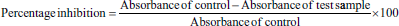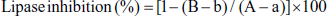Abstract
Background
Yellow horn poppy (Glaucium flavum, syn. Chelidonium flavum) in the family Papaveraceae, is growing in many geographical areas; it is mostly native to Northern Africa, Western Asia, and Europe. The plant is well known for its different types of alkaloids, including aporphines, protopines, and phenolic compounds. In Eastern Europe, the HCl and HBr salts of glaucine (the common active constituent in G. flavum) have been approved as cough suppressants, in addition to several therapeutic applications in traditional medicine. The natural products of G. flavum varied according to the geographical habitat, the stage of maturity, and the selected plant organ.
Materials and Methods
For the first time, the extraction and isolation of natural products (alkaloids and flavonoids) from dried aerial parts of the Libyan population of yellow horn poppy and testing their biological effects, which include antihistaminic, antiarthritic, and antiobesity effects.
Results
Alkaloids (glaucine and isoboldine) and flavonoid constituents (quercetin-7-O-rutinoside and rutin) have been isolated, while the biological activity results showed that quercetin-7-O-rutinoside and rutin showed half-maximal inhibitory concentration (IC50) (µg) = 29.7 and 46.6, 26.2 and 61.3, and 58.8 and 50.1 for antihistaminic, antiarthritic, and antiobesity, respectively, compared with those of glaucine and isoboldine effects, IC50 = 77.4 and 159.3, 61.7 and 118.7, and 237.8 and 157.6, respectively.
Conclusion
According to the obtained results, the antihistaminic, antiarthritic, and antiobesity effects of the flavonoid compounds were stronger than those of the alkaloidal constituents of yellow horn poppy.
Introduction
Inflammation is caused by infectious agents, environmental exposures (sun and nuclear radiation), severe and repeated injuries, gastric fluids, bile acid, and certain immune problems. Moreover, cancer is linked to oxidative stress and inflammation. In fact, during the inflammatory reaction, various molecules are produced by immune cells as chemical mediators, such as cytokines, which are involved in cancer formation mechanisms (Hanada and Yoshimura, 2002). Immune cells (e.g., mast cells) due to inflammatory reactions tend to secrete an inflammatory mediator such as histamine and cytokines (Park et al., 2008). Histamine release is responsible for a variety of inflammatory reactions (Park et al., 2008). Several clinical and preclinical trials have been conducted to detect an effective natural product that alleviates the release of histamine and other proinflammatory mediators with fewer side effects (Park et al., 2008). One of the most common inflammatory conditions is rheumatoid arthritis (RA) (Saleem et al., 2019). One-fifth of the elderly world population, mostly females, suffers from arthritis (Murugananthan et al., 2013; Mubashir et al., 2014). The severity of RA extends to include some physiological issues regarding the heart, lungs, and liver, and consequently, RA affects the quality of life (Gutiérrez-Rebolledo et al., 2018). Long-term use of common symptomatic antiarthritic drugs, for example, non-steroidal anti-inflammatory drugs, disease-modifying antirheumatic drugs, corticosteroids, immunosuppressants, and cytotoxic drugs, has developed many life-threatening side effects, including cardiovascular diseases, myocardial and hypertension, and renal and gastrointestinal complications. The adverse effects of these therapies are numerous and necessitate the development of more safe and effective treatments (Reddy et al., 2014). Nature is ever-giving, rich in beneficial natural products, and never depleted (Elisha et al., 2016). Herbal drugs are promising natural alternatives (Hasan and Alamgeer, 2018). Antiarthritic activity of herbal drugs can be tested through the method “protein denaturation assay” using the Sakat et al.’s (2010) method.
