Abstract
Background
Schisandra chinensis (S. chinensis) is a drug commonly used in the clinical treatment of cardiovascular diseases in Traditional Chinese Medicine. However, the specific components and mechanisms of its action are still unclear. We screened six kinds of lignans from S. chinensis with high content and found that schisandrol A and schisantherin A had a strong vasorelaxant effect. The purpose of this study was to investigate the relaxation and underlying mechanism of schisandrol A in the isolated thoracic aorta of rats.
Materials and Methods
Isolated rat endothelium-intact and endothelium-removed thoracic aorta strips were pre-constricted with phenylephrine (PE), and the relaxation of schisandrol A on the strips was observed. Then, the mechanism was explored by pre-incubating the strips with nitric oxide synthetase inhibitor Nɷ-nitro-l-arginine methyl ester (L-NAME), cyclooxygenase inhibitor (indomethacin), potassium channel blockers 4-aminopyridine (4-AP), barium chloride (BaCl2), tetraethylamine (TEA), and glibenclamide, respectively, and changing the calcium concentration in the bath. In addition, expressions of endothelial nitric oxide synthetase (eNOS) mRNA and protein in rat thoracic aorta were detected.
Results
Schisandrol A induced both endothelium-dependent and endothelium-independent relaxation of isolated thoracic aorta strips of rats, and the mechanism might be related to promoting the synthesis of NO, inhibiting Ca2+ release from the sarcoplasmic reticulum, and blocking the Ca2+ channels.
Conclusion
These discoveries may provide a theoretical basis for the traditional application of S. chinensis to treat cardiovascular disease.
Key Messages
Schisandrol A induced both endothelium-dependent and independent vasorelaxation of isolated rat thoracic aorta. The endothelium-dependent vasorelaxation might be related to activating eNOS to promote NO synthesis, while endothelium-independent vasorelaxation might be related to blocking calcium channel opening.
Introduction
Cardiovascular disease (CVD) has been considered one of the most common diseases threatening human health worldwide since the 21st century (Cheng et al., 2023). A variety of traditional Chinese medicines and their active ingredients have achieved good therapeutic effects in cardiovascular diseases, such as lowering blood pressure, modulating blood lipids, anti-atherosclerosis, anti-oxidation, relaxing blood vessels, and so on (Demirel, 2022a, 2022b; Jin et al., 2023; Liu et al., 2023).
In this regard, Schisandra chinensis (Turcz.) Baill. (S. chinensis), a genuine medicinal material of Changbai Mountain in China, was first recorded in Shennong’s Herbal Classic. S. chinensis is often used to relieve hypertension, coronary heart disease, and heart failure in clinics (Ko et al., 1995; Li et al., 1996; Zhaoxu et al., 2016). In Korea, S. chinensis is used to treat cardiovascular symptoms in menopause syndrome in women (Chen & Li, 1993). However, there are few studies on the material basis of S. chinensis in the treatment of cardiovascular diseases, and its mechanism of action is unclear. Rhyu et al. (2006) reported that the aqueous extract of S. chinensis could relax the isolated thoracic aorta of rats. Park et al. (2009) found that the hexane extract dilated the rat thoracic aorta concentration-dependently. Our previous study demonstrated that S. chinensis total lignans could significantly relax phenylephrine (PE) pre-contracted isolated thoracic aorta of rats. The above findings suggest that the therapeutic effects of S. chinensis on cardiovascular diseases may be related to its vasodilation. In order to further explore the specific components of S. chinensis, we screened six S. chinensis lignans with higher contents, of which schisandrol A and schisantherin A were found to have a strong relaxing effect on isolated rat vascular strips.
Schisandrol A, a biphenyl cyclooctene lignan with a molecular weight of 432.51, is a lignan monomer with a high content in S. chinensis (Figure 1). Schisandrol A is taken as an index component for evaluation of the quality of S. chinensis in Pharmacopoeia of the People’s Republic of China, and its content is stipulated not less than 0.4% in S. chinensis (Chinese Pharmacopoeia Commission, 2015). To further study the vasorelaxation effect of schisandrol A and its mechanism, a vascular tension detection experiment in vitro was employed, and a variety of tool agents such as nitric oxide synthase inhibitor, cyclooxygenase inhibitor, and potassium channel blockers were used to speculate its mechanism. This study was expected to give pharmacological evidence for the research and development of S. chinensis.
