Abstract
Background
The purpose of this study was to evaluate the effect of 18β-glycyrrhetinic acid (18β-GA) on isolated rat pulmonary artery vascular rings as well as the mechanism that lies behind the diastolic effects of this compound.
Materials and Methods
To examine the effects of various doses of 18β-GA on resting normal vascular rings, we used isolated rat pulmonary artery vascular rings. The isolated rat pulmonary artery vascular ring was precontracted with phenylephrine (PE) and potassium chloride solution (KCl), which allowed for the observation of the diastolic effects of 18β-GA, as well as the effects of various concentrations of 18β-GA on blocking the four potassium channels of glibenclamide, barium chloride (BaCl2), tetraethylamine (TEA), or 4-aminopyridine; the impact of various 18β-GA concentrations on the pulmonary artery vasodilation effect pre-use of the nitric oxide synthase inhibitors l-nitroarginine methyl ester and indomethacin.
Results
It was shown that 18β-GA has concentration-dependent diastolic effects on isolated rat pulmonary vascular rings that have been precontracted with PE and KCl but it has no effect on resting isolated thoracic aortic vascular rings.
Conclusion
The two Kir and Kv channels may be connected to the endothelium-dependent vasodilation mechanism of 18β-GA.
Keywords
Introduction
Heart disease, vascular disease, stroke, heart failure, arrhythmia, and other illnesses are included in the term “cardiovascular disease” (CVD). CVD remains the leading cause of premature death with rising medical costs (Chen et al., 2021; Mensah et al., 2019; Li et al., 2020). Pulmonary arterial hypertension (PAH) is a serious condition in which pulmonary vascular resistance is progressively increased due to complex multifactorial causes. The main pathogenesis is the proliferation of pulmonary artery smooth muscle cells, pulmonary vasoconstriction, and remodeling (Sakao & Tatsumi, 2011), which leads to an increase in pulmonary blood pressure and eventually causes the patients’ death with right heart failure (de Jesus, 2016, Galiè et al., 2016, Orriols et al., 2017). Finding pulmonary vasodilator medications is another way to treat PAH because PAH is directly associated with excessive pulmonary artery constriction. Antihypertensive medications are widely used, however, many of them have more negative effects than others. Therefore, developing novel antihypertensive medications is important (Cristo et al., 2021; Hao et al., 2020; Wang et al., 2016).
Pulmonary vasodilatation is one of the characteristics of early pulmonary vasculopathy. After receiving treatment with pulmonary vasodilators, exercise hemodynamics in patients with exercise PAH peaked, according to Wallace et al. (2018). Traditional Chinese Medicine has a long history of use in China. It has the advantages of low cost, a wide range of applications, and small side effects (Dang et al., 2020; Kosanovic et al., 2013; Tsai et al., 2008). Licorice is recorded as an herbal medicine and has been used for thousands of years in ancient China, India, and Greece medical books. It has biological activities, such as anti-inflammatory, anticancer, and antioxidant (Huan et al., 2021; Liu et al., 2021; Wang et al., 2013). The chemical compound 18β-glycyrrhetinic acid (18β-GA), which is obtained from licorice, is significant. Two trivalent nitrogen atoms and dipiperidine rings make up its basic structure. It has several pharmacological properties, including proliferative, anti-inflammatory, and antiapoptosis activities (Luo et al., 2021; Mizoguchi et al., 2014; Song et al., 2014). According to earlier research (Zhang et al., 2019), 18β-GA extracted from licorice can reduce right ventricular hypertrophy, mean pulmonary artery pressure, and right ventricular systolic pressure in PAH rats. It can also dilate the vascular rings of the thoracic aorta in rats and inhibit monocrotaline (MCT)-induced PAH. With inhibitory effects on pulmonary vascular cyclic hypoxic constriction, 18β-GA has been widely employed as a connexin inhibitor and gap junction blocker (Kizub et al., 2013, 2016).
However, the mechanism by which 18β-GA dilates pulmonary vascular rings to reduce hemodynamics in rats with PAH remains unclear. Therefore, in this article, we further investigated the mechanism of the diastolic effect of 18β-GA on pulmonary vascular rings through ion channels to provide experimental and theoretical support for the treatment of PAH with functional components of traditional Chinese medicine.
