Abstract
Background
Artichoke (Cynara scolymus L.) has the potential to treat diabetes, dyslipidemia, hypertension, and obesity. However, the evidence from previous studies is not consistent.
Objectives
This meta-analysis evaluated the efficacy of products derived from artichokes on blood glucose, lipid level, blood pressure, and anthropometric parameters.
Methodology
The literature was reviewed via international databases (PubMed, ScienceDirect, and Scopus). A total of 21 RCTs with high quality, assessed by the Cochrane risk-of-bias tool, were included.
Results
Artichoke was linked to a significant reduction in fasting blood sugar (FBS) (WMD: −3.76 mg/dL: 95%CI −7.31, −0.22), insulin level (WMD: −1.35 mIU/L: 95%CI −2.29, −0.41), and HOMA-IR (WMD: −1.00: 95%CI −1.95, −0.06). Similar results were observed for LDL-c (WMD: −12.94 mg/dL: 95%CI −18.02, −7.87), total cholesterol (TC) (WMD: −19.64 mg/dL: 95%CI −23.94, −15.35), and triglyceride (TG) (WMD: −13.36 mg/dL: 95%CI −19.06, −7.66). Moreover, participants who administered artichoke experienced a significant reduction in SBP (WMD: −1.59 mmHg: 95%CI −3.02, −0.16), body weight (BW) (WMD: −1.17 kg: 95%CI −1.75, −0.60), and BMI (WMD: −0.30 kg/m2: 95%CI −3.02, −0.16).
Conclusion
Artichoke may improve blood glucose, lipid profile, blood pressure, and anthropometric parameters. A large, well-designed RCT and head-to-head comparison using a standardized preparation of artichoke will provide definitive data on specific participants.
Introduction
Cardiometabolic outcomes, including obesity, hypertension, type 2 diabetes mellitus, and hyperlipidemia, are the leading causes of morbidity and mortality. Multiple literature reviews have established a link between these conditions and an increased risk of cardiovascular disease (CVD). The prevalence of CVDs is rapidly increasing, and they are the most common multimorbidity pattern (Xu & Cao, 2022). Death rates due to CVD have increased by 22.3% between 2007 and 2017 (Stanaway et al., 2018). Therefore, the primary aim of preventing CVD would involve the control and regulation of risk factors (Assmann et al., 2010). Davidson & Parkin (2009) posited that a previous study has provided evidence supporting the efficacy of therapies targeting blood glucose reduction in significantly ameliorating cardiovascular complications. Moreover, the research conducted by Wu et al. (2009) demonstrated that a decrease of approximately 10% in low-density lipoprotein (LDL) levels was associated with a significant reduction of 12%−20% in the incidence of ischemic heart disease among the study participants over a period of 5 years.
Consequently, there is growing interest in alternative therapies to improve cardiometabolic parameters (Nauck et al., 2021). Herbal medicine has been introduced as an adjunctive method, alongside lifestyle interventions and drug therapy, to address these issues (Kumar et al., 2021).
Artichoke (Cynara scolymus L.) has been used as an herbal remedy. It is a member of the family Asteraceae which has been used in traditional medicine for centuries worldwide (Rolnik & Olas, 2021). Over the years, artichoke has been studied extensively, with preclinical and clinical investigations reporting various pharmacological properties, including its potential to treat diabetes, dyslipidemia, hypertension, and obesity (Amini et al., 2022; Mahboubi, 2018; Sahebkar et al., 2018).
Extensive research conducted through animal studies and clinical trials has evaluated the therapeutic potential of artichokes in managing various health conditions. A previous randomized controlled trial showed that artichoke significantly reduced blood glucose, blood lipid, and blood pressure in comparison with placebo (Ardalani et al., 2020). However, prior research has demonstrated that neither artichoke nor placebo improved blood glucose (Rondanelli et al., 2011), lipid profile (Lupattelli et al., 2004), blood pressure (Rondanelli et al., 2020), and body weight (BW) outcomes (Roghani-Dehkordi & Kamkhah, 2009). A recent meta-analysis of nine randomized controlled trials (RCTs) indicated that artichoke products effectively reduce fasting blood sugar (FBS). However, this product did not improve hemoglobin A1c (HbA1c) or homeostatic model assessment for insulin resistance (HOMA-IR) (Jalili et al., 2020). The potential for improved blood glucose and lipid profiles has been identified as a result of artichoke product consumption, according to clinical research findings (Fallah Huseini et al., 2012; Rondanelli et al., 2020). However, the results obtained from the existing research demonstrate contradictory conclusions (Rezazadeh et al., 2018; Skarpañska-Stejnborn et al., 2008), predominantly as a result of the small sample sizes utilized in the majority of the conducted studies (Rangboo et al., 2016; Rondanelli et al., 2014).
A meta-analysis has suggested that artichoke may be effective in improving the lipid profile, which is a key marker for assessing cardiometabolic risk. However, comparative studies between artichoke and placebo have led to conflicting findings and generated controversy (Santos et al., 2018). Previous meta-analyses have also been limited to a meta-regression analysis among lipid profiles (Sahebkar et al., 2018), indicating the need for further research using stronger data to establish the benefits of artichoke on cardiometabolic outcomes. In light of these limitations, the authors recommend additional high-quality research to better elucidate the potential benefits of artichokes in improving cardiometabolic outcomes. Therefore, an updated systematic review and a meta-analysis of RCTs were conducted to clarify the effects of artichoke on blood glucose, lipid level, blood pressure, and anthropometric parameters.
Methodology
Search Strategies and Selection Criteria
The research adhered to the Preference Reporting Items for Systematic Review and Meta-Analysis (PRISMA) reporting standard, which ensures transparent reporting of systematic reviews and meta-analyses, and the protocol is registered to the PROSPERO database (CRD42023421339).