Excessive accumulation of fats due to metabolic disorders results in obesity (Wang et al., 2011). Globally, it was reported that more than 1.9 billion adults were overweight and of these, more than 650 million were obese (World Health Organization, 2016). Obesity represents a common risk factor for cardiovascular disorders, hypertension, dyslipidemia, type 2 diabetes mellitus, and stroke (Kim et al., 2020; The GBD 2015 Obesity Collaborators, 2017; Obata et al., 2017). Heart attack and stroke, as well as liver damage, are the most reported side effects of the existing synthetic antiobesity drugs (Sun et al., 2016). Antiobesity natural products attracted attention several years ago as safe and effective substitutes for these synthetic ones through several research studies (Amin and Nagy, 2009; Park et al., 2011). Garcinia cambogia, Plantago psyllium, and Morus alba are famous examples of antiobesity natural drugs as they regulate fat and carbohydrate metabolism (Jaber and Iraby, 2014). However, continuous research studies on the effects of G. cambogia extract revealed some safety issues related to hepatic and heart pathophysiology. Accordingly, it is required that new natural materials with good safety and efficacy be developed (Kim et al., 2020). The yellow horn poppy (G. flavum Cr., syn. Chelidonium flavum) is one of 28 Glaucium species belonging to the family Papaveraceae; it is growing in many geographical areas and is mostly native to Northern Africa, Europe, and Western Asia. The name “horned poppy” is derived from its long and pointed capsulated fruits, bearing horn-like appendices (Lapa et al., 2004; Arafa et al., 2016; Akaberi et al., 2021). The plant is well known for its different types of alkaloids, including aporphines, protopines, benzophenanthridines, benzylisoquinolines, protoberberines, and morphinanes. However, some phenolic compounds have also been reported as G. flavum natural products, for example, kaempferol, caffeic acid, catechin hydrate, syringic acid, chlorogenic acid, isoquercitrin, and trans-hydroxycinnamic acid (Boulaaba et al., 2019). Several pharmacological activities have been reported in G. flavum, for example, analgesic (Arafa et al., 2016), antioxidant (Khoshvaghti et al., 2019), antidiabetic (Darya et al., 2019), and antitumor activities (Bournine et al., 2013a, b). The herb G. flavum was used widely in Europe as a cough suppressant several decades ago. The major alkaloidal constituent and most common natural product in all plant varieties of G. flavum (i.e., glaucine) is used as an antitussive.
Our research interest is focusing mainly on promising naturally occurring drug leads that can effectively participate in coping with the above-mentioned health issues, that is, inflammatory diseases, rheumatic arthritis, and obesity, with no or minimal side effects compared with the existing drugs. The Libyan population of the herb G. flavum has been chosen to conduct preliminary chemical and biological investigations regarding our research interest. G. flavum is a well-known medicinal plant that has had many therapeutic applications in folk and traditional systems of medicine since ancient times (Ibn Sina, 1025 republished in Ibn Sina (2009); Ibn BeyyBe 1248 republished in Ibn Beyṭar (2020); Akaberi et al., 2021; Bournine et al., 2013a, b). The G. flavum natural products were varied according to the geographical habitat where the species grew, the stage of maturity, and the selected plant organ (Peled et al., 1988). Our research study focuses on the isolation of phytoconstituents from the Libyan population of G. flavum and their biological activities. To the best of our knowledge, the Libyan population of G. flavum had never been studied before, neither for phytochemistry nor pharmacology.
Materials and Methods
Materials, Plant Collection, and Extraction Procedure
The aerial parts of G. flavum Cr. were collected during the flowering stage from Derna city, east Libya, located at the geographical zone 32.767 longitude and 22.6367 latitudes. The aerial part was composed of bluish-green leaves (each leaf was deeply pinnatifid with 5–6 opposite lobes, where the lobe itself is further 1–3 lobed on the basal part), yellow flowers, and unripe, long, swollen, and pointed capsulated fruits, tapering with horn-like appendices. The plant was air dried, then finely grinded. Ten grams of plant powder were extracted with 50 mL of 70% hydroethanol for phytochemical screening.
Phytochemical Screening
The hydroethanol extract was filtered off and screened for the presence of different phytochemical compounds according to Aljameeli and Ashour (2022), where Molisch’s test was used for testing the presence of carbohydrates and/or glycosides, Dragendorff’s test for true alkaloids, AlCl3 solution for flavonoids, Borntrager’s test for anthraquinones, Keller–Kiliani test for cardiac glycosides, FeCl3 reagent for phenolics/tannins, Lieberman-Buchard’s test for sterols/triterpenes, and froth test for saponins.
