Abstract
Background
Chronic liver dysfunction (CLD) is a significant global health issue; alcohol use, obesity, drugs, and antibiotic and viral infections contribute to its development. However, changes in gut microbiota composition, primarily as people grow older, can also play a role in initiating and progressing liver dysfunction.
Purpose
This review summarizes the altered gut microbiota–mediated chronic liver dysfunction and promising therapeutic strategies for NAFLD, ALD, NASH, and liver cirrhosis.
Discussion on the chronic liver disease and its therapeutic aspects
The gut microbiota influences physiological processes, including immune system regulation, by promoting the production of pro-inflammatory markers and elevating the expression of toll-like receptors (TLRs) on hepatocytes, metabolisms of bile acids and choline, alteration in saturated aliphatic organic acid (SCFA) production, and food absorption. These alterations increase intestinal permeability, chronic inflammation, and liver dysfunction. Dysbiosis triggers inflammatory pathways, oxidative stress, and the production of toxic compounds that damage liver cells and promote chronic liver diseases such as alcoholic liver disease (ALD), non-alcoholic fatty liver disease (NAFLD), non-alcoholic steatohepatitis (NASH), and cirrhosis. Age-related changes in gut motility, secretory function, and immunological function can further decline beneficial bacterial diversity and abundance and shifts in pathogenic bacterial species’ relative abundance.
Conclusion
Dysbiosis resulting from these age-related changes may contribute to the development of CLD. Numerous therapeutic approaches are available to restore the altered gut microbiota and reduce the risk of chronic liver dysfunction.
Introduction
Chronic liver dysfunction (CLD) is a huge worldwide health issue, with an estimated 4.5 million deaths yearly owing to liver disease. Altered gut microbiota composition, particularly as people age, can have a substantial impact on the initiation and development of liver dysfunction (Sharma & Nagalli, 2022). Age-related changes in the makeup of the gut microbiota can cause dysbiosis, a condition of microbial imbalance that can result in the overgrowth of negative microbes and the depletion of helpful ones. Increased intestinal permeability brought on by dysbiosis can cause chronic inflammation and liver dysfunction by allowing toxins and bacterial byproducts to enter the circulation. Dysbiosis can also play a role in the onset of NAFLD, a disorder marked by fat buildup in the liver as well as in non-alcoholic steatohepatitis (NASH), alcoholic liver disease (ALD), and liver cirrhosis (Grosicki et al., 2018; Odamaki et al., 2016). However, it’s thought that dysbiosis can trigger inflammatory pathways, oxidative stress, and the formation of toxic compounds that can destroy liver cells and aid in the onset of liver disease (Coker et al., 2022).
To better understand the processes behind the association between the makeup of the gut microbiota and liver function and to create methods for the prevention and treatment of CLD, further study is required (Vijay & Valdes, 2022; Xie et al., 2016). This review focuses on CLD, its age-related changes in the gut microbiota, the management of CLD, and promising therapeutic strategies for various CLDs such as ALD, NAFLD, NASH, and liver cirrhosis.
Gut Microbiota Composition and Its Role in Liver Function
The diverse distribution of bacteria inside the gastrointestinal system of humans is vital for maintaining general health. Still, sometimes, it may lead to disease when there is an imbalance between good and harmful bacteria. According to a recent study, the gut microbiota’s makeup can significantly affect liver functions and changes in the gut microbiota have been associated with the onset and progression of liver disease (Islam et al., 2022).
The control of bile acid metabolism, necessary for the liver’s removal of waste products and the absorption of dietary lipids, is another function of the gut microbiota (Yoo et al., 2020). Bile acid influences the regulation of lipid metabolism as well as glucose metabolism via the activation of G-protein-coupled receptor (GPCR) and farnesoid X receptor (FXR) (Copple & Li, 2016). Upon interaction of bile acids with FXR, antimicrobial peptide angiogenin-1 is synthesized, which plays a potential role in the inhibition of the overgrowth of intestinal microbiota and improves gut barrier function by preventing bacterial uptake (Parséus et al., 2017).
Short-chain fatty acids (SCFAs) mainly include butyrate, propionate, and acetate, which are generated by gut bacteria fermentation of dietary fiber. Dietary soluble fibers undergo fermentation by gut microorganisms into SCFAs, which are known to possess health-improving benefits by sustaining the diversity of the gut microbiota and generating beneficial metabolites (Pan & Zhang, 2022). Due to their anti-inflammatory and immunomodulatory properties, SCFAs may be able to protect the liver against oxidative stress and inflammation-related harm (Al Bander et al., 2020; Anand & Mande, 2022). Butyrate has been found to regulate various tight junction proteins, such as mucin-2 and claudin-1 proteins, to improve the gut barrier and also play an anti-inflammatory role by activating peroxisome proliferator-activated receptor gamma (PPAR-γ), which further inhibits the transduction of nuclear factor kappa B (NF-kβ) (Schwenger et al., 2019). Ethanol is also a vital metabolite of the gut microbiota produced by E. coli (Schwenger et al., 2019). Acetaldehyde, the intermediate product of ethanol synthesis from carbohydrates via fermentation, may disrupt gut barrier function by damaging tight junctions, allowing microbial products to enter the bloodstream (Chaudhry et al., 2016). Alterations in the gut microbiota composition have also been linked to the onset and progression of several CLDs, mostly including ALD, NAFLD, and cirrhosis.
