Abstract
Background
Chemotherapy drugs damage intestinal cells, weakening the intestinal barrier. This damage results in higher permeability, which enables bacteria and toxins to enter the intestinal tissue.
Purpose
This study aimed to explore the protective effects of Fraxinus excelsior L. (F. excelsior) extract against Capecitabine (CT)-induced enterocolitis.
Materials and Methods
Fifty Wistar rats were divided into five groups: sham, F. excelsior (750 mg/kg orally), CT (500 mg/kg orally), and two co-treatment groups receiving CT with F. excelsior (500 and 750 mg/kg orally). After 50 days, rats were sacrificed, and blood samples were collected for various analyses. Biochemical assessments included measurements of serum nitric oxide, catalase, glutathione peroxidase, and superoxide mutase enzymes. Tissue oxidative stress was evaluated through FRAP, thiol, and TBARS levels. Pro-inflammatory cytokines were quantified using ELISA, and apoptosis was assessed through the evalution of p53/Bax/Bcl-2 pathway. Histopathological examination affirmed the preservation of tissue structure in groups treated with F. excelsior extract.
Results
F. excelsior extract reduced intestinal cell apoptosis and elevated the expression of intestinal aquaporin (AQP) genes/proteins by enhancing antioxidant enzymes and diminishing free radicals. Additionally, the extract modulated inflammatory cytokine levels, regulated antidiuretic hormone (ADH) and arginine vasopressin (AVP) levels, maintaining serum and intestinal osmotic balance. The study also revealed decreased expression of pro-inflammatory cytokines and a positive impact on water homeostasis-related genes (AQP3, AQP8, AQP10).
Conclusion
The study concludes that F. excelsior extract exhibits potential benefits in treating enterocolitis in individuals undergoing chemotherapy, emphasizing its ability to mitigate oxidative stress, inflammation, apoptosis, and maintain osmotic balance.
Introduction
Chemotherapy-induced enterocolitis (CIE) is a known complication that can occur as a side effect of cancer treatment. CIE can present with a range of symptoms, such as vomiting, diarrhea, abdominal pain, loss of appetite, rectal bleeding, nausea, and weight loss (Wang et al., 2021). The molecular mechanisms underlying CIE are complex. However, several key processes have been identified that contribute to the development of enterocolitis. Chemotherapy drugs have various detrimental effects on the intestines. They directly harm the intestinal cells, compromising the integrity of the intestinal barrier (Zhan, Chen, et al., 2023). This disruption increases permeability, allowing bacteria and toxins to leak into the intestinal tissue. Consequently, the damaged tissue triggers an inflammatory response, characterized by immune cell activation and the release of proinflammatory cytokines and chemokines. This immune response contributes to tissue damage and sustains the inflammatory process (Chen et al., 2021). The resulting excessive reactive oxygen species (ROS) production leads to oxidative damage of cellular components, further aggravating tissue injury and inflammation (Akbarali et al., 2022). Chemotherapy drugs impair the regenerative capacity of intestinal epithelial cells, impeding tissue repair, and prolonging the inflammatory response. CIE is associated with an imbalance in immune tolerance mechanisms in the intestines. This disruption results in an abnormal immune response against intestinal tissue, exacerbating inflammation, and tissue damage (Baldelli et al., 2021).
Chemotherapy drugs have been found to inhibit and reduce the expression of intestinal aquaporins (AQPs). AQPs, a group of membrane proteins, are essential for controlling the movement of water through cell membranes, such as those found in the intestines (Kamar et al., 2020). Recent evidence suggests that AQPs are implicated in the development of CIE. The disruption of AQP function causes a loss of water homeostasis and intestinal barrier function. Specifically, studies have highlighted the involvement of AQP3 and AQP8 in regulating immune cell migration, cytokine production, and inflammatory signaling pathways (Hong et al., 2020). Dysregulation of AQPs can contribute to the perpetuation of inflammation in enterocolitis. Additionally, AQPs are associated with tissue repair and regeneration processes. Altered AQP expression or function can interfere with the regenerative capacity of intestinal epithelial cells, impairing the healing process in CIE (Mosleh et al., 2023).
In addition to their apoptotic effects on tumor cells, chemotherapy drugs also trigger distinct apoptotic pathways in normal host cells, including intestinal cells. Studies show that chemotherapy medications induce apoptosis in intestinal cells by impacting the p27/PI3K pathways (Lukosiute-Urboniene et al., 2020). Moreover, these drugs induce the upregulation of p27, leading to cell cycle arrest and apoptosis in intestinal cells. Through the activation of MAPK/ MEK1/2 signaling pathways, chemotherapy drugs participate in regulating cell proliferation, survival, and apoptosis (Zhan, Yu, et al., 2023; Zhang et al., 2020). Consequently, these pathways lead to the phosphorylation and activation of downstream targets, including transcription factors and apoptotic regulators like Bax and Bcl-2, ultimately influencing intestinal cell apoptosis. Additionally, chemotherapy drugs can modulate the expression of cyclin D1, resulting in its downregulation. This disruption of cell cycle progression further promotes apoptosis in intestinal cells (Bian et al., 2021).
