Abstract
Background
Diabetes Mellitus (DM) is a syndrome that interferes with energy metabolism and is caused by a decrease and dysfunction of insulin, leading to chronic hyperglycemia. DM associated with dyslipidemia is a severe health risk, increasing the chance of cardiovascular events, such as acute myocardial infarction and stroke. The Cannabis sativa L. fixed oil (CSO) is composed of unsaturated fatty acids and can be crucial in treating metabolic alterations. In addition, the nanoemulsion of C. sativa oil (NCS) has advantages in optimizing treatments.
Objectives
This study aimed to evaluate the effects of treatments with CSO and its nanoemulsion (NCS) on induced diabetes and dyslipidemia in Wistar rats.
Materials and Methods
CSO’s physical-chemical and chromatographic characterization was performed, followed by the preparation of an NE containing 6% CSO. DM was induced in Wistar rats by intraperitoneal injection of streptozotocin (STZ) at a 55 mg/kg dose. Four days later, animals with blood glucose levels exceeding 300 mg/dL were considered diabetic. The rats were then divided into five groups (n = 5) and treated orally. The groups included a normoglycemic control group (NOR), a diabetic control group, a group treated with metformin (100 mg/kg), a group treated with CSO (400 mg/kg), and a group treated with nanoemulsion (NCS 200 mg/kg). Subsequently, the pancreas’s clinical, biochemical, and histopathological parameters were evaluated.
Results
In the chemical profile of CSO, it was observed the majority composition of palmitoleic (14.58%), oleic (12.50%), linoleic (42.40%), and linolenic (8.55%) acids. The results demonstrated that the induction of DM by STZ could reproduce the typical symptoms and clinical signs of DM. It was observed that treatments with CSO and NCS showed a significant improvement (p < 0.001) in polydipsia, polyuria, and loss of body mass, as well as a significant reduction (p < 0.001) of glucose levels in urine and blood and serum lipids. Histopathology of the pancreas revealed that treatment with CSO and NCS showed an increase in the number of cells in the islets of Langerhans and a decrease in regions devoid of cells, indicating possible cell regeneration. Moreover, insulin levels were significantly increased (p < 0.05) in the preferred groups. With dyslipidemia induced by Triton (Tyloxapol), it was observed that the treatment with CSO and NCS significantly decreased the levels of triglycerides (p < 0.05) and cholesterol (p < 0.001), as well as low-density lipoproteins (LDL) (p < 0.01).
Conclusion
Treatment with CSO and NCS under the conditions of this study demonstrated an anti-diabetic effect and the ability to act in the reduction of triglyceride, cholesterol, and LDL levels. In this respect, treatments with CSO and NCS act in the control of DM, as well as in the prevention of cardiovascular diseases.
Introduction
Diabetes mellitus (DM) is a syndrome that interferes with energy metabolism, caused by factors such as deficiency in insulin production and reduced tissue sensitivity to this hormone. Such factors lead to chronic hyperglycemia, characterized by high glucose levels in the bloodstream, which directly interferes with lipid metabolism, causing dyslipidemia, a condition in which changes in plasma lipid levels, such as cholesterol, triglycerides, and lipoproteins, are evidenced (de Oliveira Carvalho et al., 2021).
DM associated with dyslipidemia is a severe health risk. If not properly treated, it favors the installation of other co-morbidities, such as arterial hypertension and atherosclerosis, increasing the risk of cardiovascular events, mainly acute myocardial infarction, and stroke (Kimura et al., 2013). DM represents a significant public health problem. According to the International Diabetes Federation, it is estimated that more than 425 million people have DM worldwide, and there are perspectives that by 2045, DM will reach 629 million cases, with people aged between 20 and 79 years (Carvalho et al., 2018).
The increase in the prevalence of DM in the world makes this pathology the target of studies to seek new sources of treatments and contribute to reducing morbidity and mortality rates (de Oliveira Carvalho et al., 2016). In this regard, the use of medicinal plants in the preparation of medicines to treat DM and dyslipidemia is expressive. In addition, these natural resources can be safer, cheaper, and more effective when used to treat chronic diseases and associated with conventional medicines (Campos et al., 2016; Đurašević et al., 2020).
Cannabis sativa L. (CS) belongs to the Cannabinaceae family and is an herbaceous species known as “marijuana” and “hemp.” Its use is prohibited in several countries due to its capacity to produce psychoactive substances, mainly delta-9-tetrahydrocannabinol, a substance capable of causing physical and psychological dependence. When CS seeds are crushed or shredded and pressed, C. sativa L. fixed oil (CSO) is extracted. CSO does not have a psychoactive effect, and it is used as a food and culinary product in countries where the cultivation of CS is legal (Fiorini et al., 2019; Vonapartis et al., 2015).
The CSO is rich in unsaturated fatty acids (UFAs), consisting of about 80% of total fatty acids, including linoleic and linolenic acids, with proportions considered ideal for nutrition. Studies with UFAs focused on the action on glucose and lipid metabolism have demonstrated their properties in attenuating the diabetic condition and improving the lipid profile with cardioprotective properties and on the vascular endothelium (Kaushal et al., 2020; Souza et al., 2017).
Nanotechnology can be defined as the science and engineering involved in the design, synthesis, characterization, and application of materials and devices whose functional organization is at the nanometric scale. Remarkable advances were made with the help of this technology, including the development of a series of formulations with potential applications in food, cosmetics, medicines, and pesticides (Borthakur et al., 2016; Irache et al., 2011).
In the pharmaceutical field, nanoemulsions (NEs) are pharmaceutical forms produced by emulsification techniques containing dispersed systems of two immiscible liquids, often stabilized by surfactants. They comprise tiny droplets smaller than 200 nm (Aswathanarayan & Vittal, 2019). They have several advantages: greater stability, favorable organoleptic characteristics, greater penetration capacity through membranes, greater bioavailability, increased solubility in water for poorly soluble substances, controlled release, and increased therapeutic potency (Irache et al., 2011).