The study was conducted between 1st March and 5th August 2023. The comprehensive searches of three electronic databases, namely PubMed, ScienceDirect, and Scopus, were conducted up to 25th April 2023. The search strategy for this study involved the utilization of Medical Subject Headings (MeSH) to meticulously query the databases. The following MeSH terms were employed: (artichoke OR Cynara scolymus OR C. scolymus) AND (blood glucose OR fasting blood glucose OR FBS OR fasting plasma glucose OR FPG OR HbA1c OR postprandial glucose OR PPG OR lipid profile OR Low-density Lipoprotein OR LDL OR High-Density Lipoprotein OR HDL OR total cholesterol OR TC OR triglyceride OR TG OR blood pressure OR systolic blood pressure OR SBP OR diastolic blood pressure OR DBP OR Anthropological parameter OR body weight OR BW OR body mass index OR BMI). All relevant papers’ citations were also verified.
Inclusion criteria were strict and only RCTs that demonstrated the effectiveness of artichoke for any outcome, including lipid profile, blood glucose, blood pressure, and anthropological parameters, were considered eligible. No language limitations were imposed to ensure a comprehensive and inclusive analysis.
Exclusion criteria: After reviewing the entire contents of the nominated publication, the following selection conditions were used to determine whether studies should be excluded from our meta-analysis research: (1) quasi-experimental studies and (2) review articles.
Data Extraction and Quality Assessment
Two researchers (WP and KW) extracted data independently from the selected studies, in accordance with the CONSORT statement for reporting herbal medicine interventions. The extracted data consisted of study design, patient characteristics, intervention details, outcomes, and other relevant information. Following a systematic approach, all included studies were thoroughly assessed for their risk of bias using the Cochrane risk-of-bias tool for RCTs. The collected data from the studies were then provided by the researchers. Any inconsistencies that arose during the literature search or data extraction were resolved through discussion and consultation with a third researcher (BS) until a consensus was reached.
Outcomes of Interest
The study focused on assessing the impact of artichokes on several primary and secondary outcomes of interest. The primary outcomes of interest were FBS, glycated HbA1c, the HOMA-IR, fasting insulin levels, LDL, high-density lipoprotein (HDL), total cholesterol (TC), triglycerides (TGs), systolic blood pressure (SBP), diastolic blood pressure (DBP), BW, and body mass index (BMI). The secondary outcomes included laboratory findings.
Statistical Analysis
The study employed various statistical techniques to analyze the data. Pooled effects were computed and stratified based on the outcome data. Weighted mean difference (WMD) was used to summarize data for continuous outcomes, and summary statistics for dichotomous outcomes were presented as a risk ratio (RR) with a 95% confidence interval (CI). To determine statistical heterogeneity across studies, the chi-squared test and I2 test were used, and a p-value of <0.05 was considered significant. If heterogeneity was observed, subgroup analyses were conducted to identify its cause. The random effect model was used to combine the findings of separate investigations. Publication bias was assessed using funnel plots and the Egger test, with a p-value of <0.05 indicating statistical significance. Meta-regression was employed to evaluate the relationship between computed WMD and the dosage and duration of artichoke supplementation. Statistical analysis was performed using Revman® version 5.3 (Cochrane Collaboration) and STATA software version 15. Sensitivity analysis was conducted using fixed effect models, including the one-study-removal (leave-one-out) method, to ensure the reliability of the findings. Subgroup analyses were also carried out based on treatment duration, artichoke dosage, and health condition for each of the primary outcomes to provide a more comprehensive understanding of the data.
Results
Initially, the researchers retrieved 870 articles through three electronic searches. Following the screening process based on the inclusion criteria, 26 articles met the eligibility criteria. However, after a thorough evaluation of the full text, five studies were excluded for the following reasons: three studies did not report relevant outcomes and two studies were quasi-experimental in design (Figure 1).
A PRISMA Flow Diagram Describing the Selection Process for Identifying Studies Included in the Meta-analysis.
