Abstract
Background
Cryptotanshinone (CTS) is a monomer possessing a diverse array of pharmacological properties, primarily derived from Salvia miltiorrhiza. CTS was examined for its impact on rats with depression subjected to chronic unpredictable mild stress (CUMS) in this study.
Materials and Methods
Depression-like behavior of rats was established by isolated feeding combined with CUMS for 28 days, with CTS (7/14/21 mg/kg) or fluoxetine (5 mg/kg) administered once daily during this time. A comprehensive evaluation was performed, including body weight assessment, sucrose preference test, forced swimming test, and open field test. Hematoxylin-eosin staining was conducted to detect pathological changes in hippocampal CA1 neurons. Western blot analysis was employed to assess the brain-derived neurotrophic factor (BDNF), tyrosine kinase receptor B (TrkB), Ras-related C3 botulinum toxin substrate 1 (Rac1), and Cofilin expression levels in the hippocampus.
Results
The depression rat model was successfully constructed. CTS can significantly improve impaired neurons in the hippocampal CA1 area, depression-like behaviors, and weight loss in CUMS-induced rats. Moreover, CTS reversed the down-regulation of BDNF/TrkB and Rac1/Cofilin in the CUMS-induced rats’ hippocampus.
Conclusion
CTS demonstrates the therapeutic potential for depression by improving depression-like behaviors and ameliorating impaired neurons in the hippocampal CA1 area, and the mechanisms may involve the up-regulation of BDNF/TrkB and related signaling proteins Rac1/Cofilin.
Introduction
Depression is a prevalent clinical psychiatric disorder distinguished by its widespread occurrence, high rates of recurrence, suicide, and disability, impacting over 300 million individuals globally. The global incidence of depression experienced a notable 18.4% rise from 2005 to 2015. Southeast Asia and the Western Pacific are the regions with the highest number of cases, with 50% of patients living in these two regions. Dense populations in the region and overwork caused by stressful lives are important reasons for the concentration of people with depression (WHO, 2017). Patients with depression suffer from depressed mood or impaired thinking and cognitive abilities (Zhang et al., 2022). Pharmacotherapy is the primary treatment of depression. Traditionally, antidepressants commonly used in clinical practice include drugs that inhibit serotonin reuptake, norepinephrine reuptake, monoamine oxidase, and tricyclic antidepressants which can effectively alleviate depression but have long treatment cycle, many adverse effects and low efficacy (He et al., 2022; Trivedi et al., 2006). Cryptotanshinone (CTS), a biologically active quinone compound derived from Salvia miltiorrhiza, has been the subject of extensive research as a pharmacologically active compound, showing its effectiveness in treating a variety of illnesses and disorders, including anti-cancer, visceral protective, cardioprotective, anti-metabolic disorders, neuroprotective, and anti-fibrosis activities (Li et al., 2021; Wu et al., 2020). CTS is widely used in Asian countries to treat cardiovascular diseases, and it has been found to be helpful in treating central nervous system disorders, such as Alzheimer’s disease (Maione et al., 2018). Recently, we investigated the potent activity of CTS in mice with different depression models induced by various drugs such as corticosterone, reserpine, and 5-HTP (Chen, Chen, et al., 2021; Chen, Liao, et al., 2021). Moreover, CTS is also effective in chronic unpredictable mild stress (CUMS)-induced depression mice (Wang et al., 2021). However, the positive control group is lacking, and the dose-dependent effects are still needed to be elucidated. Besides, the impact of CTS on body weight, the morphology of the hippocampal CA1 area, and Racl and Cofilin levels in hippocampal tissue remained ambiguous.
The neurotrophic hypothesis based on the brain-derived neurotrophic factor (BDNF), a crucial indicator in understanding depression and researching antidepressant drugs, has received increasing attention, thereby promoting neuron growth, development, repair, and regeneration in the brain after injury (He et al., 2022; Meshkat et al., 2022).