Alcoholic Liver Disease
Alcohol consumption is the primary cause of ALD, which leads to gut dysbiosis by promoting bacterial translocation (migration of live intestinal microbes or their products to mesenteric lymph nodes or other locations) and inflammation by causing injury to the intestinal barrier (Adhikary et al., 2023; Leclercq et al., 2014). Toxic bacterial byproducts such as endotoxins, lipopolysaccharides (LPS), various pathogen-associated molecular patterns (PAMPs), and bacterial DNA contribute to the onset of ALD (Hartmann et al., 2015) by inducing inflammation and liver damage, increasing ROS to promote pro-inflammatory responses, and enhancing intestinal epithelial permeability (Hartmann et al., 2015; Liu et al., 2021). Furthermore, it has been reported that a leaky gut and enhanced level of bacterial products in serum were observed in ALD patients compared to healthy control (Bajaj, Heuman, Hylemon, Sanyal, White, et al., 2014; Parlesak et al., 2000). In mice experiments, after the injection of alcohol into the gastrointestinal tract, a decreased level of Firmicutes phylum and Lactobacillus spp. was observed and also increased levels of Enterococcus spp., Corynebacterium spp., Akkermansia muciniphila, and Alcaligenes spp. were observed (Bull-Otterson et al., 2013; Hartmann et al., 2013).
It is also reported that ALD patients with higher gut permeability have an enhanced level of phenol and lower level of methyl-phenol, indole, and 3-methyl-indole concentration in feces compared with those with lower gut permeability. In alcoholic patients, a decreased level of several saturated aliphatic organic acids (SCFAs) such as propionate and isobutyrate, and a reduced level of natural suppressants such as caryophyllene and of hepatic steatosis attenuators such as camphene, dimethyl-disulfide, and dimethyl-trisulfide were observed, and increased levels of oxidative stress biomarkers such as tetradecane were also observed (Couch et al., 2015). Microbial taxa that have been found in abundance in ALD patients are Proteobacteria, Lachnospiraceae, Streptococcus, Enterobacteria, Klebsiella, Lactococcus, Lactobacillus, Shuttleworthia, Rothia, Gemella, Actinomyces, Desulfovibrio, Candida albicans, and Candida dubliniensis, among others, and those that have been found in lower numbers are Bacteroidetes, Ruminococcaceae, Alistipes, Coprococcus, Paraprevotella, Prevotella, and Faecalibacterium, among others (Leclercq et al., 2020) (Table 1).
Existing Clinical Manifestations of Chronic Liver Dysfunction and Alteration of the Gut Microbial Taxa and Compositions.

Non-alcoholic Fatty Liver Disease
Unhealthy fat builds up in the liver of people who drink little to no alcohol, a condition known as NAFLD, which is the hallmark of NAFLD. In NAFLD, a decrease in the number of helpful bacterial species, such as Bacteroidetes, and an increase in the number of possibly hazardous bacterial species, such as Prevotella and Porphyromonas spp., in comparison to healthy controls have been reported (Hrncir et al., 2021). By encouraging inflammation, oxidative stress, insulin resistance, hyperlipidemia, and hypertension cause changes in the makeup of the gut microbiota, which has been demonstrated to contribute to the onset of NAFLD (Hrncir et al., 2021; Riaz Rajoka et al., 2018). An increase in Gram-negative bacteria, predominantly Proteobacteria (phylum), and a decrease in Gram-positive bacteria, mostly Firmicutes (phylum), were also found (Yang et al., 2017). The metabolomic profiles of antibiotic-treated or germ-free mice showed that gut microbiota-produced metabolites such as SCFAs, lithocholic acids (LCA), deoxycholic acids (DCA), and trimethylamine (TMA) are involved in the development of NAFLD (Marion et al., 2019; Studer et al., 2016). Trimethylamine (TMA) is a byproduct of the conversion of choline through gut microorganisms. Trimethylamine N-oxide (TMAO) is potentially generated in the liver via the oxidation of TMA. High TMAO concentrations in the liver have been associated with atherosclerosis and other metabolic diseases such as obesity and NAFLD (Krueger et al., 2021). Ethanol may influence the permeability of the gut alongside being readily absorbed, instantly detrimental to the liver (Abouelkheir et al., 2023). Fructose has been found to play a crucial role in the development of NAFLD and NASH at both clinical and preclinical levels, and it is further reported to disrupt tight junctions, thereby enhancing intestinal barrier permeability (Jensen et al., 2018; Li et al., 2019). Gut microbiota–induced choline deficiency also contributes to NAFLD pathogenesis by decreasing its availability (Dumas et al., 2006) and increasing TMAO levels in mice fed a high-fat diet (Gao et al., 2014). Ethanol may influence the permeability of the gut alongside being readily absorbed, which is instantly detrimental to the liver (Abouelkheir et al., 2023). Children who suffered from the inflammatory variant of NAFLD, NASH, had higher blood ethanol levels than healthy children (Zhu et al., 2013). The microbial taxa that are found in greater proportions in NAFLD patients are Bacteroides, Proteobacteria (phylum), Escherichia, Enterobacteriaceae (family), Peptoniphilus (genus), and Dorea, among others, and those that are found in lower proportions are Eubacterium (genera), Rikenellaceae, Faecalibacterium, Anaerosporobacter, Ruminococcaceae (family), and Coprococcus, among others (Sookoian et al., 2020). One study reported a dysbiotic biomarker that may be used to characterize NAFLD: an increased ratio of Bacteroidete to Firmicutes (B/F), which means an increased abundance of Bacteroidete and a decreased abundance of Firmicutes (Gkolfakis et al., 2015). A decrease in the abundance of Ruminococcaceae UCG-010 of the Ruminococcaceae family was observed in NAFLD individuals. Faecalibacterium prausnitzii (a butyrate-producing bacterium) is less abundant in obese children associated with NAFLD or without NAFLD (Albhaisi & Bajaj, 2021). In obese NAFLD individuals, a lower abundance of Bacteroidetes, Gemmiger, and Oscillospira was found to have a significant association with the exacerbation of liver fat fraction (Monga Kravetz et al., 2020) (Table 1).