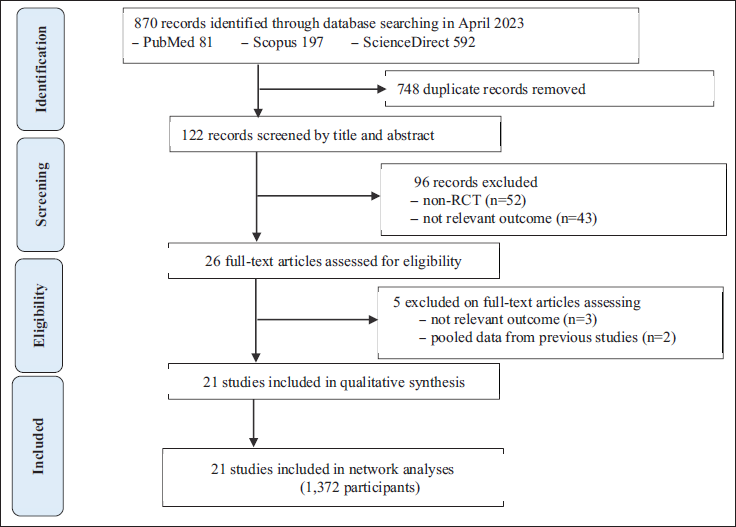
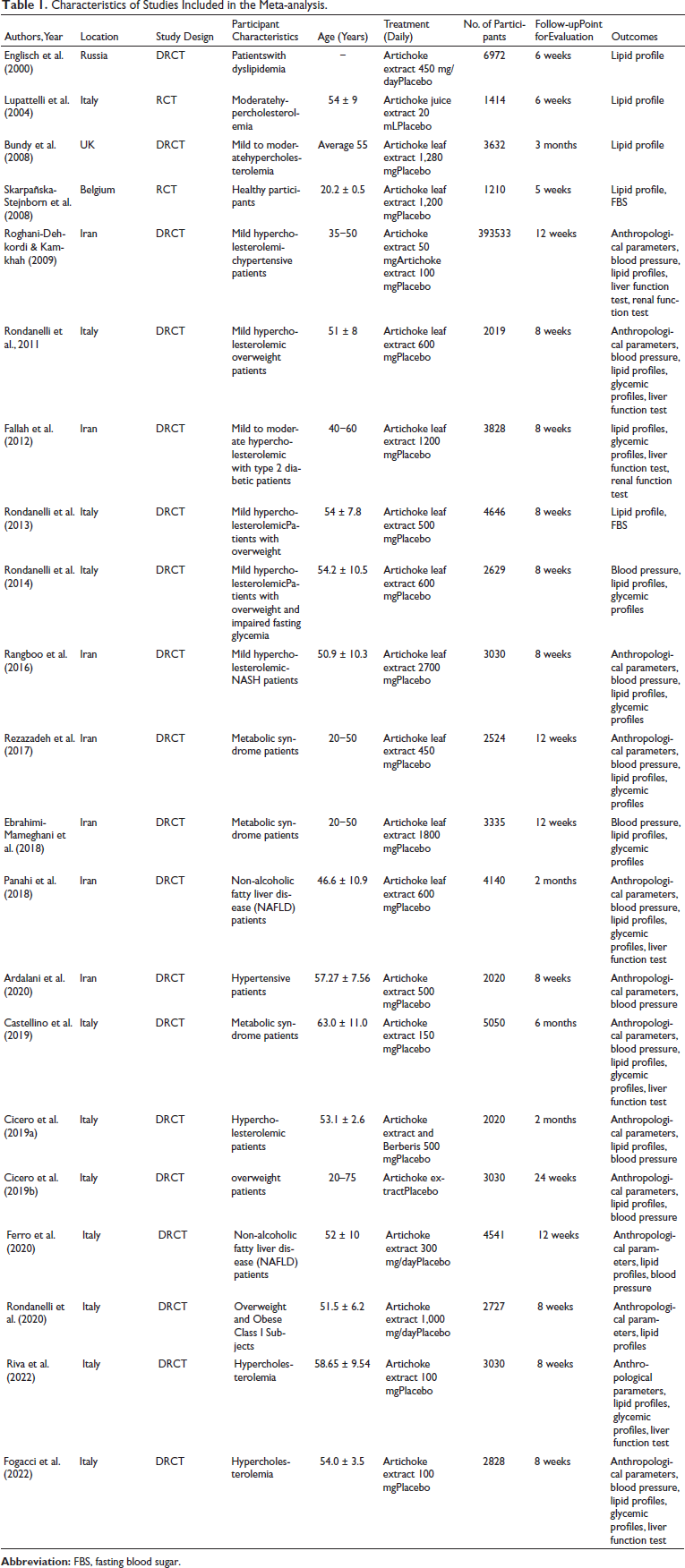
Table 1 presents a comprehensive summary of the key features of the studies included in this meta-analysis. Of the 21 studies included, six were conducted in Iran, six in Italy, and one each in India, the United Kingdom, Belgium, and Russia. The studies were conducted between 2004 and 2022. Most of the studies were single-center and had fewer than 100 participants. The total sample size across all studies was 1,372, including 812 patients with hyperlipidemia from 12 studies, 217 patients with metabolic syndrome from three studies, and 254 overweight or obese participants from four studies. The ages of participants ranged from 20 to 60 years. Artichoke extract doses varied across the 13 studies, ranging from 50 to 2,700 mg per day.
Characteristics of Studies Included in the Meta-analysis.
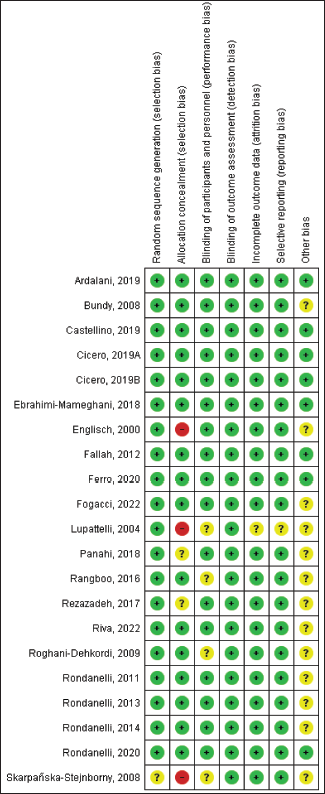
Quality Assessment
Upon evaluation of the randomization and reporting methods used in the 21 studies, only three were identified to have a high risk of bias (Englisch et al., 2000; Lupattelli et al., 2004; Skarpañska-Stejnborn et al., 2008). The majority of the studies were found to have a low risk of bias with regard to allocation concealment, blinding of participants and personnel, and blinding of outcome assessment. The selective reporting of most studies was adequately described. The quality of the included studies was deemed to be relatively high based on the Cochrane Handbook for Systematic Reviews of Interventions (Cumpston et al., 2019) as shown in Figure 2.
Primary Outcomes
Anti-hyperglycemic Effect
Nine research reports provided blood glucose results in a variety of outcomes, including FBS, HbA1c, serum fasting insulin, and HOMA-IR.
