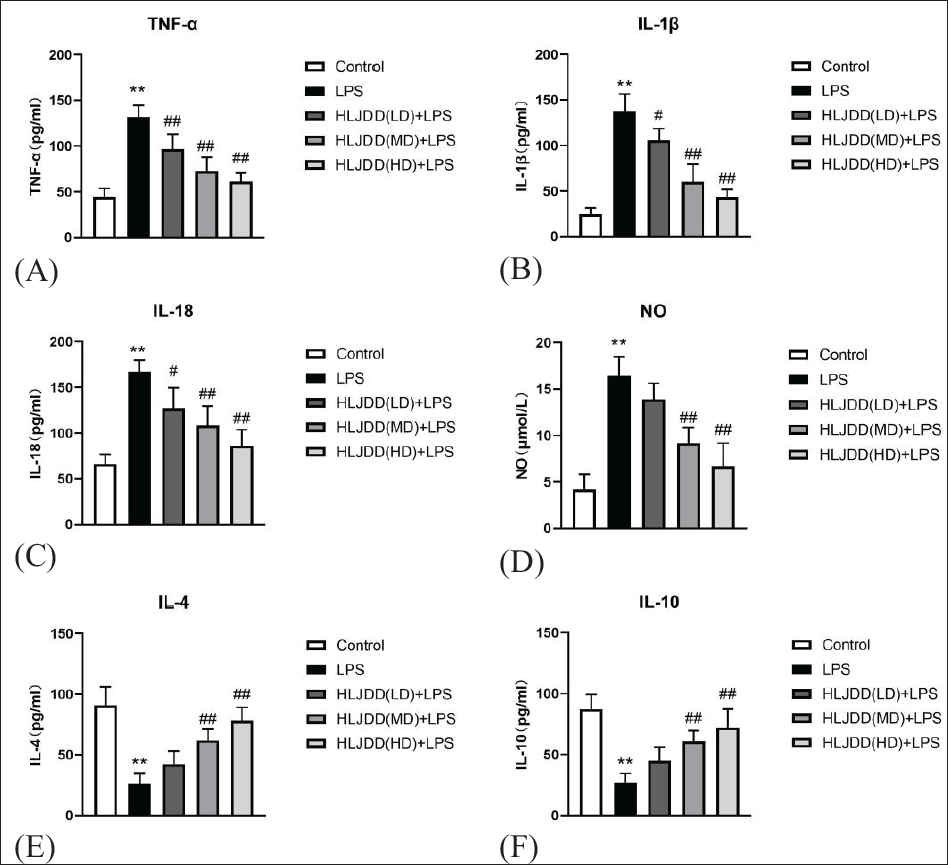
Effects of HLJDD Treatment on LPS-induced Inflammatory Response in BV2 Cells
To evaluate HLJDD’s impact on inflammatory responses, ELISA was employed to measure both proinflammatory and anti-inflammatory cytokine concentrations in LPS-stimulated BV2 microglia. LPS (1 µg/mL) substantially increased the secretion of TNF-α, IL-18, IL-1β, and NO compared to control cells (p <.01). In contrast, 24 h of treatment with medium and high doses of HLJDD significantly suppressed LPS-induced secretion of these proinflammatory factors (p <.01) (Figure 2A–D). Furthermore, HLJDD enhanced the secretion of IL-4 and IL-10, which are key anti-inflammatory cytokines, in LPS-activated BV2 cells (p < 0.01) (Figure 2E and F). These results suggested that HLJDD suppressed neuroinflammatory responses in LPS-activated microglia.