Non-alcoholic Steatohepatitis
NASH is a kind of liver disease characterized by inflammation, damage to liver cells, and fat buildup in the liver. It is thought to be an intermediary stage between NAFLD and cirrhosis (Tsay & Lim, 2022). It is frequently observed in people who have type 2 diabetes, high blood pressure, and high cholesterol levels and is strongly correlated with obesity, insulin resistance, and metabolic syndrome (Santos et al., 2022). Altered permeability of the intestinal barrier, alteration in the metabolism of bile acids and choline, secretion of endogenous ethanol, production of inflammatory cytokines, and elevated expression of toll-like receptor (TLR) on hepatocytes are the leading mechanisms to disrupt the gut–microbiome–liver axis, thereby promoting the progression of NAFLD to NASH (Jayakumar & Loomba, 2019; Ma et al., 2017; Mouzaki & Loomba, 2020). The abundance of the genus Prevotella was found in NASH and obese patients. Furthermore, one pediatric study reported a greater abundance of ethanol-producing bacteria, such as the Escherichia genus, in NASH patients than in obese patients (Zhu et al., 2013). Other investigations have found an elevated ethanol level in NASH patients compared to obese people, which may be a source of ROS generation contributing to hepatic inflammation (Zhu et al., 2013).
Endotoxins activate TLRs, leading to the release of inflammatory cytokines such as TNF-α and IL-8. This triggers the migration of neutrophils and monocytes to the liver (Singh et al., 2011). Similarly, gut-derived metabolites may activate TLRs, causing inflammation and the development of NAFLD to NASH and, subsequently, fibrosis (Henao-Mejia et al., 2013). Bacteroides have been found in more numbers in NASH patients, and this abundance of Bacteroides is associated with the production of higher levels of sugars such as glucose and fructose, DCA, and D-pinitol and decreased production of SCFAs and amino acids (Zhao et al., 2013). Fructose has also been found to stimulate the inflammation and fibrosis of hepatic cells in NASH patients (Abdelmalek et al., 2010). Zhu et al. (2013) reported that NASH patients exhibited a higher abundance of Proteobacteria, Enterobacteriaceae, and Escherichia while a reduced abundance of Alistipes, Oscillospira, Blautia, Eubacterium, Bifidobacterium, and Coprococcus compared to healthy individuals (Zhu et al., 2013). Another study reported a greater number of Ruminococcus, Dorea, and Blautia and lower number of Oscillospira associated with pediatric NASH (Del Chierico et al., 2017) (Table 1).
Liver Cirrhosis
Liver cirrhosis (Table 1) is one of the detrimental types and occurs at the end stage of liver fibrosis, which may develop in the later stages of NAFLD and NASH if patients are not properly treated (Liu & Chen, 2022). Several cascading events, such as bacterial overgrowth and dysbiosis, and diseases, such as viral hepatitis, autoimmune hepatitis, sclerosing cholangitis, steatohepatitis, and Wilson’s disease, are associated with the progression of cirrhosis (Lee & Suk, 2020; Trebicka et al., 2021). Acute-on-chronic liver failure (ACLF) is the most critical form of cirrhosis, associated with a higher rate of systemic inflammation with proven bacterial infection (Trebicka et al., 2021). The initial stages of liver damage occur due to oxygen-free free radicals and inflammatory materials, which ultimately activate Kupffer cells and inflammatory cells. Following that, the stellate cells in the liver are also activated. The basic pathogenesis of liver fibrosis is as follows (Bataller & Brenner, 2005; Trebicka et al., 2021): Hepatic stellate cells, found in the Disse space (also known as the perisinusoidal space), significantly involve liver fibrosis (Trebicka et al., 2021). A previous study found that a significant alteration in the immune system in cirrhosis potentially involves gut dysbiosis, which means the alteration of the microbial community in saliva, serum, ascites, intestinal mucosa, and stool (Gressner et al., 2002).
One study conducted on cirrhosis patients by analysing recto-sigmoid mucosa-microbiota found a greater abundance of pathogenic bacteria such as Enterococcus sp., Burkholderia sp., Clostridium sp., and Proteus sp., while it also found reducing number of many autochthonous bacteria including Dorea sp., Subdoligranulum sp., Blautia, Roseburia, Faecalibacterium, and Incertae Sedis XIV in cirrhosis patients compared to control groups (Bajaj et al., 2012). Chen et al. (2011) performed one study by taking cirrhosis patients, where they found a greater abundance of Dialister sp., Atopobium sp., Veillonella sp., Megasphaera sp., and Prevotella (Table 1).
