Abstract
Background
Isoproterenol is a drug used for the treatment of bradycardia that has many side effects due to its non-selective β-adrenoceptor agonist properties.
Aim
In this study, we evaluated an in vivo experimental model for isoproterenol (ISO) cardiac injury using BALB/c mice and protective treatment with a combined optimized dose of Arctiin (AR) and Lactobacillus plantarum (LP) (ARLP; 30 mg/kg b.w. AR and viable 106 CFU/mL LP p.o.) together.
Introduction
Isoproterenol causes cardiac injury in many instances. Arctiin is a naturally occurring lignin glycoside present in many herbal medicinal plants that has many beneficial effects for humans. Lactobacillus plantarum is yet another beneficial probiotic that benefits largely to humans.
Materials and Methods
Male BALB/c mice in appropriate groups (6 animals each) were treated with ARLP for 7 days with ISO administration (100 mg/kg i.p.) on days 5 and 6. On the 8th day, animals were sacrificed. Serum and heart samples were collected for further processing. CK-MB, cardiac troponin, AST, ALT, LDH, GST, GPx, CAT, SOD, GSH, GSSG, MDA, nuclear factor kappa B (NFκB), TNF-α, and IL-6 by ELISA and gene expressions of nuclear factor erythroid 2-related factor 2 (Nrf2) were estimated in samples.
Results
Mice administered with ISO showed a prominent rise in the serum marker enzyme activities and tissue oxidative stress markers (MDA and GSSG). Furthermore, marked reductions in body weight and enzymatic and non-enzymatic antioxidant levels were noticed in ISO toxicity. Alterations in proinflammatory cytokines (TNF-α and IL-6) and Nrf2 levels by RT-PCR were analyzed. Histopathology of the heart also indicated cardiac injury in ISO-intoxicated mice. ARLP pretreatment prevented all these cardiac injuries and exerted significant (p ≤ 0.05) prophylactic protection toward cardiac tissue. Further, we analyzed the possible interactions between AR and LP in vitro, to understand the influence of ARonLP, which showed no significant suppressive/antibacterial activity on LP.
Conclusion
In conclusion, ARLP exerts a strong cardioprotective and anti-inflammatory activity in cardiovascular injury.
Introduction
Heart failure is the eventual result of multiple cardiac dysfunctions, health imbalances, and inflammation (Tang et al., 2019). It has been termed an “epidemic,” which affects 5.6 million people in the United States each year. More than 23 million people are affected worldwide (Roger, 2013). Clinical management of acute heart failure itself meets several challenges because of the diverse population among patients, incomplete understanding of pathogenesis, and absence of strong evidence-based guidelines for treatment (Gheorghiade & Pang, 2009). Looking at the clinical condition as a disease, patients as victims, not only leads to the mortality and morbidity of the patients (Kurmani & Squire, 2017). This makes the treatment of most coronary artery diseases more expensive for the common man.
Heart failure is not only associated with cardiac overload or injury but also with various other factors collectively referred to as biomarkers. These biomarkers include enzymes, hormones, and biological substances that indicate cardiac stress, malfunction, or myocyte injury (Braunwald, 2008). Individuals with heart failure are typically identified with the presence of systemic inflammation, marked by the elevated levels of several inflammatory cytokines. However, the mechanism for systemic inflammation was not known so far (Yndestad et al., 2006).
Plant-derived antioxidants are increasingly employed against several cardiovascular diseases. These natural bioactive sources have gained sufficient significance in reviving the traditional system of medicine. Herbal medicines have played an important role in health care for a large population of the world throughout history. Statistical studies reveal phytochemical medicines as the “choice of drug” during the past 20th century. There is a wide range of medicinal plants that have been used for the treatment of heart diseases (Shah et al., 2019).
Arctiin is an active compound extracted from Arctium lappa L. Well-documented literature regarding significant anti-inflammatory, anti-viral, antioxidant, anti-tumor, and anti-cardiac hypertrophy has been found. The report specifically points out the anti-inflammatory effects of arctiin caused by lipopolysaccharide (LPS) and the influenza virus (Zhang et al., 2020). Arctiin has been reported to present an inhibitory effect on myocardial fibrosis and attenuate myocardial hypertrophy via suppressing inflammatory response (Li et al., 2017).
A previous investigation on the mice model revealed that arctiin can potentially reduce tissue dysfunction and oxidative stress by scavenging reactive oxygen species (ROS), restoring mitochondrial functions and diminishing necroptosis. The study also reveals a mechanism that is, induction of nuclear factor kappa B (NF-κB) deactivation, which explains its anti-inflammatory and anti-viral properties (Chen et al., 2020). A recent review on the pharmacological properties of arctigenin and arctiin discloses the ameliorative effects of arctiin associated with metabolic disorders and central nervous system (CNS) dysfunctions moderately due to their anti-inflammatory properties (Gao et al., 2018).