Capecitabine (CT) is a frequently prescribed oral chemotherapy medication employed in managing a range of cancer types, such as brain, colorectal, and gastric cancers. It is predominantly utilized in treating metastatic breast cancer, colorectal cancer, and gastric cancer, commonly serving as an initial treatment or as additional adjuvant therapy (Alzahrani et al., 2023). It can be administered as a single agent or in combination with other chemotherapy drugs or targeted therapies. Common side effects of CT include nausea, fatigue, diarrhea, hand-foot syndrome (palmar-plantar erythrodysesthesia), stomatitis, dermatitis, myelosuppression (reduced blood cell counts), and increased susceptibility to infections (Deneken-Hernandez et al., 2022). The mail CT metabolite in the body is fluorouracil (5-FU). The metabolites of 5-FU, including fluorouridine triphosphate and fluorodeoxyuridine monophosphate, are further incorporated into ribonucleic acid (RNA) and DNA, respectively, interfering with their synthesis and leading to cytotoxic effects. The active metabolite 5-FU acts as an antimetabolite, inhibiting the synthesis of DNA and RNA (Derissen et al., 2016). It interferes with the normal functions of these nucleic acids, disrupting cell division, and leading to cell death. Additionally, 5-FU inhibits the enzyme thymidylate synthase, which is involved in the synthesis of thymidine, a building block of DNA. By inhibiting thymidylate synthase, 5-FU reduces the availability of thymidine for DNA synthesis, further impeding cell proliferation. The exact mechanisms by which CT leads to enterocolitis are not fully understood but may involve a combination of immune dysregulation, oxidative stress, and direct toxic effects on the intestinal mucosa (Aarnoutse et al., 2022; Gu et al., 2021; Terranova-Barberio et al., 2017). Various studies have shown that extracts of plants contain bioactive compounds such as polyphenols. These compounds, through various mechanisms and synergistic effects, can protect normal cells from inflammatory, oxidative, and apoptotic damage alongside the therapeutic effects of chemotherapeutic drugs on tumor cells (Jain et al., 2016).
Fraxinus excelsior L. (F. excelsior), commonly known as European ash or common ash, is a deciduous tree species belonging to the Oleaceae family. The leaves of F. excelsior are compound and pinnate, composed of multiple leaflets arranged in an opposite arrangement along a central axis called a rachis. Each leaf typically consists of seven to thirteen leaflets, although the number can vary. F. excelsior is native to most of Europe and Asia. It is commonly found in deciduous forests, woodlands, and riverbanks (Thomas, 2016). In traditional medicine, the leaves of this plant have been used for various medicinal purposes. They are believed to possess therapeutic properties in the treatment of conditions, such as diabetes, nausea, vomiting, digestive disorders, diarrhea, fatty liver, and various tumors, particularly hepatocellular carcinoma and colon cancer. Liquid chromatography-electrospray ionization-mass spectrometry studies of F. excelsior leaf extract have revealed a high content of polyphenolic compounds, including catechin, p-coumaric acid, isoquercitrin, daidzein, kaempferol, scopoletin, caffeic acid, genistein, and quercetin (Qazi et al., 2018). Polyphenolic compounds, known for their anti-inflammatory, antioxidant, and immunomodulatory properties, have been studied for their potential protective effects against enterocolitis. By reducing oxidative stress, polyphenols can safeguard against intestinal damage and inflammation (Li et al., 2022). Moreover, they have the ability to boost the generation of anti-inflammatory cytokines and reduce the secretion of proinflammatory cytokines. This aids in maintaining a balanced immune response and relieving inflammation (Ul Islam et al., 2021).
In this study, our objective was to examine the effects of F. excelsior on Capecitabine (CT)-induced enterocolitis in rats. We employed biochemical, molecular, and histopathological approaches to assess the impact of F. excelsior on CIE caused by CT.
Materials and Methods
Preparation of F. excelsior Extract
Fresh leaves of F. excelsior (2,000 g) were dried at 35°C after being collected and confirmed by a botanist. Then, with the help of a soil grinder, a powder was prepared from the leaves and added to 70% ethanol/water (v.v). After incubation at 35°C for 72 h, it was filtered by a paper filter, and the resulting mixture was compressed by a rotary evaporator. The resulting extract (200 g) was kept at 4°C (Parveen et al., 2016).
Experimental Design
Before commencing the research, the rats were acclimatized to the experimental surroundings for 72 h. Rats displaying symptoms of illness, particularly digestive issues, such as diarrhea and anorexia, were excluded from the study. All rats were cared for by international standard guidelines, protocols, and ethical regulations outlined by the QingDao Municipal Hospital ethics committee. The rats were housed in standard laboratory conditions (at 25 ± 2°C, 12/12 dark/light cycle, and 45 ± 3% relative humidity). They were provided ad libitum access to standard rodent pellets and tap water. Subsequently, a total of 50 adult male Wistar rats (185 ± 10 g) were divided into five groups and observed for 50 days.
Sham group, administered with distilled water (1 mL/orally for 50 days). Seven-hundred fifty F. excelsior group, administered with 750 mg/kg of F. excelsior orally for 50 days. CT group, administered with 500 mg/kg of CT orally on days 10, 20, 30, 40, and 50. Co-treatment group 1 (CT + 500 F. excelsior), receiving CT on days 10, 20, 30, 40, and 50 along with 500 mg/kg of F. excelsior orally for 50 days. Co-treatment group 2 (CT + 750 F. excelsior), receiving CT on days 10, 20, 30, 40, and 50 along with 750 mg/kg of F. excelsior orally for 50 days.
LD50 technique was used to select the effective dose of nontoxic treatment along with the pilot study and similar studies conducted on CT and F. excelsior. In this study, administration of CT and F. excelsior was done every day during specific and fixed hours, 9 am and 3 pm, respectively (Abbasi et al., 2017; Sakai et al., 2021).
Acute Toxicity Test (LD 50 ) for F. excelsior
Lork’s two-step method was used to evaluate the LD50 of F. excelsior. Briefly, first, nine rats in three separate groups received doses of 25, 250, and 2,500 mg/kg of F. excelsior through intraperitoneal (i.p.) injection and the rats were monitored for possible death or toxic effects within 24 h. Then three rats in three separate groups received concentrations of 50, 500, and 5,000 mg/kg of F. excelsior through i.p. injection and were monitored again within 24 h. Finally, the highest safe dose (D safe) and the lowest death (toxic) dose (D toxic) obtained after monitoring the rats were placed in Lork’s formula to calculate the LD50 of F. excelsior.
LD50 = (D safe × D toxic) 1/2
During the study period, the survival rate was drawn with the help of the Kaplan–Meier curve (Zhang et al., 2023).