Given this context, applying nanotechnology to increment pharmaceutical forms containing CSO is a way to optimize its therapeutic effect. Therefore, it is necessary to conduct studies that seek to verify the pharmacological activities of medicinal plants and their formulations so that we can benefit from their therapeutic properties without the risk of health impairment. Thus, this study aimed to evaluate the effects of treatments with CSO and its nanoemulsion of C. sativa oil (NCS) on induced diabetes and dyslipidemia in Wistar rats.
Materials and Methods
Acquisition of CSO
The CSO (Lot 124563) was purchased from Just Hemp Foods (Niverville, Canada). According to the company’s quality certificate, the CSO was extracted by cold pressing the seeds of C. sativa.
Physical-chemical and Chemical Characterization of the CSO
The physical-chemical characterization of density, color, acid number (Cd3d-63), saponification index (Cd 1c-85), and iodine index (Cd 3a-94) was carried out according to the methodologies described in AOCS (2003).
The chemical composition of the CSO was analyzed by gas chromatography coupled with mass spectrometry. A 5 mL sample of CSO was esterified according to the methodology described by Rosset et al. (2013). It was injected into Shimadzu/GC 2010 equipment, Shimadzu/AOC-5000 auto-injector, and mass detector (Shimadzu MS2010 Plus) with electronic impact (70 eV) equipped with a DB-5MS fused silica column (Agilent Advanced J&W; 30 m × 0.25 mm × 0.25 µm). The parameters were as follows: split ratio 1:30; Helium as carrier gas (65 kPa); injection volume of 1.0 µL; injector temperature at 250°C; detector temperature, 250°C; initial column temperature, 60°C for 1 minute; heating rate, 3°C min−1 at 290°C. The total analysis time was 46.67 minutes. The compounds were identified through the fragmentation pattern compared with the mass spectrum library contained in the equipment (NIST 5.0).
Obtaining, Characterizing, and Stability of the CSO NE (NCS)
The NE was prepared using a low-energy methodology adapted from Gumiero and da Rocha Filho (2012) for a final mass of 20 g, 90% water, 6% CSO, and 4% polysorbate 80 (Tween) were used. Initially, a phase was prepared by adding CSO and surfactant. The mixture was stirred using a magnetic stirrer (800 rpm) for 30 minutes at 60°C. Then, the aqueous phase was added at a flow rate of 4.0 mL/min with stirring and constant temperature for 60 minutes. The stability of the NE was evaluated at 0, 7, and 30 days after preparation, using macroscopic analysis of color, visual aspect, phase separation, and sedimentation. Particle size and polydispersity index were evaluated according to the methodology previously described by Borges et al. (2017), physical-chemical tests (Bernardi et al., 2011), centrifugation, and thermal stress according to Gumiero and da Rocha Filho (2012). The NEs were kept at room temperature (25°C ± 2°C) in glass test tubes with screw caps during the analysis period.
Ethical Considerations and Animals Used
This project was submitted to the Animal Use Ethics Committee of the Federal University of Amapá, CEUA-UNIFAP, and was approved on August 28 under protocol number 015/2018.
In this study, male Wistar rats from the Multidisciplinary Center for Biological Research in the Area of Science in Laboratory Animals, CEMIB—University of Campinas, UNICAMP, were used at 21 days of age. During the study, the animals were housed in a polyethylene box with stainless steel bars measuring 60 cm × 50 cm × 22 cm. They were kept in an air-conditioned environment at the Drugs Research Laboratory of the Federal University of Amapá, where they remained for a period of acclimatization, grouped in groups of five animals per box, kept under controlled temperature conditions (21°C ± 0.5°C), with free access to food and potable water, with a 12-hour light/dark cycle and cage cleaning.
Induction and Experimental Design of DM
DM induction was performed in animals fasting for 16 hours by intraperitoneal (i.p.) injection of streptozotocin (STZ) (SIGMA-Aldrich Inc., St. Louis, MO, USA) dissolved in 0.01 M sodium citrate buffer (pH 4.5), with an administered dose of 55 mg/kg in a volume of 1 mL/kg of body weight. Four days after STZ injection, animals with blood glucose above 300 mg/dL were considered diabetic (de Oliveira Carvalho et al., 2021).
The diabetic animals were randomly divided into five groups (n = 5) and treated orally for 30 days. The animals’ body mass, water intake, urine volume, and food intake were evaluated daily.
The groups are:
The normoglycemic group was treated with 0.5 mL/animal-vehicle solution (Tween 4%) (NOR). Diabetic group treated with 0.5 mL/animal-vehicle solution (Tween 4%) (diabetic control group (DTC)). The diabetic group was treated with metformin 100 mg/kg in 0.5 mL of distilled water (MET). The diabetic group was treated with CSO 400 mg/kg vehicle (1 mL/animal) in 4% Tween solution (CSO). Diabetic group treated with NCS 200 mg/kg (NCS).
Biochemical and Histopathological Evaluation of Animals with DM
On the 30th day, whole blood samples were collected from the retro-orbital plexus of the left eye. The collected blood was centrifuged for 10 minutes at 5,000 rpm, and then the analyses were performed using reagent kits from the industry Doles Reagents and Lab Equipment. Ltd. (Goiânia-GO, Brazil). The samples were analyzed in a UV–vis model UVmini-1240 spectrophotometer (Shimadzu Corporation, Kyoto, Japan) (Souza et al., 2017). For biochemical analyses, tests were performed for triglycerides, total cholesterol (TC), aspartate aminotransferase (AST), alanine aminotransferase (ALT), urea, creatinine, glycosuria, and insulin.