Materials and Methods
Instruments
Hv-4 thermostatic perfusion system for isolated tissues and organs (Chengdu Taimeng Technology Co., Ltd); Jz-100 muscle tension transducer (Beijing Xinhang Xingye Science and Trade Co., Ltd.); Bl-420s biological function experimental system (Chengdu Taimeng Technology Co., Ltd); Hh-s24 ultra-thermostatic water bath (Heze Dahua Instrument Co., Ltd.).
Medicines and Reagents
18β-GA (Shanghai Yuanye Biotechnology Chemical Co., Ltd); phenylephrine (PE, Shanghai Hefeng Pharmaceutical Co., Ltd); acetylcholine chloride (Beijing Solabao Technology Co., Ltd); glibenclamide (Gli, Shanghai Yien Chemical Technology Co., Ltd); barium chloride (BaCl2, Shanghai Yuanye Biotechnology Chemical Co., Ltd.); tetraethylammonium (TEA, Shanghai bide Medical Technology Co., Ltd); 4-aminopyridine (4-AP, Tokyo Huacheng Industrial Zhushi Society);
The k-H nutrient solution was prepared, using 1000 mL double steamed water and adding 0.35 g KCl, 0.29 g MgSO47H2O, 0.28 g CaCl2, 0.16 g KH2PO4, 2.0 g glucose, 6.92 g NaCl and 2.21 g NaHCO3 into the beaker, respectively. The mixture was swirled until it dissolved entirely. For immediate use, adjust pH to 7.4 with 10 mmol/L NaOH; for later use, keep in a water bath at a constant 37°C.
Animals
Specific pathogen-free healthy adult male Sprague-Dawley (SD) rats (weights: 220–280 g, ages: six to eight weeks) were provided by the Experiment Animal Center of Ningxia Medical University (Ningxia, China). All the experiments were approved by the Experimental Animal Committee of Ningxia Medical University (Certificate No. SCXK (Ning) 2015-0001). All the rats were housed under specific conditions (12 h light-dark cycles, 20°C–24°C) and had access to food and water.
Methods
Preparation of Isolated Rat Pulmonary Artery Rings
Male SD rats (n = 6) were positioned in the supine position on the operating table and intraperitoneally injected with a 20% urethane solution (100 mg/kg). After the rats were anesthetized, the pulmonary artery was removed. The left upper lobe pulmonary artery and its branches were removed. The secondary branches of the left upper lobe pulmonary artery (about 1–1.5 mm in diameter) were cut into 2–4 vascular rings about 3 mm in length and placed in Krebs salt solution at 37°C. Do not pull too hard during the operation to avoid vasoactivity damage. The isolated rat pulmonary vascular ring was fixed in a bath with a volume of 10 mL, and the mixed gas (95% O2 + 5% CO2) was continuously injected into the bath. The tension load of the vascular ring was adjusted to 1 g. The temperature was maintained at 37°. For 30 min, Krebs and Celsius solutions were swapped out every 15 min. The responsiveness of pulmonary artery rings to vasoactive substances was measured in vitro by precontracting them with 70 mmol/L KCl. Pulmonary vascular rings were precontracted with 1 µmol/L PE, and vascular endothelial integrity was detected by adding acetylcholine (10 µmol/L) after 15 min of stable vasoconstriction. The endothelium was considered intact when the diastolic amplitude exceeded 85%.
Pulmonary Arterial Ring Tensions at Rest
Rat plumomatic artery rings that were isolated and given cumulative doses of 18β-GA (10−4 mol/L,1.76 × 10−4 mol/L,3.0 × 10−4 mol/L,5.6 × 10−4 mol/L,10−3 mol/L) were found to be endothelial and fully functioning. Two groups—one receiving the medication and one receiving a control—were created from the isolated pulmonary artery vascular rings of rats. The pulmonary vascular ring was stabilized with PE and KCl, and the control group received the same volume of 18β-GA. The diastolic rate was computed once the tension shift was noted.
Effect of K+ Channel on the Vasorelaxation Due to Betaine in Isolated Rat Pulmonary Artery Rings
To explore the vasorelaxation effect of K+ channel on 18β-GA, the pulmonary vascular rings were incubated with BaCl2 (1 mmol/L), TEA (10 mmol/L), 4-AP (5 mmol/L), and Gli (10 µmol/L) for 20 min after precontraction with PE (1µmol/L). After stable vasoconstriction was achieved, 18β-GA or K-H solution was added cumulatively.
