Abstract
Background
Beta-amyloid peptide (Aβ) induces oxidative stress, contributing to Alzheimer’s disease (AD) initiation and progression. This study aims to explore Thunbergia laurifolia (T. laurifolia) leaf extract’s protective effects on Aβ25–35-induced oxidative stress and cell injury in SH-SY5Y cells and investigate underlying mechanisms.
Materials and Methods
SH-SY5Y cells were treated with T. laurifolia leaf extract in the presence or absence of Aβ25–35. After 24 h, neuroprotective effects were assessed using cell viability and lactate dehydrogenase (LDH) assays. Caspase-3/7 activity, intracellular reactive oxygen species (ROS) levels, catalase (CAT), and superoxide dismutase (SOD) activities were measured to examine mechanisms. Total flavonoid and phenolic content assays were performed.
Results
The findings showed that exposure to Aβ25–35 led to a notable rise in oxidative stress in SH-SY5Y cells, as evidenced by increased levels of ROS. Additionally, Aβ25–35 treatment increased caspase-3/7 activity and LDH release and decreased cell viability. However, T. laurifolia extract effectively suppressed ROS production, attenuated caspase-3/7 activity, and concentration-dependently reduced Aβ25–35-induced neurotoxicity. LDH release decreased, and cell viability increased. SOD and CAT activities also increased after T. laurifolia treatment. The extract had total phenolic and flavonoid contents of 178.5 ± 6.86 and 32.51 ± 1.26 mg/g, respectively.
Conclusion
T. laurifolia extract demonstrated neuroprotective effects against Aβ25–35-induced injury in SH-SY5Y cells. These effects were attributed to reduced oxidative stress, elevated SOD and CAT activity, and suppressed caspase-3/7 activity. T. laurifolia extract shows potential as an alternative or therapeutic approach to AD mediated by Aβ. Nevertheless, further research is needed to elucidate the mechanism by which T. laurifolia ameliorates neuronal cell death induced by Aβ25–35.
Introduction
Alzheimer’s disease (AD) is a progressive disorder that causes neuronal cell death which mostly affects the elderly. Aging is considered to be a very important risk factor for the disease. As a result of a lack of effective treatments, AD has become the most destructive disease in the world. The pathology of AD is characterized by the deposition of extracellular amyloid beta (Aβ) peptides and intracellular accumulation of neurofibrillary tangles primarily composed of hyperphosphorylated tau, together with increased neuroinflammation as well as neuronal loss (Bronzuoli et al., 2016; Raskin et al., 2010). Although the exact etiology of AD remains obscure, the accumulation of Aβ has been proposed as one of the key roles in the disease onset. Several studies have demonstrated that the excessive production of Aβ induces free radical oxidative stress, inflammation, an increase in intracellular Ca2+, and neurotoxicity (Butterfield et al., 2001, 2002, 2013; Bharadwaj et al., 2009; Gray & Patel 1995; Muthaiyah et al., 2011). Thus, the therapeutic approach in the prevention and treatment of AD is to find substances or herbs that can attenuate Aβ-induced oxidative damage.
Thunbergia laurifolia Lindl. (T. laurifolia) or Rang Chuet is classified as a member of the family Acanthaceae and is widely distributed in the north of Thailand. Various parts of the plant possess medicinal properties and are used in traditional medicine. The major constituents of T. laurifolia leaf extract include phenolic compounds, such as caffeic acid, protocatechuic acid, chlorogenic acid, rosmarinic acid, and vinixin. There are also reported findings of such flavonoids as apigenin, catechin, isoquercetin, quercetin, and rutin in T. laurifolia leaf extract (Chuthaputti 2010; Junsi et al., 2017; Oonsivilai et al., 2007; Preechasuk et al., 2020; Woottisin et al., 2020;). Additionally, chlorophyll derivatives have an established bioactivity that is consistent with detoxification attributed to T. laurifolia leaf (Fahey et al., 2005). In Thai traditional medicine, T. laurifolia leaves are used as an antidote and treatment for drug addiction (Thongsaard & Marsden, 2002; Thongsaard et al., 2005). The plant has also been reported to have anti-microbial, anti-inflammatory, anti-diabetic, and anti-pyretic properties as well as detoxifying and non-toxic effects (Aritajat et al., 2004; Chivapat et al., 2009; Kanchanapoom et al., 2002). Recently, we demonstrated that leaves of T. laurifolia exert anti-oxidative and anti-inflammatory activity through the inhibition of Akt and the ERK1/2-mediated NF-κB pathway in LPS-stimulated BV2 cells (Mairuae et al., 2020). While various medicinal properties of T. laurifolia leaf have been discovered, the impact of T. laurifolia leaf extract on Aβ-induced oxidative stress remains unexplored. Hence, the objective of this study is to explore the neuroprotective effects and the antioxidant mechanism of T. laurifolia leaf extract when exposed to Aβ in neuroblastoma SH-SY5Y cells. The total phenolic and flavonoid contents of the plant extract were also determined.