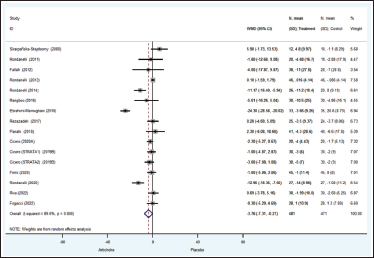
Fourteen investigations with a total of 952 patients reported FBS results (Cicero et al., 2019a; Cicero et al., 2019b; Ebrahimi-Mameghani et al., 2018; Fallah Huseini et al., 2012; Ferro et al., 2020; Fogacci et al., 2022; Panahi et al., 2018; Rangboo et al., 2016; Rezazadeh et al., 2018; Riva et al., 2021; Rolnik & Olas, 2021; Rondanelli et al., 2011, 2013, 2014). The effects of artichoke on FBS were significantly different from those of the control group (WMD = −3.76, 95%CI: −7.31, −0.22, p = 0.038) (Figure 3). The artichoke group had non-significantly lower HbA1c readings than the placebo group (WMD = −0.22%; 95%CI: −0.57, 0.13; p = 0.21). In addition, the pooled analysis revealed that the WMD of insulin levels in participants treated with artichoke differed from those in the placebo group (WMD −1.35 mIU/L; 95%CI: −2.29, −0.41; p = 0.005). There is statistically substantial variability in these results.
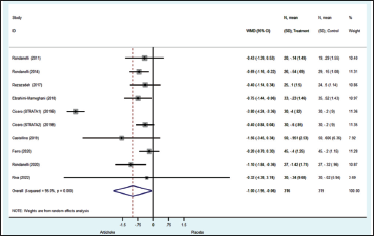
Compared to placebo, the artichoke intervention lowered HOMA-IR significantly (WMD = −1.00; 95%CI = −1.95, −0.06; p = 0.037). There was no evidence of heterogeneity for these outcomes (Figure 4).
Anti-hyperlipidemic Effect
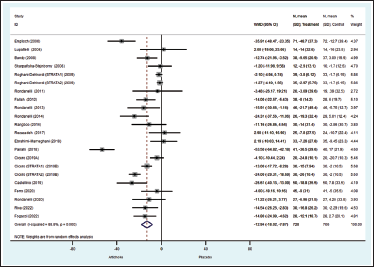
Twenty trials (Bundy et al., 2008; Castellino et al., 2019; Cicero et al., 2019a; Cicero et al., 2019b; Ebrahimi-Mameghani et al., 2018; Englisch et al., 2000; Fallah Huseini et al., 2012; Ferro et al., 2020; Fogacci et al., 2022; Lupattelli et al., 2004; Panahi et al., 2018; Rangboo et al., 2016; Rezazadeh et al., 2018; Riva et al., 2021; Roghani-Dehkordi & Kamkhah, 2009; Rondanelli et al., 2011, 2013, 2014, 2020; Skarpañska-Stejnborn et al., 2008) documented artichoke’s clinical therapeutic effect on lipid profile. LDL levels in the artichoke-treated group were considerably lower than those in the placebo group (WMD −12.94 mg/dL; 95%CI −18.02, −7.87; p < 0.0001), with signs of heterogeneity across trials (I2 = 90%, p < 0.00001) (Figure 5). The meta-analysis of 20 trials revealed a substantial reduction in TC when compared to the placebo group (WMD −19.64 mg/dL; 95%CI −23.94, −15.35; p < 0.00001), with indications of heterogeneity across studies (I2 = 83.3%, p < 0.00001). TG levels were dramatically reduced in the artichoke-treated group compared to the placebo group (WMD −13.36 mg/dL; 95%CI −19.06, −7.66; p < 0.0001). The pooled data indicated heterogeneity across trials (I2 = 81.90%, p < 0.00001).
In contrast to the placebo, however, artichoke had no influence on HDL levels. The pooled WMD was 0.21 mg/dL (95%CI: −1.52, 1.95; p = 0.81), and there was evidence of heterogeneity across trials (I2 = 99.72%, p < 0.00001).
Anti-hypertensive Effect
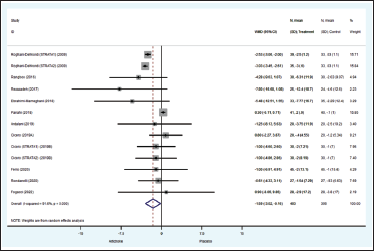
The aggregated meta-analysis revealed that the artichoke group had substantially lower SBP than the placebo group (WMD −1.59 mmHg; 95%CI −3.02, −0.16; p = 0.029) (Figure 6). Nevertheless, the administration of artichoke did not result in a significant reduction in DBP when compared to the placebo group (WMD −1.02 mmHg; 95%CI −2.207 to 0.159; p = 0.09), despite considerable heterogeneity observed across the trials.
Anti-obesity Effect
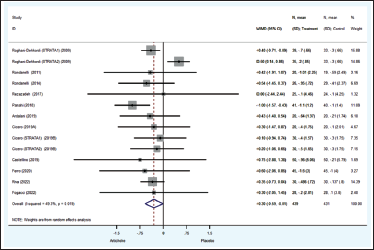
Eleven studies (Ardalani et al., 2020; Castellino et al., 2019; Cicero et al., 2019a; Cicero et al., 2019b; Ferro et al., 2020; Panahi et al., 2018; Rezazadeh et al., 2018; Riva et al., 2021; Roghani-Dehkordi & Kamkhah, 2009; Rondanelli et al., 2014, 2011) with a total of 870 patients reported BW and BMI. The aggregated findings revealed that the impact of artichoke on BW differed significantly from that of the control group (WMD −1.17 kg; 95%CI −1.75, −0.60; p < 0.001). This result exhibited no statistically significant heterogeneity. While there was no statistically significant difference between the artichoke group and the control group with regard to BMI (WMD −0.30 kg/m2; 95%CI −0.59, −0.01; p = 0.042), heterogeneity across trials (I2 = 49.3%, p = 0.019) was present (Figure 7).