Serum Concentrations of Antidiuretic Hormone and Arginine Vasopressin Hormones
After sacrificing the rats (preanesthesia/anesthesia using ketamine in xylazine 50 mg/kg and 30 mg/kg), blood was taken from the serum, and heart specimens were isolated using a centrifuge. Then, using commercial ELISA kits, serum levels of antidiuretic hormone (ADH) and arginine vasopressin (AVP) hormones were measured as instructed (Özcan Türkmen et al., 2023).
Superoxide Dismutase, Catalase), and Glutathione Peroxidase Serum Activity
To measure serum levels of superoxide dismutase (SOD), glutathione peroxidase (GPx), and catalase (CAT) activity, commercial ELISA kits from R&D Systems, Inc. were used as instructed (Khanjar, 2021).
Serum Levels of Nitric Oxide
ZellBio ELISA kit was applied to measure nitric oxide (NO) serum concentrations as an indicator of free radical production as instructed. As per the kit instructions, 50 µL of serum and standard samples (assay buffer) were added to the wells of the 96-well plate, and then 10 µL of nitric reductase solution was transferred to each well. After incubation (20 min/37°C), 25 µL of kit solutions B and A were added to the wells and incubated again (20 min/37°C). Finally, the mixture absorbance was read at 540 and 570 nm by a UV-visible spectrophotometer (V-630 Bio; JASCO, US) (Shahraki et al., 2020).
Intestine Ferric Reducing the Ability of Plasma Levels
One of the methods of measuring total antioxidant capacity is ferric reducing the ability of plasma (FRAP) evaluation. Briefly, first, 100 mg of tissue after homogenization at 4°C was mixed with 200 µL of cold PBS, and 100 µL of it was mixed with 10 µL of FRAP solution. Following incubation of the extract mixture (15 min/25°C) and with 12,000 g for 10 min centrifuging it, the supernatant absorbance was read at 593 nm using a UV-visible spectrophotometer (V-630 Bio; JASCO, US) (Shahraki et al., 2020).
Intestine Lipid Peroxidation Levels
To determine lipid peroxidation (LPO) levels, the concentration of thiobarbituric acid reactive substances (TBARS) in tissue was measured. Following the FRAP protocol, 100 µL of homogenized intestine tissue was added to 100 µL of TBARS solution. This solution underwent incubation (37°C/30 min). The supernatant’s absorbance was measured using a UV-visible spectrophotometer (at 593 nm, V-630 Bio; NY, US) (Shahraki et al., 2020).
Intestine Thiol Levels
To measure tissue thiol level as an important tissue antioxidant index, 100 µL of intestine-homogenized tissue mixture was mixed with 5,5-dithio-bis-2-nitrobenzoic acid (20 µL), and after incubation (37°C/15 min) and centrifugation (12,000 g/5 min), the supernatant wavelength was 412 nm. It was read by a UV-visible spectrophotometer (V-630 Bio; JASCO, US) (Shahraki et al., 2020).
Serum Concentrations of IL-1β, IL-6, CXCL-1, and TNF-α
To measure serum concentrations of anti/pro-inflammatory cytokines, commercial ELISA kits from R&D Systems, Inc. were used as instructed (Qi et al., 2022).
Intestine Total RNA Extraction
The Canvas Biotech Total RNA Purification Kit was employed for isolating total RNA, following the manufacturer’s protocol. Initially, 50 mg of intestine tissue was homogenized, and 300 µL of buffer BLY was combined with 10 µL of β-mercaptoethanol and incubated (15 min/4°C). The sample then underwent centrifugation (10,000 g/2 min), and the supernatant was transferred to a filter column. After another centrifugation at the same speed and duration, 300 µL of 70% ethanol was added to the filtrate, followed by an additional centrifugation step. To conclude, 50 µL of deionized water was mixed with the pellet, followed by storing at −70°C. The RNA concentration was measured using a Nanodrop (Thermo Fisher Scientific spectrophotometer), and the RNA quality was measured through 2% agarose gel electrophoresis (Koral et al., 2021).
Expression of Aquaporin’s and Apoptosis-related Genes with Quantitative Real-time PCR
qScriber cDNA synthesis kit was a kit that was used to prepare a cDNA template according to the instructions of the kit. Extracted RNA (1 µg) was added to 10-µL qScriber reaction master mix, 8 µL deionized water, and 1 µL of both oligo/random hexamer primers. Then the resulting mixture was placed in a temperature cycle including 20 min at 55°C, 99°C for 5 min, and 10 min at 30°C. Primer-3 software and Gene Runner version 6.5.52 were used to design primers. NCBI database was applied for the designed blast primers. The β-actin gene was considered the internal reference and the expression of other genes was measured relative to it. The quantitative real-time PCR mixture contained cDNA (1 µL), SYBR Green Master Mix (10 µL), and reverse/forward primers (1 µL). Forty-two standard temperature cycles with an annealing temperature of 62°C (1 min) are considered and finally, the threshold cycle (CT) of Bax, AQP3, p53, Caspase-3, AQP8, Bcl-2, and AQP10 was registered, and fold formula change was used to calculate the gene relative expression and considering ∆∆Ct.
∆∆Ct = [(CtS − CtHG) − (CtS − CtC)]; S: sample, HG: house kepping, C: control (Koral et al., 2021).
Expression of Aquaporin-related Proteins in the Intestine with Western Blotting
Western blot analysis evaluated the protein expression levels of AQP3, AQP10, and AQP8. Briefly, a homogenized tissue sample from the intestine was prepared. Subsequently, 100 mg of the specimen was added to PBS (50 µL) and radio-immunoprecipitation assay buffer (100 µL). The resulting mixture was centrifuged, and then 20 µL of the loading buffer was mixed with a polyvinylidene fluoride membrane containing primary antibodies (Cambridge Bioscience, UK) targeting AQP10 (Cat. No. HPA062621; 1:200), AQP3 (Cat. No. RC201856; 1:200), and AQP8 (Cat. No. HPA046259; 1:200). The mixture, along with an SDS polyacrylamide gel (10%), was separated using appropriate techniques. After incubation for 12 h at 4°C, the membrane underwent incubation using 40 min Horseradish peroxidase–conjugated antibody. Finally, the protein band signals were documented and assessed by Bio-Rad software, utilizing an enhanced chemiluminescence reagent (e-BLOT company, China). For further analysis, Image J software was employed (32).