Materials and Methods
T. laurifolia Leaf Extraction
T. laurifolia leaves were obtained in Mahasarakham Province, Thailand. Identification was performed by Mahasarakham University (MSU), Faculty of Medicine, Division of Applied Thai Traditional Medicine. An herbarium specimen, no. MSUT_7233 was deposited at the Faculty of Science. The extraction of T. laurifolia was described in a previous publication (Mairuae et al., 2020). Initially, T. laurifolia leaves were dried, weighed, and chopped to prepare an ethanolic extract. Next, they were macerated in 95% (v/v) ethanol at room temperature for 7 days. Subsequently, the extract underwent filtration, concentration via a rotary evaporator, and ultimately, lyophilization.
Cell Cultures Assay
Neuroblastoma SH-SY5Y cells were cultured in DMEM medium supplemented with 10% FBS, 1% penicillin-streptomycin, and 1% non-essential amino acids. They were kept in a humidified atmosphere with 5% CO2 at 37°C. The cell density for each experiment was appropriately adjusted. Before starting each experiment, the culture medium in each well was entirely replaced with fresh medium containing Aβ, with or without T. laurifolia leaf extract. To assess the cytotoxicity of Aβ25–35 and T. laurifolia leaf extract, the MTT assay was employed.
Cell Cytotoxicity Assay
The MTT assay was employed to assess the in vitro cytotoxicity of Aβ25–35 and T. laurifolia leaf extract. SH-SY5Y cells were seeded in 96-well plates at a density of 1 × 104 cells per well and cultured as described above. These cells were exposed to varying concentrations of Aβ25–35 (10–30 µM) and T. laurifolia leaf extract (0–100 µg/mL) in an FBS-free medium for 24 h. To investigate the protective effects of T. laurifolia leaf extract against Aβ-induced neurotoxicity, cells were treated with Aβ25–35, both with and without the addition of T. laurifolia leaf extract, for 24 h. Following this treatment period, the medium was replaced with an MTT reagent at a final concentration of 0.5 mg/mL. The cells were subsequently incubated for 2 h at 37°C in a 5% CO2 incubator. After incubation, the MTT reagent was removed, and 100 µL of dimethyl sulfoxide was added to dissolve the insoluble purple formazan product. Absorbance was measured at 570 nm using a Synergy-4 plate reader (BioTek Instruments, Inc., Winooski, VT, USA). The results were expressed as a percentage of the control.
Lactate Dehydrogenase (LDH) Assay Analysis of Cell Damage
A density of 1 × 104 cells/well was used to plate the cells in 96-well plates. Subsequently, the cells were cultured and subjected to the treatments as described above. Following a 24-h incubation period, the extent of Aβ-induced cell injury (20 µM) was assessed by measuring the amount of intracellular enzyme LDH released into the culture medium, as previously described in another study (Mairuae et al., 2019). Briefly, culture medium (100 µL) was collected from each well and transferred to a new 96-well plate, and 100 µL reaction mixture was added to each well and incubated for 30 min at 37℃. Absorbance was measured at 492 nm using a microplate reader. The quantity of LDH released was then expressed as a percentage of the untreated control.