Therefore, our research created a rat depression model using CUMS and solitary feeding, and then different doses of CTS were administrated, and treatment duration was prolonged to observe the effects of CTS on behavioral and hippocampal tissues. In order to enhance comprehension of its mechanism of action, we assessed the expression of proteins relevant to the BDNF/tyrosine kinase receptor B (TrkB) pathway in the hippocampus, thereby offering a theory-based foundation for developing innovative anti-depressant medications.
Materials and Methods
Animal Administration
The tests were conducted using male Sprague-Dawley rats of SPF-grade quality (Zhejiang Vital River Laboratory Animal Technology Co., Ltd., America, License No. SCXK (Zhe) 2019-0001), weighing 100–120 g. The rodents were kept in a controlled environment at 25°C ± 2°C and 60% ± 10% humidity, meanwhile subjected to 12-hour light and dark cycles. They had unrestricted access to food and water. Experiments were carried out in compliance with global animal regulations and were sanctioned by the Ethical Review Committee of Quanzhou Medical College (approval number: 2021006). Each rat was utilized in a single experiment and euthanized upon completion of the study.
Drug Administration and Animal Grouping
CTS (Shanghai Yuanye Bio-Technology Co., Ltd.) and fluoxetine (FLX) capsule (Eli Lilly and Company, Ltd.) as positive control were dissolved in sterile normal saline prior to use. Six groups of 10 rats each were randomly selected from 60 rats: Naive (normal control + saline), depression model (solitary + CUMS + saline), treatment (solitary + CUMS + CTS 7/14/21 mg/kg), positive control (solitary + CUMS + FLX) groups. A amount of 7/14/21 mg/kg of CTS was administered orally by gastric gavage, while 5 mg/kg of FLX or normal saline was administered. The appropriate drug was given to each group once daily for 28 days starting from CUMS. The body weight of all groups was recorded once a week. The naive group was group-housed and did not receive any stimulation during the experiment. The remaining groups of rats were held in a single cage (solitary) and subjected to CUMS for 28 consecutive days. CUMS was conducted and improved according to previous studies (Lei et al., 2022). The mild stress included clip tail for 1 minute, tilting the cage at 45° for 24 hours, cold water (4°C) and hot water (45°C) swimming for 5 minutes each, water deprivation, and fasting for 24 hours, horizontal shaking for 10 minutes, wet cage housing for 24 hours, mothball stimulation for 12 hours, unfamiliar objects for 24 hours, and reversal of day and night. To ensure the unpredictability of the experiment, rats were subjected to one to two times randomly scheduled, mild stressors that were not repeated. Upon completion of the CUMS protocol, the sucrose preference test, forced swimming test (FST), and open field test (OFT) proceeded sequentially. After behavioral evaluations, their brains were immediately harvested for subsequent experiments.
Sucrose Preference Test
In the adaptation phase, all rodents were provided with 1% sucrose solution in two bottles for one day, followed by replacing two bottles (1% sucrose solution vs. water) for one day and deprived of food and water on the third day. During the test, each cage received two bottles (1% sucrose solution vs. water). All rodents were provided unrestricted access to drinks for 2 hours. The sucrose preference index was calculated by weighing the sucrose and water bottles before and after the test. The formula is as follows: Preference for sucrose (%) = consumption of sucrose solution/consumption of sucrose solution and water × 100%.
Forced Swimming Test
During the test, rodents were plunged individually into a water-filled cylindrical vessel made of transparent glass. The diameter of the forced swimming bucket is 40 cm and the height is 50 cm. The water was 30 cm deep and 22°C. The test lasted a total of 6 minutes, during which the initial 2 minutes were for getting used to the environment, followed by 4 minutes of immobility.
Open Field Test
During testing, rodents were put in the central region of the open field box (100 × 100 × 40 cm) from the same position under quiet conditions and moved freely for 5 minutes. The opaque box is divided into a 60 × 60 cm central area and the surrounding border zone. After each rat finished the experiment, alcohol was used to wipe the bottom of the box to remove residual odors that interfered with the rats’ behavior. Duration and bouts for the animals to stay within the center grid during the last 3 minutes were recorded and analyzed.