Secondary Outcome
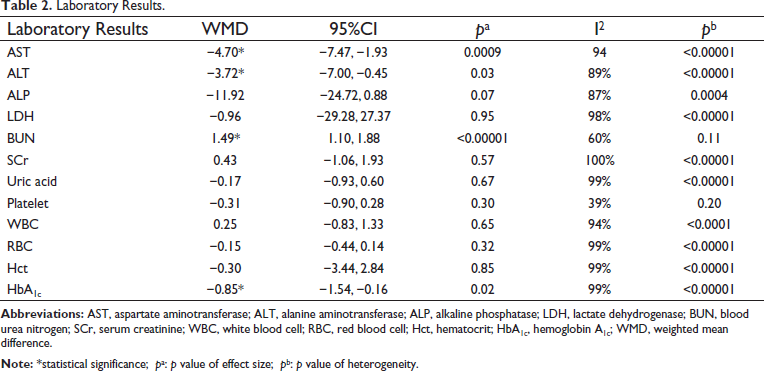
The pooled research revealed that individuals treated with artichoke had significantly lower liver function tests. The estimated WMD effects on AST and ALT were −4.70 U/L (95%CI −7.47 to −1.93; I2 = 94.0%) and −3.72 U/L (95%CI −7.00 to −0.45; I2 = 89%), respectively. In addition, there were statistically significant changes in Hgb levels (WMD −0.85 g/dL; 95%CI −1.54 to −0.16; I2 = 99%). Table 2 provides further information and evidence of heterogeneity for all laboratory findings.
Laboratory Results.
Sensitivity Analysis and Subgroup Analysis
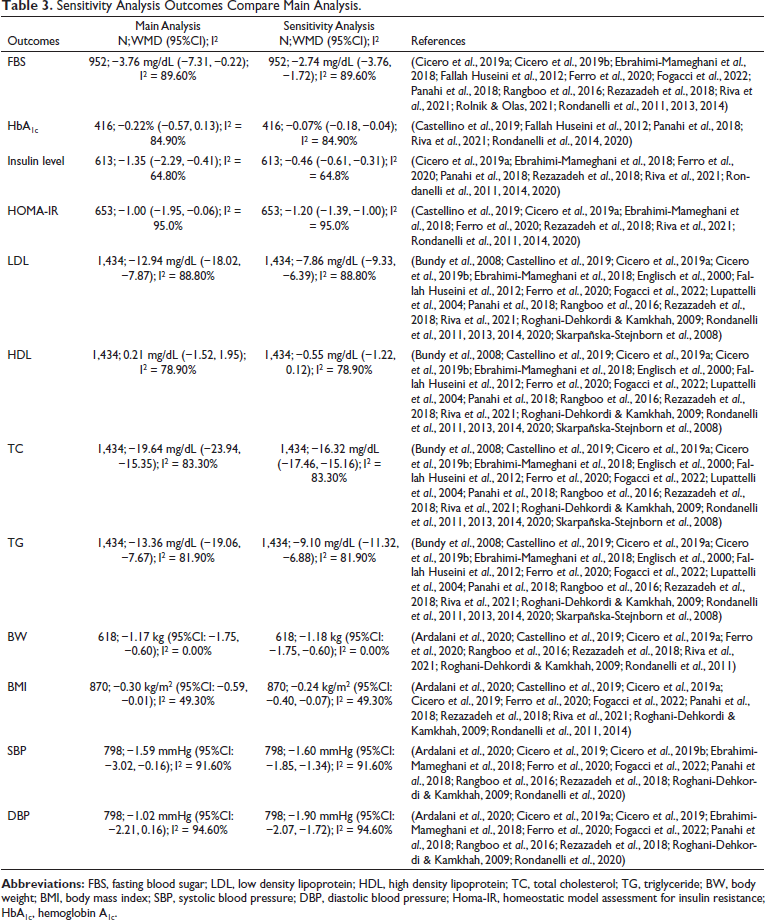
In this research, we examined the effect of removal on study findings and heterogeneity by using the one-study-removed method, in which one study is omitted at a time. Compared to the main analysis, the one-study-eliminated method did not reveal any differences in any of the outcomes. In addition, the analyses from the random effect model were replaced with the fixed effect model in order to assess the sensitivity of each outcome in the present research. The fixed effect model of the sensitivity analysis produced diverse BMI results (Table 3).
Sensitivity Analysis Outcomes Compare Main Analysis.