Caspase-3/7 Activity Detection in Cell Culture
Caspase-3/7 activity was assessed using Caspase-Glo® 3/7 kits from Promega (Madison, WI, USA) following the manufacturer’s guidelines, as described previously (Mairuae et al., 2019). Briefly, the Caspase-GloR 3/7 buffer and lyophilized Caspase-GloR 3/7 substrate were brought to room temperature. The Caspase-GloR 3/7 buffer was then transferred to the vial containing the Caspase-GloR 3/7 substrate, and equal volumes of this reaction mixture were added to the samples. After a 30-min incubation period, luminescence measurements were taken, and the results were expressed as a percentage relative to the untreated control.
Intracellular ROS Assay
Cell plating was done in 96-well plates at a density of 1 × 104 cells per well, and they were then cultured and treated as explained earlier. After 24 h of treatment, the fluorescent probe 2′,7′dichlorofluorescein was used to measure intracellular ROS levels. Briefly, the cells were cultured with 10 µM DCFH-DA for 20 min at 37℃ in a 5% CO2 incubator. Then, the cells were treated with a medium containing Aβ25–35 (20 µM), in the presence or absence of T. laurifolia extract for 24 h. Subsequently, fluorescence intensity was measured at excitation and emission wavelengths of 495 and 525 nm, respectively.
Superoxide Dismutase (SOD) and Catalase (CAT) Activity Assay
In 96-well plates, cells were seeded at a density of 1 × 105 cells/well, after which they were grown and treated as previously mentioned. After 24 h of treatment, superoxide dismutase activity and catalase activity were examined by using a kit from Biovision Inc. and Cayman Chemical Company, respectively. The methods were as described previously (Mairuae et al., 2019). Briefly, after 24 h of treatment, the cells were harvested using a rubber policeman and gathered through centrifugation (2,000× g for 10 min at 4°C). The resulting cell pellets were homogenized in a cold assay buffer and then subjected to centrifugation at 10,000× g for 15 min at 4°C. The supernatant was collected for subsequent analysis. Superoxide dismutase (SOD) and catalase (CAT) activity were determined using a commercially available assay kit in accordance with the manufacturer’s instructions. The results were expressed as a percentage relative to the untreated control.
Determination of Phenolic and Flavonoid Content
The phenolic and flavonoid compounds of the T. laurifolia extract were examined using the aluminum chloride colorimetric method and the Folin‑Ciocalteu method, respectively. The total flavonoid content in the T. laurifolia leaf extract was determined using the aluminum chloride colorimetric method. In summary, 100 µL of 1 mg/mL T. laurifolia leaf extract was blended with 0.9 mL of a flavonoid mixture (comprising 10% aluminum hydroxide and 1 M potassium acetate, with dilutions of 0.1, 0.1, and 4.3 mL). This mixture was then incubated for 30 min at room temperature in the absence of light, and the absorbance intensity was subsequently measured at 450 nm. The total flavonoid content was calculated based on a calibration curve, and the results were expressed as mg of rutin equivalent per gram of dry weight.
The total phenolic content in the T. laurifolia leaf extract was determined using the Folin-Ciocalteu method. In summary, 100 µL of 1 mg/mL T. laurifolia leaf extract was thoroughly mixed with 0.45 mL of the Folin-Ciocalteu reagent for 5 min, followed by the addition of 0.45 mL of 60 g/L sodium bicarbonate. The mixture was then left in the dark for an additional 1 h at room temperature, and the absorbance intensity was measured at 650 nm. The total phenolic content was calculated from the calibration curve, and the results were expressed as mg of gallic acid equivalent per gram of dry weight.
Statistical Analysis
A one-way ANOVA assay followed by the Bonferroni post hoc test was used to compare between control and treatment groups. A p-value ≤0.05 was considered significant.
Results
Aβ25–35-induced Toxicity in SH-SY5Y Cells
Twenty-four hours after treatment with Aβ, cell toxicity was measured by MTT assay. As shown in Figure 1, Aβ25–35 treatments were toxic to cells at a concentration of 20 µM. Aβ 20 µM concentration of Aβ25–35 was therefore used in subsequent assays.