Hematoxylin-Eosin (HE) Staining
Deeply anesthetized rodents were first treated with transcardial perfusion with phosphate buffer solution and subsequently fixed in 4% paraformaldehyde (PFA). The left and right hippocampi were rapidly taken out from rats and fixed at 4°C overnight with 4% PFA, dehydrated stepwise in increasing concentrations of ethanol, embedded in paraffin, and sliced (4 µm thick). The morphological features of the neurons in the hippocampal CA1 area were assessed by HE staining, and the hippocampal slices were observed under a microscope (Olympus, Tokyo, Japan). Other parts of hippocampus sections were independently processed for western blotting analysis.
Western Blotting Analysis
The samples were lysed with a homogenizer and the lysate was incubated on ice for 30 minutes, then centrifuged at 4°C for 5 minutes at 10,000g. The resulting supernatant was used for immunoblot analysis. In equal amounts, protein samples were loaded into 12% SDS-PAGE gels, separated, and subsequently transferred onto a PVDF membrane (Millipore). The membranes were first incubated with 5% skim milk powder (w/v) at room temperature for 1 hour, followed by overnight incubation with the following antibodies at 4°C: anti-BDNF (GR3217060-6, Beyotime, China), anti-TrkB (GR3280550-1, Beyotime, China), anti-Cofilin (CT7017A, Beyotime, China), anti-Ras-related C3 botulinum toxin substrate (Rac1) (LA77201, Beyotime, China), anti-β-actin (ab6276, Abcam, UK). After that, the membranes were treated with a HRP-labeled secondary antibody for a duration of 2 hours at ambient temperature, prior to being washed and exposed to ECL solution (Pierce) to induce chemiluminescence. Protein bands were visualized on X-ray film and analyzed with ImageJ software.
Statistical Analysis
GraphPad Prism 9.3.0 was used to analyze the data, which are shown as mean ± standard error of measurement and analyzed through the application of one-way analysis of variance (ANOVA) with the Dunnett post hoc tests to compare multiple datasets. Statistical significance was determined at p < 0.05.
Results
CTS Reversed the Weight Loss of the Depression Model Rats
As demonstrated in Figure 1, during the observation period, each group of rats gained weight gradually, with the naive group experiencing the greatest growth in body weight and the depression model rats experiencing the least. After being subjected to CUMS and administered (28th day), the model group demonstrated a statistically significant reduction in body weight (p < 0.01 vs. naive). CTS (14, 21 mg/kg) and FLX (5 mg/kg) significantly reversed body weight (p < 0.01, p < 0.05 vs. depression model).
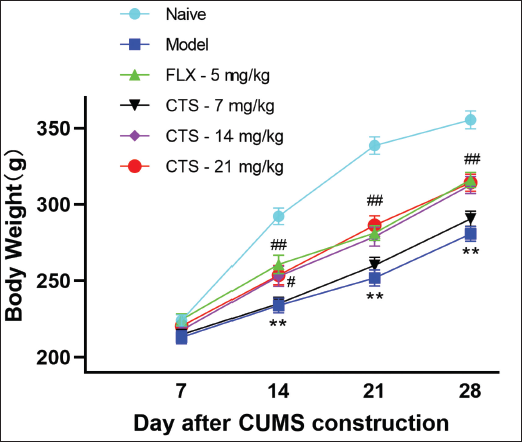
CTS Attenuated Depressive-like Behaviors
According to our study, CTS can significantly improve sucrose preference in depression-related behaviors. Rodents subjected to CUMS demonstrated a notable reduction in sucrose preference (p < 0.01 vs. naive; Figure 2a). All doses (7, 14, 21 mg/kg) of CTS and FLX reversed the CUMS-induced decline in sucrose preference (p < 0.05, p < 0.01 vs. depression model; Figure 2a).