Intestine Histopathology
To evaluate the level of tissue destruction and histopathological changes of the intestine in different groups after removing the intestinal tissue and fixing it in 10% formalin, 5-micrometer slices were prepared from the tissues during the routine process of tissue preparation and stained with hematoxylin and were stained with eosin. By an optical microscope (model No. BX61TRF; Olympus, Japan) attached to the Moticam 1080N BMH camera system, intestinal villi, intestinal glands, the amount of lymphocyte infiltration, the amount of intestinal villus of the intestinal wall, and the structure of the villi were evaluated with 40×, 100×, and 400× magnifications (Koral et al., 2021).
Statistical Analysis
Group comparisons were conducted using one-way analysis of variance, followed by Newman–Keuls post hoc tests for in-depth comparisons. Significance was considered at p values < 0.05. The normality and homogeneity of the data were assessed using the Kolmogorov–Smirnov test, with a p value above 0.05 indicating normality. Results are presented as mean ± standard deviation (SD). Data analysis was carried out using SPSS 16, and GraphPad Prism software 9 was utilized for creating graphs.
Results
LD 50 of F. excelsior
After conducting a 24-h monitoring of groups treated with F. excelsior, the results revealed that the safe dosage (D safe) was determined to be 2,500 mg/kg, while the toxic dosage (D toxic) was found to be 5,000 mg/kg of F. excelsior. Using Lork’s formula, the LD50 of F. excelsior was calculated to be 3.535 mg/kg. Consequently, it suggests that lower doses than the LD50 of F. excelsior are applicable in animal studies.
Body Weight and Rat Survival Rate
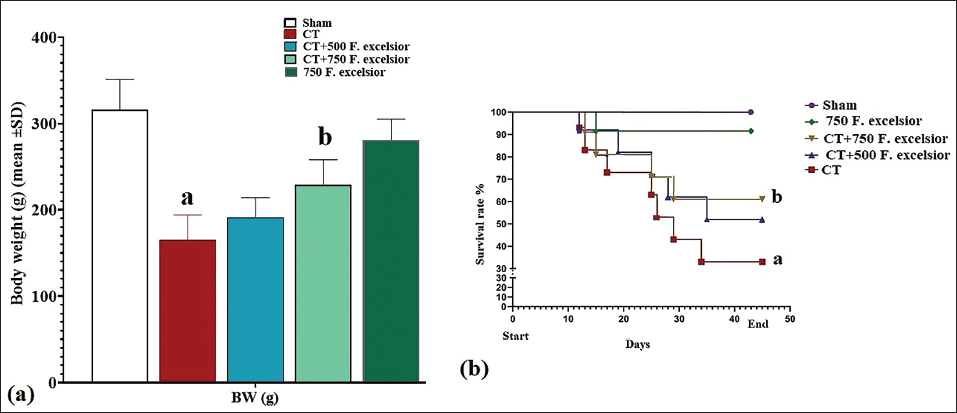
Treatment with CT resulted in a statistically significant (p < 0.05) decrease in rat weight compared to the sham group. Moreover, the survival rate of rats in the CT group exhibited a noteworthy reduction (p < 0.05) compared to the sham group. Conversely, F. excelsior demonstrated a dose-dependent effect when administered at 500 and 750 mg/kg (in the CT+500 and 750 mg/kg F. excelsior groups), leading to a significant (p < 0.05) increase in rat weight compared to the CT group. Additionally, the survival rate of rats in the F. excelsior groups significantly improved (p < 0.05) compared to the CT group (Figure 1).
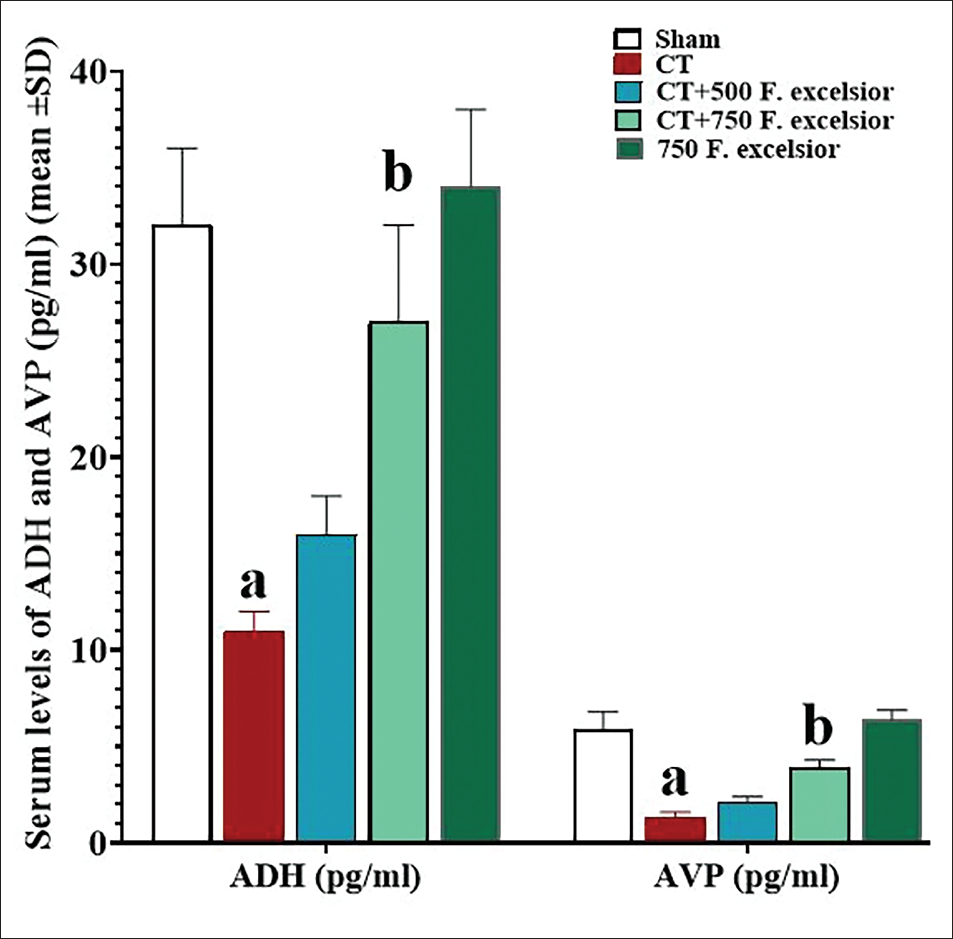
Serum Levels of ADH and AVP
The analysis of the serum ADH and AVP data indicated a significant decrease (p < 0.05) in both parameters within the CT group than in the sham group. However, it is noteworthy that F. excelsior demonstrated a dose-dependent effect in elevating the levels of these serum parameters than in the CT group. Interestingly, the increase in the serum level of ADH was found to be significant (p < 0.05) specifically in the CT+ 750 mg/kg F. excelsior group (Figure 2).