Isolation Procedure
G. flavum powder (450 g) was extracted with 70% hydroalcohol, then the extract (GFE) was evaporated under vacuum while 10 g of solid mass was precipitated out and separated. The concentrated mother liquor was defatted with petroleum ether (pet. ether) from which 55 g of defatted extract was obtained. The solid mass (10 g) was purified and recrystallized from dichloromethane/methanol (DCM/MeOH) to afford an 8.1 g needle crystalline substance (G1). Forty grams of defatted extract was chromatographed (column 1) using silica gel particles (Si gel) as a stationary phase with different mobile phase combinations [pet. ether/ethyl acetate (EtOAc) ↓ DCM/MeOH], where 25 fractions, 10 mL each, were obtained [fr.1 ↓ fr25]. The fractions obtained with [pet. ether/EtOAc, 1:1 and DCM/MeOH, 98:2], [fr1↓ fr5] were concentrated affording crystalline substance (G1, 3.5 g) while the mother liquor (10.5 g, fr1m) was chromatographed again (column 2) using Si gel particles and DCM 100%, and then DCM/MeOH, 8:2 as mobile phases where 1–17 and 18–,39 fractions were eluted, respectively. Fractions 9–11 of column 2 were chromatographed again (column 3) using Si gel and DCM/MeOH, 8:2 as mobile phase to afford (G2). The flavonoid-rich fractions (fr15–fr17) from column 1 were chromatographed using the Si gel column and DCM/MeOH (9:1 and 8:2) as mobile phases. The first four fractions were purified again with a Sephadex column using methanol as a mobile phase where two flavonoid constituents were obtained (GF1 and GF2).
Antihistaminic Test
Antihistaminic activity of yellow horn poppy natural products was conducted using rat peritoneal mast cells (RPMCs), according to Kim et al. (2008). Briefly, the isolated RPMCs were centrifuged at 400 rpm for 15 min at 4°C after suspension in metrizamide-tyrode buffer. Then the cells in the pellet were washed and resuspended in 1 mL of Tyrode buffer. The o-phthaldialdehyde spectrofluorometric procedure was used for measuring the histamine contents, according to Kim et al. (2008). RPMCs were incubated with a test sample (0.00–1.0 mg/mL) for 10 min at 37°C before the addition of compound 48/80 (5 µg/mL) and incubation for an additional 10 min. The cells were separated from the released histamine by centrifugation at 400 rpm for 5 min at 4°C. The fluorescent intensity was measured at emission (438 nm) and excitation (353 nm) using a spectrofluorometer (RF-5301 PC; Shimadzu).
Antiarthritic Activity Test
It was conducted through a “Protein denaturation assay using egg’s albumin.” In brief, the assay was performed according to the method described by Saleem et al. (2019). The 5 mL reaction mixture consisted of 0.2 mL egg albumin, 2.8 mL phosphate-buffered saline (pH 6.4), and 2 mL of sample solution at 0.00–1000 µg/mL concentrations. The standard solution contained diclofenac sodium instead of a test sample. These solutions were incubated at 37°C for ¼ hour, followed by heating at 70°C for 5 min. Then the solutions were cooled to room temperature and their absorbance was measured at 660 nm. The test was performed in triplicate and the percentage inhibition of protein denaturation was determined.
Antiobesity Test
The activity of yellow horn poppy natural products as inhibitors for pancreatic lipase was measured using the substrate p-nitrophenyl butyrate (PNPB) as described by Kim et al. (2020) with slight modification. Briefly, an enzyme buffer was prepared by adding 30 µL of pancreatic lipase (Sigma-Aldrich, St. Louis, MO, USA) in 10 mM of morpholine propane sulfonic acid and 1 mM of ethylene diamine tetraacetic acid (pH 6.8) to 850 µL of Tris buffer (100 mM of Tris–HCl and 5 mM of CaCl2; pH 7.0). Then, 100 µL of sample or orlistat was mixed with 880 µL of enzyme buffer and incubated for 15 min at 37°C. After incubation, we added 20 µL of the substrate solution (10 mM of PNPB in dimethyl formamide), and the enzymatic reactions were allowed to proceed for 30 min at 37℃. Pancreatic lipase inhibitory activity was determined by measuring the hydrolysis of PNPB to p-nitrophenol at 405 nm using a microplate reader (Infinite M200 PRO; Tecan Austria). Inhibition of lipase activity was expressed as a percentage decrease in OD. The inhibitory activity (%) was calculated according to the formula:
where A is the activity of the enzyme without the inhibitor, a is the negative control without the inhibitor, B is the activity of the enzyme with the inhibitor, and b is the negative control with the inhibitor.
Statistical Analysis
The obtained data was analyzed using Microsoft Excel (version 365) software. The data was presented as mean ± standard deviation (SD) and percentage values.
Results
Phytochemical Screening
Phytochemical screening tests revealed the presence of phytochemical compounds related to alkaloids, flavonoids, phenolic compounds, sterols/triterpenes, glycosides, saponins, and the absence of anthraquinones and cardiac glycosides.
Isolated Compounds
Two aporphine alkaloids (glaucine and isoboldine) and two flavonol glycosides (querctin-7-O-rutinoside and rutin) were isolated and identified from the Libyan population of G. flavum (Figure 1).