Effects of L-NAME and INDO on Vasorelaxation Due to 18β-GA in Isolated Rat Pulmonary Artery Rings
The isolated pulmonary vascular rings from the rats were separated into a treatment group and a control group after being stabilized for 1 h in a 37°C K-H solution. For the drug group, rings were stimulated with L-NAME (0.1 mmol/L) and INDO (10 µmol/L) for 20 min, and PE was applied to precontract the rings. After stable vasoconstriction was reached, 18β-GA or K-H solution was added cumulatively. The vasodilation rate was calculated for each group based on the maximum constriction amplitude (100%) induced by PE or KCl after drug administration.
Statistical Analyses
The data were analyzed using SPSS 24.0 statistical software and presented as mean ± SEM. The independent sample t-test was used to compare two samples. In all statistical tests, p < 0.05 was considered statistically significant.
Results
Vasodilatory Effects of Cumulative Concentrations of 18β-GA on Isolated Pulmonary Artery Vascular Rings in Normal Rats
Comparing normal rats to the control group, cumulative doses of 18β-GA had no discernible impact on isolated pulmonary arterial rings (Figure 1).
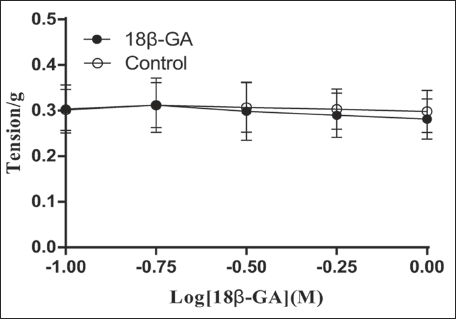
Effects of Cumulative Concentrations of 18β-GA on Isolated Pulmonary Artery Vascular Rings in Normal Rats With PE or KCl Precontraction
In this study, isolated rat pulmonary vascular rings that have been precontracted with PE and KCl exhibit a concentration-dependent vasodilatory response to 18β-GA. The results showed that 18β-GA had a significant vasodilatory effect on normal pulmonary arterial rings preconstricted by PE and KCl in the concentration range of 10−4 to 10−3 