A thiopental overdose euthanized the animals. The animals’ pancreas was collected for histopathological analysis and then fixed in 10% buffered formaldehyde for 24 hours. The samples were dehydrated in an increasing series of alcohols (70%, 80%, 90%, and 100%), clarified with xylene, impregnated, and embedded in paraffin at 60°C. The histopathological slides were sectioned using a semi-automatic rotary microtome model CUT 5062 (SLEE) with 5 µm cuts and stained using the Hematoxylin–Eosin method (Vasconcelos et al., 2011).
Treatments and Induction of Dyslipidemia by Triton
The animals were randomly divided into five groups (n = 5) and orally treated for 10 days. On the seventh day of treatment, dyslipidemia was induced by i.p. administration of Triton WR1339 (Tyloxapol, Sigma-Aldrich Chemical Co. St. Louis, MO, USA), 150 mg/kg dissolved in 0.9% NaCl. The groups are: Normal Group–Treated with 0.5 mL/animal of distilled water, without inducing dyslipidemia–(NOR); Triton Group–Treated with 0.5 mL/animal of distilled water, induction of dyslipidemia (TRI); CSO Group–Treated with 400 mg/kg of CSO, induction of dyslipidemia (CSO); NCS Group–Treated with 200 mg/kg of NCS, induction of dyslipidemia (NCS); Group SIM–Treated with 20 mg/kg of simvastatin (SIM), induction of dyslipidemia (SIM).
Biochemical Analysis of Animals with Dyslipidemia
On the 10th day of treatment, the animals were anesthetized with thiopental intraperitoneally and submitted to collect 1.5 mL of blood through the retro-orbital plexus. Subsequently, the collected blood was centrifuged for 10 minutes at 5,000 rpm. Then, biochemical analyses of triglycerides, TC, low-density lipoproteins (LDL), and high-density lipoproteins (HDLs) were performed. Tests were performed using reagent kits from the industry Doles Reagents and Lab Equipment. Ltd. (Goiânia-GO, Brazil). The samples were analyzed using a UV-mini-1240 UV–vis spectrophotometer (Shimadzu Corporation, Kyoto, Japan).
Statistical Analysis
All experimental quantitative results were expressed as mean ± standard deviation for statistical analysis. One-way analysis of variance was used, followed by Tukey’s test for multiple comparisons. Results with significance levels of p < 0.05 were considered statistically significant. The statistical program used was Prism (version 5.03).
Results
Physical-chemical and Chemical Parameters of the CSO
In the evaluation of the physical-chemical and chemical parameters (Table 1), it was observed that the CSO is a dark yellow viscous liquid with a density of 0.949 ± 0.08 g/cm3. The saponification index and unsaponifiable material in the CSO were 191.6 ± 2.08 mgKOH/g and 0.19% ± 0.04%, respectively. The iodine index obtained a value of 146.3 ± 4.16 gI2/100 g. Regarding the acidity index, a value of 2.7 ± 0.12 mgKOH/g was observed.
Physico-chemical and Chemical Parameters of the CSO.
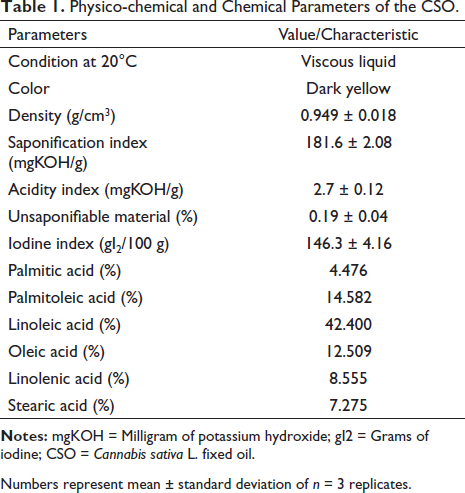
Numbers represent mean ± standard deviation of n = 3 replicates.
The chemical profile of CSO (Table 1) was obtained by gas chromatography. A composition of mostly UFAs was observed, with a majority of palmitoleic (14.58%), oleic (12.50%), linoleic (42.40%), and linolenic (8.55%) acids.
Obtaining, Characterizing, and Stability of the NCS
The NE was obtained using low energy input methodology and 90% water, 5% CSO, and 5% polysorbate 80. Immediately after preparing the NCS (first day), the particle size, polydispersion index (PDI), and stability were characterized (Table 2) over 30 days. The NCS did not change when subjected to centrifugation and thermal stress tests, and there was no separation of phases and color changes, with a final density of 0.993 ± 0.02 g/cm3, viscosity 0.889 ± 0.13, and pH 4.28 ± 0.04.
Parameters Obtained from the Characterization and Stability of the Nanoemulsion of C. sativa Fixed Oil (NCS) Performed During a Period of 30 Days.
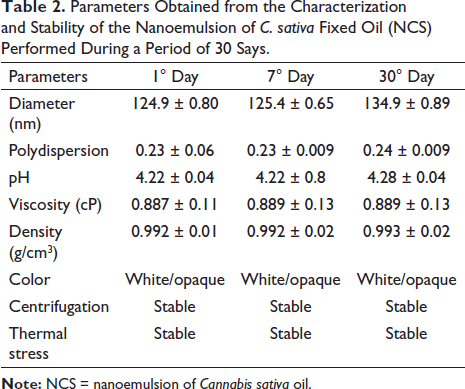
Initially (first day), the NCS presented a droplet size of 124.9 ± 0.80 nm with a PDI of 0.23 ± 0.06. At the end of the 30 days of evaluation (Table 2), it was observed that the average diameter of the droplets was 134.9 ± 0.89 nm, with a PDI of 0.24 ± 0.009. There was a slight increase in droplet size. However, the results revealed that the NCS showed stability with droplet size below 140 nm and with the monomodal distribution.