NO Serum Levels and Serum Activity of CAT, GPx, and SOD
Capecitabine, through the induction of free radicals, led to a notable increase in NO serum levels compared to the sham group, whereas F. excelsior exhibited a dose-dependent decrease in NO levels when compared to the CT group. This reduction was statistically significant (p < 0.05) at 750 mg/kg (in the CT+750 mg/kg F. excelsior group). Additionally, CT significantly decreased the serum activity of antioxidant enzymes in comparison to the sham group. Conversely, F. excelsior dose-dependently raised the serum levels of all three enzymes compared to the CT group, with a significant elevation (p < 0.05) observed at 750 mg/kg (in the CT+750 mg/kg F. excelsior group) when compared to the CT group (Figure 3a).
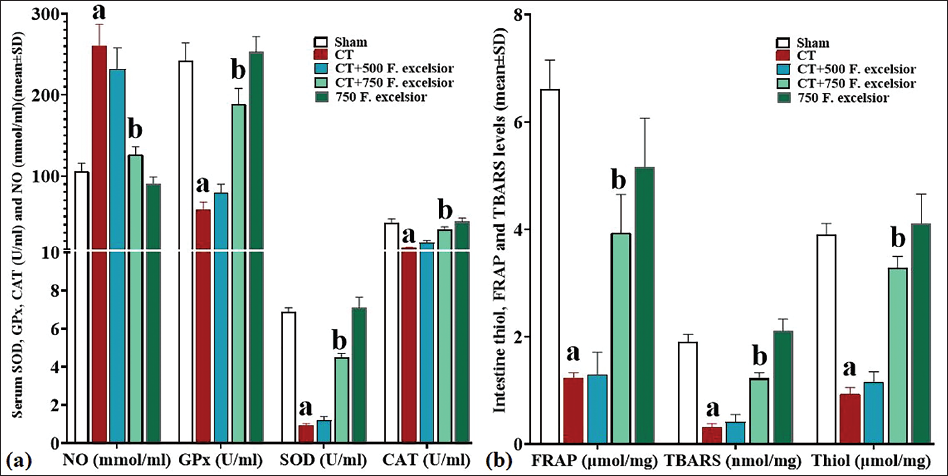
TBARS, Thiol, and FRAP Levels in the Intestine
Thiobarbituric acid reactive substances, thiol, and FRAP levels served as comprehensive indicators of total antioxidant capacity, as well as LPO. The findings revealed that CT led to a significant (p < 0.05) reduction in the tissue levels of all three factors compared to the sham group. Conversely, F. excelsior, with its rich antioxidant content, was able to dose-dependently increase the concentrations of all three factors when compared to the CT group. This elevation was statistically significant (p < 0.05) at 750 mg/kg (in the CT+750 mg/kg F. excelsior group) and demonstrated the most pronounced change (p < 0.05) in comparison to the CT group (Figure 3b).
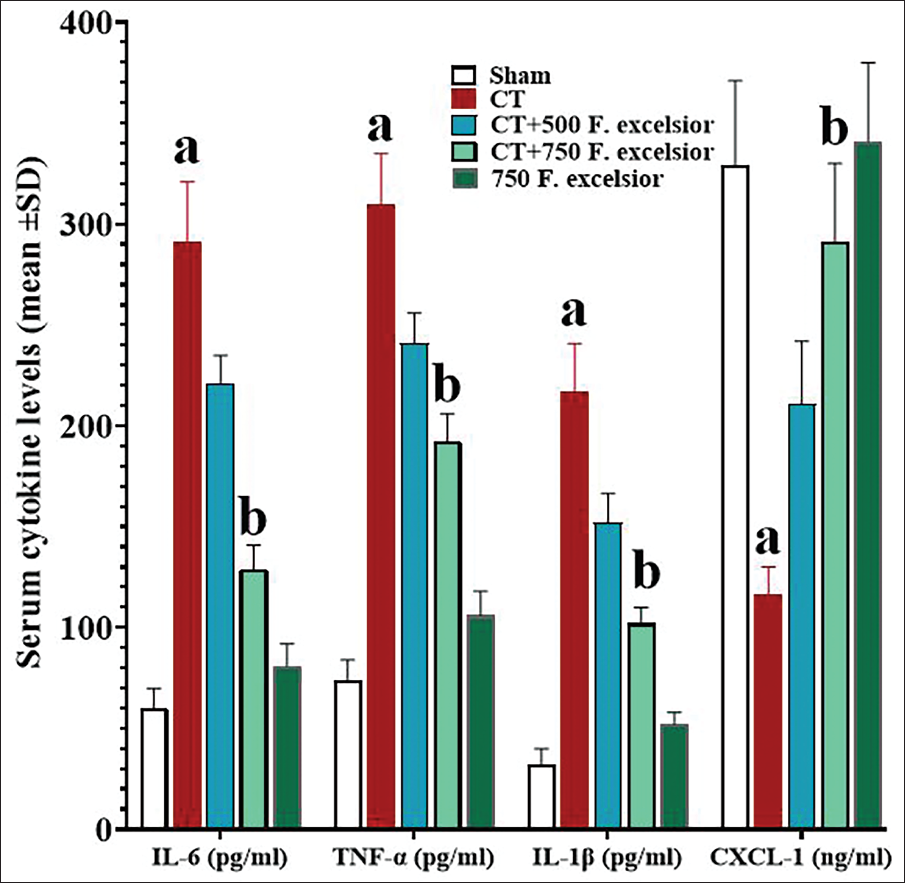
Serum concentrations of IL-1β, IL-6, CXCL-1, and TNF-α
By inducing inflammatory effects, CT increased the concentrations of proinflammatory cytokines and inhibited systemic anti-inflammatory cytokines, so that the serum concentrations of all three proinflammatory cytokines were significantly increased (p < 0.05) in comparison to the sham group, whereas CXCL-1 concentrations decreased significantly. However, the results of the study revealed that F. excelsior has powerful anti-inflammatory effects. The dose-dependently increased the serum concentrations of CXCL-1 and decreased the concentrations of all three proinflammatory cytokines compared with the CT group. These alterations were found to be significant (p < 0.05) in the 750 mg/kg (in the CT+ 750 mg/kg F. excelsior group) of this plant, in comparison to the CT group (Figure 4).