(A) Schisandra chinensis (Turcz.) Bail. (B) The Chemical Structure of Schisandrol A.
Materials and Methods
Chemicals and Reagents
Schisandrol A (No. wkq18022702) was purchased from Chengdu Pufeide Biotech Co., Ltd. (Sichuan, China). Phenylephrine, L-NAME, indomethacin, glibenclamide, 4-aminopyridine (4-AP), and barium chloride (BaCl2) were obtained from Sigma Aldrich (St. Louis, MO, USA). ACh was supplied by Sinopharm Chemical Reagent Co., Ltd., (Beijing, China). A polyclonal antibody of endothelial nitric oxide synthetase (eNOS) was bought from ABclonal Biotechnology (Wuhan, China). Schisandrol A was dissolved with DMSO, and the final concentration of DMSO was controlled within 0.1% (v/v).
Animal Handling
Male SD rats (250–300 g) were purchased from Changchun Yisi Experimental Animal Technology Co., Ltd. [License No.: SCXK (Ji) 2018-0007] and fed in a laboratory under conditions of 20 ± 2°C and 50 ± 10% relative humidity. The rats were permitted to eat and drink freely. Adapting feeding for 1 week, rats could be used for the experiment.
Preparation of Isolated Rat Thoracic Aorta Strips
The preparation process referred to our previous report (Yang et al., 2020). In brief, rats were anesthetized with 100 mg/kg urethane, and then the thoracic aorta was quickly cut and placed in a pre-cooled (4°C) modified Ringer-Locke solution (Demirel, 2022). After carefully removing the surrounding tissues, the vascular strips about 3 mm in width and 0.8 cm in length were prepared by helical cutting. The endothelial tissue of some strips was gently wiped off with a small cotton ball to prepare endothelium-denuded vascular strips for observing the endothelium-independent relaxing effect. Subsequently, each specimen was vertically installed in a 10-mL bath tube containing nutrient solution with pH 7.4, in which the temperature was constantly kept at 37.0°C, and 95% O2 and 5% CO2 were aerated. Resting tension should be adjusted to 1.5 g to obtain a maximal contraction. The specimen was equilibrated at least 30 min before the beginning of the formal experiment, and the fresh solution was replaced every 10 min.
After the stabilizing of specimen tension, 30 mM KCl solution was used for pre-stimulating the strip twice to activate them. Then, phenylephrine (10−6 M) was added into the bath to constrict the strip after its tension was stabilized again, and ACh (10−9–10−5 M) was applied after its tension reached the peak to detect the integrity of vascular endothelium. If the maximum relaxation rate induced by ACh was greater than 90%, the specimen was considered with an intact endothelium, and if less than 10%, without endothelium. Papaverine (10−4 M) was applied to produce the maximum diastolic response as a reference. Then, the relaxation induced by the experimental drug was calculated as a percentage relative to the relaxation induced by papaverine.
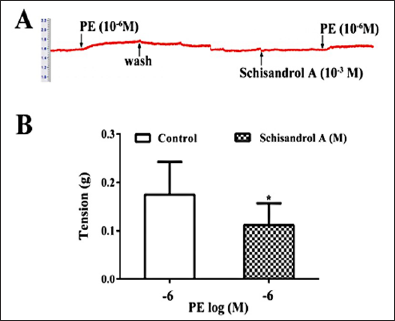
Effect of Schisandrol A on PE-pre-contracted Isolated Rat Thoracic Aorta
Schisandrol A at a cumulative concentration of 10−7–10−3 M was successively added to the bath tube after the aortic strip was pre-contracted by 10−6 M phenylephrine, and the tension changes were recorded for investigating vasodilation of schisandrol A on endothelium-intact and -denuded aortic strips. The percentage of vasodilation induced by schisandrol A was computed according to the following formula: (maximal tension by phenylephrine − minimal tension by schisandrol A)/(maximal tension by phenylephrine − minimal tension by PA) × 100% (Yang et al., 2020). Finally, the dose–response curve was drawn based on the relaxation rate of the aortic strips.