Lactobacilli, a type of bacteria that produces lactic acid, is part of the indigenous microbial community found in the small and large intestines of both humans and animals. Lactobacilli, predominantly non-pathogenic micro-organisms, have been observed to generate antibiotic compounds including bacteriocins and hydrogen peroxide (H2O2). Consequently, since the early 1900s, it has been hypothesized that they engage in combat with pathogenic bacteria and provide advantageous effects on human health (Herías et al.,1999). Lactobacillus plantarum is a common species of Lactobacilli and is reported to have extensive biological activities (Peng et al., 2014). Therefore, in this study, we hypothesized that L. plantarum can be beneficial in enhancing the cardioprotective properties of Arctiin.
Therefore, the current study aims to investigate the cardioprotective potential of Arctiin with L. plantarum against the pathogenesis of ISO-induced cardiac dysfunction in mice with symptoms of acute heart failure.
Materials and Methods
Chemicals
Arctiin of >98% purity was purchased from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). Mouse tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6), ELISA kits were purchased from BioLegend (CA, USA). ISO and other chemicals of analytical grade were purchased from Sigma Aldrich, USA.
Collection of Bacterial Strain
L. plantarum was collected from ATCC, USA. The strain was grown on Rogosa agar plates (Difco Labs, MI, USA).
Animal Model
24 healthy BALB/c mice, 8 weeks old that weighed 20 ± 3 g were obtained from the institutional animal facility and housed under standard laboratory conditions. Animals had free access to water and fed pellets for an acclimatization period of 7 days in the animal house before investigations. All experimental study protocols adopting ARRIVE guidelines were reviewed and approved by the institutional animal care and use committee prior to initiation of the research (Approval Number: SXCT2021-2R).
Experimental Design
The rodents were separated into 4 groups each containing 6 animals. The treatment protocols of the respective groups were as follows:
The body weight of all the rodents was recorded daily until the end of the experimentation. The animals were sacrificed by cervical decapitation on the 8th day under anaesthesia using pentobarbital sodium (40 mg/kg). Blood and heart tissue samples were rapidly collected as per the need for various assays.
Organ Index
Heart tissue collected was rinsed with ice-cold potassium chloride (KCl) and was blotted dry with sterile filter paper and weighed to determine the organ index (heart to body weight). After weighing the organ was homogenized using 0.15 M KCl in a homogenizer. The contents were then centrifuged at 3000 rpm for 15 min to collect the supernatant. The collected supernatant was stored at −80°C for further analysis.
Serum Biochemical Markers
The collected blood samples post-decapitation were centrifuged to separate the serum for the biochemical analysis. Cardiac markers creatine phosphokinase (CK-MB), lactate dehydrogenase (LDH), and troponin-T were determined according to the method of Okinaka et al. (1961). Serum alanine transaminase (ALT) and aspartate transaminase (AST) were assayed as per the protocol suggested by Reitman and Frankel (1957). The tests were carried out with the help of commercially available kits and following the manufacturer’s instructions. The total protein in the samples was determined by the standard protocol of Lowry et al. (1951).
Oxidative Stress Markers
The heart tissue homogenate and serum collected were tested for malondialdehyde (MDA), which gives an estimation of thiobarbituric acid (TBARS) related products in the sample. For this, the heart tissue was homogenized and centrifuged at a low rpm for 30 min. In samples in which TBARS was observed, the serum was mixed with 0.1 mL of 8.1% SDS, 1 mL of 20% acetic acid, and 1 mL of 8% thiobarbituric acid (TBA). The prepared mixture was heated for 1 h. The tubes were cooled post-incubation and centrifuged further for 15 min. Absorbance at 532 nm was measured using the supernatant. TBARS levels were recorded as U/l in serum.
Li et al. (2012) described the method for the estimation of oxidized glutathione (GSSG). This method was adopted with a modification. The sample was mixed with metaphosphoric acid (MPA) and M2VP for further oxidation to determine the absorbance at 370 nm. The observed values were recorded as nmol/g of tissue.
Enzymatic and Non-enzymatic Antioxidants
Homogenate of the heart tissue was centrifuged and the supernatant collected was used to quantify the level of antioxidants (enzymatic and non-enzymatic).
The superoxide dismutase (SOD) value was estimated as per the protocol of Marklund and Marklund (1974). The technique detects the enzyme with its capability to prevent superoxide-reduction. A unit of SOD was assumed to be the quantity required to inhibit 50% pyrogallol oxidation. The sample and reagents were well mixed, and incubated at 37°C for 20 min. The absorbance was observed at 450 nm and recorded as U/mg of protein.