Chemical Structure of the Isolated Compounds.
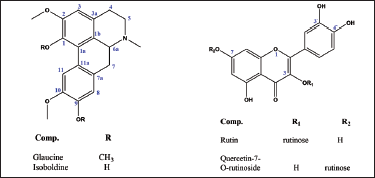
Compound (G1), the major phytoconstituent of GFE (11.62 g), was obtained as buff crystalline needles, positive to Dragendorff’s reagent (gave orange red spot on thin-layer chromatography (TLC), R
f
= 0.75, mobile system: dichloromethane/methanol; 9/1), electrospray ionization mass spectrometry (ESI-MS) (m/e): 355 [M]+; 1H NMR spectrum (dimethyl sulfoxide (DMSO)-d6) showed four singlet signals resonating at δH (ppm) = 3.85 (
Compound (G2), (37.6 mg), was obtained as amorphous white powder, positive to Dragendorff’s reagent (gave orange red spot on TLC, R
f
= 0.65), (mobile system: dichloromethane/methanol; 9/1), ESI-MS (m/e): 327 [M]+; 1H NMR spectrum (DMSO-d6) showed two singlet signals resonating at δH (ppm) = 3.84 (
Antihistaminic Test
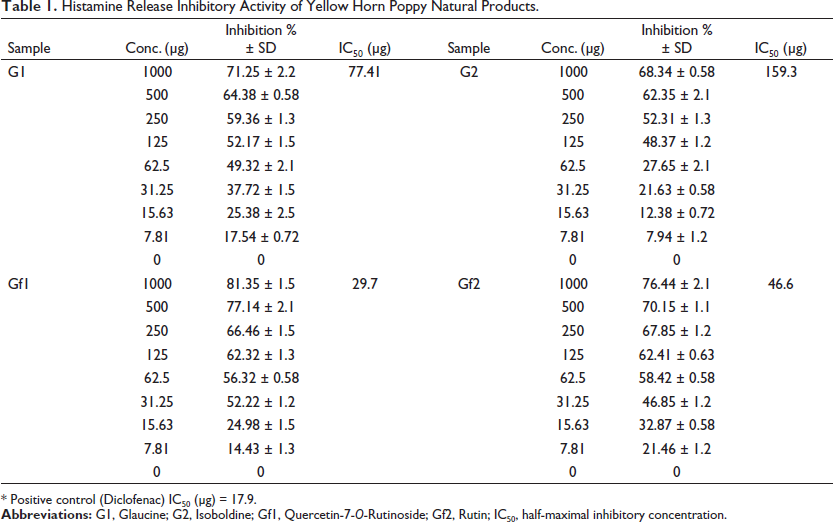
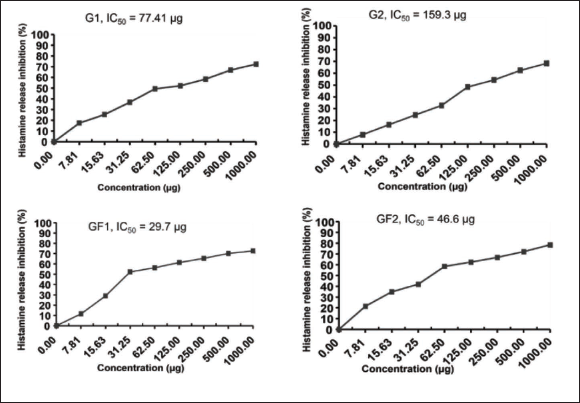
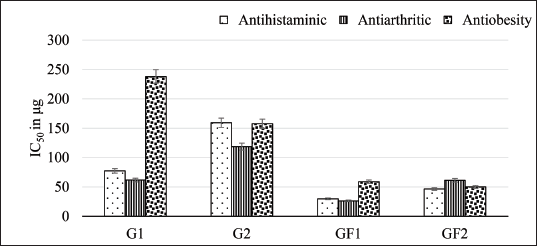
The results of the inhibitory effects of G. flavum natural products against rat peritoneal mast cell histamine release, according to Kim et al. (2008), are summarized in Table 1. The flavonoid (GF1) shows the minimum inhibitory concentration (IC50 = 29.7 µg) due to its higher inhibitory activity compared with other natural products, and then, the flavonoid (GF2) shows an IC50 = 46.6 µg, while the alkaloids G1 and G2 showed weak inhibitory effects, where IC50 were 77.41 and 159.3 µg, respectively.