Expression of Intestine Cas-3, AQP3, Bax, AQP8, Bcl-2, AQP10, and p53 Genes
Capecitabine induced a notable (p < 0.05) rise in the levels of all three pro-apoptotic genes (Cas-3, Bax, and p53) as compared to the sham group. Concurrently, there was a significant decrease (p < 0.05) in the expression of the anti-apoptotic Bcl-2 gene. Conversely, F. excelsior exhibited the ability to counteract these effects by diminishing the expression of the three pro-apoptotic genes (Cas-3, p53, and Bax) and enhancing the expression of the anti-apoptotic Bcl-2 gene through the inhibition of apoptosis. These alterations were statistically significant (p < 0.05) at 750 mg/kg (in the CT+750 mg/kg F. excelsior group) of this plant when compared to the CT group (Figure 5a).
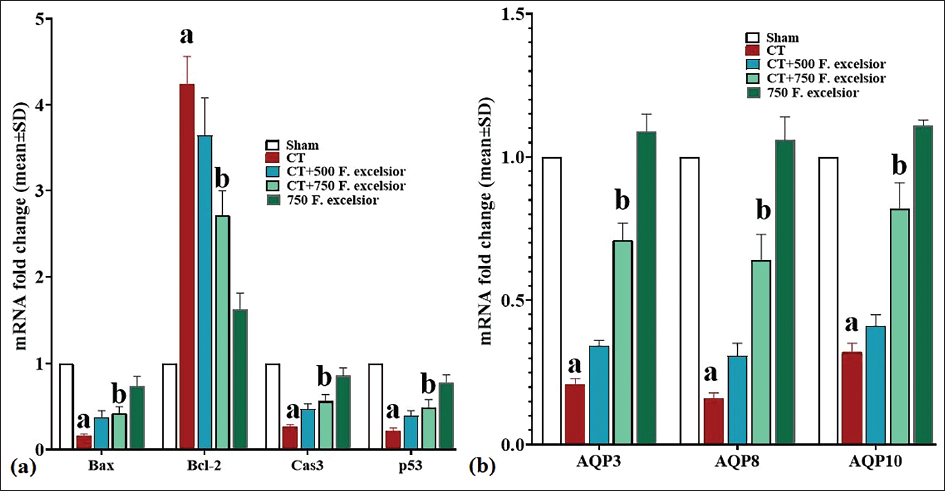
One of the major factors contributing to the disturbance in osmotic balance among patients receiving CT is the impairment in the functioning of AQPs. In this study, CT indicated a significant reduction (p < 0.05) in the gene expression of all three AQPs (AQP3, AQP8, and AQP10) when compared to the sham group. However, F. excelsior demonstrated its ability to effectively increase the expression of all three AQP genes in the 750 mg/kg dosage (in the CT+ 750 mg/kg F. excelsior group) of this plant, which was significant (p < 0.05) in comparison to the CT group (Figure 5b).
