Abstract
Objective
To investigate the role of hydrogen sulfide (H2S) in human internal mammary arteries (IMA) and its interaction with endothelial nitric oxide synthase (eNOS) and phosphodiesterase (PDE)5A activity.
Methods
Human IMA segments from patients undergoing coronary artery bypass grafting (CABG) were studied by myography for acetylcholine and sodium hydrosulfide (NaHS)-induced relaxation. Locations of 3-mercaptopyruvate sulfurtransferase (3-MPST) and cysteine aminotransferase (CAT) were examined immunohistochemically. Levels of H2S, eNOS, phosphorylated-eNOSser1177, and PDE5A were measured.
Results
In IMA segments from 47 patients, acetylcholine-induced relaxation (resistant to NG-nitro-L-arginine and indomethacin) was significantly attenuated by aminooxyacetic acid or L-aspartate (CAT inhibitors), iberiotoxin (large-conductance calcium-activated K+ channel blocker), TRAM-34 plus apamin (intermediate- and small-conductance Ca2+-activated K+ channel blockers) or glibenclamide (ATP-sensitive K+ channel blocker). 3-MPST and mitochondrial CAT were found in endothelial and smooth muscle cells while cytosolic CAT was located only in endothelial cells. Acetylcholine significantly increased the H2S levels. The H2S donor, NaHS, increased eNOS phosphorylation and down-regulated PDE5A.
Conclusions
Human conduit artery endothelium releases H2S under basal and stimulated conditions, involving the 3-MPST/CAT pathway, eNOS phosphorylation, PDE5A activity, and potassium channels. These findings may provide new therapeutic targets for treating vasospasm in CABG grafts and facilitate the development of new vasodilator drugs.
Introduction
Hydrogen sulfide (H2S) plays regulatory roles in the cardiovascular, nervous, and immune systems. 1 In the brain, H2S is mainly generated by cystathionine β-synthase, while in the cardiovascular system it is mainly produced by cystathionine ɣ-lyase.2,3 Vascular endothelium has been demonstrated to produce H2S through metabolism by 3-mercaptopyruvate sulfurtransferase (3-MPST) and cysteine aminotransferase (CAT), and in a mouse model, H2S was shown to be a smooth muscle relaxant released from the endothelium. 4 Further, 3-MPST produces H2S from 3-mercaptopyruvate, which is generated by CAT from L-cysteine and α-ketoglutarate in the presence of the cofactors thioredoxin and dihydrolipoic acid. 5
Cyclic guanosine monophosphate (cGMP) is synthesized by soluble guanylyl cyclases (sGCs) and degraded by phosphodiesterases (PDEs), and plays an important role in nitric oxide (NO)-stimulated vasorelaxation.6,7 H2S and NO are suggested to be mutually required to evoke angiogenesis and vasodilatation, and although H2S does not directly activate sGC, it maintains a tonic inhibitory effect on PDE5, thereby delaying the degradation of cGMP. 1 Exposure of endothelial cells to H2S has been shown to increase intracellular cGMP in a NO-dependent manner, and activate protein kinase G (PKG) and its downstream effector, vasodilator-stimulated phosphoprotein. 1 In addition, oligopeptide DT-2 (a PKG-I inhibitor) was found to reduce the sodium hydrosulfide (NaHS)-induced vasorelaxation in both endothelium-intact or denuded rat aortic rings, 8 suggesting that H2S may induce relaxation by stimulating PKG-I. 8
Hydrogen sulfide has been shown to relax rat aortic tissues via opening of ATP-sensitive K+ (KATP) channels in the sarcolemma,2,9 leading to a decrease in blood pressure in rats. 10 Moreover, activation of voltage-gated potassium channels may be another key mechanism in the vasorelaxation induced by H2S, 11 and co-application of Ca2+-activated K+ (KCa) channel blockers (apamin and charybdotoxin) has been reported to reduce H2S-induced relaxation of endothelium-intact aortic tissues. 12 Therefore, the present authors hypothesized that H2S may induce vasorelaxation by stimulating different potassium channels. In a recent study of human mesenteric arteries, H2S-induced relaxation was demonstrated to be endothelium-dependent and to involve mechanisms related to the stimulation of KATP channels. 13
To date, published work regarding the release of H2S from endothelium in the human conduit arteries, and the related mechanism of H2S-induced relaxation, remains scarce. The present study examined the role of H2S in human conduit arteries, using internal mammary arteries (IMA), and the possible mechanisms related to a cGMP-dependent pathway and potassium channels.