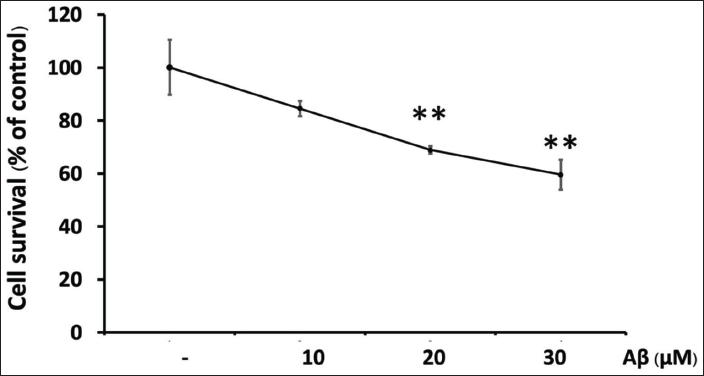
Effects of T. laurifolia Leaf Extract in SH-SY5Y Cells
T. laurifolia at concentrations ranging from 0 to 100 µg/mL were applied to SH-SY5Y cells to determine the potential cytotoxic effect. Cell viability was evaluated by MTT assay after 24 h of treatment. The results indicated that T. laurifolia extract was non-toxic to cells at concentrations up to 100 µg/mL (Figure 2). Therefore, 50 and 100 µg/mL of T. laurifolia extract was used in subsequent assays.
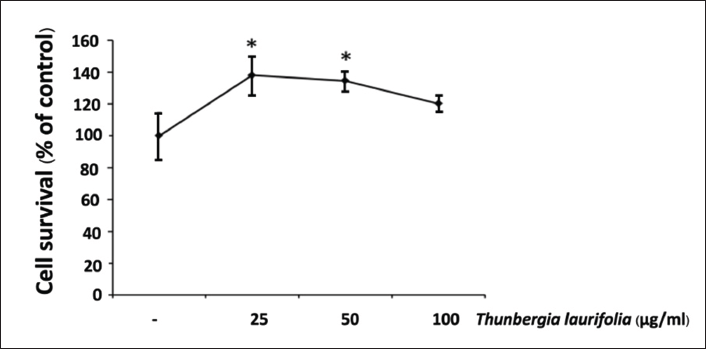
Aβ25–35-induced Cytotoxicity was Prevented by T. laurifolia Leaf Extract in SH-SY5Y Cells
The cytoprotective effect of T. laurifolia extract against Aβ25–35-induced cytotoxicity was examined by treating SH-SY5Y cells with 20 µM of Aβ25–35 in the presence or absence of T. laurifolia extract for 24 h. As shown in Figure 3, the treatment of SH-SY5Y cells with 20 µM Aβ25–35 for 24 h resulted in cytotoxicity, as observed by reduced cell viability compared with the control group. Conversely, when the cells were exposed to T. laurifolia extract, cell viability increased, suggesting a concentration-dependent cytoprotective effect. As an additional measure to confirm the cytoprotective effect of T. laurifolia extract, LDH was assayed, being another marker of cell toxicity. The results were in line with those obtained from the MTT assay. After exposing SH-SY5Y cells to 20 µM Aβ25–35, there was a notable increase in LDH release into the medium compared with the control group. Nonetheless, treatment with T. laurifolia extract exhibited a concentration-dependent decrease in Aβ25–35-induced LDH release (Figure 4).