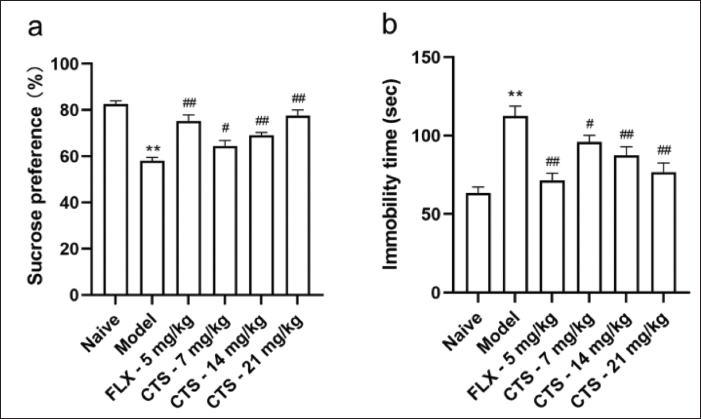
Furthermore, rodents exposed to CUMS exhibited depression-like behaviors, as evidenced by the prolonged FST immobility time (p < 0.01 vs. naive; Figure 2b). The immobility time was significantly reduced by CTS and FLX as demonstrated in Figure 2b (p < 0.05, p < 0.01 vs. model), suggesting that CTS could improve behavioral despair induced by CUMS.
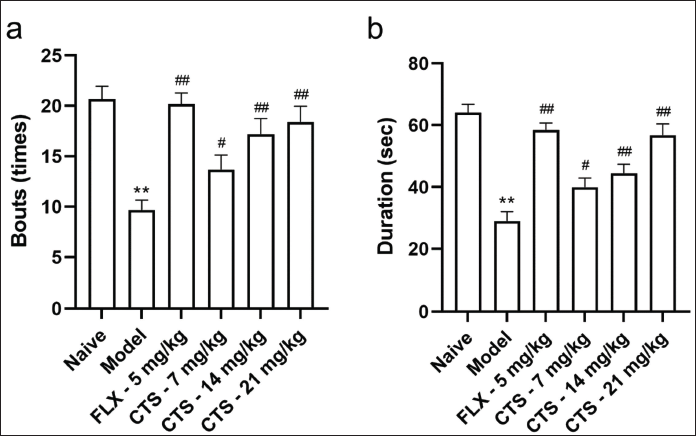
For anxiety-like behaviors (OFT), the model group rats had fewer entries (Figure 3a) and a shorter stay (Figure 3b) in the central zone (p < 0.01 vs. naive). As shown in Figure 3, after administrating, duration and bouts for the animals to stay within the center grid were found to be significantly increased in the groups administered with FLX and CTS at varying doses (7, 14, 21 mg/kg) (p < 0.05, p < 0.01 vs. model).