Age-related Changes in Gut Microbiota Composition
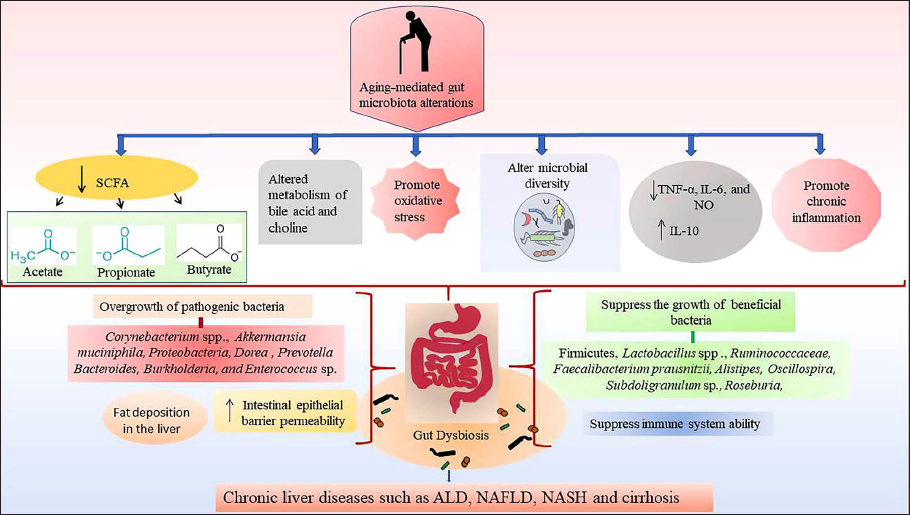
In general, the phases of age-related changes in the composition of the gut microbiota may be divided as infancy, adulthood, and elderly. In the first few years of life, the gut microbiota is quickly evolving and is greatly impacted by nutrition, delivery method (vaginal or Caesarean), and early exposure to antibiotics and drugs (La-Ongkham et al., 2020). The gut microbiota stabilizes and diversifies as people age (La-Ongkham et al., 2020). In elderly individuals, the diversity of the gut microbiota declines and a shift toward potentially hazardous microorganisms occurs. Changes in nutrition and a reduction in immunological function are suggested to be contributing factors to this shift (Kim & Jazwinski, 2018; Rinninella et al., 2019).
Age-related changes are characterized by a rise in the number of potentially dangerous bacterial species, such as Proteobacteria and Firmicutes, and a decrease in the abundance of helpful bacterial species, such as Bifidobacterium and Lactobacillus (Xu et al., 2019). A study performed high-throughput sequencing of the V3 and V4 regions of 16S rRNA sequencing to study the enrichment of gut bacteria in infants, adults, and elderly people, and the analysis was made based on the bacterial co-abundance group (CAG), which is defined by the Kendall rank correlation coefficient between the bacterial genera. It was observed that the transition of different CAGs from infants to elderly people is dominated by distinctive bacterial CAGs, such as elderly-linked CAGs dominated by Clostridiaceae and Bacteroides; Lachnospiraceae CAGs are abundant in adult-linked CAGs; Enterobacteriaceae CAGs are predominantly abundant in infant and elderly-linked CAGs; and Bifidobacterium CAGs are abundant in infant-linked CAGs (Odamaki et al., 2016).
Aging-related declines in microorganism levels, including Bacteroides, Faecalibacterium, Bifidobacterium, and Clostridium cluster XIVa, were observed in another study, consistent with prior research (Candela et al., 2014; Mäkivuokko et al., 2010; Salazar et al., 2013). Biagi et al. (2010) reported that in centenarian people, there is increased abundance of pathogenic bacteria belonging to the Proteobacteria phylum, whereas they found lower abundance of several butyrate-producing bacteria such as Ruminococcus obeum, Roseburia intestinalis, Eubacterium ventriosum, Eubacterium rectale, and Eubacterium hallii, all belonging to the Clostridium cluster XIVa, and Papillibacter cinnamovorans and Faecalibacterium prausnitzii of the Clostridium cluster IV. Conversely, they also found an increased abundance of several butyrate-producing bacteria Anaerotruncus colihominis and Eubacterium limosum (Biagi et al., 2010). So, dysbiosis, a condition of microbial imbalance that can contribute to the development of a number of age-related disorders, including CLD, might result from changes in the makeup of the gut microbiota that are age-related. Dysbiosis has been proven to aid in the onset and progression of cirrhosis by promoting oxidative stress and inflammation, which are late-stage liver diseases defined by the formation of scar tissue in the liver. Dysbiosis can result in greater intestinal permeability, which makes it possible for toxins, byproducts of bacteria, and other hazardous chemicals to enter the circulation. This can cause oxidative stress and chronic inflammation, both of which are key factors in liver failure (Meng et al., 2022). A reduction in immunological function is one of the main causes of age-related alterations in the composition of the gut microbiota. Dysbiosis is brought on by the immune system’s decreasing ability as we age to manage bacterial growth in the gut. Alterations in gut motility and secretory function brought on by aging can potentially contribute to the emergence of dysbiosis (Maynard & Weinkove, 2018; Salazar et al., 2019).
SCFA (such as acetate, propionate, and butyrate) generation can modulate the gut microbiota by lowering pH, protecting against pathogen overgrowth, and stimulating the development of good bacteria pertaining to the phylum Firmicutes (Duncan et al., 2009; Zimmer et al., 2012). Moreover, SCFAs play a major role in modulating intestinal transit time, insulin response, and immune response through the inhibition of the generation of various inflammatory mediators such as tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and nitric oxide (NO) and stimulation of IL-10 secretion (Gao et al., 2009; Vinolo et al., 2011).