Effect of Treatments on Parameters of Rats with DM–Evaluation of Clinical Parameters of Animals with DM
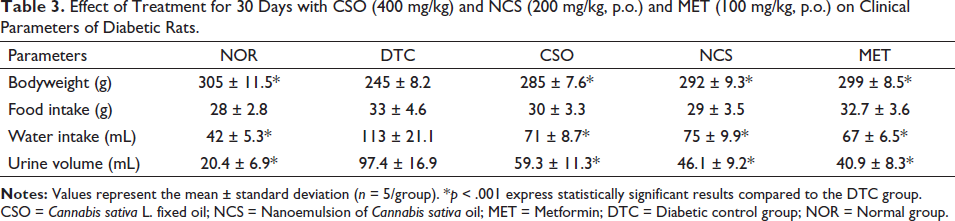
In the evaluation of the clinical parameters of diabetic animals (Table 3), when comparing the DTC group with the NOR group, it is observed that the DTC group significantly increased (p < 0.001) water consumption and urinary excretion volume. On the other hand, there was a significant decrease (p < 0.001) in body weight during treatment. The groups of diabetic animals treated with both CSO and NCS were able to attenuate significantly (p < 0.001) the values of water consumption and urinary volume, as well as being able to reduce significantly (p < 0.001) the loss of body weight when compared to the DTC group. The group treated with MET also attenuated the parameters, showing a statistical difference of p < 0.001 in the evaluated parameters.
Effect of Treatment for 30 Days with CSO (400 mg/kg) and NCS (200 mg/kg, p.o.) and MET (100 mg/kg, p.o.) on Clinical Parameters of Diabetic Rats.
Effect of Treatments on Biochemical Parameters of Rats with DM
As for the biochemical results (Table 4), it was demonstrated that in most parameters, there was no significant difference between the groups. No changes in urea, creatinine, transaminases (AST and ALT), or bilirubin were observed at the end of the treatments. A study reports that there may be changes in these parameters when the diabetic condition becomes chronic, and indicates the possibility of renal dysfunction mainly due to changes in urea and creatinine levels, while changes in AST and ALT transaminases accompany liver damage (Sá et al., 2015). Therefore, no results observed showed kidney and liver damage in this study. In the study conducted by Souza et al., (2017) using the fixed oil of the species Euterpe oleracea, rich in UFAs, there were no changes in kidney and liver functions.
Effect of Treatment for 30 Days with CSO (400 mg/kg) and NCS (200 mg/kg, p.o.) and MET (100 mg/kg, p.o.) on Biochemical Parameters of Rats with DM.
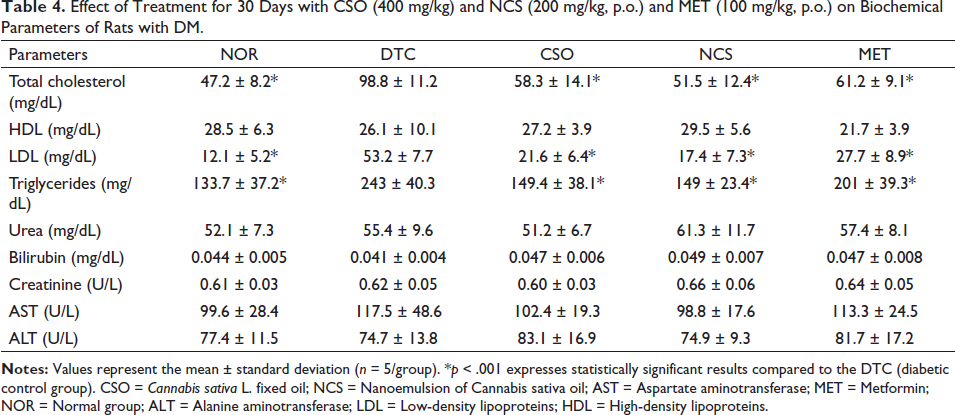
For the levels of triglycerides and cholesterol (Table 4), it was observed that the DTC group showed a significant increase (p < 0.05) in comparison to the NOR group, demonstrating values of 243 ± 40.3 and 98.8 ± 11.2 mg/dL, respectively. On the other hand, the groups treated with CSO and NCS showed a significant difference (p < 0.05) in the levels of triglycerides and cholesterol when compared with the DTC group, indicating that these treatments were able to avoid the increase of these parameters in the animals with DM, a fact which was also observed in the group treated with MET. Regarding HDL levels, treatments did not show a significant difference between groups. However, for LDL values, it was observed that the DTC group showed a significant increase (p < 0.05), with a value of 53.2 ± 7.7 mg/dL. The groups treated with CSO, NCS, and MET avoided this increase, showing slightly lower values, 21.6 ± 6.4, 17.4 ± 7.3, and 27.7 ± 8.9 mg/dL, respectively.
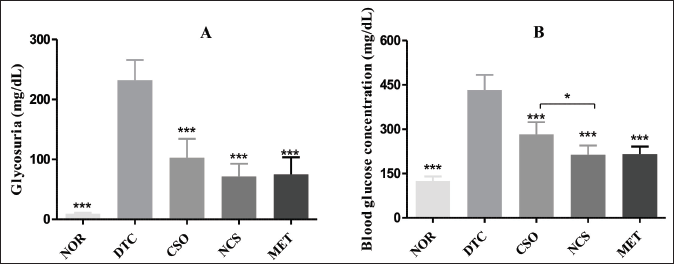
In the assessment of glycosuria and glycemia (Figure 1), it was observed that during treatment, the NOR group presented glycemia of 125.30 ± 15.1 and glycosuria of 9.60 ± 1.5 mg/dL. On the other hand, the DTC group demonstrated high levels of glycemia and glycosuria with extremely significant values (p < 0.001), with means of 432.00 ± 51.60 and 232.00 ± 33.80 mg/dL, respectively. The group treated with CSO was able to reduce significantly (p < 0.001) the levels of glycemia (282.3 ± 41.47 mg/dL) and glycosuria (102.8 ± 31.67 mg/dL) in animals with DM, as well as the group treated with NCS that also showed a significant reduction (p < 0.001), with blood glucose values of 213.5 ± 31.20 mg/dL and glycosuria of 71.50 ± 21.40 mg/dL. The MET group presented similar results to the NCS group, with glycosuria of 75.0 ± 28.6 mg/dL and blood glucose of 215.9 ± 25.30 mg/dL.