Aebi (1974), suggested the method for the estimation of catalase (CAT). The test sample was incubated with H2O2 and sodium chloride (substrate) at 37°C for 1 min. The solution was incubated until the appearance of yellow colouration, and the absorbance was measured at 405 nm. A unit of CAT is the enzyme activity needed to decompose 1 µM of H2O2 per min. The absorbance was recorded as µM of H2O2 decomposed/min/mg of protein.
Glutathione peroxidase (GPx) was estimated with the protocol adopted from Flohé and Günzler (1984). The supernatant was mixed with PBS, Sodium azide, reduced glutathione (GSH) and H2O2 and made up to 1 mL. The solution was incubated for 15 min at 37°C. The reaction was stopped using 5% TCA. The solution tubes were centrifuged at 10,000g for 8 min and the supernatant was collected. The absorbance of the supernatant was measured at 420 nm and recorded as µM of GSH oxidized/min/mg of protein.
Total glutathione-S-transferase (GST) levels were estimated according to Habig et al. (1974). The sample was mixed with 1-chloro-2,4-dinitrobenzene followed by 10 nM 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) for 24 h. After the reaction, the absorbance was measured at 340 nm and recorded as nanometer of 1-Chloro-2,4-dinitrobenzene (CDNB) conjugated/min/mg of the protein.
GSH was estimated by adopting the method of Aebi (1974). The samples were diluted using phosphate EDTA buffer and incubated for 5 min at room temperature. The solution was mixed well and incubated for 15 min again. The resulting solution was used to measure the absorbance at 350 nm and the value was recorded as mg/dL for serum and µmol/g for heart tissue.
Quaife and Dju (1949) describe the protocol for the quantification of vitamins C and E content using the principle of colorimetry. The assay detects the compound as there is a decrease in the concentration of tocopherols when the ferric ions in the solution are converted to ferrous ions indicated by a red colouration. The sample was added with ethanol and mixed vigorously. Xylene was then added to the resulting mixture and incubated in the dark. The solution was centrifuged for 30 min and ferric chloride solution was added. The solution was mixed well and the absorbance was recorded at 562 nm and expressed as µmol/L for both vitamins E and C.
Inflammatory Cytokines
The supernatant from the centrifuged heart tissue homogenate was used to estimate the titre values of the TNF-α, and IL-6 through the method of ELISA following the protocol provided by the manufacturer (BioLegend CA, USA).
Quantitative PCR
The nuclear factor erythroid 2-related factor 2 (Nrf2) gene was quantitatively amplified using RT-PCR. RNA was isolated from the tissue homogenate using TRIZOLTXT reagent and treated with DNase-I to remove traces of DNA as per the protocol provided by the manufacturer. The extracted RNA was checked using a spectrophotometer at 260 nm for quality confirmation and stored at −80°C. cDNA was synthesized using 1 µg of total RNA isolated with a commercial kit. The primers for Nrf2 are 5′-TAGTGCCCCTGGAAGTGTCA-3′ (upstream) and 5′-TTGGGATTCACGCATAGGAG-3′ (downstream) was used. PCR was carried out keeping GAPDH as the internal control (IC). The amplification of the gene was quantified using the SYBR green quantitative polymerase chain reaction (qPCR) master mix.
Histopathology
Tissue sections of the heart were prepared and immersed in 10% buffered formalin for 2 days and serially dehydrated in graded ethanol. Upon further dehydration with formalin, tissue sections were fixed in paraffin wax. With the help of a microtome, tissue sections of 5 µm thickness were sliced and stained with hematoxylin and eosin for microscopic examination. Microscopic examination was done under 40X magnification to study the histological changes in experimental groups of animals.
Antibacterial Activity Assay
Stock solutions of Arctiin were prepared for anti-bacterial activity assay. Culture plates of L. plantarum was prepared using a growth medium, and the well-diffusion method was used with appropriate negative and positive controls. 10 µL sterile water was added as negative control and antibiotic solution was added as positive control. Arctiin at concentrations 2.5 mg/mL and 5 mg/mL was added. The plates were covered aseptically and incubated upright at 37°C in a thermostat-controlled hot-air incubator. The plates were observed for inhibition of bacterial growth after 24 h.
Results
Effect on Bodyweight of Mice
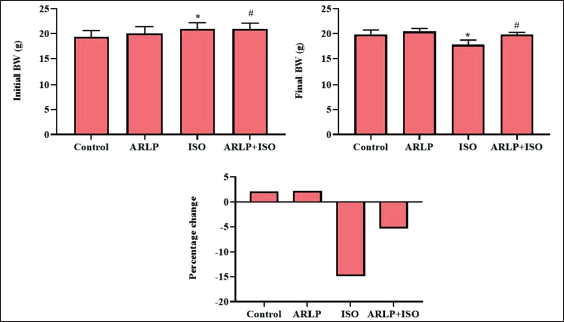
Change in the percentage of body weight was calculated from the recorded data and depicted in Figure 1, shows comparable values in all 4 groups. No significant change was observed in the index values between group I (Control) and II (ARLP). However, a significant (p≤0.05) decrease in the value (14.97%) was observed in group III (ISO). Group IV (ARLP+ ISO) showed a recovering value in comparison to group III. A 5.34% variation in the body weight was observed in the group after treatment, indicating the curative effect of Arctiin against the toxic shock produced by ISO.