Blocking NO and PGI2 Production
The aortic strip with intact endothelium was pre-incubated with 10−4 M L-NAME or 10−5 M indomethacin for 10 min to investigate whether endothelium-derived NO and PGI2 pathways could be involved in the vasorelaxation induced by schisandrol A. Then, 10−6 M phenylephrine was applied to induce pre-contraction. After the tension of the strip was stable, schisandrol A at a cumulative concentration of 10−7–10−3 M was added into the bath tube to observe the vasorelaxation.
Blocking Four Types of Potassium Channels
It was unclear whether potassium channel activation was involved in the vasorelaxation induced by schisandrol A. Therefore, four potassium channel blockers, namely, 10−3 M BaCl2 for inward rectifier potassium channel (KIR), 10−3 M 4-AP for voltage-dependent potassium channel (KV), 10−3 M TEA for calcium-activated potassium channel (KCa2+), and 10−6 M glibenclamide for ATP-sensitive potassium channel (KATP), were pre-incubated with endothelium-denuded aortic strips for 20 min, respectively. Then, phenylephrine was used to pre-contract the strips. Schisandrol A at a cumulative concentration of 10−7–10−3 M or the vehicle was added to the bath tube after the strip tension was stable.
Intracellular Calcium Release Test
Intracellular calcium is involved in the contraction of smooth muscle, and phenylephrine activates the α-receptor, promotes the release of calcium ions from the sarcoplasmic reticulum (SR), and then induces muscle contraction. The present experiment was performed to investigate whether schisandrol A could cause the Ca2+ release from SR. Vascular strips without endothelium were incubated in Ca2+-free nutrient solution (containing 10−3 M EGTA) for 30 min, and then 10−6 M phenylephrine was added into the bath to induce the first transient constriction (T1). Subsequently, the strips were washed with Ca2+-containing nutrient solution three times to supplement the intracellular Ca2+ loss and with Ca2+-free nutrient solution twice in succession (Yang et al., 2020). Then, schisandrol A (10−3 M) was applied and incubated for 15 min, and the second transient vasoconstriction (T2) in strips was induced through 10−6 M phenylephrine. The value (T1–T2) was calculated as the tension difference.
Extracellular Calcium Influx Observation
Endothelium-removed vascular strips were incubated in Ca2+-free nutrient solution (containing 10−3 M EGTA). Notably, 10−6 M phenylephrine or 60 mM KCl was used to induce a basic constriction, and then 10−6–10−2 M CaCl2 was applied in turn to draw a concentration-response curve. The relationship between the vasorelaxation induced by schisandrol A and the vasocontraction induced by phenylephrine or KCl was observed. The specimen was pre-incubated with 10−3 M schisandrol A for 15 min before using phenylephrine or KCl. The vasocontraction induced by the highest concentration of CaCl2 (10−2 M) was taken as 100%, and the inhibitory rate of schisandrol A could be calculated based on it.
Total RNA Preparation and RT-PCR
Total RNA was prepared, and RT-PCR was performed as described in our previous study (Yang et al., 2020). In brief, the endothelium-intact aortic segment was pre-incubated with 10−3 M schisandrol A at 37°C for 15 min first, and then total RNA was extracted through the use of Trizol reagent, reverse transcribed, and amplified by following the instructions of HiScriptۚ II One-Step RT-PCR Kit. Specific primer sequences for rat eNOS were as follows: forward: 5′-CGCCTACCCAAGAAACACCT-3′ and reverse: 5′-GCTGACTCCCTCCCAGTCTA-3′, and the amplification conditions were denaturation at 94°C, annealing at 56°C, and extension at 72°C for 30 s, with 30 cycles. To confirm the PCR results, the agarose gel electrophoresis on 5 µL of PCR products was performed at 100 V for 40 min. The images of the results were observed and analyzed by the Tanon 1600 Gel Image system (Shanghai Tianneng Tech. Co. Ltd., China).
Western Blot Detection
The endothelium-intact aortic segment was pre-incubated with 10−3 M schisandrol A at 37°C for 15 min, and then total proteins were obtained by using Trizol reagent (Beytime Biotechnology, Shanghai, China). The protein concentration of the sample was measured by the BCA method, separated by SDS-polyacrylamide gel electrophoresis, and transferred onto the PVDF membrane, and then the membrane was blocked with 5% skim milk powder for 1.5 h. Subsequently, the membranes were incubated with the primary antibodies p-eNOS (1:1000 dilution) and GAPDH (1:20000 dilution), respectively, at 4°C overnight. After washing three times, the membranes were incubated with the secondary antibody routinely in a shaker at room temperature for 1 h. The images were acquired by chemiluminescence, visualized by autoradiography, and quantified densitometrically (Image J). Values for p-eNOS were normalized to GADPH.