A healthy gut microbiota composition may be maintained and the risk of age-related illness can be decreased by preventative strategies such as dietary changes, exercise, and probiotic supplementation. In addition, therapeutic approaches like fecal microbiota transplantation (FMT) may help in the treatment of dysbiosis and related conditions such as CLD (Salazar et al., 2019).
Claesson et al. (2012) discovered that a decrease in microbial diversity was associated not only with diet but also with increased frailty, markers of inflammation, and poorer health indices. Residence location and antibiotic therapy also had an impact on gut microbiota modification (Albouery et al., 2019).
In fact, alterations in the makeup of the gut microbiota that are brought on by aging may be a factor in a number of health problems, such as immune system problems, metabolic abnormalities, and digestive problems. To properly comprehend the effects of these modifications and to create therapies that would support a healthy gut microbiota in aging populations, more study is nonetheless required (Figure 1) (Al Bander et al., 2020).
Chronic Liver Disease Due to Aging-mediated Alterations of Gut Microbial Taxa and Compositions. Aging Causes Decreased Production of SCFA such as Acetate, Propionate, and Butyrate and Altered Bile Acid and Choline Metabolism and Promotes Oxidative Stress and Chronic Inflammation, Which Ultimately Causes Gut Dysbiosis by Fostering the Growth of Pathogenic Bacteria and Suppressing the Beneficial Bacteria; as a Result, Chronic Liver Disease Occurs.
Management of Chronic Liver Dysfunction by Targeting Gut Microbiota
Dietary Modification
The gut microbiome has seven times more genetic material than humans, providing many therapeutic possibilities (Qi et al., 2020). It is reported that the abundant intake of branched-chain amino acids (BCAAs) such as valine, leucine, and isoleucine and proteins derived from vegetables are beneficial for cirrhosis patients. Dietary intake of high amounts of BCAAs may decrease the chances of liver failure with advanced-stage cirrhosis and reduce the frequency of hepatic encephalopathy (HE) (Fukushima et al., 2003). Deficiencies of vitamins and minerals are also noticeable in CLD patients, and the supplementation of thiamine, vitamin K, and fat-soluble vitamin can be used against the severely alcoholic, cholestatic, and hemorrhagic situation of the liver, respectively (Atarashi et al., 2017; Satish et al., 2021). Studies also reported that vitamin D and calcium may be used as supplementation against cholestasis (Francis, 2006). SCFAs can inhibit histone deacetylase and activate several G-protein-coupled receptors such as GPR41, GPR43, and GPR109a to regulate liver metabolism and immune function. Additionally, acetate and propionate have been found to suppress the lipogenesis pathway of liver and lipid accumulation (Sahuri-Arisoylu et al., 2016). Butyrate was found to inhibit steatohepatitis via the modulation of gut microbiota composition, enhancement of glucagon-like peptide-1 receptors (GLP-1R), and reduction of inflammation and oxidative stress-mediated damage of the liver (Jin et al., 2015).
Antibiotics
Antibiotics can potentially reduce the level of harmful bacteria and the inflammatory signal from the gut microbiota. Rifaximin is one of the potent antibiotics that promote the growth of good bacteria. In contrast, it reduces the level of harmful bacteria (Zhang et al., 2015). It also inhibits toxin secretion from intestinal microbiota and reduces pro-inflammatory cytokine levels (Zhang et al., 2015). Bajaj et al. (2013) reported that treating rifaximin in cirrhosis patients reduces the Veillonellaceae level and increases the Eubacteriaceae level.
Probiotic
Many studies have reported that probiotics improve liver health by promoting the generation of SCFAs to suppress apoptosis, improve intestinal epithelial permeability, and repair damaged mucosal barriers (Kanauchi et al., 1999). The probiotic Lactobacillus GG (LGG) was used against the alcoholic steatohepatitis rat model, and it was found that it improved liver injury via the reduction of intestinal leakage and inhibited the generation of intestinal oxidative stress (Forsyth et al., 2009). The use of Lactobacillus casei Shirota against alcoholic cirrhosis was found to alter the secretion of IL-10 and TL-4 expression to restore phagocytosis by neutrophils (Stadlbauer et al., 2008). A phase I study was done on cirrhosis patients to study the effect of probiotic LGG and reported that LGG can alter the microbiota, reduce the secretion of TNF-α and endotoxin, and reduce gut dysbiosis (Bajaj, Heuman, Hylemon, Sanyal, Puri, et al., 2014). Probiotics’ efficacy in treating liver cirrhosis may be classified into two categories: enhancing liver function and reducing pathogen infection, hepatic encephalopathy, and other consequences (Liu et al., 2022).
Prebiotics
Prebiotics are nondigestible but fermentable dietary compounds fermented by bacteria that aid in peristaltic digestion and selectively increase the proliferation of gut bacteria. Some prebiotic foods, such as lactulose and pectin, can acidify the intestine, block urease-producing bacteria, and reduce ammonia transport into the blood (Zhao et al., 2015). The primary treatment for HE patients is targeting the gut to lower colonic bacterial production and blood ammonia levels. Pectin may restore the Bacteroides level and inhibit liver damage in mouse models (Rocco et al., 2021). One meta-analysis study reported that prebiotic treatment improves the health of NAFLD patients by reducing serum cholesterol, liver enzymes, and triglycerides and regulating the gut flora balance (Phongsamran et al., 2010). Fructooligosaccharide (FOS) has the potential to decrease cholesterol accumulation and also facilitate fatty acid oxidation via the upregulation of peroxidase expression and downregulation of sterol regulatory element-binding protein-2 (SREBP-2) in the liver (Ferrere et al., 2017).