Effect on Enzymatic Antioxidants Activities
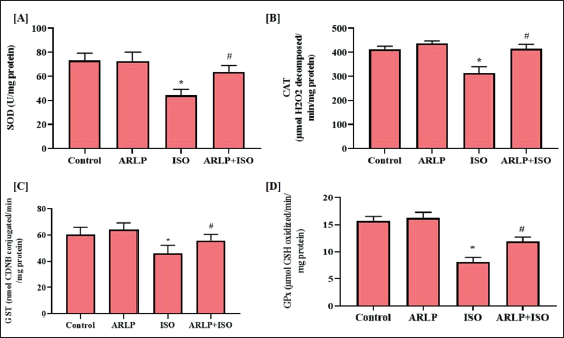
Activities of antioxidant enzymes such as SOD, CAT, GST, GPx were analyzed in the supernatant of the collected from the tissue homogenate. Figure 2 (A-D) demonstrates the activities of these enzymes as observed in their respective assays. All enzyme concentrations were reduced significantly in group III treated with ISO that induced abnormality in the heart. ARLP group II showed no significant reduction and were closer to the normal values as observed in the control group I. ARLP and ISO treated (group IV) showed a reduction in the toxicities of these enzyme activities when compared to the ISO (group III), indicating the curative effect of ARLP.
Effect on Biochemical Markers, Oxidative Stress Markers and Non-enzymatic Antioxidants
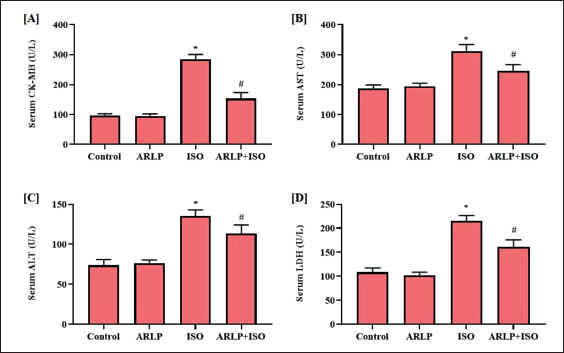
Highly elevated serum CK-MB (Figure 3A) activities were observed in group III intoxicated with ISO when compared to control. Group IV treated with ARLP also showed a relatively higher value of CK-MB, however, the value was observed to ameliorate towards the normal level when compared to the controls and group III. Figure 3B shows the activities of AST activity in the serum sample of the experimental rodents. AST levels are comparable in groups I (control) and II (ARLP). The higher AST elevation was observed in groups III. However, in group IV, a decrease in the activities of AST indicated the effect of ARLP in the test animals. Similarly, ALT activities (Figure 3C) were also elevated in group III which received ISO. Group IV showed a reduction in the ALT activities when compared to the controls and group II, indicating the cardioprotective potential of the ARLP treatment. Serum LDH results showed comparable results between groups I and II (ARLP) (Figure 3D). LDH activities were increased significantly in rodents administered with ISO (Group III). When comparing groups III, it was observed that group IV had a decreased LDH activities, but higher than the groups I and II.