Statistical Analysis
Statistical analysis was performed by use of SPSS software (Windows 22.0 version). All data were shown as mean ± SD (standard deviation). The statistical significance between the two groups was compared by Students’ t-test, and that of multiple comparisons was analyzed using a one-way ANOVA. A value with p < 0.05 was considered a significant difference in statistics.
Results
Relaxation of Schisandrol A on PE-pre-contracted Isolated Rat Thoracic Aorta Strips
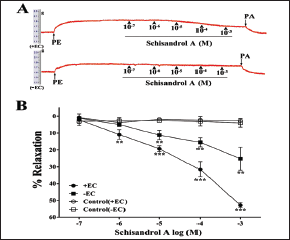
Schisandrol A (10−7–10−3 M) could induce a remarkable and concentration-dependent relaxation of both endothelium-intact and endothelium-removed aortic strips pre-contracted with phenylephrine. The maximum relaxation rates reached 52.79 ± 1.43% and 25.18 ± 6.83% for schisandrol A in comparison with 2.65 ± 1.53% and 3.47 ± 2.25% for the control (Figure 2), indicating that endothelium might be partly involved in the vasorelaxation induced by schisandrol A.
Effects of Blocking NO and PGI2 Production on Schisandrol A-induced Vasorelaxation
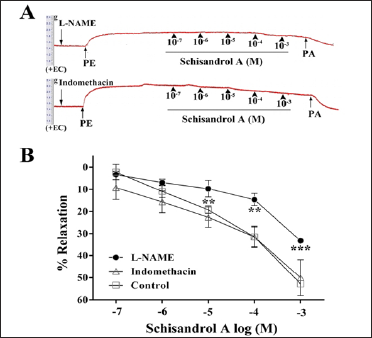
As shown in Figure 3, pre-incubation with L-NAME could obviously reduce the relaxation induced by schisandrol A with Emax of 33.24 ± 1.04%, compared with the control group with Emax of 52.79 ± 3.27% (p < 0.001). However, pre-incubation with indomethacin had no significant effect on vasorelaxation by schisandrol A. These suggested that the endothelium-dependent vasodilation of schisandrol A might be mainly related to the increase of NO synthesis, but not the PGI2 synthesis.
Effects of Four K+ Channel Blockers on Schisandrol A-induced Vasorelaxation
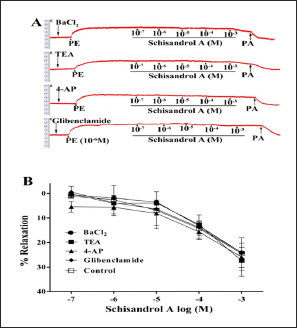
In order to clarify whether the K+ channel would be involved in the regulation of vasodilation induced by schisandrol A, four kinds of K+ channel blockers, BaCl2, TEA, 4-AP, and glibenclamide were incubated with endothelium-removed thoracic aorta strips, respectively. The results showed that none of the four K+ channel blockers produced an effect on schisandrol A-induced vasorelaxation (Figure 4), indicating that the potassium channel or currents might not be involved in this relaxation.
Effect of the Calcium Release from SR on Schisandrol A-induced Vasorelaxation
To elucidate if the vasorelaxation produced by schisandrol A would be related to the inhibition of Ca2+ release from SR of smooth muscle cells, endothelium-removed aortic strips were incubated with a Ca2+-free nutrient solution and contracted by phenylephrine, so as to promote Ca2+ release from SR into the cytoplasm. The results showed that pre-treatment with schisandrol A (10−3 M) remarkedly affected phenylephrine-induced vasoconstriction (Figure 5), indicating that the endothelium-independent vasodilation of schisandrol A might be related to the inhibition of Ca2+ release from SR.