Synbiotics
Synbiotics, which consist of good bacteria in combination with both probiotics and prebiotics, play a role in regulating the functional gene expression of intestinal flora. It is reported that synbiotics may have the potential to reduce insulin resistance by modifying the gut flora (Ghosh et al., 2022). Additionally, they reduce the inflammatory response, fecal SCFA level, and colitis reaction in the liver and promote the tight junction of the gut barrier and fatty degeneration. One clinical study reported that after treatment of NASH patients with a combination of both Bifidobacterium longum and FOS for six months, a significant reduction of serum aspartate aminotransferase (AST), LPS, lipid denaturation, NASH activity index, and homeostasis model assessment of insulin resistance (HOMA-IR) was observed (Malaguarnera et al., 2012). The two potential mechanisms of synbiotic influence on liver disease are the changes in gut microbiota structure and alterations of gut microbiota–derived metabolite, which have anti-inflammatory and immunomodulatory effects. There have been no known side effects with this method (Liu et al., 2022).
Fecal Microbiota Transplantation
FMT is the process of transplantation of bacteria from the fecal matter of a healthy donor to the recipient (Xu et al., 2021). FMT restores intestinal floral health by producing antimicrobial peptides; minimizing the transfer of endogenous ethanol, endotoxin, and other metabolites to the liver; reducing metabolic chemical-mediated liver injury; and effectively reducing inflammatory signals and cell proliferation (Liu et al., 2022). One study reported that transplantation of FMT from alcohol-resistant mice to alcohol-sensitive mice prevented alcohol-induced injury of the intestine’s fatty liver to some extent. It was also noticed that there was a decrease in the Bacteroides level and an increase in the level of Actinobacteria and Firmicutes (Shasthry, 2020). Another study reported that transplantation of fecal bacteria improved gut health, enhanced intestinal epithelial barrier function, and reduced liver steatosis in high-fat-diet-induced NAFLD mice (Zhou et al., 2017). As a result, FMT can be utilized to cure and manage CLD effectively. According to preliminary fundamental studies, FMT can replenish probiotics in the gut ecology (Lechner et al., 2020; Liu et al., 2021). In a recent study, FMT from donors enriched with Lachnospiraceae and Ruminococcaceae has reduced serum IL-6- and LPS-binding protein levels while increasing butyrate, suggesting fecal bacteria transplantation as a promising and safe therapy for cirrhosis (Bajaj et al., 2021).
Promising Therapeutic Strategies Against NAFLD
According to research, the treatment goals of NAFLD fall into four classes: lipotoxicity and cell death, metabolism of carbohydrates and lipids, extracellular matrix deposition (ECM) and anti-fibrosis, and inflammation (Inci et al., 2023).
Lipotoxicity and Cell Death–based Agents
The mediators that cause lipotoxicity include saturated free fatty acids (FFAs), unesterified cholesterol, diacylglycerols, sphingolipids, ceramide, and lysophosphatidylcholine (Farrell et al., 2018). Lipotoxicity-mediated ER stress by continuous activation of the unfolded protein response (UPR) and mitochondrial dysfunction cause an imbalance in oxidant and antioxidant levels, promoting oxidative stress, which further activates c-Jun N-terminal kinase (JNK) and other signaling pathways to induce apoptosis (Arroyave-Ospina et al., 2021). Both ER stress and oxidative stress together can cause the occurrence and development of NAFLD (Fujii et al., 2018). In the reduction of oxidative stress and hepatic inflammation, vitamin E is a potential antioxidant (Nagashimada & Ota, 2019). One meta-analysis study reported that vitamin E administration in NAFLD patients significantly reduced liver enzymes (such as AST and ALT). The anti-inflammatory, antioxidant, and anti-apoptotic properties of vitamin E make it a potential therapeutic approach for NAFLD patients (Perumpail et al., 2018). Silymarin is a potent bioactive compound that has antioxidant, anti-inflammatory, and anti-fibrosis properties that can alleviate the manifestations of NAFLD patients by reducing the levels of glucose-regulated protein 78 (GRP78) and X-box binding protein 1 (XBP-1), the common ER stress proteins (Sahin et al., 2020). A meta-analysis study reported that, compared to placebo treatment, silymarin treatment in NAFLD patients showed a significant reduction of the transaminase level (Kalopitas et al., 2021). Apart from this, numerous drugs are also available to target lipotoxicity and cell death, such as ornithine aspartic acids, resveratrol, niacin, anthocyanin, piceatannol, and polydatin, which target oxidative stress and reduce inflammation and fibrosis (Fu et al., 2022).
Carbohydrate and Lipid Metabolism–based Agents
The drugs that target carbohydrate and lipid metabolism are the most potent therapeutic approaches for treating NAFLD. The lowering of intrahepatic FFAs is a possible NAFLD therapeutic option, which can be accomplished by enhancing insulin sensitivity, fatty acid oxidation, or fatty acid export and storage by peripheral tissues, as well as lowering de novo lipogenesis (DNL) (Neuschwander, 2010).