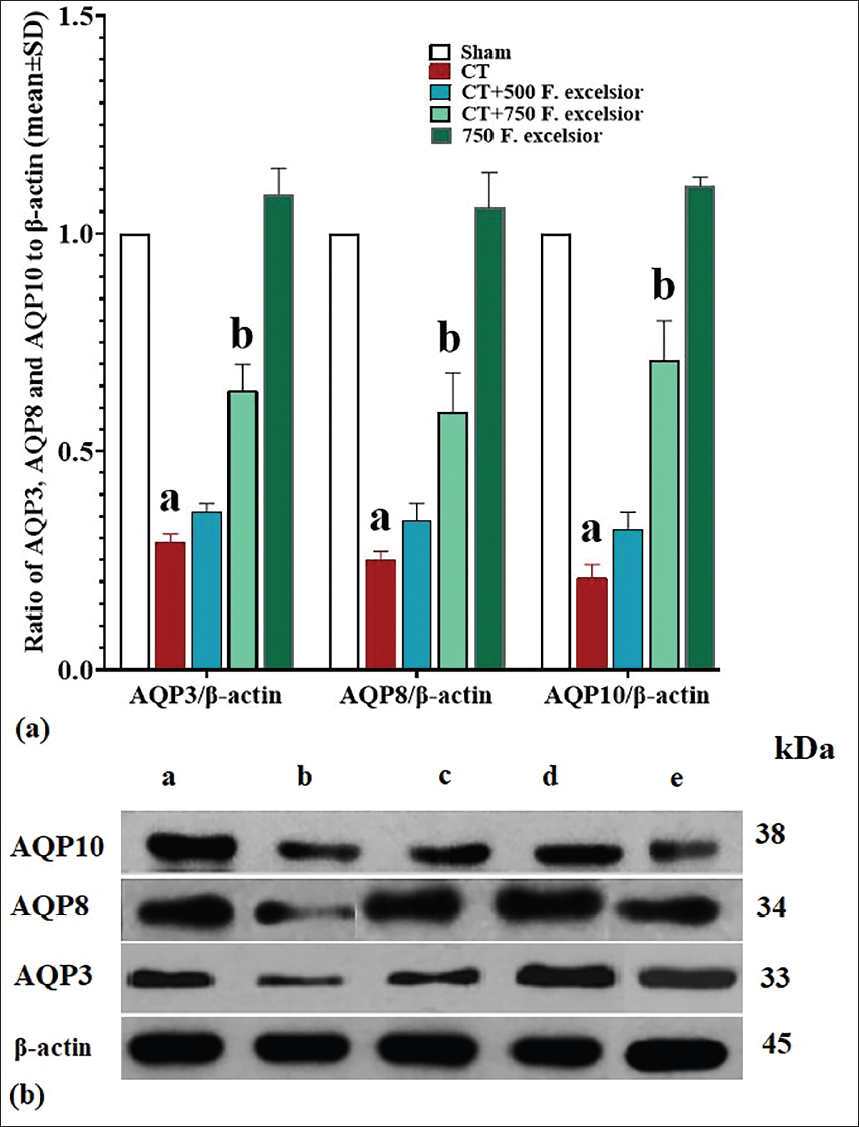
Expression of Intestine AQP3, AQP8, and AQP10 Proteins
To better assess the impact of CT on the expression of AQP proteins in the intestine, we examined the expression levels of AQP3, AQP8, and AQP10. The results of this analysis revealed that CT had a significant reduction (p < 0.05) in the protein expression of all three AQPs in comparison to the sham group. However, in the CT+750 mg/kg F. excelsior group, the expression of all three AQP proteins increased significantly (p < 0.05) compared to the CT group. This notable increase was observed with a dosage of 750 mg/kg of F. excelsior and demonstrated statistical significance (p < 0.05) (Figure 6).
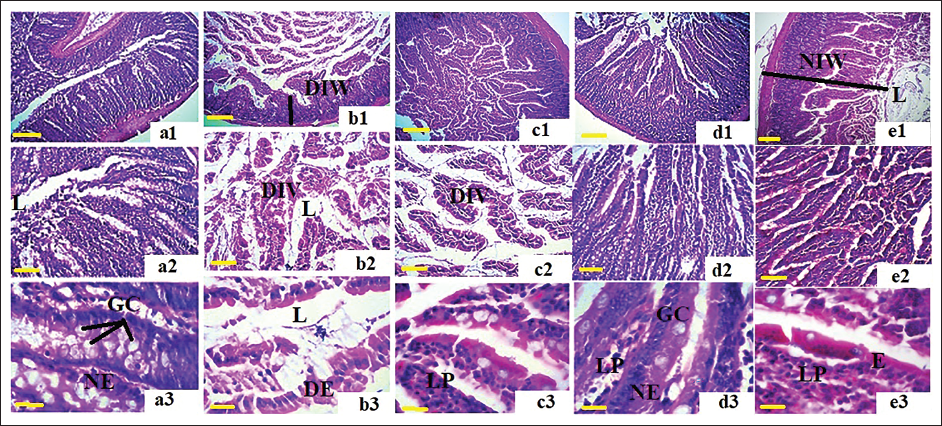
Intestine Histopathological Evaluations
The histopathological examination of the intestine in the different groups revealed that CT caused severe damage at the tissue level. It resulted in the separation of the mucosa from the lamina propria and submucosa, leading to the disruption of the arrangement of intestinal villi. The underlying tissue showed signs of edema and inflammation. Moreover, the intestinal lumen exhibited uprooted villi and floating epithelial cells, indicating the degradation of the basal and epithelial membranes and the presence of apoptotic bodies within the lumen. On the other hand, in the groups that received F. excelsior, the integrity of the intestinal villi was clearly observed, and this effect was observed to be dependent on the dosage. There was a significant reduction in tissue edema and lymphocyte infiltration. Furthermore, the groups receiving F. excelsior displayed a normal architecture of absorptive epithelium (enterocytes) adjacent to a thin lamina propria and a submucosa rich in glands and vessels. This suggests the protective role of F. excelsior in preserving the structural integrity of the intestine (Figure 7).