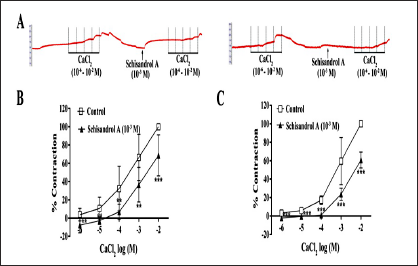
Effect of Extracellular Ca2+ Influx on the Schisandrol A-induced Vasorelaxation
To investigate whether extracellular Ca2+ influx was associated with schisandrol A-induced vasorelaxation, the activities of receptor-operated calcium channel (ROCC) and voltage-dependent calcium channel (VDCC) were observed. Phenylephrine can promote the influx of extracellular Ca2+ into the cytoplasm by activating ROOC, while a high concentration of KCl can promote Ca2+ influx by opening VDCC. Therefore, phenylephrine and KCl were employed to contract the endothelium-removed aortic strips in a Ca2+-free solution separately. It was found that schisandrol A not only weakened the contraction produced by CaCl2 + phenylephrine but also inhibited the contraction produced by CaCl2 + KCl (Figure 6), indicating that endothelium-independent vasodilation by schisandrol A might be partially related to double blockade of VDCC and ROCC.
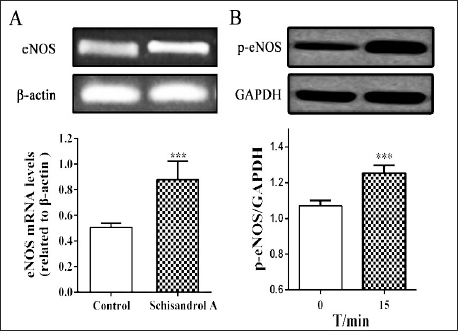
Effect of Schisandrol A on eNOS Expression
To explore the effect of schisandrol A on eNOS activity, the expressions of eNOS mRNA and protein in isolated thoracic aorta were further detected. As shown in Figure 7, schisandrol A treatment upregulated expression levels of eNOS mRNA and p-eNOS after incubation for 15 min, indicating that schisandrol A might further promote NO synthesis by regulating eNOS activity to induce its endothelium-dependent vasorelaxation.
Discussion
Schisandra chinensis has been applied in the treatment of a variety of diseases, and it is mainly distributed in Northeast China, Russia, Japan, and the Far East of South Korea (Panossian & Wikman, 2008; Rybnikář et al., 2019). The Treasure of Eastern Medicine, a Korean traditional medicine book, also introduced the cardiovascular effects of S. chinensis and clearly pointed out that S. chinensis could restore blood pressure and heart rhythm in patients with hypertension. Clinically, S. chinensis is often used in compound preparations, such as Shengmai Powder, to treat cardiovascular diseases in China (Jiang et al., 2020; Li et al., 1996). However, there are few reports on the material basis and mechanism of S. chinensis impacting cardiovascular function. In recent years, S. chinensis fruit extracts have attracted attention because of their potential roles in treating cardiovascular diseases such as myocardial infarction and hypertension (Alexander & Wang, 2012; Panossian & Wikman, 2008; Park et al., 2012). Furthermore, aqueous extract, hexane extract, gomisin A, and gomisin J from S. chinensis have shown relaxant effects on isolated thoracic aorta and mesenteric arteries (Park et al., 2007, 2009, 2012; Rhyu et al., 2006). With the progress of separation and analysis methods, lignans have been identified as a group of the main bioactive components in S. chinensis (Bártlová et al., 2002; Panossian & Wikman, 2008; Yang et al., 2022), containing a higher content of schisandrol A, schisandrol B, schisantherin A, schisandrin A, schisandrin B, schisandrin C, and so on (Chun et al., 2014). It was found in our experiments that total lignans and several monomers could induce vasorelaxation, of which schisandrol A (52.79 ± 1.43%) and schisantherin A (43.56 ± 2.17%) had stronger vasodilation, followed by schisandrol B (33.04 ± 2.21%), while schisandrin A, schisandrin B, and schisandrin C had no significant influence on the tension of isolated rat thoracic aorta (Yang et al., 2020). These results, along with the findings of other researchers, may provide evidence that lignans should be the main active substance for S. chinensis to exert its vasorelaxation. In this study, it was confirmed that schisandrol A induced both endothelium-dependent and -independent vasorelaxation in isolated rat thoracic aorta pre-contracted by PE. It was also found that the relaxation in the endothelium-intact aorta was significantly greater than that in the endothelium-denuded aorta.