Histamine Release Inhibitory Activity of Yellow Horn Poppy Natural Products.
Antiarthritic Activity Test
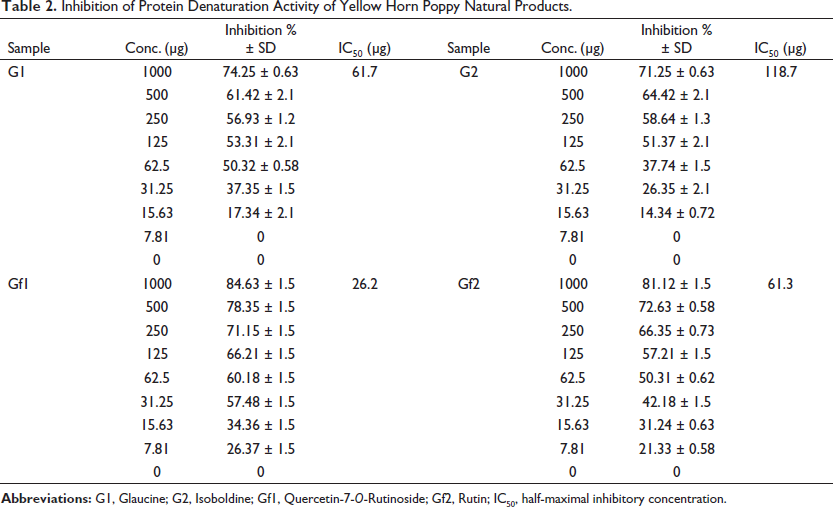
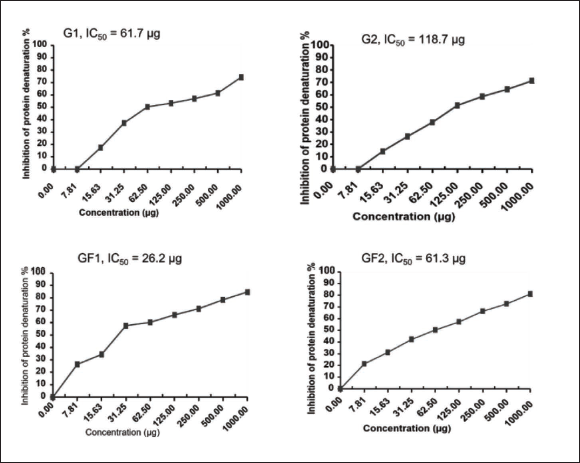
The results of the inhibitory effects of G. flavum natural products against albuminous protein denaturation according to the Saleem et al. (2019) procedure are summarized in Table 2. The flavonoid (GF1) shows the minimum inhibitory concentration (IC50 = 26.2 µg) due to its higher inhibitory activity compared with other natural products. The flavonoid (GF2) shows an IC50 = 61.3 µg, while G1 and G2 showed IC50 = 61.7 and 118.7 µg, respectively.
Inhibition of Protein Denaturation Activity of Yellow Horn Poppy Natural Products.
Antiobesity Test
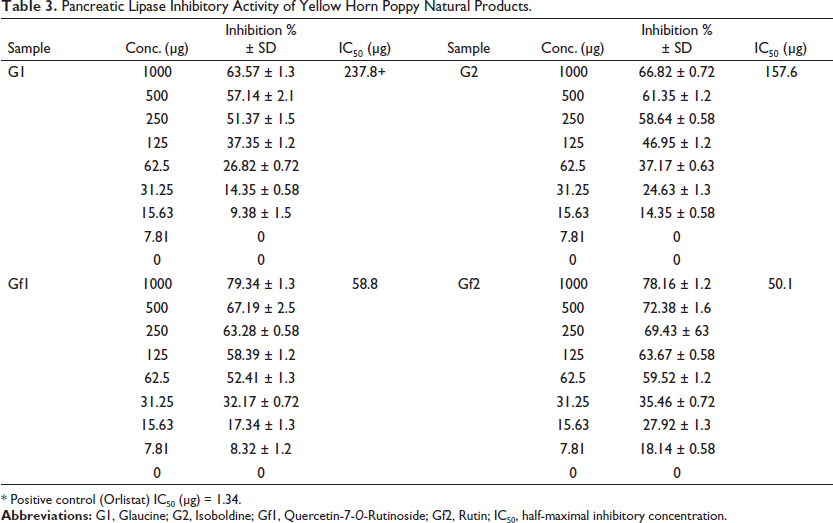
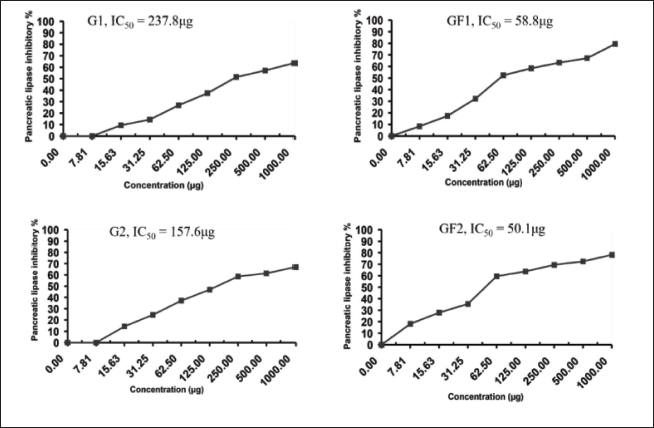
The results of the inhibitory effects of G. flavum natural products against pancreatic lipase activity using Kim et al.’s (2020) procedure are summarized in Table 3. The flavonoid (GF2) shows the minimum inhibitory concentration (IC50 = 50.1 µg) compared with other natural products. The flavonoid (GF1) shows an IC50 = 58.8 µg, while G1 and G2 showed IC50 = 237.8 and 157.6 µg, respectively.
Pancreatic Lipase Inhibitory Activity of Yellow Horn Poppy Natural Products.
Discussion
G. flavum, like other Papaveraceous species, is rich in alkaloidal constituents, especially Aporphine Alkaloids. Alkaloidal constituents of G. flavum attract the most research interests due to their abundancy in the plant organs, promising and wide scope of pharmacological activities as well as their therapeutic applications both in traditional and modern medical systems. Comparatively, Phenolic constituents of G. flavum, have been studied very rare. Different varieties or population of G. flavum from different geographical areas have been studied previously for their phytochemical and pharmacological activities. However, the Libyan population of G. flavum had never been studied neither for its phytochemical nor pharmacological activities. One of G. flavum’s therapeutic benefits was the anti-inflammatory effects. The question is remained “which chemical constituent of G. flavum is responsible for this anti-inflammatory effect?” Therefore, the present research was conducted both to investigate the chemical constituents of the Libyan population of G. flavum as well as