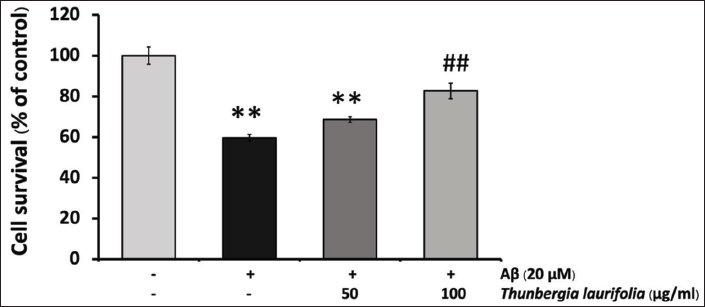
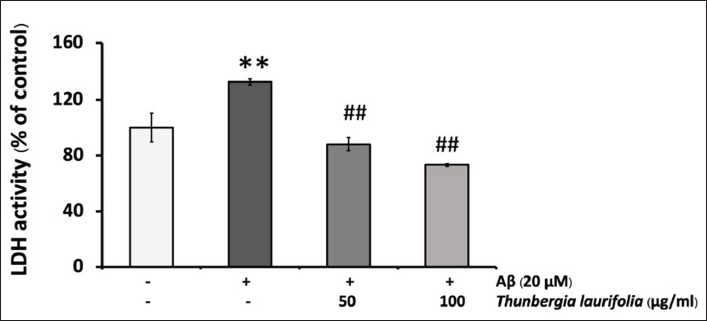
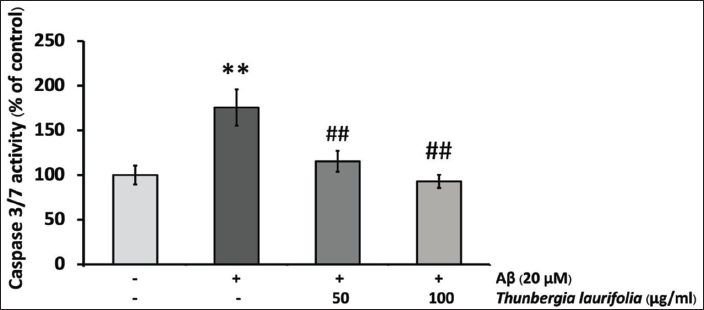
T. laurifolia Leaf Extract Reduced Aβ25–35-induced Caspase-3/7 Activity
Numerous studies have documented that Aβ-induced neuronal cell death involves the activation of the caspase pathway. In our investigation of the protective mechanism of T. laurifolia extract, we measured caspase-3/7 activity. Upon Aβ25–35 treatment, there was a substantial increase in caspase-3/7 activity. Nevertheless, the presence of T. laurifolia extract effectively blocked the induction of caspase-3/7 activity (Figure 5).
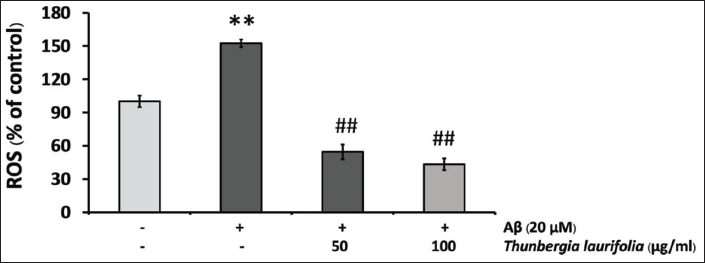
Effects of T. laurifolia Leaf Extract on ROS Production Induced by Aβ25–35
To evaluate the effect of T. laurifolia extract on Aβ25–35-induced oxidative stress, levels of intracellular ROS production were determined using the fluorescent probe DCF. As shown in Figure 6, when cells were exposed to Aβ25–35 for 24 h, ROS levels were significantly increased compared with the control group. However, ROS levels were significantly decreased in a concentration-dependent manner after treatment with T. laurifolia extract.