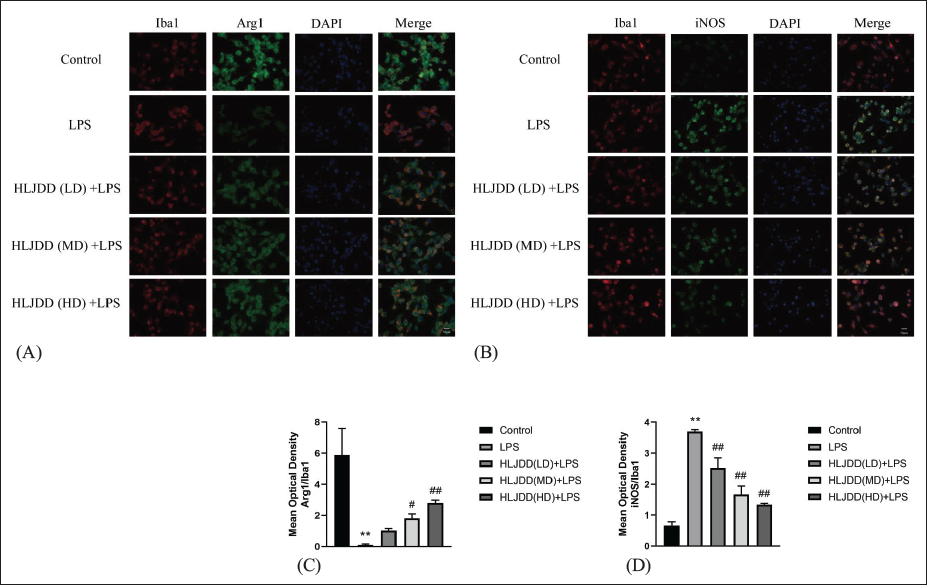
Effects of HLJDD Treatments on LPS-induced Microglial Activation and Polarization
To further assess whether the anti-inflammatory effects of HLJDD were related to microglial activation and M1/M2 phenotypic switch, we next used double immunofluorescence for Iba1, microglial activation marker (red), together with iNOS, M1 marker (green), or Arg-1, M2 marker (green), respectively.
As shown in Figure 3, BV2 cells showed significantly higher expression of the M1-microglia marker after LPS in comparison to untreated controls (p < 0.01). Interestingly, this increase in immunostaining for iNOS was dose-dependently suppressed by HLJDD treatment (p < 0.01), suggesting that the anti-inflammatory role of HLJDD by suppressing M1-microglia polarization. However, a significant increase in M2-related markers was observed in LPS-stimulated BV2 cells after exposure to HLJDD (MD, HD) therapy (p < 0.01 or p < 0.05). These findings demonstrate that HLJDD alters microglial polarization by promoting an M2 phenotype while attenuating the M1 phenotype.
The Effect of HLJDD Treatment on NLRP3 Inflammasome Activation Induced by LPS Expression
Activation of the NLRP3 inflammasome is critical for the modulation of microglial polarization states. NLRP3 expression was detected by qPCR and Western blotting to confirm whether HLJDD regulates microglial polarization through NLRP3 inflammasome activation. The results, outlined in Figure 4, demonstrated that LPS stimulation upregulated both gene and protein expression of NLRP3 (p < 0.01), which was significantly attenuated after HLJDD (MD, HD) treatment (p < 0.05 or p < 0.01). Thus, administration of HLJDD significantly inhibited LPS-induced NLRP3 inflammasome activation.

Discussion
A classic TCM formula, HLJDD, known for its heat-clearing and detoxifying properties, is often applied in the treatment of various diseases with inflammation, including pneumonia (Li et al., 2021), atherosclerosis (Cai et al., 2021; Jiang et al., 2019) and atopic dermatitis (Chen et al., 2021). The existing research has demonstrated that HLJDD can ameliorate inflammation, and enhance humoral and cellular immune responses through regulating the polarization of macrophages (Li et al., 2013; Xu et al., 2021). Nevertheless, the effects involved in HLJDD treatment of microglia remain poorly understood. The present study examined the phenotypical and functional properties of LPS-stimulated BV2 cells treated with different concentrations of HLJDD. The study reports on observations of upregulated inflammatory responses after exposure to LPS (1 µg/mL) in BV2 cells and then a significant reduction in the addition of HLJDD treatment. Moreover, elevated levels of NLRP3 inflammasome activation could also be reduced through HLJDD treatments that correlate with microglial phenotype shifts.