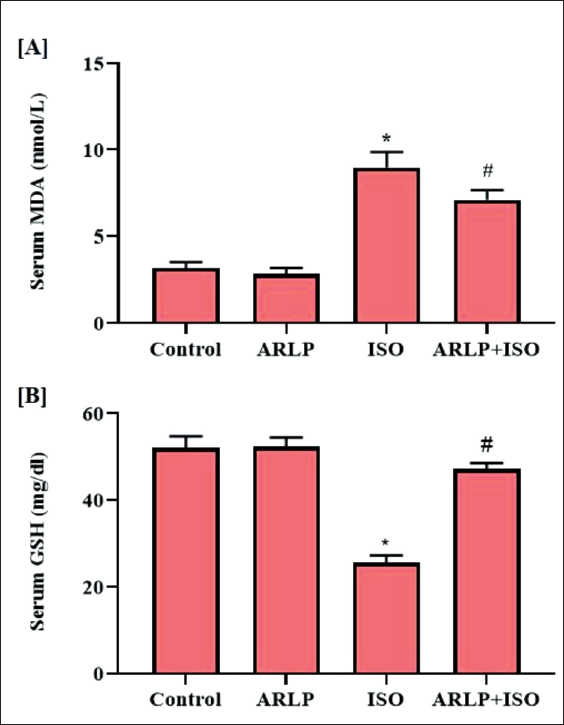
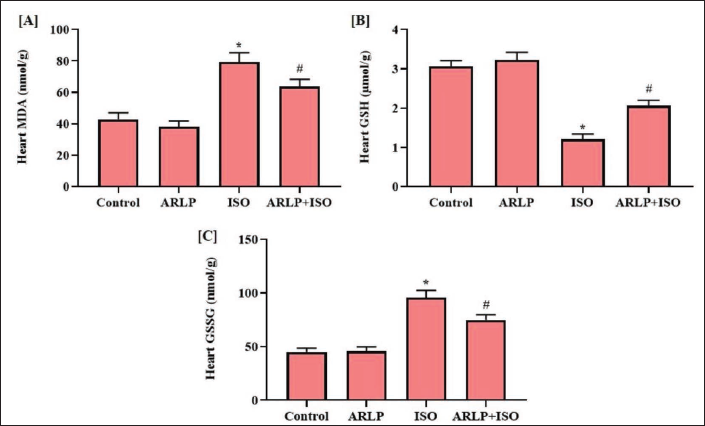
Malondialdehyde (MDA) and oxidized glutathione (GSSG) are biomarkers that indicate the prevalence of oxidative stress in the body through lipid peroxidation. All the experimental animals were screened for the presence of these biomarkers. It was observed that all the groups showed the presence these metabolites in the serum and heart tissue as shown in Figures 4 and 5. Groups I and II showed no difference in MDA levels. ISO group III presented a definite increase in MDA concentration in the serum and heart tissues. However, when compared to Group III, group IV showed a restrained MDA level. GSSG levels in the tissue samples were altered almost similar as observed for MDA levels. GSH is a natural non-enzymatic antioxidant produced in the body. Increased GSH levels in the serum are indicative of a healthy body. Experimental groups I and II showed a fairly normal level of GSH except Group III which was decreased due to ISO intoxication. Group IV that received ARLP along with ISO also showed a relatively significant higher level of GSH that was quite close to the control group I. Heart tissue homogenate showed a diminished concentration of GSH when compared to serum values and the least of which, was observed in group III, which was almost similar to the results of the serum. Group IV showed a normalized GSH level in comparison to group III.
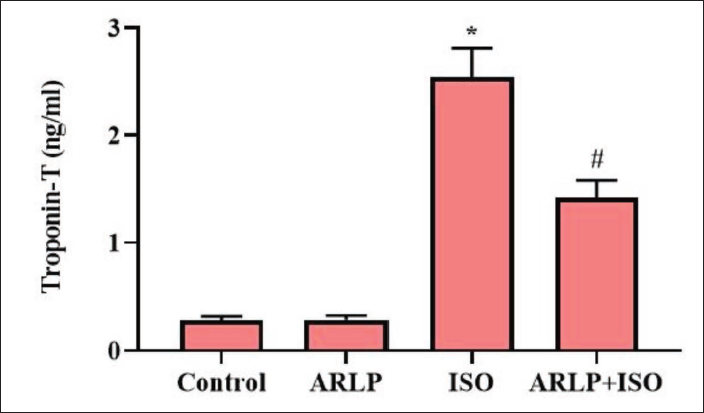
Highly elevated troponin-T levels were observed in group III intoxicated with ISO when compared to control (Figure 6) . Group IV treated with ARLP also showed a relatively higher value of troponin-T, however, the value was observed significantly ameliorated towards the normal level.
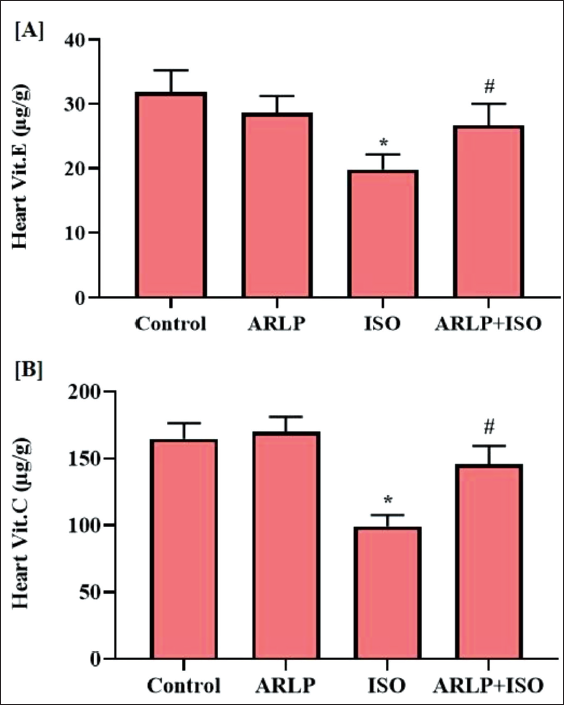
Vitamin E and C content were estimated from the heart samples taken from all the experimental animals. Results of the analysis showed that group III to witness a substantial reduction in levels of vitamin E and C as shown in Figure7A and 7B. Levels of vitamins in group IV were normalized when compared to the group III. Group II vitamin levels were similar to concentrations as in the control group of animals.