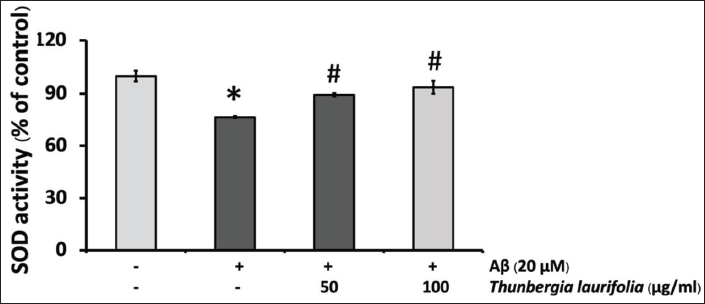
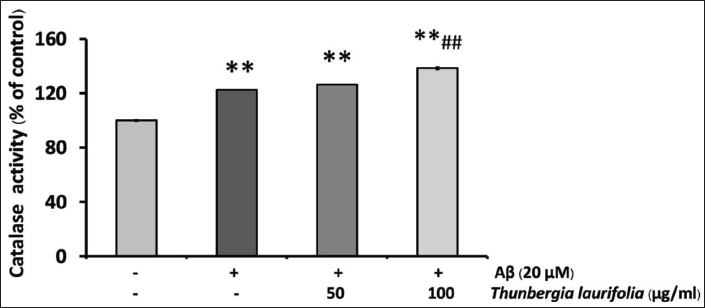
Effects of T. laurifolia Leaf Extract on the Activity of SOD and CAT in SH-SY5Y Cells After Treatment with Aβ25–35
The result showed that SOD activity was decreased after exposure to Aβ25–35, compared with that of the control. When cells were treated with T. laurifolia for 24 h, SOD (Figure 7) and CAT (Figure 8) activities were significantly increased in a concentration-dependent manner. CAT activity increased beyond that of Aβ treatment and the control group.
The Contents of Total Phenolic and Flavonoid Content in T. laurifolia Extract
Phenolic and flavonoid compounds found in plants are well-known for their potent antioxidant properties. Hence, it was justified to ascertain the levels of these phytochemical classes in the T. laurifolia leaf extract. To quantify the phenolic and flavonoid contents in T. laurifolia extract, gallic acid and rutin were used as standards in colorimetric assays. Table 1 shows that the values obtained in dried extract for gallic acid and rutin were 178.5 ± 6.86 and 32.51 ± 1.26 mg/g, respectively.
Total Phenolic and Flavonoid Content of T. laurifolia Extract.

Discussion
AD is the most common neurodegenerative disease associated with aging. In future, the human lifespan is expected to increase and the number of people at risk of this disease will be elevated (Ferri et al., 2005). One of the key events of AD is Aβ deposition which plays a critical role in the origination and progression of nervous tissue damage. Increased production of ROS mediated by Aβ can trigger oxidative stress, which causes functional and structural change in essential macromolecules leading to cell death. Hence, the attenuation or suppression of Aβ-induced ROS production may be a crucial tool for protecting neuronal cells from oxidative damage and providing a therapeutic strategy in AD treatment. Currently, the use of medicinal plants has considerably increased due to their minimal side effects and low cost as compared with synthetic medicine. In our study, we have explored if T. laurifolia leaf extract can protect SH-SY5Y cells from damage caused by Aβ25–35-induced toxicity and oxidative stress. We demonstrated that the level of ROS production significantly increased after treatment with Aβ25–35. Our result was consistent with a report that Aβ25–35 significantly increased ROS production in SH-SY5Y cells (Jia et al., 2021; Wang et al., 2016).
Furthermore, the treatment with T. laurifolia leaf extract significantly suppressed ROS production induced by Aβ25–35 in a concentration-dependent manner. Moreover, we noted that treatment with T. laurifolia leaf extract in the presence of Aβ25–35 resulted in a more substantial decrease in ROS levels compared with the baseline control group. In normal physiological cells, there exists a baseline level of ROS. This decline in ROS levels in the treatment groups than the baseline control group can be attributed to several potential mechanisms. For instance, the presence of antioxidant properties in the T. laurifolia leaf extract, such as flavonoids or phenolic compounds, may have played a key role in reducing ROS levels by effectively scavenging free radicals.
Another contributing factor could be the regulation of enzymes. It is plausible that the treatment influenced the activity of enzymes involved in ROS production, including enzymes such as SOD and CAT. Alterations in enzymatic activity have the potential to lead to reduced ROS levels.
Furthermore, it is important to consider that the concentration of the T. laurifolia leaf extract treatment may also have a significant impact on the observed effects. Therefore, we further investigate the antioxidant properties in the T. laurifolia leaf extract. In this study, the T. laurifolia leaf extract exhibited strong antioxidant properties based on total phenolic content and total flavonoid content. A higher total phenolic content was detected in this study (178.5 ± 6.86 mg/g) compared with previous studies (Oonsivilai et al., 2008; Posridee et al., 2022).