Effect of Treatments (o.a.) for 30 Days with OCS (400 mg/kg, o.a.) and NCS (200 mg/kg, o.a.) and MET (100 mg/kg, o.a.) on Glycosuria (A) and Glycemia (B) of DM mice. Results Represent the Mean ± Standard Deviation (n = 5/Group). **p < 0.01 and ***p < 0.001 Represent Statistically Significant Results Compared to the DTC Group. CSO = Cannabis sativa L. fixed oil; NCS = Nanoemulsion of Cannabis sativa oil; MET = Metformin; DTC = Diabetic control group; NOR = Normal group.
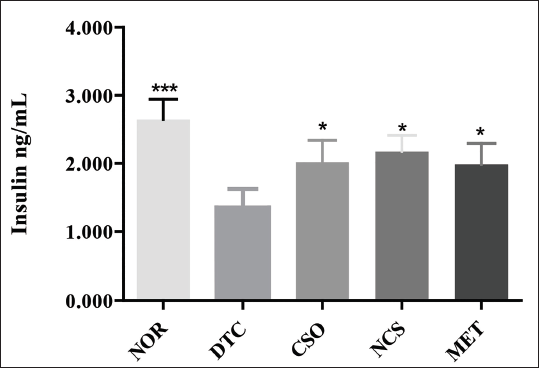
Regarding the quantification of insulin levels (Figure 2), it was observed that the NOR group had the highest mean insulin levels, in the range of 2.645 ± 0.29 ng/mL. In contrast, the DTC group showed a significant reduction, with a mean of 1.352 ± 0.27 ng/mL. In the groups treated with CSO, NCS, and MET, insulin levels were significantly increased (p < 0.05), with mean values of 2.019 ± 0.31, 2.175 ± 0.23, and 1.985 ± 0.31 ng/mL, respectively.
Effect of Treatments (o.a.) for 30 Days with CSO (400 mg/kg, o.a.) and NCS (200 mg/kg, o.a.) and MET (100 mg/kg, o.a.) on Insulin Levels in Rats with DM. Results Represent Mean ± Standard Deviation (n = 5/Group). *p < 0.05, **p < 0.01, and ***p < 0.001 Represent Statistically Significant Results Compared to the DTC Group. CSO = Cannabis sativa L. fixed oil; NCS = Nanoemulsion of Cannabis sativa oil; MET = Metformin; DTC = Diabetic control group; NOR = Normal group.
Histopathological Evaluation of the Pancreas of Animals with DM
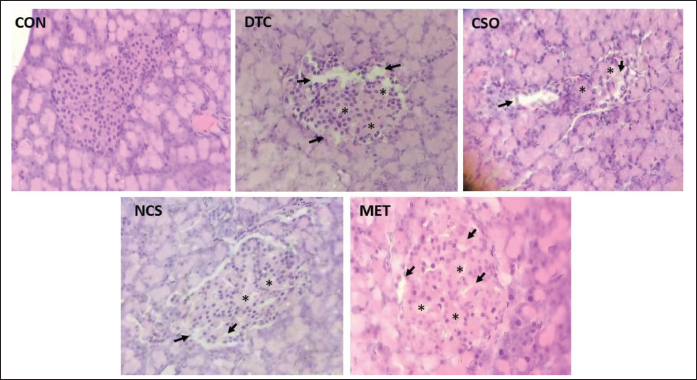
The histopathological results (Figure 3) showed that DM induction with STZ could promote necrosis of pancreatic β cells with partial destruction of the islets of Langerhans, observed mainly in the photomicrography of the DTC group, where there is a reduction in cell population in the pancreatic islets. On the other hand, it is possible to notice that the groups treated with CSO and NCS presented an increase in the number of cells in the islets of Langerhans and showed a decrease in regions devoid of cells, indicating possible cell regeneration. These findings confirm the previously described inferences about the increased insulin levels and decreased blood glucose levels.
Histopathological Photomicrographs of the Pancreas, Showing Islets of Langerhans, Stained with H&E, 400× Magnification. Normal Group (NOR), Showing Regions with a Normal Number of Cells in the Islets. Diabetic Control Group (DTC), Showing Regions with the Absence of Cells Due to Destruction by STZ (Black Arrows) and Regions with a Reduced Number of Cells, Possibly Regenerated Cells (Asterisks). Oil-treated Group (CSO), Nanoemulsion-treated Group (NCS) and Metformin-treated Group (MET). CSO = Cannabis sativa L. fixed oil. NCS = Nanoemulsion of Cannabis sativa oil; MET = Metformin; STZ = Streptozotocin.