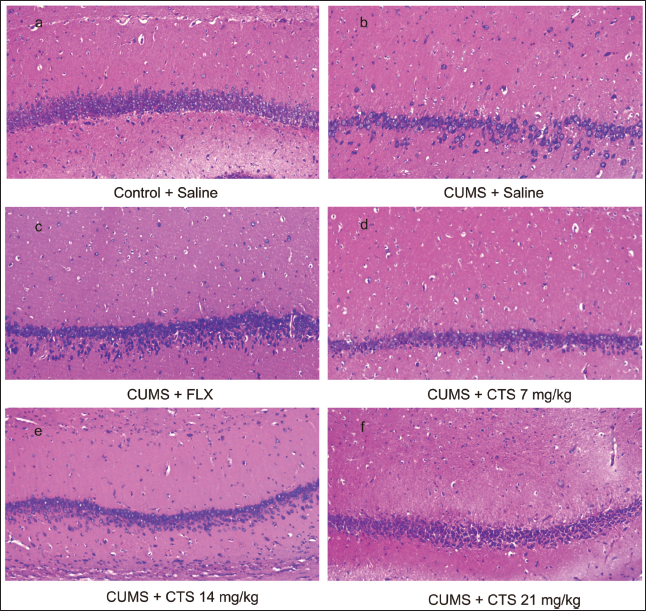
CPT Rescued Impaired Neurons in Hippocampal CA1 Area
Three rats from each group had their hippocampus randomly selected for an immunohistochemical experiment with HE staining to assess the impact of CTS on the morphology of the hippocampal CA1 area. Figure 4a illustrates that the neurons in the hippocampal CA1 region of the healthy control rats had normal morphology, neat arrangement, well-defined hierarchy, large and round nuclei, and clear nucleoli. The model group’s hippocampal CA1 area had a significantly reduced number of neurons, a disordered hierarchy, irregular morphology, and fixed and shrunken nuclei (Figure 4b). These pathological changes in rat hippocampal CA1 neurons were significantly ameliorated by FLX (Figure 4c) and high-dose (21 mg/kg) CTS (Figure 4f) and only mildly ameliorated by lower doses (7, 14 mg/kg) of CTS (Figure 4d and e).
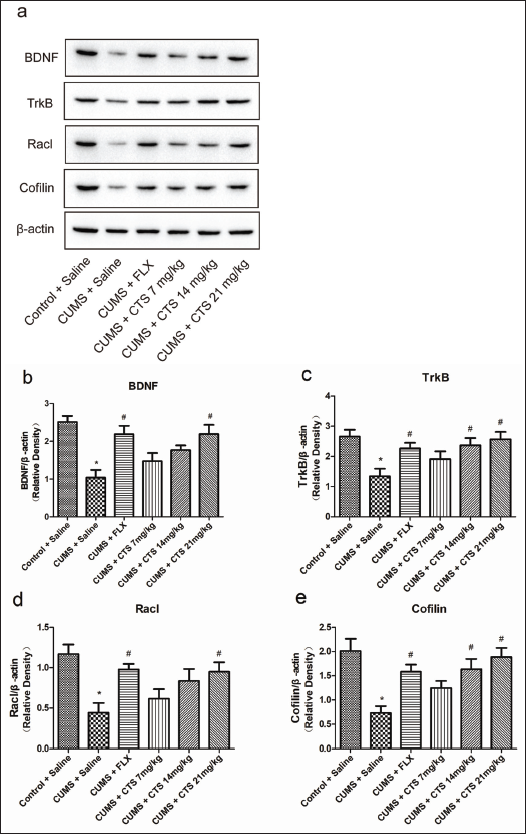
CTS Impact on BDNF/TrkB Pathway Protein Levels in Rat Hippocampus
After treatment with high-dose (21 mg/kg) CTS and FLX (p < 0.05 vs. model; Figure 5b–e), the BDNF, TrkB, Racl, and Cofilin levels in the hippocampal were significantly increased. The results verified that CUMS significantly reduced these protein levels. However, medium-dose (14 mg/kg) CTS restores only TrkB and Cofilin (p < 0.05 vs. model; Figure 5c and e), not BDNF and Racl, and none of the lower doses (7 mg/kg).
Discussion
Rodents exposed to CUMS can simulate the behavioral changes, and pathophysiological states of depression triggered by various chronic life stresses in humans. The animals exhibit typical symptoms of depression, such as lack of pleasure and interest, and behavioral despair, which are closest to the clinical development of depression. This animal model is frequently utilized in research to investigate the etiology of depression and for drug screening purposes (Antoniuk et al., 2019). In our study, we combined this method with solitary to induce a rat depression model and evaluate CTS’ antidepressant potential. CTS is a natural compound mainly in S. miltiorrhiza which is an important medicinal plant in China (Li et al., 2021). The results demonstrated that after being subjected to CUMS (28th day), the model group had slower body weight gain, longer time of forced swimming immobility, decreased sucrose water preference, shorter duration staying in the central zone of the opaque box, and less central zone bouts, which were reversed significantly after the long-term administration of CTS, and that high dose (21 mg/kg) of CTS were close to the positive control FLX in improving the above depression-like behaviors. It indicates that CTS has antidepressant activity.