Variations in quantitative differences may be due to differences in the growth location, extract preparation, extractant type, drying methods, harvesting time, and stage of leaf development. Our study strongly suggests that T. laurifolia leaf extract possesses robust antioxidant properties, as also indicated by previous studies (Jirabanjerdsiri et al., 2022; Posridee et al., 2022).
As mentioned above, phenolic and flavonoid compounds are well-known antioxidants, and numerous studies have been published on their antioxidant activity in plant foods such as vegetables and fruits. Phenolic compounds are effective hydrogen donors, which makes them good antioxidants, whereas flavonoids act as free radical scavengers that can inhibit the formation of free radicals such as peroxyl radicals, superoxide anion, and hydroxyl radicals (Ratty & Das, 1988). Based on the total phenolic content and antioxidant action mechanism, T. laurifolia leaf extract was proficient in chemical antioxidant activities, hydrogen atom transfer, singer electron transfer, non-radical redox potential, and metal chelation capacity (Sunthrarak & Oonsrivilai, 2022; Zeb, 2020). Thus, reducing ROS induced by Aβ25–35 may be due to the phenolic and flavonoid compounds present in T. laurifolia leaf extract.
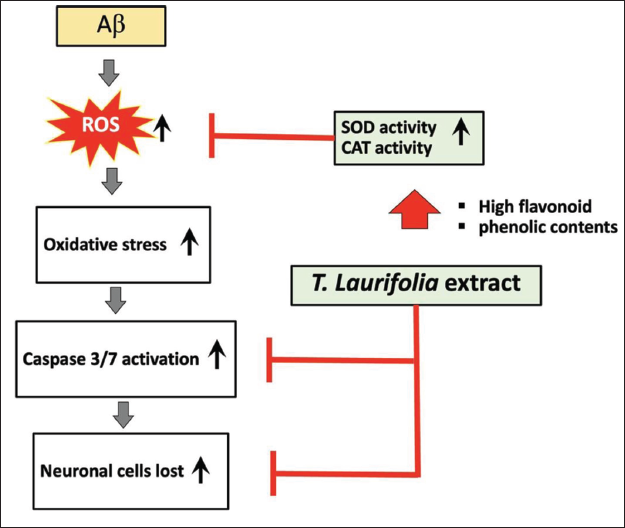
A hypothetical diagram demonstrating the signaling pathways through which T. laurifolia leaf extract shields cells from damage brought on by Aβ. Aβ is known to enhance the generation of ROS in neuronal cells, causing oxidative stress that in turn activates caspases-3/7 (and other caspases) and ultimately results in cell death. Increased SOD and CAT activity, reduced ROS generation, and decreased caspases-3/7 activity are all effects of T. laurifolia leaf extract. Increases in phenolic and flavonoid content, as well as SOD and CAT activity, may all help to reduce the generation of ROS. Reduced oxidative stress and caspase-3/7 activation may result in lessened cell death if ROS production falls.
Antioxidants could conceivably be used to prevent AD by inhibiting the detrimental effects of excess ROS by inducing endogenous enzymes such as SOD and CAT. SOD represents the primary line of antioxidant defenses by dismutation of O2. In the second step, CAT is responsible for eliminating H2O2 into water, thus preventing the production of OH. The balance between ROS production and the endogenous cellular antioxidant system is necessary for neuron oxidative metabolism (Tong et al., 2018).
In our study, SOD activities significantly decreased after treatment with Aβ25–35 which shows that the overproduction of ROS cannot be abolished by endogenous antioxidant enzymes possibly due to the direct toxic effect of Aβ25–35 or their metabolites. This result is in agreement with a previous study (Mairuae et al., 2019).
It is noted that the activity of CAT was increased beyond the control group after treatment with Aβ25–35 which could be a compensatory mechanism against Aβ25–35 or a direct induction from Aβ25–35. However, these effects might result from unknown factors as well (Mairuae et al., 2019). When cells are treated with T. laurifolia leaf extract, we found that the levels of SOD and CAT significantly increased, indicating that T. laurifolia extract may activate the SOD and CAT enzymatic activities and enable the cells to manage with Aβ‑induced ROS production.