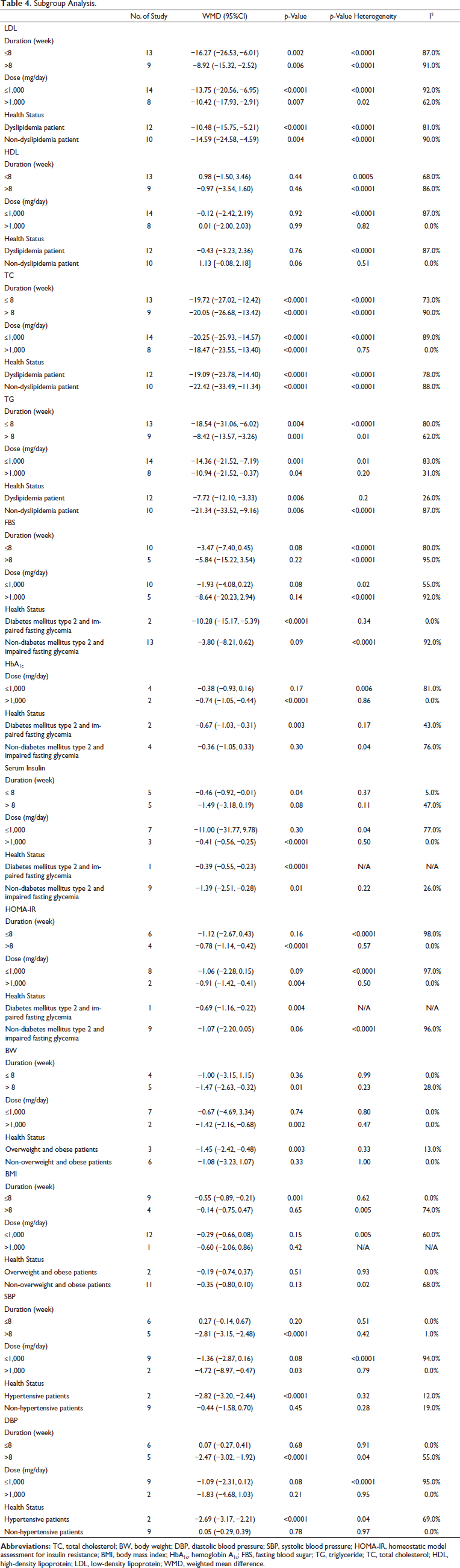
The analysis of subgroups based on treatment duration, dosage, and health condition revealed that the therapeutic effectiveness of artichoke doses more than 1,000 mg per day and treatment durations longer than 8 weeks improved LDL, TC, TG, HbA1c, serum insulin, HOMA-IR, SBP, DBP, and BW. While artichoke resulted in considerably improved outcomes for participants with type 2 diabetes and impaired fasting glycemia, as well as overweight and obese individuals and hypertensive patients, the results were not statistically significant (Table 4)
Subgroup Analysis.
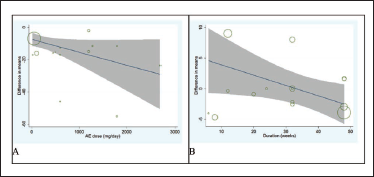
Meta-regression
Meta-regression was used to evaluate the effect of possible variable responses on the primary outcomes of the included trials. The findings demonstrated a significant correlation between dosage and changes in mean TC (slope: −7.37; 95%CI: −14.31, −0.44; p = 0.039) (Figure 4A), SBP (slope: −3.12; 95%CI: −4.89, −1.35; p = 0.008) and DBP (slope: −3.12; 95%CI: −3.82, −1.76; p = 0.002). In addition, a significant correlation was detected between duration and changes in mean LDL (slope: −2.51; 95%CI: −4.21, −1.79; p = 0.007) (Figure 8), TC (slope: −2.57; 95%CI: −4.45, −1.69; p = 0.011), and DBP (slope: −0.07; 95%CI: 0.11, 0.04; p = 0.002) levels.
Publication Bias
Egger’s test was used to investigate publication bias for FBS (intercept, −1.35; SE = 2.09; 95%CI, −6.17, 3.47, t = −0.65, p = 0.540), HbA1c (intercept, −2.08; SE = 2.16; 95%CI, −8.07, 3.91, t = −0.97, p = 0.389), HOMA-IR (intercept, 1.75; SE = 3.50; 95%CI, −6.31, 9.82, t = 0.50, p = 0.630), HDL (intercept, 0.85; SE = 0.84; 95%CI, −0.89, 2.59, t = 1.02, p = 0.320), TC (intercept, −1.10; SE = 0.67; 95%CI, −2.45, 0.35, t = −1.56, p = 0.134), TG (intercept, −1.11; SE = 0.63; 95%CI, −2.43, 0.21, t = −1.76, p = 0.094), SBP (intercept, −0.12; SE = 1.26; 95%CI, −2.90, 2.65, t = −0.10, p = 0.924), DBP (intercept, 1.05; SE = 1.53; 95%CI, −2.31, 4.40, t = 0.69, p = 0.507), BW (intercept, 0.01; SE = 0.45; 95%CI, −1.02, 1.04, t = 0.02, p = 0.982), and BMI (intercept, −0.49; SE = 0.69; 95%CI, −1.99, 1.00, t = −0.72, p = 0.484). We discovered indications of publication bias using this method for insulin level (intercept, −1.37; SE = 0.43; 95%CI, −2.36, −0.38, t = −3.20, p = 0.013) and LDL (intercept, −2.21; SE = 1.01; 95%CI, −4.30, −0.10, t = −2.19, p = 0.041). In addition to utilizing funnel plots for all of the analyzed outcomes, we visually inspected the plots to identify publication bias.
Discussion
This study conducted a meta-analysis and meta-regression of several RCTs that investigated the impact of artichokes on cardiovascular outcomes. In total, 12 different outcomes were assessed, and the results suggest that taking artichoke supplements can significantly enhance various parameters such as blood glucose, lipid profile, anthropometric measures, and blood pressure.
The results of our study are consistent with previous clinical research, systematic reviews, and meta-analyses that have examined the hypolipidemic efficacy of artichoke. For example, Sahebkar et al. (2018) conducted a meta-analysis that used artichoke extract and found a significant reduction in LDL, TC, and TG levels with artichoke extract administration. However, no significant changes were observed in HDL levels compared to the placebo group. Another study by Santos et al. (2018) demonstrated the beneficial effects of artichoke extract on improving the lipid profile in individuals with various health conditions, including hypercholesterolemia, metabolic syndrome, overweight, type 2 diabetes mellitus, non-alcoholic fatty liver disease, and non-alcoholic steatohepatitis. These findings support the use of artichoke extract as a natural approach to manage dyslipidemia and related health conditions. Furthermore, our meta-analysis contributes to the expanding body of evidence on the cardiovascular benefits of artichoke extract by demonstrating improvements in blood glucose, anthropometric parameters, and blood pressure, in addition to the lipid profile.