The use of FXR agonists such as a steroidal agonist, obeticholic acid (OCA), has already been used in phase III clinical trials of NASH treatment but has not been approved by the FDA. In contrast, tropifexor is a nonsteroidal agonist, and a phase II clinical trial is ongoing to treat NASH and primary biliary cirrhosis (Koutsounas et al., 2015; Kowdley et al., 2020). Peroxisome proliferator-activated receptors (PPARs) primarily belonging to the nuclear receptor family have three subclasses, PPAR-α, -β, and -γ, which are found to be involved in fatty acid absorption, metabolism, lipid transport, α-oxidation, and β-oxidation; deploying anti-inflammatory effects; and improving insulin sensitivity, respectively. Therefore, the development of PPAR agonists could be effective against NAFLD and NASH (Fernández-Miranda et al., 2008). PPAR-γ agonists such as pioglitazone and PXL065, which is an R-stereoisomer of pioglitazone, are very much effective against NAFLD and NASH; saroglitazar is both a PPAR-α and a PPAR-γ agonist (Kumar et al., 2020), and elafibranor is a PPAR-α and -δ agonist (Ratziu et al., 2016); they are very much effective against NAFLD and NASH. Glucagon-like peptides enhance insulin secretion, and therefore, GLP-1R agonists such as liraglutide and semaglutide are used as antidiabetic drugs potentially effective against NAFLD (Armstrong et al., 2016; Newsome et al., 2021). N-3 polyunsaturated fatty acids (n-3 PUFA) effectively treat NAFLD by inhibiting the DNL of hepatocytes and promoting fatty acid oxidation (Climax et al., 2020). Various agonists of thyroid hormone receptor-β (THR-β), such as resmetirom, VK2809 (MB 07811), ASC41, and TERN501, to treat NAFLD are now being developed (Kim et al., 2018).
Aramchol (3β-arachidonoyl amide cholic acid) suppresses the expression of hepatic stearoyl-CoA desaturase (SCD1) (Bhattacharya et al., 2021) and is found to improve steatohepatitis and fibrosis in animal studies (Aljohani et al., 2019), and a clinical trial study reported it to be safe to use as it efficiently reduces fat accumulation in NAFLD patients (Fu et al., 2022).
Inflammation-based Agents
Numerous drugs are already discovered by targeting inflammatory markers and inflammation in the liver, mainly caused by macrophages. Infiltration of Ly-6C + monocyte is one of the crucial factors in the development of NAFLD by facilitating hepatitis and subsequent progression of fibrosis (Soleimani et al., 2019). The hepatic macrophages of NASH patients show an apparent inflammatory character, which may be due to an excessive accumulation of lipids and FFAs (Jindal et al., 2015). Selonsertib potentially inhibits the apoptotic signal-regulated kinase 1 (ASK1) and regulates the p38 kinase and JNK via the mitogen-activated protein kinase (MAPK) pathway, which involves pro-inflammatory and fibrotic changes. Selonsertib has alleviated liver fibrosis in many NASH patients with stage 2 or 3 fibrosis, showing its promise as a future NAFLD therapy (Harrison et al., 2020). Namodenoson, an A3 adenosine receptor agonist, improves ALT and AST levels, reduces fat accumulation and inflammation, and benefits NAFLD patients with cardiovascular and diabetic comorbidities (Cross et al., 2002; Safadi et al., 2021).
Promising Therapeutic Strategies for ALD
In the therapy of AH, corticosteroids and pentoxifylline are utilized, both of which try to alleviate inflammatory conditions. Corticosteroids suppress cytokine synthesis by transcriptional regulation, while pentoxifylline has a similar impact via phosphodiesterase (PDE) inhibition (Taïeb et al., 2000; Yan et al., 2021).
Glucocorticoid is one of the best treatment approaches against severe and acute alcoholic hepatitis (SAH and AAH), which can inhibit inflammation by suppressing the immune response and pro-inflammatory cytokine secretion (Taïeb et al., 2000). Metadoxine can treat acute alcoholism via promoting ethanol excretion and lowering serum ethanol levels, but it is unable to treat hepatic steatosis or improve inflammation (Vonghia et al., 2008). Additionally, some natural compounds are also effective in treating ALD by suppressing oxidative stress and apoptosis, inhibiting inflammation, and improving lipid metabolism. Curcumin, dihydroquercetin, and tanshinone IIA potentially improve lipid metabolism via the regulation of NF-E2-related factor 2 (Nrf2)-FXR pathway, SIRT1-AMPK pathway, and LXRα/SREBP1 pathway (Lu et al., 2015; Zhang et al., 2018; Gao, Chen, et al., 2021). Silymarin, isoorientin, and oleanolic acids are essential natural compounds that suppress oxidative stress. Anthocyanin, baicalin, puerarin, sophoronol, and catechin are involved in the suppression of inflammation. Programmed cell death inhibitor compounds are corosolic acid, gastrodin, and ursolic acid (Yan et al., 2021). Berberine is one of the bioactive compounds that can increase the level of Akkermansia muciniphila bacteria to regulate metabolic disbalance and reduce inflammation (Li et al., 2020). Naringenin, naringin, hesperidin, quercetin, dihydromyricetin, glycycoumarin, lycopene, and limonoids have multiple therapeutic mechanisms to treat ALD (Yan et al., 2021).