Endothelial-dependent vasorelaxation is primarily mediated by endothelial-derived relaxing factors (EDRFs), such as nitric oxide (NO), PGI2, and endothelial-derived hyperpolarizing factor (EDHF), of which NO and PGI2 are the major vasodilators (Luksha et al., 2009).
Endothelial-independent vasorelaxation is primarily mediated by regulating various ion channels on the vascular smooth muscle cell membrane (VSCM), such as potassium and calcium channels. Four kinds of potassium channels, KIR, KV, KATP, and KCa2+, have been found now on VSCM (Dogan et al., 2019). The opening of these potassium channels can induce cell membrane hyperpolarization, reduce calcium influx, and lead to vasorelaxation. In this study, pre-incubating with four inhibitors of potassium channels, namely, BaCl2 (KIR inhibitor), TEA (KV inhibitor), 4-AP (KATP inhibitor), and glibenclamide (KCa2+ inhibitor), did not affect the vasodilation of schisandrol A on the endothelium-removed aortic strips, indicating that the endothelium-independent vasodilation of schisandrol A may not be related to the potassium channel opening. The calcium channel is the main ion channel to maintain vascular tension, mainly including ROCC and VDCC (Sandoo et al., 2010). Phenylephrine can activate ROCC and then regulate the inflow of extracellular calcium, resulting in vasocontraction (Chen et al., 2009). Our study found that the incubation of schisandrol A with phenylephrine could significantly reduce the vasoconstrictive effect of CaCl2, suggesting that ROCC may be involved in schisandrol A-induced vasorelaxation. Phenylephrine activates the specific receptor of the sarcoplasmic reticulum to give rise to Ca2+ release, which increases the concentration of intracellular calcium and then causes a transient contractile response (Senejoux et al., 2013). Pre-incubation with schisandrol A significantly inhibited the transient vasocontraction of phenylephrine on the strips, suggesting that schisandrol A-induced vasodilation may be related to its inhibiting the calcium release from the sarcoplasmic reticulum. High potassium promotes VDCC opening and depolarizes cell membranes (Hu et al., 2018). These results demonstrated that schisandrol A could significantly weaken the vasoconstriction caused by CaCl2 under the condition of high potassium, indicating that the vasodilation induced by schisandrol A may be related to its inhibiting VDCC opening.
Our previously published data showed that schisantherin A, another main lignan ingredient of S. chinensis, could cause an endothelium-dependent relaxation via NO and PGI2 pathways, as well as an endothelium-independent relaxation by the blockade of VDCC (Yang et al., 2020). The results regarding the mechanism are different from the vasodilation mechanism of schisandrol A. Further studies are needed to elucidate the structure–effect relationship regarding the different monomers of S. chinensis and the pathways involved in their activating eNOS or blocking calcium channels.
Summary
In sum, this study demonstrates that schisandrol A can induce both the endothelium-dependent and -independent vasorelaxation on isolated rat thoracic aorta for the first time, and the synthesis of NO and the activity of calcium channel may contribute to the mechanism. The results may provide experimental evidence for the research and development of S. chinensis. However, the specific mechanism of this effect is still unclear, and further research is necessary to evaluate the effect of schisandrol A on vasorelaxation.
Footnotes
Acknowledgments
We thank all our colleagues working at the Jilin Schisandra Development and Industrialization Engineering Research Center, Beihua University.
Abbreviations
S. chinensis: Schisandra chinensis; PE: Phenylephrine; L-NAME: Nω-nitro-L-arginine methyl ester; 4-AP: 4-aminopyridine; BaCl2: Barium chloride; TEA: Tetraethylamine; eNOS: Endothelial nitric oxide synthetase; ROCC: Receptor-operated calcium channel; VDCC: Voltage-dependent calcium channel; EDRFs: Endothelial-derived relaxing factors; NO: Nitric oxide; EDHF: Endothelial-derived hyperpolarizing factor; sGC: Soluble guanosine cyclase; COX: Cyclooxygenase; VSCM: Vascular smooth muscle cell membrane.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal experiments were approved by the Experimental Animal Ethics Committee of Beihua University (Jilin, China). All animal experiments were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Funding
This work received financial support from the Natural Science Foundation of Jilin Province (No. 20210101429JC) and the Beihua University Graduate Innovation Program Project (No. 2022029).