mol/L. When the cumulative concentration was at its maximum, the vasodilatory effects of 18β-GA were most noticeable. There are significant differences in the control group and the results were statistically significant (p < 0.05 or p < 0.01, Figure 2A and B).
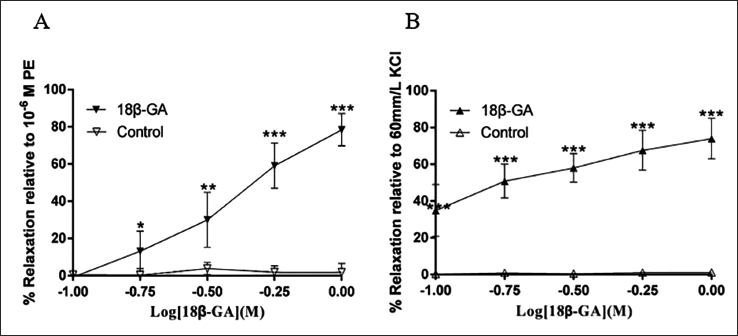
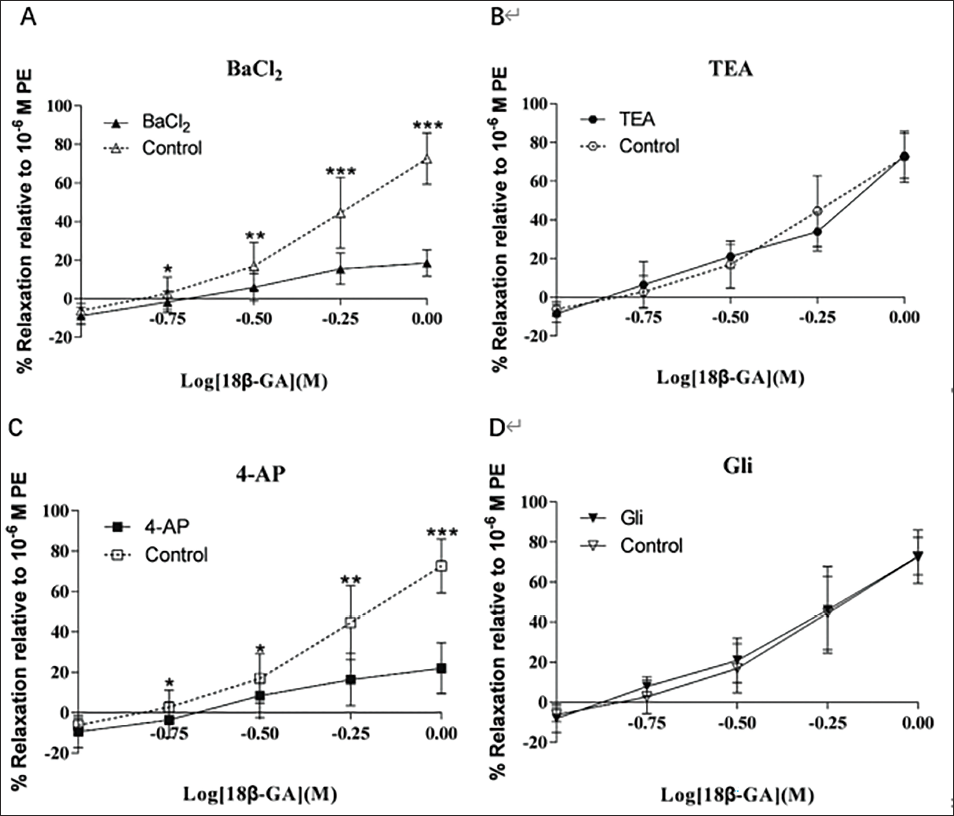
Effects of K+ Channels on 18β-GA-induced Relaxation in the Isolated Rat Pulmonary Artery Vascular Rings
In pulmonary artery vascular rings, the inward rectifier potassium (Kir) channel inhibitor BaCl2 and the voltage-operated potassium (Kv) channel inhibitor 4-AP inhibited 18β-GA-induced vasodilation (p < 0.05 and p < 0.01, Figure 3A and B). In contrast, the potassium channel inhibitor tetraethylammonium (TEA), which activates KCa by Ca2+, and the ATP-sensitive potassium (KATP) channel inhibitor Gli had no significant effect on 18β-GA-induced vasodilation (p > 0.05, Figure 3C and D). These findings imply that Gli and TEA had no impact on the vasodilatory action of 18β-GA on the pulmonary artery vascular rings.
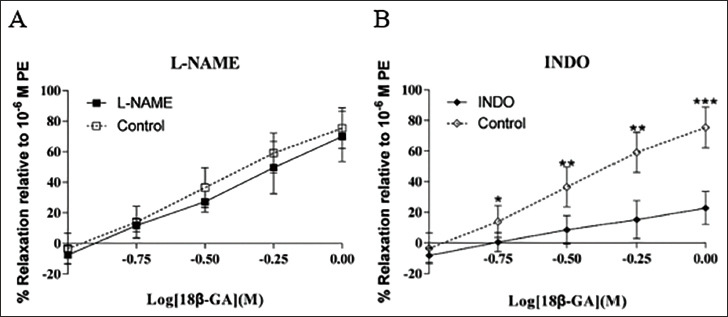
Vasorelaxation Effects of L-NAME and INDO on 18β-GA in the Isolated Pulmonary Artery Vascular Rings
Compared with the 18β-GA group, L-NAME did not inhibit the diastolic effect of 18β-GA on rat-isolated pulmonary vascular rings (p > 0.05, Figure 4A), indicating that the diastolic effect of 18β-GA on pulmonary vascular rings was not affected by L-NAME. Compared with the 18β-GA group, the diastolic effect of 18β-GA on pulmonary vascular rings in the INDO group was significantly inhibited (p > 0.05, Figure 4B), suggesting INDO has an effect on the relaxation of 18β-GA pulmonary vascular rings.