Lipopolysaccharide, a bacterial endotoxin, is extensively utilized for experimental research on microglial activation (Zhang et al., 2019). Microglia’s activation and subsequent polarization are critical in the neuroinflammatory process, playing a pivotal role in the immune response (Kwon & Koh, 2020). M1-polarized microglia are characterized by increased production of proinflammatory cytokines, including IL-1β, IL-6, and TNF-α. While important for host defense and phagocytic functions, these factors can cause damage to tissues outside of hemorrhagic lesions (Subhramanyam et al., 2019). M2-polarized microglia release IL-4 and IL-10, cytokines with anti-inflammatory properties that inhibit proinflammatory immune activities and enhance the expression of genes associated with repair (Colonna & Butovsky, 2017). As a result of LPS treatment, the M1 marker iNOS was significantly increased, while the M2 marker Arg-1 was significantly decreased in BV2 cells. However, treatment with HLJDD significantly inhibited both the iNOS expression and the proinflammatory cytokines production, while promoting the Arg-1 expression. These results indicated that HLJDD suppressed LPS-induced inflammatory responses and may exhibit neuroprotective effects.
NLRP3, a major member of the NLR family of cytosolic pattern recognition receptors, is important for detecting exogenous and endogenous ligands that stimulate innate immune responses within the CNS (Haneklaus & O’Neill, 2015). NLRP3 inflammasome’s activation is crucial for the production of biologically active proinflammatory cytokines, notably IL-1β and IL-18. IL-1β regulates gene expression of fever and pain threshold. Additionally, its receptor engagement promotes the migration of immune cells into infected or injured tissues, mediated by the response of endothelial cells (Hanslik & Ulland, 2020). IL-18 is also a co-stimulatory cytokine that mediates adaptive immunity and is crucial for interferon-gamma production (Kelley et al., 2019). To identify whether NLRP3 inflammasome activation is involved in HLJDD’s anti-inflammatory properties, the study of LPS-treated BV2 cells was conducted to examine its effects on NLRP3 gene and protein expression as well as downstream signaling molecules. The data indicate that LPS triggers the NLRP3 inflammasome within BV2 cells. In contrast, HLJDD significantly reduces both NLRP3 inflammasome activity and the related release of inflammatory cytokines, IL-18 and IL-1β, by microglia. This evidence highlights HLJDD’s capability to diminish microglial activation and its anti-inflammatory effects, which are likely due to the inactivation of the NLRP3 inflammasome. Research by Jin et al. (2019) demonstrates that baicalin effectively suppresses NLRP3 inflammasome activity and interrupts the TLR4/NF-κB signaling pathway, leading to a reduction in microglial activation and inflammation. Similarly, Tian et al. (2022) found that berberine facilitates the shift of microglia from the M1 phenotype to the M2 phenotype, enhancing its anti-inflammatory actions. Based on these findings, it is hypothesized that HLJDD’s ability to alleviate inflammation is possibly due to its constituent bioactive elements
Conclusion
The current investigation examines the potential impact of HLJDD on neuroinflammation, focusing particularly on its ability to modulate microglial responses and NLRP3 inflammasome activation. Our findings suggest that HLJDD treatment effectively mitigates LPS-induced neuroinflammatory reactions by regulating proinflammatory cytokines and inhibiting microglial activation alongside NLRP3 inflammasome activation. The findings highlight HLJDD’s promising role in the treatment of neuroinflammatory conditions, including Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, amyotrophic lateral sclerosis, and various acute cerebral disorders. Nevertheless, further exploration of the mechanistic underpinnings of HLJDD in clinical settings and additional studies are warranted.
Abbreviations
AD: Alzheimer’s disease; Arg-1: Arginase-1; CNS: Central nervous system; DAMPs: Damage-associated molecular patterns; HD: High dose; HLJDD: Huanglian Jiedu decoction; iNOS: Inducible nitric oxide synthase; IFN-γ: Interferon-gamma; IGF-1: Insulin-like growth factor 1; IL-1β: Interleukin-1β; LD: Low dose; MD: Medium dose; MS: Multiple sclerosis; NLRP3: Nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing 3; PAMPs: Pathogen-associated molecular patterns; PD: Parkinson’s disease; PRRs: Pattern recognition receptors; TCM: Traditional Chinese medicine; TNF-α: Tumor necrosis factor alpha ; SDS-PAGE: Sodium dodecyl sulphate-polyacrylamide gel electrophoresis; HRP: Horseradish peroxidase; DAPI: 4’,6-diamidino-2-phenylindole.
Authors’ Contribution
Junli Wang and Rui Wang contributed equally to this work.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The research is funded by Youth project of Wuhan Municipality’s Health and Family Planning Commission (Grant No. WZ21Q08). Wuhan Application Foundation Frontier Project (Grant No. 2020020601012302).