Evaluation of Triton-induced Dyslipidemia
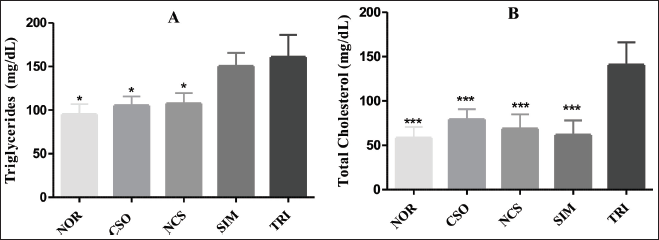
Regarding Triton-induced dyslipidemia, the results reveal that the TRI group significantly increased the levels of triglycerides (p < 0.05) and TC (p < 0.001) (Figure 4). On the other hand, it was observed that treatments with CSO and NCS e were able to decrease significantly (p < 0.05) triglyceride levels when compared to the TRI group. Furthermore, the groups treated with CSO and NCS were also able to reduce significantly (p < 0.001) TC levels, with values of 79.2 ± 13.62 and 68.73 ± 16.22 mg/dL, respectively. The group treated with SIM significantly reduced cholesterol levels, representing 61.80 ± 16.25 mg/dL. Thus, it was evidenced that the treatments with CSO and NCS prevented Triton-induced hyperlipidemia.
Effect of Treatments (o.a.) for 10 Days with CSO (400 mg/kg) and NCS (200 mg/kg) and SIM (20 mg/kg) on Levels of Triglycerides (A) and Total Cholesterol (B) in Rats with Triton-induced Dyslipidemia (i.p.). Results Represent Mean ± Standard Deviation (n = 5/Group). **p < 0.01 and ***p < 0.001 Represent Statistically Significant Results Compared to the TRI Group. CSO = Cannabis sativa L. fixed oil; NCS = Nanoemulsion of Cannabis sativa oil; MET = Metformin; DTC = Diabetic control group; NOR = Normal group.
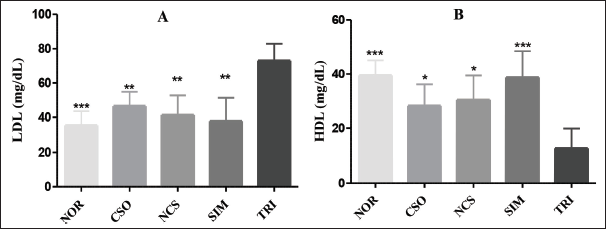
Regarding the values of LDL and HDL (Figure 5), it is observed that the TRI group showed a significant decrease (p < 0.001) in HDL in comparison to the NOR group. The SIM group avoided the HDL decrease, presenting a value close to the one observed in the NOR group, being 38.76 ± 9.66 mg/dL. The CSO and NCS groups also avoided the reduction in HDL levels, although to a lesser extent and significance (p < 0.05).
As for LDL levels, it was possible to observe that the TRI group presented an extremely high result (72.94 ± 9.90 mg/dL) compared to the other groups. The groups treated with CSO and NCS demonstrated the ability to reduce significantly (p < 0.001) LDL values, being 46.52 ± 8.35 and 41.39 ± 11.4 mg/dL, respectively. The group treated with SIM also decreased the LDL levels, with 37.68 ± 13.71 mg/dL.
Effect of Treatments (o.a.) for 10 Days with CSO (400 mg/kg) and NCS (200 mg/kg) and SIM (20 mg/kg) on LDL (A) and HDL (B) Levels in Rats with Dyslipidemia Triton-induced (i.p.). Results Represent Mean ± Standard Deviation (n = 5/Group). **p < 0.01 and ***p < 0.001 Represent Statistically Significant Results Compared to the TRI Group. CSO = Cannabis sativa L. fixed oil; NCS = Nanoemulsion of Cannabis sativa oil; MET = Metformin; DTC = Diabetic control group; NOR = Normal group; LDL: Low-density lipoproteins; HDL: High-density lipoproteins.
Discussion
In this study, the quality control of the CSO was carried out by evaluating the physical-chemical and chemical parameters. Freire et al. (2013) report that there are chemical changes that occur in fixed oils and fats that can influence their quality, mainly reactions of hydrolysis and auto-oxidation catalyzed by the action of heat and humidity, with the formation of free fatty acids, monoacylglycerol, and diacylglycerol, lipid auto-oxidation associated with the reaction of oxygen with UFAs.
A study by Latif and Anwar (2009) evaluating the physical-chemical parameters of the CSO corroborates our results since they observed similar results for the parameters of density, iodine index, and unsaponifiable material. On the other hand, a superior result was observed for the saponification index, which could be explained by the absence of long-chain fatty acids with 20 Carbons.
The saponification index indicates the relative amount of high molecular weight fatty acids and low molecular weight fatty acids, as low molecular weight fatty acids require more potassium hydroxide for saponification. Thus, the saponification index is inversely proportional to the molecular weight of triglyceride fatty acids (Souza et al., 2017).
In the study by Mikulcová et al. (2017), the acidity index results were lower than those reported in our study, with values ranging from 1.7 to 0.7 mg KOH/g. The acid number indicates the formation of free fatty acids resulting from the breakdown of triglycerides during extraction and exposure to light and heat. However, acidity is a variable that also depends on the processing and extraction of lipids, considering that while extracting raw vegetable oil, some species may contain high levels of phenolic compounds and organic acids that may contribute to the process of acidity, stability, and antioxidant potential (Farhoosh et al., 2009).
Other studies agree with this study’s composition and percentage of fatty acids (de Oliveira Carvalho et al., 2021; Gao & Birch, 2016; Teh & Birch, 2013). However, the concentrations of these fatty acids can be influenced by different extraction processes, cultivation sites, and seasonality (Antoniassi et al., 2013).
The development of NEs for application in pharmaceutical or nutraceutical products results in several techniques that seek to incorporate low aqueous solubility substances in formulations with droplets on a manometric scale ranging from 20 to 200 nm and coexisting in a continuous phase stabilized by surfactants (Maali & Mosavian, 2013; Pereira et al., 2016).