Discussion
In China, licorice (Glycyrrhiza uralensis Fisch) is one of the most popular herbal remedies. According to studies (Kwon et al., 2020; Li et al., 2019; Petramfar et al., 2020). Radix glycyrrhizae offers anti-inflammatory, antioxidant, anticancer, and other pharmacological properties with fewer adverse effects. 18β-GA is the main active ingredient of Radix glycyrrhizae. It is a pentacyclic triterpenoid compound formed by glycyrrheic acid releasing the disaccharide chain. It has anti-allergy, anti-inflammation, antivirus, antioxidation, and other pharmacological effects (Hasan et al., 2015; Parida et al., 2014). It has been found that 18β-GA has therapeutic effect on PAH in rats (Zhang et al., 2019), but its therapeutic effect on pulmonary artery and its specific mechanism remains unclear. To determine if 18β-GA has diastolic effects on isolated pulmonary vascular rings in rats and its potential mechanism, this study set out to answer such questions.
Blood vessel components known as vascular smooth muscle cells (VSMCs) are crucial for maintaining healthy vascular homeostasis (Hwang & Cho, 2020). According to Hungerford and Little (Hungerford & Little, 1999; Yang & Hori, 2021), atherosclerosis and PAH are two vascular disorders that can develop in response to changes in the contractility of VSMCs. PE mainly acts on the α1 adrenergic receptor of VSMCs to activate the phospholipase C signaling pathway. In addition, it activates receptor-dependent calcium channels and voltage-dependent calcium channels (VOCC) on the cell membrane. At the same time, it increases the uptake of extracellular calcium and stimulates the release of intracellular calcium, which leads to vasoconstriction (Lee et al., 2021; Liao et al., 2021). KCl activates voltage-dependent calcium channels (VDCC) by depolarizing smooth muscle cells which opens calcium channels and allows extracellular calcium to flow in, thereby increasing intracellular calcium concentrations and leading to vasoconstriction (Akata, 2007). Vascular contraction occurs even though PE and KCl have different contractile mechanisms. In this study, PE and KCl precontracted rats’ isolated pulmonary artery rings respond to 18β-GA in a concentration-dependent manner by vasodilating.
Potassium channels are known to play an important role in the regulation of vascular contraction and vasodility (Guntur et al., 2021). Changes in the activity of potassium channels can lead to changes in cell membrane potential, thereby inhibiting extracellular calcium influx and causing vasodilation. There are four kinds of potassium channels in vascular smooth muscle, namely ATP-sensitive potassium channel (KATP), inward rectifier potassium channel (Kir), Ca2+ activated potassium channel (KCa), and voltage-sensitive potassium channel (Kv) (Zhang et al., 2021; Zingman et al., 2007). TEA and BaCl2 are KCa and Kir blockers, respectively. Kv and KATP blockers are 4-AP and Gli, respectively. After the vascular ring was incubated with the four potassium-channel blockers, TEA and Gli were unable to stop the vasodilation brought on by 18β-GA. However, 4-AP and BaCl2 suppressed the vasodilatory effects of 18β-GA, indicating that these effects may be connected to the voltage-sensitive potassium channel (Kv) and the inward recirculating potassium channel (Kir).
It is well known that vascular endothelium plays an important role in the contraction and relaxation of blood vessels. Vascular endothelium mainly regulates the structure and function of blood vessels through endocrine and paracrine activities (Kusayanagi et al., 2015; Mitchell et al., 2008). Healthy endothelial cells release a variety of substances that relax blood vessels, mainly represented by nitrogen monoxide (NO) and prostaglandin-I-2(PGI2) (Esmaeili et al., 2017; Skogastierna et al., 2011). Numerous physiological homeostasis mechanisms in the body involve nitric oxide. Vascular endothelial cells can activate guanylate cyclase by secreting NO, which boosts the body’s production of cGMP. The decrease of calcium ions in the cytoplasm of vascular smooth muscles promotes myosin dephosphorylation, which leads to relaxation of VSMCs, vasodilation, and lower blood pressure (Koress et al., 2016). PGI2 is the endothelial vasodilation mediator and is the main product of arachidonic acid. Cyclooxygenase-2 (COX-2) and PGI2 synthase work together to create it. According to studies, patients with PAH had less PGI2 expression in their pulmonary artery endothelial cells. PGI2 is regarded as having a preventive impact in vascular disorders as it is a potent vasodilator of vascular smooth muscle that can decrease platelet aggregation. In the body, PGI2 acts on the corresponding IP receptors by activating adenylate cyclase and increasing the content of cAMP, which causes a vasodilation response (Huang et al., 2015; Liu & Zhou, 2021; Sugita et al., 2016). INDO is a Cyclooxygenase (Cox) inhibitor. INDO acts on Cox and inhibits the release of PGI2 from the endothelium in the intact vascular rings (Kasznicki & Wiktorowska-Owczarek, 2001). In the experiment, we noticed that the vasodilation effect of 18β-GA was diminished when the vascular ring was incubated with INDO (a cyclooxygenase inhibitor), which suggests that the vasodilation effect of 18β-GA is connected to the release of PGI2 and that its vasodilatory effect may be endothelium-dependent.