Discussion
CT-induced Enterocolitis
The results of the present study indicate that CT and its metabolites induce enterocolitis in recipients through distinct mechanisms. CT, like certain other chemotherapy drugs, can affect the secretion of ADH and AVP through various molecular pathways. Animal studies have revealed that certain chemotherapeutic agents, including cyclophosphamide and 5-fluorouracil (5-FU), can disrupt the regulation of ADH and AVP release, resulting in fluid balance and electrolyte abnormalities. Chemotherapy-induced nausea and vomiting can trigger dysregulation of ADH and AVP secretion, leading to fluid retention and hyponatremia. This effect is particularly noticeable with highly emetogenic chemotherapy regimens (Sobol et al., 2023). Additionally, chemotherapy-induced mucositis and gastrointestinal disturbances can indirectly impact fluid intake and absorption, thereby influencing ADH and AVP regulation. Our study demonstrated that CT caused dysregulation of ADH and AVP secretion, leading to decreased serum levels at the end of the study (Huang et al., 2022). Numerous studies have shown that 5-FU can generate ROS and free radicals, which can cause oxidative stress and damage cellular components, such as proteins, DNA, and lipids (Aarnoutse et al., 2022). CT can activate various signaling pathways by releasing free radicals, including mitogen-activated protein kinases and nuclear factor kappa B (NF-κB). Moreover, CT has been found to inhibit the proliferation of normal cells through the NF-κB/p53/Cas-3 and WNT/β-catenin pathways, while enhancing endogenous apoptotic pathways (Gu et al., 2021; Terranova-Barberio et al., 2017). Xiang et al. (2020) also demonstrated that CT inhibits cyclins through the MAPK and AKT/IRS-1 pathways, causing dysregulation of cyclin-dependent kinases and ultimately inhibiting the intestinal parenchymal cell cycle (Almaimani et al., 2022; Xiang et al., 2020). Österlund et al. (2007) with randomized trial data conducted a study on colorectal cancer chemotherapy using CT and found that it led to severe diarrhea and disrupted the balance of water and electrolytes, impacting the overall well-being and treatment outcomes of patients (Österlund et al., 2007). Furthermore, Rtibi et al. (2018) investigated the effects of CT in rats and revealed that this chemotherapy drug not only hindered the activity of endogenous antioxidant enzymes, such as GPx, CAT, and SOD but also led to intestinal mucosal damage and disrupted fluid accumulation and electrolyte secretion (Rtibi et al., 2018). We found that CT treatment resulted in a decrease in total antioxidant capacity, as indicated by reduced levels of intestinal FRAP, TBARS, and total thiol. This decrease was related to the production of free radicals, specifically an increase in NO levels. Moreover, CT treatment was found to enhance the mitochondrial apoptotic pathway activation, demonstrated by an upregulation of Bax, p53, and Caspase-3 gene expression in rats. These effects were accompanied by the inhibition of antioxidant enzyme activity, including GPx, CAT, and SOD. In clinical studies conducted by Klimko et al. (2021), it was observed that patients undergoing CT treatment experienced significant adverse effects, including severe diarrhea and injuries to the gastrointestinal tract, such as mucositis, enterocolitis, and, although rare, ileitis. These complications have been found to disrupt the efficacy of CT in tumor treatment. They suggested the potential use of supplements or medications to manage diarrhea and restore electrolyte balance in patients receiving CT (Klimko et al., 2021). Another mechanism contributing to the onset of diarrhea following CT treatment is the mitotic arrest of crypt cells in the G2 phase. Additionally, recent research has highlighted abnormalities in goblet cells associated with CT usage, warranting further investigation. Among the various mechanisms impacted by CT, one notable aspect is the dysfunction, expression, and repair of AQPs, with a particular focus on AQP3 (Alzahrani et al., 2023). Trigueros-Motos et al. (2012) demonstrated that this chemotherapy drug not only inhibits the expression of AQP3 but also induces apoptosis in enterocyte cells by enhancing the p53-dependent apoptotic pathway. Consistent with prior research, our present study similarly found that CT suppresses the expression of all three AQPs (AQP3, AQP8, and AQP10) present in intestinal cells (Trigueros-Motos et al., 2012).
F. excelsior Ameliorates Enterocolitis Induced by CT
The present study demonstrates that F. excelsior exhibits a dose-dependent ameliorating effect on CT-itxtnduced enterocolitis, particularly at a dose of 750 mg/kg. This effect is mediated through various mechanisms. F. excelsior contains several polyphenol compounds, particularly flavonoids, which exert multifactorial effects. These compounds not only synergistically enhance the efficacy of chemotherapy drugs in suppressing tumor cells but also protect the function and structure of parenchymal cells against the oxidative, inflammatory, and apoptotic damage caused by chemotherapy drugs. An in vivo study demonstrated that the extract of this plant has protective effects on the liver against carbon tetrachloride (CCl4) toxicity. They observed that the plant extract increased the activity of SOD, CAT, glutathione reductase, peroxidase, and glutathione-S-transferase. Additionally, it enhanced the total antioxidant capacity and increased tissue TBARS levels in the liver (Younis et al., 2016). Numerous in vitro and in vivo studies have highlighted the protective effects of F. excelsior. Ahmadianmoghadam et al. (2022) assessed the plant’s protective effects against oxidative stress, memory impairment, and withdrawal syndrome due to morphine. They declared that the plant extract significantly enhanced the brain’s total antioxidant capacity while reducing free radicals, and LPO (malondialdehyde and NO production) (Ahmadianmoghadam et al., 2022). In addition to its protective effects, another in vitro study showed that compounds derived from F. excelsior can suppress the growth of various cancer cell lines, including MCF-7 (breast ductal carcinoma), SW742 (colorectal adenocarcinoma), PLC/PRF/5 (liver hepatoma), and A375 (melanoma cancer). This property can complement the action of chemotherapy drugs in inhibiting the growth of diverse tumors, further enhancing the therapeutic outcomes (Sardari et al., 2009). In our study, F. excelsior effectively enhanced the total antioxidant capacity (FRAP, TBARS, and thiol). Furthermore, it demonstrated the ability to decrease tissue and systemic levels of free radicals (NO) while also attenuating inflammatory pathways and the release of associated cytokines.
Conclusion
F. excelsior exhibits dose-dependent ameliorating effects on CT-induced enterocolitis through its polyphenol compounds, including flavonoids.
Footnotes
Abbreviations
ADH: antidiuretic hormone; AVP: Arginine vasopressin; AQP: Aquaporin; CAT: Catalase; CIE: Chemotherapy-induced enterocolitis; COX-2: Cyclooxygenase-2; CT: Capecitabine; CXCL1: Chemokine (C-X-C motif) ligand; DNA: Deoxyribonucleic acid; DTNB: 5,5-dithio-bis-2-nitrobenzoic acid; 5-FU: Fluorouracil; F. excelsior.: Fraxinus excelsior L.; FRAP: Ferric-reducing antioxidant power; FUTP: Fluorouridine triphosphate; HRP: Horseradish peroxidase; iNOS: Inducible nitric oxide synthase; MAPK: Mitogen-activated protein kinase; MDA: Malondialdehyde; RNA: Ribonucleic acid; TPTZ: 2,4,6-tripyridyl-riazine.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This research adhered to ethical guidelines for the use of animal subjects. Animal welfare considerations were taken into account, minimizing pain, distress, and suffering. Ethical approval was obtained from the Qingdao Municipal Hospital, Qingdao for animal research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