Promising Therapeutic Strategies for NASH
Cenicriviroc has a great impact on the regulation of inflammation of NASH patients and has been found to reduce circulatory biomarkers such as C-reactive protein (CRP), soluble cluster differentiation 14 (CD14), IL-6, and IL-10 (Friedman et al., 2018). Elafibranor, a dual PPAR/agonist, is another potential medication currently being studied for treating NASH. The GOLDEN-505 clinical trial study found improvement in steatosis; reduction in the level of inflammatory markers such as CRP, haptoglobin, and fibrinogen in serum; reduction of liver fibrosis; and improvement in liver enzyme levels such as ALT, ALP, and gamma-glutamyl transpeptidase (GGTP) and lipid profile such as LDL, HDL, and triglyceride in NASH patients after elafibranor treatment (Ratziu et al., 2016). Obeticholic acid, a potent FXR agonist, can reduce liver fibrosis, steatosis, hepatocyte ballooning, hepatic inflammation, and serum ALT level. It can be used to treat NASH patients with fibrosis (Abenavoli et al., 2018; Roy et al., 2022). Pioglitazone and vitamin E have shown greater efficacy in the treatment of both diabetic and prediabetic NASH patients by reducing GGTP, AST, ALT, and ALP levels in serum, serum triglyceride level, and insulin resistance. GLP-1 agonist is a potent therapeutic option to treat NASH patients. Liraglutide is a GLP-1 analog that improves lipid transport, DNL, β-oxidation, reduction of fibrosis, liver enzymes in serum, and lipid profile (Nevola et al., 2023).
Promising Therapeutic Approach to Cirrhosis
Cirrhosis of the liver is the last stage of CLD, characterized by irreversible extracellular matrix deposition and structural damage to the liver (Cai et al., 2021). Cellular cross-talk, microenvironmental signals, and intracellular signaling are important in developing liver fibrosis and cirrhosis (Gao, Wei, et al., 2021). In this issue, two reviews discussed how the critical kinds of liver cells, such as HSCs, hepatocytes, liver sinusoidal endothelial cells, cholangiocytes, and inflammatory cells, contribute to the pathophysiology and development of liver cirrhosis. The vital signaling pathways that lead to hepatic fibrosis and potential treatment targets have been fully characterized (Gu et al., 2021; Zhang et al., 2021). Furthermore; Li et al. (2022) emphasized the role of mitochondrial dysfunction and hypoxia-inducible factor-1 (HIF1)–induced oxygen imbalance in liver fibrosis metabolism and immunity.
Current therapies, such as tenofovir disoproxil fumarate (TDF) for anti-HBV infection, have shown promise in relieving liver fibrosis via antiviral-independent mechanisms. According to genomic research, TDF may alter the hepatic immune response and metabolic processes via mmu-miR-155-5p-NF-B signaling (Duan et al., 2021).
Conclusion
The gut microbiota influences liver function through multiple mechanisms, including the production of metabolites (bacterial toxin, LPS, SCFA, fructose, and glucose), regulation of the immune system, and metabolism of bile acids, and the age-related alterations of these factors can induce gut dysbiosis, which ultimately leads to chronic inflammation in liver, causing the development of ALD, NAFLD to NASH, and more severe liver cirrhosis and liver fibrosis.
To prevent and manage CLD associated with changes in the gut microbiota, it is necessary to employ strategies that restore microbial balance and reduce inflammation. These strategies may involve dietary modifications such as incorporating prebiotics, probiotics, and synbiotics and using antibiotics that promote the growth of beneficial gut bacteria. Additionally, FMT and clinical trial–tested bioactive compounds offer hope for restoring gut microbiota and treating dysbiosis-related conditions, such as NAFLD, ALD, NASH, and cirrhosis.
Future Directions
The human gut is the habitat for trillions of gut bacteria and also allows their diversification to create a complex community. This complex community regulates normal physiology and disease susceptibility through their metabolic activity and host interactions. Our primary concern is to understand the mechanism of gut dysbiosis and its functional consequences that cause CLD. An in-depth study is still required to explore the dysbiosis mechanism and its impact on intestinal homeostasis to create a condition for the occurrence of CLD beyond causing an increase in intestinal epithelial permeability. More studies are required to discover the mechanism of involvement of genetic factors, such as single-nucleotide polymorphisms, gene copy numbers, and so forth, in shaping gut microbial composition and disease susceptibility or resistance (Ghosh et al., 2022).
The extensive integration of multiple symptom shifts in gene expression, gut flora, and metabolic traits associated with the discovered NAFLD subtypes will influence the extent to which progress can be achieved in the future. Based on different traits and the application of contemporary tools to directly interfere with specific gut bacteria, this will deliver new insights as well as better treatment for CLD. In the years to come, it will be crucial to comprehend whether CLD results from changes in the gut microbiota and its metabolites in relation to the gut-liver axis. Further research is necessary to elucidate the underlying causes of CLD from innovative perspectives centered on discrepancies in the gut microbiota and metabolites, along with generating novel therapeutic strategies based on research findings.
Highlights
A comprehensive explanation of how aging and gut microbiota relate has been provided.
Studies on the association between the gut microbiome and liver diseases have been related and explained.
Therapy-based studies have been analysed for NAFLD, ALD, NASH, and cirrhosis. Alterations in the gut microbiota composition are associated with the onset and progression of various chronic liver diseases, including ALD, NAFLD, and cirrhosis.
Dysbiosis is characterized by microbial imbalance, contributes to various age-related disorders including chronic liver disease (CLD) by promoting oxidative stress and inflammation.
Age-related alterations in gut microbiota composition are partly due to declines in immunological function and alterations in gut motility and secretory function.
Footnotes
Acknowledgments
The authors are thankful to the First Affiliated Hospital of Heilongjiang University of Chinese Medicine, Heilongjiang, China, for providing the infrastructural support.
Authors’ Contributions
Conceptualization and editing of the MS: Gang Zhao; writing original draft: Han Yu, Zhijun Wang. The content of the submitted manuscript was approved by all the authors. Han Yu and Zhijun Wang contributed equally.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