Jalili et al. (2020) have suggested that the administration of artichoke products can significantly reduce FBS. However, other glycemic indices such as fasting insulin, HOMA-IR, and HbA1c did not show significant changes after the administration of artichoke and its products. Our meta-analysis results demonstrated that consuming artichokes can lead to improved blood glucose levels and reduced insulin resistance. One possible mechanism for this effect is the presence of bioactive compounds in artichokes that have a hypoglycemic effect by inhibiting gluconeogenesis and the glycolytic process, as well as suppressing the glucose-6-phosphate-translocase enzyme. Artichoke extract also acts as an inhibitor of the intestinal glucose carrier. Additionally, caffeoylquinic acid, another component found in artichokes, can inhibit α-glucosidase and exert a synergistic effect to control the glycemic index (Rondanelli et al., 2013). These findings align with previous studies (Jalili et al., 2020; Rondanelli et al., 2013) which demonstrated the hypoglycemic effect of artichokes and their products.
Another mechanism by which artichoke acts as a hypoglycemic agent is through inhibiting PTP1B, according to quercetin, a compound found in artichoke. (Ruiz-Cano et al., 2014). The previous experiment showed that quercetin compound inhibiting PTP1B can improve insulin resistance and can be effective in treating type 2 diabetes and obesity (Choi et al., 2008). The recently reported recommended PTP1B inhibitors offer new perspectives for the development of new drugs to treat type 2 diabetes (Saeed et al., 2021).
Various studies have investigated the potential mechanisms responsible for the blood glucose-lowering and lipid-reducing effects of artichokes. One mechanism that has been explored is the reduction of insulin resistance, as prebiotics present in artichokes have been shown to improve glucose response and decrease post-prandial insulin secretion. Artichokes contain prebiotics such as inulin and fructooligosaccharides, which are non-digestible and reach the lower gastrointestinal tract as substrates. These prebiotics bind bile acids and reduce solubilized cholesterol, leading to decreased plasma concentrations of TC, LDL, and TGs (Yeo et al., 2009). Artichoke extracts have also demonstrated protective properties by reducing intracellular reactive oxygen species (ROS) production and inhibiting the oxidation of LDL in human cells (Miláčková et al., 2017; Zapolska-Downar et al., 2002). Additionally, a study by Gebhardt (Gebhardt, 1998) has shown that artichokes can indirectly inhibit HMG-CoA, a key enzyme in cholesterol biosynthesis, contributing to its lipid-lowering effects.
Furthermore, our results suggest that artichokes may have a favorable effect on anthropometric parameters. Studies conducted in animals have indicated that artichoke leaf extract can significantly decrease serum levels of TGs, TC, and LDL-C while increasing HDL levels in mice that were fed a high-fat diet (Azeem et al., 2016). Additionally, artichoke has been shown to increase lipolysis and decrease lipogenesis in mice, which results in enhanced energy utilization in the liver and brown adipose tissue, as well as the stimulation of lipolysis in white adipose tissue (Mahboubi, 2018).
The study findings suggest that the outcomes were not significantly affected by the exclusion of any individual study, indicating the reliability of the results. However, the results of sensitivity analyses using fixed effect models for BMI were inconsistent, suggesting that additional investigations may be required to comprehensively assess the effects of artichoke on BW.
Our results showed that artichoke decreased SBP significantly. A recent meta-analysis has reported that the consumption of artichokes is associated with a significant reduction in both systolic and DBP in hypertensive individuals (Moradi et al., 2021). This hypotensive effect may be due to the presence of phenolic compounds, such as cynarin, chlorogenic acid, luteolin, cynaroside, and caffeic acid, which have antioxidant properties, can potentially reduce oxidative stress, and subsequently lower blood pressure (Baradaran et al., 2014; Halvorsen et al., 2002; Rangboo et al., 2016). Artichoke extract has also been shown to decrease the expression of inflammatory enzymes, possibly by inhibiting the activity of NF-κB in human cells, suggesting that artichoke consumption may have anti-inflammatory effects and contribute to improved cardiovascular function (Miláčková et al., 2017). Overall, the abundant phenolic compounds and anti-inflammatory properties of artichokes make them a promising dietary intervention for improving blood pressure regulation and cardiovascular health.
The potential mechanisms of artichoke in treating cardiometabolic outcomes were improved glucose metabolism and insulin sensitivity, which could be significant for individuals with prediabetes and type 2 diabetes. It also contributes to the improvement of lipid profiles by reducing LDL cholesterol levels through the inhibition of synthesis and the promotion of excretion. Furthermore, artichoke has the ability to regulate blood pressure by inducing vasodilation. In addition, it is worth noting that artichoke exhibits anti-inflammatory characteristics and has been found to have an impact on the composition of gut bacteria.
Nevertheless, the previously documented clinically noteworthy enhancement was characterized by a reduction of 20 mg/dL in FBS, a decrease of 10 mg/dL in LDL, a decline of 40 mg/dL in TG, and an increase of 5 mg/dL in HDL-C. According to the reference provided, a reduction in SBP or DBP by 5 mmHg and a moderate weight loss of 5% to 10% were found to be related to notable improvements in clinical outcomes (Macek et al., 2020). Therefore, artichoke can be recommended as a dietary or complementary intervention in the prevention and management of cardiometabolic disorders.