The hippocampus is a key brain region for emotion regulation, memory, learning, and neurofactor secretion. It is sensitive to chronic stress and easily damaged; damage to this region is closely related to depression occurrence and development (Liao et al., 2021). Recently, the
[h]ippocampal neuronal regeneration disorder hypothesis suggests that altered neuronal plasticity can cause depression, as evidenced by decreased nerves in the brain or atrophy of related important brain regions and impaired intra-neuronal signaling pathways (Duman & Aghajanian, 2012; Serafini et al., 2014). Chronic stress causes abnormal functional morphological changes in the hippocampus, such as volume reduction, neuronal loss or circuit disruption, reduced neurogenesis, and dendritic retraction (Boldrini et al., 2013). Among them, hippocampal neuronal damage in CA1 and CA3 regions is the main pathological alteration in depression (Liang et al., 2022), while antidepressant treatment increases hippocampal tissue volume in depressed patients, repairs neuronal damage, and gradually restores normal morphology (Zhou et al., 2020). This experiment indicated that neuronal damage occurred in the CUMS-induced rats’
CA1 hippocampus, presenting decreased neuronal cells, scattered distribution, and nuclear consolidation. This result was consistent with the literature, and the long-term administration of CTS could reverse the neuronal damage in this region, exhibiting a uniform distribution of neuronal cells, an absence of neuronal cells, and decreased crumpled necrotic cells.
Numerous studies have demonstrated that BDNF plays a crucial role in regulating synaptic plasticity, with the highest levels found in the cortex and hippocampus, regulating dendritic and axonal growth, synaptic transmission, and long-range enhancement in the hippocampus. BDNF treatment increases neuronal dendritic spine density and promotes its regeneration (Leal et al., 2017). BDNF expression is down-regulated, accelerating neuronal apoptosis, decreasing synaptic plasticity, and causing neuronal atrophy, ultimately leading to depression (Sharma et al., 2017). However, long-term antidepressant administration normalizes BDNF expression in the hippocampus (Han et al., 2020). BDNF-specific receptor TrkB binds to it, further dimerizes and phosphorylates tyrosine residues, triggering intracellular cascade reactions and activating downstream signaling pathways (Tacke et al., 2022), causing up-regulation of synaptic-related protein expression and promoting synaptogenesis (Zhang et al., 2016).
Numerous anti-depressants can play a therapeutic role by targeting TrkB and then activating the BDNF/TrkB pathway, with TrkB in precursor cells being the target of BDNF released from dentate granule neurons. After the movement of TrkB from dentate granule neuron precursor cells, the antidepressant effects of drugs can be attenuated. However, it does not occur when BDNF is removed. The dimerized TrkB transmembrane domains cross each other in the transmembrane region to create a pocket for antidepressants. Different classes of antidepressants, such as FLX, promethazine, and ketamine, can bind directly to TrkB, facilitating the translocation of TrkB to the plasma membrane and the retention of the cytoplasm. BDNF released from active synapses can bind to TrkB on the cell surface and trigger the activation of the BDNF/TrkB signaling pathway. However, the TrkB receptors at inactive synapses can remain in an inactive state since no BDNF can be released. The conformation of the TrkB dimer depends on the membrane thickness, which is associated with cholesterol concentration, and TrkB at intermediate cholesterol concentrations appears to be more conducive for the pathway activation. However, the affinity of antidepressants for TrkB is much lower than their affinity for the 5-hydroxytryptamine transporter, and it always needs several weeks of dose accumulation before achieving the required concentration of chemotherapeutic drugs in the brain. Therefore, typical antidepressants tend to have a slow onset of action (Castren & Monteggia, 2021).