ROS and caspase activation has been implicated in Aβ-induced neurotoxicity, resulting in the loss of neuronal cells and a gradual loss of cognitive function (Deshpande et al., 2006; Shelat et al., 2008; Sponne et al., 2004). We explored the T. laurifolia leaf protective effects on the activation of caspases-3/7, which play crucial roles in neuronal cell apoptosis. The result demonstrated that Aβ25–35 increased caspase-3/7 activities in SH-SY5Y cells. However, treatment with T. laurifolia leaf extract significantly attenuated Aβ-induced caspase-3/7 activity. In this study, the MTT assay and LDH release test are also used for the evaluation of the neuroprotective effect of T. laurifolia leaf extract against the Aβ25–35-treated decrease in cell viability. The results showed that T. laurifolia leaf extract decreased Aβ25–35-induced SH-SY5Y cell death in MTT assay. Additionally, further confirmation was provided by the LDH assay which is a cell toxicity indicator; T. laurifolia leaf extract-treated cells reduced Aβ-induced LDH release.
The reduction in LDH levels may suggest reduced cellular damage and enhanced cell viability in cells treated with T. laurifolia leaf extract, consistent with the findings from the MTT assay. We also observed that treatment with T. laurifolia leaf extract at a concentration of 100 µg/mL in the presence of Aβ25–35 resulted in a notably greater reduction in LDH release compared with the control group. This observation is likely due to the elatively high concentration of T. laurifolia leaf extract, which effectively lowered LDH release below the baseline control levels. Our finding suggests that T. laurifolia leaf extracts can protect SH-SY5Y cells against Aβ-induced cellular damage. Based on the results of this study, it appears that the concentration of T. laurifolia leaf extract capable of safeguarding neuronal cells from Aβ-induced injury may be 100 µg/mL, as demonstrated by its ability to reduce neuronal cell death following exposure to Aβ25–35.
Conclusion
To the best of our knowledge, this is the first report that T. laurifolia leaf extract protects SH-SY5Y cells against Aβ25–35-induced injury. The mechanisms may be related to its ability to increase CAT and SOD enzyme activity and/or its phenolic and flavonoid contents leading to the inhibition of ROS production and reduced oxidative stress, as well as attenuating caspase-3/7activity. The present data suggest that T. laurifolia leaf constituents may be useful in the prevention or treatment of AD mediated by Aβ.
Summary
T. laurifolia treatment demonstrates a concentration-dependent suppression of ROS production in neuroblastoma cells induced by beta-amyloid peptide.
T. laurifolia treatment leads to a significant increase in CAT activity in neuroblastoma cells exposed to beta-amyloid peptide.
T. laurifolia treatment results in a notable increase in SOD activity in neuroblastoma cells induced by beta-amyloid peptide.
T. laurifolia treatment shows a concentration-dependent reduction in LDH release in neuroblastoma cells induced by beta-amyloid peptide.
T. laurifolia treatment exhibits a concentration-dependent decrease in caspase3/7 activity in neuroblastoma cells induced by beta-amyloid peptide.
T. laurifolia treatment leads to a concentration-dependent improvement in cell viability in neuroblastoma cells induced by beta-amyloid peptide.
The extract contains a total phenolic content of 178.5 ± 6.86 mg/g and a total flavonoid content of 32.51 ± 1.26 mg/g.
Abbreviations
AD: Alzheimer’s disease; DMSO: Dimethyl sulfoxide; CAT: Catalase activity; DMEM: Dulbecco’s Modified Eagle’s medium; FBS: Fetal bovine serum; ROS: Reactive oxygen species; SD: Standard deviation; SOD: Superoxide dismutase.
Footnotes
Acknowledgments
The authors thank Dr. Adrian R. Plant (Mahasarakham University) for language editing the manuscript.
Authors’ Contributions
We declare that this work was done by the authors named in this article and all liabilities pertaining to claims relating to the content of this article will be borne by the authors.
Declaration of Conflicting Interests
The authors indicated no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research project was financially supported by the Faculty of Medicineand the grant from Mahasarakham University.