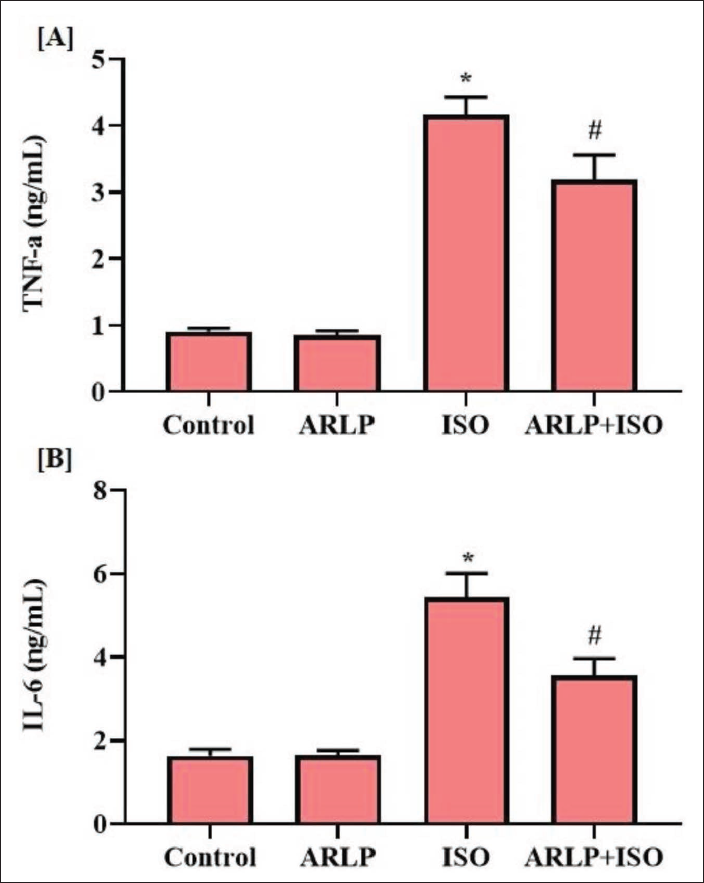
Effect on Inflammatory Cytokines
Cytokine (TNF-α and IL-6) levels in the serum were estimated to assess the anti-inflammatory potential of ARLP (Figure 8A and B). In comparison to the control mice, group III treated with ISO showed considerably elevated levels of TNF-α and IL-6, indicating the presence of severe inflammatory responses. However, group IV treated with ARLP showed a reduced cytokine level in comparison to group III ISO administered group.
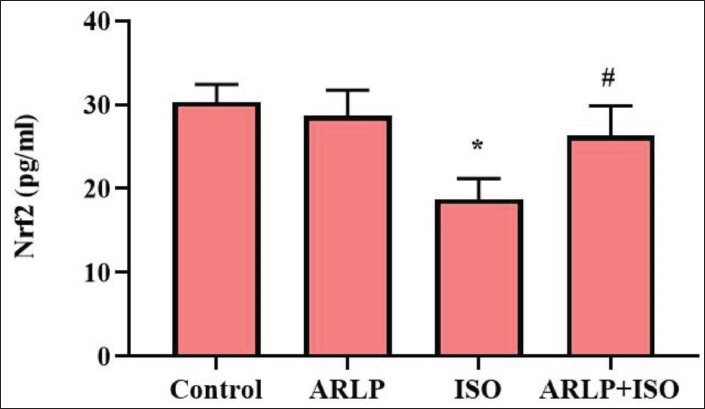
Effect on Nrf2 Level
Expression of Nrf2 was assessed using the RT-PCR technique to estimate the expression product of the gene. In Figure 9, results showed that ARLP alone does not affect the expression of Nrf2. Significant inhibition of the target gene was observed in group III during ISO intoxication. As seen in the results, ARLP treatment in ISO intoxication showed an improvement in the Nrf2 gene expression, indicating significant toxicity removal in group IV.