Antidepressants can ameliorate depressive symptoms in animal models through the modulation of the BDNF/TrkB pathway (Chen, Liu, et al., 2021; Rafalo-Ulinska et al., 2022; Wu et al., 2023), reducing neuronal apoptosis, regulating synaptic plasticity, and repairing damaged neurons (Liu et al., 2023). In our investigation, it was observed that CUMS resulted in a reduction of BDNF and TrkB levels in the rat hippocampus, while subsequent CTS treatment resulted in an increase in these levels. This suggests that the upregulation of BDNF and TrkB may be integral to the antidepressant mechanisms of action associated with CTS.
The Rac1-Cofilin pathway downstream of BDNF/TrkB may be involved in depression development. Several antidepressant medications may work by regulating this pathway (Adu-Nti et al., 2021; Lu et al., 2019). BDNF interacts with TrkB, leading to TrkB phosphorylation and subsequent activation of Rac1 (Sasaki et al., 2021) and LIM kinase 1, thereby modulating Cofilin phosphorylation (Lu et al., 2019), contributing to neural cytoskeleton remodeling, improving neuroplasticity and participating in postsynaptic and membrane transport of AMPA receptors during synaptic tractability (Cao et al., 2017). In our research, it was observed that CUMS reduced the Rac1 and Cofilin levels within the rodent hippocampus, an effect that was subsequently reversed following CTS treatment. It indicates that solitary combined with CUMS decreased the Rac1 and Cofilin phosphorylation levels within the rodent hippocampus, and the activation of the downstream Rac1-Cofilin signaling pathway of BDNF/TrkB by CTS may be a potential reason for its amelioration of depression, which requires additional investigation.
Conclusion
This study demonstrates that CTS exhibited a notable antidepressant impact on rats induced with CUMS, as evidenced by improving depression-like behaviors and ameliorating impaired neurons in the hippocampal CA1 area. The mechanisms may involve the up-regulation of BDNF/TrkB and related signaling proteins Rac1/Cofilin. These results demonstrate CTS’ antidepressant-like effects and possible mechanism of action, which support that CTS might be a potential therapeutic drug for further development as an antidepressant agent.
Summary
CTS can ameliorate depressive symptoms, and weight loss in rats subjected to CUMS, and a high dose (21 mg/kg) of CTS after 28 days of continuous administration improved depression-like behaviors similar to the positive control FLX. Meanwhile, CTS can significantly ameliorate impaired neurons in the hippocampal CA1 region of rats exposed to CUMS. Further, CTS can reverse the down-regulation of BDNF/TrkB and downstream proteins Rac1/Cofilin in the CUMS-induced rats’ hippocampus.
Abbreviations
BDNF: Brain-derived neurotrophic factor; CTS: Cryptotanshinone; CUMS: Chronic unpredictable mild stress; FLX: Fluoxetine; FST: Forced swimming test; HE: Hematoxylin-eosin; OFT: Open field test; Rac1: Ras-related C3 botulinum toxin substrate 1; TrkB: Tyrosine kinase receptor B.
Footnotes
Acknowledgments
The authors express their gratitude to the supporting staff at the Department of Pharmacy of Quanzhou Medical College, the Pharmaceutical Department of Quanzhou First Hospital (Affiliated to Fujian Medical University), the Pharmaceutical Department of Fujian Medical University Affiliated Second Hospital, for their technical assistance in this research project.
Declaration of Conflicting Interests
The authors have no conflicts of interest related to the research, authorship, or publication of this article.
Ethical Approval and Informed Consent
Ethical clearances and informed consent were obtained from all participants before starting the study.
Funding
Quanzhou City Science & Technology Program of China, Science and Technology Agency of Quanzhou City of Fujian Province (Grant number: (2021N130S); Fujian Educational and Scientific Research Project for Young and Middle-aged Teachers, Education of Fujian Province (Grant number: JAT171155; JAT220721); Quanzhou Medical College Research Project, Quanzhou Medical College (Grant number: XJJK2307; XJK1920B).