Gap junctions play an important role in vascular regulation, transmitting vasodilatory signals between endothelial cells and smooth muscle cells, both of which are critical for the integrity of gap junctions (Figueroa & Duling, 2009; Segal, 2005). While slit junctions play an important role in the vasoconstrictor response, application of slit blockers can inhibit acute and chronic cerebral vasospasm after subarachnoid hemorrhage in rabbits (Hong et al., 2008). In previous studies, 18β-GA was used as a potent gap junction blocker in the vasculature (Gupta et al., 2008). Through the use of 18β-GA, a gap linkage blocker, pathological changes brought on by the transmission of harmful information from disturbed cells to normal cells through gap links can be inhibited in disease conditions. Therefore, we hypothesize that 18β-GA, through inhibiting gap junction proteins, may also play a significant role in the aberrant constriction of pulmonary blood vessels.
Our previous study found that 18β-GA has some therapeutic effects on rats with MCT-induced PAH (Zhang et al., 2019, 2019), and some diastolic effects on isolated rat thoracic aortic vascular rings (Zhang et al., 2019). Consequently, we verified the diastolic effects and mechanism of 18β-GA on the pulmonary vascular in rats by using isolated rat pulmonary vascular rings. One of the effective modalities for the treatment of PAH is to dilate the blood vessels to improve Hemodynamics (Hu et al., 2022). This study confirmed that 18β-GA had vasodilatory effects on isolated rat pulmonary vascular rings, which may provide a basis for the further study of 18β-GA and showed that 18β-GA could treat PAH through vasodilation.
Conclusion
The findings of this study demonstrate that 18β-GA relaxes the pulmonary artery vascular ring in PE and KCl preconstricted rats in a concentration-dependent manner, and the mechanism of vasodilation may include Kv and Kir. This study has some limitations. The effect of 18β-GA on the isolated pulmonary vascular rings of normal rats was studied in vitro, but it cannot completely reflect the state of PAH. Nevertheless, this study may provide further experimental and theoretical basis for the follow-up study of 18β-GA in the treatment of PAH, and further studies are still needed to determine the mechanism of the action of 18β-GA in the treatment of PAH.
Abbreviations
4-AP: 4-aminopyridine; Indo: Indomethacin; KCl: Potassium chloride; L-NAME:
Authors’ Contributions
All authors contributed to the study conception and design. Huijuan Yang, Cong Li, and Jinrui Jiang perform a literature search. Ru Zhou, Zhiqiang Hu carried out project management. Huijuan Yang, Yuxin Liu, and Mei Hai, jointly completed the experimental research and data collection. Originals processed by Yuxin Liu draft. Ru Zhou and Huijuan Yang perform writing original article and editing. All authors commented on the final draft and approved the draft.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
All procedures for the animal study were reviewed and approved by the Ningxia Medical University, Yinchuan, Ningxia, China. All the experiments were approved by the Experimental Animal Committee of Ningxia Medical University (Certificate No. SCXK (Ning) 2015-0001).
Funding
This research was supported by the 2017 Ningxia Hui Autonomous Region Science and Technology Innovation Leader Training Project (Grant No. KJT2017005), the Ningxia Hui Autonomous Region Key Research and Development Project (Grant No. 2019BFG02027), the 2019 College Students’ Innovation and Entrepreneurship Training Program (No.201910752029).
Human and Animal Rights
In this study, no humans were employed. All animal research methods complied with the guidelines outlined in the eighth edition of Guide for the Care and Use of Laboratory Animals published by the National Academy of Sciences, the National Academies Press, Washington, D.C.