Artichoke had no major side effects in any of the trials. Additionally, the renal and hepatic function meta-analysis showed no significant changes across the groups. However, prior investigations have linked artichoke treatment to diarrhea, weakness, hunger, and flatulence. Note that these adverse occurrences are rare (Kraft, 1997). Concerns have been raised about artichoke and diabetic and hypertension medicines. Metformin, gliclazide, enalapril, hydrochlorothiazide, amlodipine, and aspirin with artichoke may cause distal muscle pain, severe anemia, standard biochemical liver cholestasis, alkaline phosphatase elevation, and hyperleukocytosis. Patients have been admitted to emergency departments due to these side effects. We hypothesized that artichoke-derived phenolic and flavonoid compounds may disrupt metabolic processes largely mediated by the CYP450 3A4 and 2C9 enzymes (Campos et al., 2018). Therefore, using artichoke in those on the above drugs may require long-term laboratory monitoring.
In this study, subgroup analyses based on treatment duration, dosage, and health condition showed that higher doses of artichoke (>1,000 mg/day) and longer treatment durations (>8 weeks) were linked to improved outcomes for several markers of cardiovascular health, including LDL, TC, TG, HbA1c, serum insulin, HOMA-IR, SBP, and DBP. Furthermore, artichoke consumption was found to be especially advantageous for individuals with type 2 diabetes and impaired fasting glycemia, as well as those who were overweight or obese and hypertensive. However, the results were not statistically significant for these subgroups.
The results suggest that artichoke consumption could have a favorable effect on several cardiovascular health markers, particularly when higher doses and longer treatment durations are employed. Nevertheless, additional research is necessary to comprehend the precise mechanisms underlying artichoke’s impacts and to clarify its potential advantages for specific subgroups of individuals. It should be emphasized, however, that the lack of statistical significance in certain subgroup analyses may be attributed to the relatively small sample sizes and the heterogeneity of the studies included. This highlights the need for larger and more homogenous studies in the future.
The present study utilized the meta-regression analysis to determine the relationship between artichoke dosage, duration of treatment, and changes in the primary outcomes of the included studies. The findings revealed that higher dosages of artichoke led to greater reductions in mean TC, SBP, and DBP, emphasizing the crucial role of artichoke dosage in its therapeutic effectiveness for cardiovascular health. Furthermore, longer treatment periods produced greater reductions in mean LDL, TC, and DBP, suggesting that a prolonged period of artichoke treatment may be necessary to achieve significant improvements in cardiovascular health. These results have important implications for future clinical studies aimed at investigating the potential of artichoke as a therapeutic agent for cardiovascular health, indicating that higher dosages and longer treatment periods may be required to achieve notable improvements in various markers of cardiovascular health.
The current meta-analysis indicates a favorable effect of artichoke on the outcomes examined, despite the heterogeneity observed and the relatively small sample sizes of the included studies. The variations in participant characteristics, artichoke types, and treatment duration may have contributed to the heterogeneity. Moreover, the therapeutic effects of artichoke may depend on several factors such as harvesting and storage conditions, plant part used, and extraction methods (Matthes & Honermeier, 2007). Other potential sources of heterogeneity include variability in study design, co-interventions, and patient compliance. Several RCTs combined artichoke with other medications or herbal remedies, which may have affected the observed effects. However, this approach also increases the generalizability of the results to real-world practice. Strengths of the study include the use of RCT data and appropriate study designs to assess the intervention’s impact.
Limitations
Several limitations of our study need to be taken into account. Firstly, the number of studies included in our meta-analysis was relatively small, which may increase the possibility of small-study effects. Secondly, our study only used data from previously published systematic reviews and meta-analyses, which might have excluded essential information from primary studies that have not yet undergone these types of evaluations. Additionally, the included studies employed various doses, preparations, and dosing regimens of artichoke, which were not accounted for in the analysis and could have influenced the outcomes. Therefore, future studies should consider investigating these factors more comprehensively. Finally, the long-term safety and benefits of artichoke are still unclear, as most included studies reported only the short-term use of up to six months, and only a few reported on the long-term safety of artichoke.
Conclusion
In conclusion, the consumption of artichokes has the potential to enhance blood glucose levels, lipid profiles, anthropometric parameters, and blood pressure. Nevertheless, it does not necessarily exert an impact on HDL levels and DBP. In addition to the varying products used, there are also methodological constraints that add unpredictability to the results, such as the fluctuating duration of the trials. However, based on our analysis, we conclude that further experiments are necessary, albeit with modifications to the research design such as the establishment of a standardized formulation and dosage protocol for artichoke. Furthermore, according to our analysis, large-scale RCTs examining the impact of artichoke on long-term cardiometabolic outcomes could provide valuable insights.
Footnotes
Abbreviations
RCTs: Randomized controlled trials; FBS: Fasting blood sugar; FPG: Fasting plasma glucose; PPG: Postprandial blood glucose; HBA1c: Hemoglobin A1c; HOMA-IR: Homeostatic model assessment for insulin resistance; LDL-c: Low-density lipoprotein cholesterol; HDL-c: High-density lipoprotein cholesterol; TC: Total cholesterol; TG: Triglyceride; SBP: Systolic blood pressure; DBP: Diastolic blood pressure; BW: Body weight; BMI: Body mass index; PRISMA: Preference reporting items for systematic review and meta-analysis; MESH: Medical subject headings; WMD: Weighted mean difference; Ci: Confidence interval; RR: Risk ratio; PTP1B: Protein tyrosine phosphatase 1B; ROS: Reactive oxygen species
Acknowledgments
We would like to thank Prof. Arian Roderick Plant for language assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Not applicable.
Funding
This research project was financially supported by Mahasarakham University (Grant Number 2566).