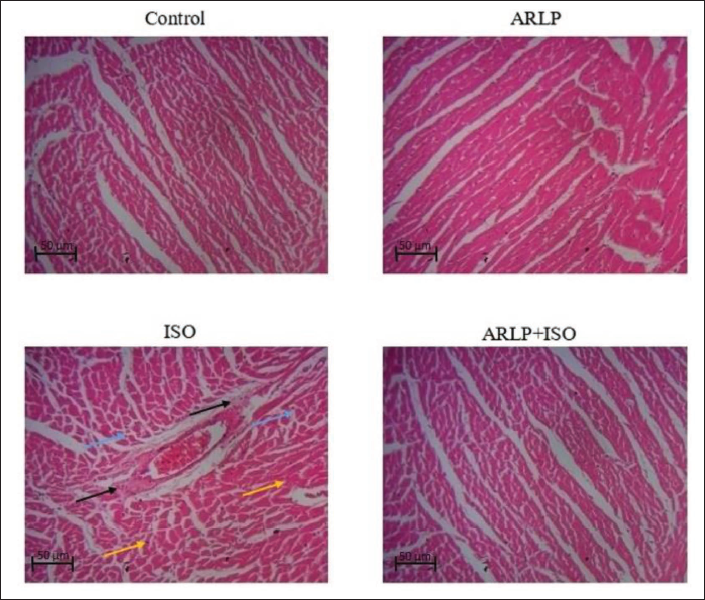
Histopathology
Microscopic examination of the tissue sections revealed evidence of cardiac injury during ISO administration as depicted in Figure 10. Results of group I (control) and group II (ARLP) showed normal striated myocardial tissue sections. Injury and inflammation were observed in ISO administration in group III tissues. The infarction area presented with focal necrosis in the myocardium, and spots of inflammation, recognized from irregular cells and displaced nuclei. The examination reported vacuolar changes due to edema and infiltration of leukocytes in the affected area. A significantly lower degree of inflammation was observed in group IV which was treated with ARLP, in comparison to group III treated with ISO. The results thus revealed the cardioprotective activity of ARLP against ISO-induced cardiac injuries.
Antibiogram
Antibiogram of arctiin against L. plantarum is shown in Figure 11. After incubation for 24 h, plates were observed for growth inhibitory activity of arctiin against the L. plantarum species used in this study. The well (Well-1) loaded with negative control presented no zone of inhibition. Also, no zone of inhibition was observed at both of the concentrations (Well-2 and Well-3) of arctiin against L. plantarum that was used in the present study. Positive control (Well-4) in the plate showed a clear zone of inhibition of 25 mm diameter around the well.
Antibiogram of Arctiin Against L. plantarum. The Arctiin did not Show Toxicity to the Growth of L. planetarum.

Discussion
Isoproterenol is a synthetic agonist that binds to the β-adrenoceptor. It has been reported to induce myocardial infarction in experimental animals as a response to an imbalance between the production of free radicals and antioxidative defence mechanisms. It induces an acute condition that is accompanied by elevated cardiac markers, lipid peroxides and inflammatory cytokines that damage the cardiac function. The pathophysiological events that cause heart failure in the mice model are concurrent to those of human myocardial infarction (Zhou et al., 2008). In the current investigation, we have aimed to induce heart failure conditions in rodents through ISO administration and study the curative effects of ARLP on it.
The organ index of the rodents treated with ISO witnessed a sharp decline in ISO toxicity. Biochemical profiling presented an elevation in all serum biochemical parameters. CK-MB isoenzyme is assayed to achieve an idea of the extent and persistence of infarction (Prabhu et al., 2006). Elevated levels of this enzyme indicate intense infarction condition of the heart in the experimental rodents.
Troponin-T is a specific biomarker that indicates cardiac injury. It is released into the circulation when myocytes are damaged due to oxygen deficiency (Panda & Naik, 2008). LDH, another biomarker was also elevated upon isoproterenol induction. Due to the injured myocardium, these biomarker enzymes including transaminases (AST and ALT) leaked into the circulation (Prince, 2011).
MDA is a marker for lipid peroxidation in the infarcted tissue, whereas, GSSG marks the presence of oxidative stress (Fathiazad et al., 2012; Saravanan et al., 2013). Increased levels of this molecule as evident from the assay results indicated the lipid peroxidation induction due to isoproterenol. The above-assessed parameters thus reveal the presence of cardiac dysfunction due to oxidative stress and inflammation. Rodents that received ARLP with ISO intriguingly suppressed the elevated biochemical profile during the investigation.
Antioxidant enzymes were estimated from the heart tissue homogenate. Glutathione-dependent enzymes (GPx and GSH), as well as anti-peroxidative enzymes (CAT and SOD), were immensely reduced upon isoproterenol intoxication. This may be a result of the non-available substrate, that is, glutathione indicative of the presence of oxidative stress (Farvin et al., 2004).
Vitamin E is a fat-soluble dietary supplement while vitamin C is a water-soluble dietary supplement for the body of most mammals. However, rats can produce their own vitamin C and act as antioxidants that scavenge for ROS and reduce symptoms of inflammation (Ognjanović et al., 2003). It was observed that the level of all antioxidant enzymes recovered along with the concentration of vitamins E and C upon administration of ARLP, which otherwise, was significantly suppressed in its absence.