the biological activities of the isolated phytoconstituents. As a result of this study, four natural products have been isolated and identified from the Libyan variety of the yellow horn poppy. However, the biological activity study of the isolated compounds showed stronger effects of the flavonoid constituents versus the alkaloid ones as explained in Figures 2 to 5, where quercetin-7-O-rutinoside and rutin showed IC50 = (29.7 &46.6), (26.2 &61.3) and (58.8 & 50.1) for antihistaminic, antiarthritic and anti-obesity respectively, compared with those of glaucine and isoboldine effects, IC50 = (77.4 &159.3), (61.7, 118.7) and (237.8 & 157.6) respectively.
Conclusion
The study confirmed the presence of alkaloid constituents (glaucine and isoboldine) in addition to flavonoid constituents (quercetin-7-O-rutinoside and rutin) for the first time in the Libyan variety of Glaucium flavum. The biological activity testing of these phytoconstituents, showed stronger bioactivities of the flavonoid constituents compared with those of the alkaloid ones in course of the inflammatory conditions i.e., antihistaminic and antiarthritic as well as anti-obesity. Accordingly, it worth recommended to serve yellow horn poppy flavonoids as alternative natural anti-inflammatory agents, in addition, further anti-inflammatory studies are also recommended in the near future for yellow horn poppy natural products especially for the flavonoid constituents. The synergistic effects of different phytoconstituents must be taken into consideration during future experimental trials.
Footnotes
Acknowledgments
The authors gratefully acknowledge the approval and support of this research study by grant no. PHAR-2022-11-1703 from the Deanship of Scientific Research at Northern Border University, Arar, Kingdom of Saudi Arabia.
Authors’ Contributions
AM: materials, isolations, experimental trials, results, and manuscript. AM and SS: data collection, results interpretation, and discussion.
Declaration of Conflicting Interests
The authors indicated no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Grant no. PHAR-2022-11-1703 from the Deanship of Scientific Research at Northern Border University, Arar, Kingdom of Saudi Arabia.
Statement of Informed Consent and Ethical Approval
Before beginning the study, the participants provided the necessary ethical clearances and informed consent.