In the study by Gumiero and da Rocha Filho (2012), where an NE with Babassu oil (Orbignya oleifera) rich in fatty acids was developed, it was observed that the NE also did not present physical-chemical changes after centrifugation and thermal stress tests. On the other hand, Jarzębski et al. (2021) demonstrated the importance of viscosity in the stability of NEs, where the tendency of changes in their values generated instability in the formulations during the storage period. The decrease in viscosity of NEs can be caused by two factors: the diffusion of water molecules from the internal to the external aqueous phase or their coalescence. These findings, in part, reinforce that the results obtained from the NCS are stable since no relevant changes in viscosity were observed.
The studies by Jarzębski et al. (2021) and de Oliveira Carvalho et al. (2021) corroborate our results, as they developed NEs from CSO, and stability tests demonstrated that the formulations showed stability with droplet diameters ranging from 129 to 209 nm and monomodal distribution.
Fathordoobady et al. (2021), applying high energy methods to obtain CSO NEs, report that the ultrasound method is efficient for obtaining NEs, as it produced formulation with an average droplet size <180 nm and PDI of 0.15 ± 0.04. On the other hand, the microfluidic approach technique produced smaller particles than ultrasonication, with good stability observed during storage at room temperature and mean droplet size <62.0 nm.
Other studies report that the stability of NEs depends in part on the size of the droplets formed. NEs with relatively large droplets (greater than 200 nm) can quickly lose stability, leading to phase separation or cremation (Mikulcová et al., 2017; Santos Rodrigues et al., 2020).
DM induced by STZ leads to a very complex pathophysiological condition, which results in alterations of several clinical parameters, promoting the loss of body weight and exaggerated increase in the consumption of water, food, and urine excretion (de Oliveira Carvalho et al., 2021; Suanarunsawat et al., 2016).
Studies report that increased water consumption and diuresis are directly related to increased blood glucose. High blood glucose leads to an imbalance in blood osmolality, causing water to pass from the intracellular to the extracellular environment, leading to dehydration and excessive water consumption (Mahendran et al., 2014; Sultan et al., 2014). This fact can be observed in the DTC group and justifies the results of the CSO, NCS, and MET groups, which attenuated water consumption and excretion of urine.
The study carried out by Pwaniyibo et al. (2020) corroborates with the results obtained in this work since it was demonstrated that the animals with DM and high hyperglycemia present a significant decrease in body weight. The treated animals that decreased glycemia showed an increase in body weight. Sultan et al. (2014) demonstrated that the treatment with Nigella sativa fixed oil of chemical composition like CSO improved the body weight loss of animals with DM.
DM is directly related to the elevation of triglycerides and cholesterol serum levels, and the reduction of insulin production is the main factor contributing to the increase in lipid levels. Hyperglycemia contributes to hepatocyte hyperactivity because, with the lack of insulin or its peripheral resistance, lipids become one of the primary energy sources, mobilizing lipids into the bloodstream as free fatty acids (Jay & Ren, 2007; Ozen et al., 2012). These plasma lipid alterations associated with increased LDL increase the risk of developing vascular diseases.
In this study, it was observed that CSO and NCS treatments could significantly attenuate triglyceride and cholesterol levels, as well as LDL values. This fact can contribute to reducing the risks of cardiovascular diseases caused by DM. Other studies with fixed oils rich in UFAs have already demonstrated the ability to reduce lipid levels (Al Jamal & Ibrahim, 2011). In a study by Sultan et al. (2014), significantly reduced cholesterol, triglycerides, and LDL levels.
Reducing glucose levels in blood and urine reinforces the anti-diabetic effects promoted by treatments with CSO and NCS. In this sense, the optimization and effectiveness of NCS are observed, as it produced an anti-diabetic effect with a lower dose than the treatment with CSO. The study by Yuvashree et al. (2020) corroborates our results since it evaluated the anti-diabetic effect of the NE of Allium sativum (garlic) oil and demonstrated its effectiveness in reducing blood glucose and other parameters in induced diabetic animals with STZ.
Studies report that UFAs act as essential antioxidants, mainly to prevent lipid peroxidation, as they have one or more double bonds that are susceptible to hydrogen loss for reactive oxygen species (ROS) (Jarzębski et al., 2019). In part, the anti-diabetic effects observed in the CSO and NCS groups may be related to the antioxidant capacity of these treatments, as they contain high levels of UFAs, mainly linoleic and linolenic fatty acids. In the study conducted by Prada et al. (2019), the anti-diabetic effect of the nanoformulation containing Cassia grandis was demonstrated, emphasizing that this effect is partly due to the significant antioxidant action of the nanoformulation.
Studies have shown that UFAs have great potential in treating DM, as administering an α-linolenic acid agonist significantly decreased plasma glucose and increased insulin release in mice (Moran et al., 2014). Furthermore, a diet rich in α-linolenic acid has been shown to improve insulin sensitivity by increasing glucose transporter (GLUT) 4 expression in cells from STZ-induced diabetic rats (Khamaisi et al., 1999). A study by Belury et al. (2018) validates our results, showing that linoleic acid intake is associated with decreased DM risks, better glycemic control, and insulin sensitivity.
Other studies propose that antioxidant compounds protect pancreatic β cells against oxidative damage and stimulate glucose-induced insulin secretion through the recruitment of GLUT 2 signaling (Dewanjee et al., 2020; Shenoy et al., 2018).
By associating the glycemic and insulin levels of the groups, it is possible to infer that the treatments with CSO and NCS contributed substantially to the improvement of the diabetic condition by increasing the elevation of insulin levels. The study carried out by de Oliveira Carvalho et al. (2021) demonstrated that the treatment with formulations containing extract of the species Calophyllum brasiliense presented an anti-diabetic effect by restoring the β-pancreatic cells and contributing to the decrease of glycemic levels.