Isoproterenol may inhibit the expression of TLR4 in the cardiac tissue and reduce the protein content of TLR adaptor proteins. As a result, TNF-α and IL-6 levels were observed to be substantially elevated (Soraya et al., 2012). Treatment with ARLP also suppressed the accretion of TNF-α and IL-6, at the inflamed area, to suppress the oxidative state and heal the intoxicated myocardium. The accumulation of the pro-inflammatory cytokines IL-6 may also arise as an inflammatory response to the cardiac injury caused by isoproterenol (Li et al., 2018).
Nrf2 is an induced transcription factor that is essential for the maintenance of redox signalling as a response to oxidative stress within cells. Nrf2 gene expression augmented the transcription activation of the genes for the production of antioxidant enzymes and its expression profiling explains the potential of the drug to show ameliorative effect at the gene level. The target gene (Nrf2) was seen to be inhibited or obstructed in ISO intoxication (as shown in Figure 7). This indicated the influence of ISO and its pathophysiology at the genetic level. Adequate expression levels of Nrf2 can relatively induce the downstream expression of antioxidants, enhance glutathione levels and prevent necrosis (Shanmugam et al., 2019). ARLP treatment promoted the upregulation of the gene expression, indicated by the higher level of Nrf2 in comparison to the control. This showed that ARLP was capable of inducing the production of natural antioxidant molecules in the host body through Nrf2 gene expression.
Histological evidence from the microscopic examination also reveals consensus results that showed signs of injury in the striated heart tissue. Myonecrosis and edema (infiltration of cytokines) due to myocarditis were also reported in the ISO-treated specimens. Degeneration of myocardial cells, fractured tissue fibers, and infiltration of inflammatory cells, have been previously reported in the isoproterenol induction study (Jiang et al., 2020). Upon ARLP administration, tissue cross-sections presented a lower degree of injury in the myocardium with more regularly placed cells and definite nuclei. Thus, the results indicated the potential of ARLP to treat cardiac dysfunction.
Previous studies on ISO-induced myocardial infarction also report the manifestation of ischemia, necrosis along other cardiac dysfunctions at a high dosage, which resemble the pathological alterations seen in human myocardial infarction (Li et al., 2012).
Recent studies with arctiin against cardiac injury describe the therapeutic properties of the natural compound in reducing necroptosis by suppressing necroptosis-relevant proteins RIPK1, p-RIPK1, MLKL, RIPK3, in the bloodstream and nearby injured tissue. Regulated necrosis is abbreviated as necroptosis which depends on RIPK1/RIPK3/MLKL pathways and is responsible for the myocardial and cerebral injury. The study reported notable elevation in the protein levels in ischemia-induced rat heart and hypoxia-induced cardiomyocyte (Chen et al., 2020).
Antibiogram results for arctiin presented no zones of inhibition when compared to the controls. This indicates that arctiin does not inhibit or suppress the growth of L. plantarum used. Thus, it was evident that the L. plantarum species used in the study might not be inhibited in combination with Arctiin in vivo. Perhaps, current findings revealed the therapeutic potential of Arctiin in combination with L. plantarum, thus the development of ARLP for the present study.
Conclusion
The current finding was reported for the first time that treatment of arctiin in combination with L. plantarum was an effective treatment against the cardiac injuries induced by ISO in mice models by influencing the NF-κB/Nrf2 network of genes and cytokines. Also, this natural drug was capable of inhibiting ISO-induced oxidative stress through activation of various strategies, including elevation of antioxidant enzymes and suppression of biochemical markers. Thus, it is evidenced to be a potential candidate in the treatment of cardiac dysfunction that may otherwise lead to heart failure.
Footnotes
Abbreviations
AR: Arctiin; LP: Lactobacillus plantarum; ISO: Isoproterenol; CK-MB: Creatine phosphokinase; AST: Aspartate transaminase; ALT: Alanine transaminase; LDH: Lactate dehydrogenase; SOD: Superoxide dismutase; CAT: Catalase; GST: Glutathione-S-transferase; GPx: Glutathione peroxidase; GSH: Reduced glutathione; GSSG: Oxidized glutathione; MDA: Malondialdehyde; qPCR: Quantitative polymerase chain reaction; TNF-α: Tumor necrosis factor-α; IL-6: Interleukin-6; ROS: Reactive oxygen species; CNS: Central nervous system; CFU: Colony forming unit; H2O2: Hydrogen peroxide.
Acknowledgments
All authors would like to acknowledge the institutional research facilities for the completion of this research work.
Data Availability
All data generated or analyzed during this study were included in this manuscript. Further enquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was supported by Researchers Supporting Project number (RSP2023R230) King Saud University, Riyadh, Saudi Arabia.
Statement of Ethics and Patient Consent
Not applicable (No human/patients were involved in the research).