Dewanjee et al. (2020) report that plant-derived anti-diabetic agents can reduce diabetic complications through glycemic control, antioxidant mechanisms, and inhibition of mechanisms that lead to the destruction of pancreatic β-cells mediated by pathological processes involved in inflammation, fibrosis, and apoptosis.
Studies report that STZ induces DM through a mechanism that involves the cytotoxic action of STZ on pancreatic β cells, in which there is transport of STZ to the intracellular environment via the GLUT 2. In the intracellular environment, it induces the excessive formation of ROS, causing oxidative damage to β cells through the activation of apoptosis with a significant decrease in insulin production and, consequently, the onset of the clinical picture of DM (Ali et al., 2020; Eleazu et al., 2013).
In the study carried out by Hamden et al. (2010) using fenugreek oil in the treatment of diabetic rats, it was demonstrated that there was a significant reduction in blood glucose levels due to the restoration of β cells in the pancreatic islets. In addition, the reduction of oxidative damage was essential for this restoration. Other studies describe that vegetable oils with a high UFA content have a high antioxidant potential, which contributes mainly to increasing the body’s antioxidant activity, favoring the restoration and regeneration of β-pancreatic cells from oxidative damage (Durašević et al., 2019; Suanarunsawat et al., 2016).
Dyslipidemia is a metabolic disorder characterized by abnormal blood concentrations of lipids and lipoproteins. It is defined by an increase in TC or low-density lipoprotein (LDL) and triglyceride levels and a decrease in HDL levels (Mantovani & Pugliese, 2021).
Triton (Tyloxapol) is a non-anionic surfactant with a polymeric structure used in several studies to induce dyslipidemia in animal models. Its mechanisms of action are the ability to inhibit the enzyme lipoprotein lipase (LPL), responsible for the hydrolysis of triglycerides present in plasma lipoproteins, and the stimulation of hydroxymethyl glutaryl coenzyme A reductase (reductase), an enzyme involved in the biosynthesis of hepatic cholesterol (Bertges et al., 2011; Castro et al., 2012).
Several studies corroborate the results obtained in this study, showing that fixed vegetable oils rich in UFAs contribute substantially to the reduction of cholesterol and triglyceride levels. Souza et al. (2017) demonstrated the ability of E. oleracea fixed oil to reduce cholesterol and lipid levels in a Triton induction model. Abdou et al. (2018) demonstrated that soybean oil rich in UFAs could attenuate the high lipid values and improve the antioxidant defense in rats with hyperlipidemia induced by Triton WR-1339. A study demonstrates the benefits of C. sativa seed oil against hyperlipidemia and protection against cardiovascular diseases (Kaushal et al., 2020).
In part, the mechanisms by which UFAs reduce the lipid profile involve the activation of peroxisome proliferator-activated receptor (PPAR) isoforms. UFAs generally have more affinity for the PPARα isoform, promoting increased intravascular lipolysis and clearance of TG-rich particles due to the regulation of the LPL gene. In addition, it participates in the control of the transport and absorption of fatty acids and increases the activity of β-oxidation enzymes in mitochondria and peroxisomes (Brito et al., 2013; Echeverría et al., 2016; Kimura et al., 2013).
The nuclear receptor PPARγ has an affinity for some types of UFAs, such as linoleic acid, which, upon activation of PPARγ, reduces plasma lipid levels, increasing the LPL enzyme-mediated hydrolysis of triglyceride-rich lipoproteins, with concomitant increases in the transcription of the FATP gene (Semple et al., 2006; Słowikowski et al., 2020).
Reducing plasma lipid levels is essential for endothelial protection and control of atherosclerotic disease and for reducing the risk of complications, such as acute myocardial infarction and systemic arterial hypertension (Mantovani & Pugliese, 2021). The results evidenced by the CSO and NCS treatments in reducing cholesterol, triglycerides, and LDL levels are data that reinforce the protective effect against cardiovascular diseases. In addition, it is known that decreasing LDL-cholesterol levels protect against cardiovascular diseases.
Conclusion
Given the results obtained in this study, the treatments with CSO and NCS were able to improve clinical and biochemical parameters, as well as possibly contribute to the partial restoration of pancreatic cells—thus increasing insulin levels—and significantly decrease serum lipid levels, both in animals with DM and in animals with Triton-induced dyslipidemia. Therefore, it is concluded that the treatments with CSO and NCS, under the conditions of this study, demonstrated an anti-diabetic effect and the ability to reduce triglyceride, cholesterol, and LDL levels. In this respect, treatments with CSO and NCS act in the control of DM, as well as in the prevention of cardiovascular diseases.
Abbreviations
ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; CSO: Cannabis sativa L. fixed oil; DM: Diabetes mellitus; DTC: Diabetic control group; GLUT: Glucose transporter; HDL: High-density lipoproteins; LDL: Low-density lipoproteins; MET: Metformin; NCS: Nanoemulsion of C. sativa oil; NOR: Normoglycemic control group; PPAR: Peroxisome proliferator-activated receptor; STZ: Streptozotocin; TC: Total cholesterol; UFAs: Unsaturated fatty acids.
Footnotes
Acknowledgments
We thank CNPq—National Research Council and CAPES—Higher Education Personnel Foundation for their research support. Process Number 88882.430015/2019-2019-01 and 88887.478228/2020-00.
Authors’ Contribution
All authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Animal manipulations and handling processes for all experiments were approved by the Ethics Committee of Animals Use from the Federal University of Amapá (CEUA, UNIFAP), on August 28 under protocol number 015/2018.
Funding
This work was partly supported by the National Council for Scientific and Technological Development—CNPq No. 12/2020—Proc.: 403587/2020-4, Master’s and Doctoral Program for Innovation—MAI/DAI.