Patients and methods
Study population and vessel preparation
Fresh human IMA segments were obtained from patients undergoing coronary artery bypass graft (CABG) surgery at TEDA International Cardiovascular Hospital, Chinese Academy of Medical Sciences, Tianjin, China between January 2016 and June 2017. Inclusion criteria comprised elective CABG surgery and patients receiving standard preoperative drug therapy including antiplatelet, antiangina, or antihypertensive drugs, as appropriate. Human IMA tissue was obtained whenever appropriate patients became available; thus, no specific study-group allocation procedure was performed and IMA segments were randomly allocated into the different groups. Patients who underwent emergency surgery, or who underwent simultaneous CABG plus other surgery, such as valve replacement, were excluded.
Approval to use the discarded vessels was obtained from the Ethics Committee of TEDA International Cardiovascular Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College Sciences, and the patients provided written informed consent for their tissue to be included.
Following collection, the vessels were immediately immersed in 4 °C Krebs solution and transferred to the laboratory within 5–10 min. The vessels were placed in a glass dish filled with Krebs solution and carefully cleaned of fat and connective tissue. In some IMA ring segments, the endothelium was denuded by gently rubbing the intimal surface with a thin polyethylene tube. In the remaining segments, great care was taken not to touch the inner surface of the blood vessels. This technique allowed the experiments to be performed with functionally intact endothelium, as shown previously, 14 compared with denuded IMA.
Vessels were cut into 3 mm-long rings and mounted in an organ-bath containing Krebs solution (composition as previously described),9,15 maintained at 37 °C and bubbled continuously with 95% oxygen/5% CO2. In addition, protein was extracted from some vessels incubated for ≥ 60 min, to perform Western blotting and enzyme-linked immunosorbent assay (ELISA), and other vessels were used to measure H2S concentration. Vessels harvested from three patients were immediately immersion-fixed in 4% formaldehyde for immunohistochemistry.
H2S-induced relaxation
Myography
Human IMA ring segments were suspended on wire hooks in a 6 ml bath within a myograph system modified for large vessel studies (model 610M; DMT, Aarhus, Denmark). Each ring segment resting unstretched on the wire hooks was equilibrated in Krebs solution for ≥1 h, as previously described.16,17 For inhibitor experiments, the 3-mm IMA ring segments from the same patient were allocated to different groups, including a control, so that each vascular ring had its own control from the same patient.
Normalization
The IMA ring segments were normalized as previously described.16,17 Briefly, each ring segment was stretched in progressive steps every minute to determine the individual length-tension curve. A computer iterative fitting program, Myodaq and Myodata, version 2.01 (Maastricht University, Maastricht, the Netherlands) was used to determine the exponential curve pressure and internal diameter by recording the internal diameter (µm) and corresponding wall tension (mN/mm2) at the end of each step. Based on length-tension curves, stretching was stopped when the transmural pressure of the rings reached 100 mmHg. The rings were then released and set at 90% of the internal diameter at 100 mmHg. 18 This degree of passive tension was then maintained throughout the experiment.
Precontraction and relaxation
Following equilibration for 45 min in the organ chamber, IMA ring segments were precontracted using a previously determined concentration (–8 log M)15,19 of U-46619 (Cayman Chemical, Ann Arbor, MI, USA). When the U-46619-induced contraction reached a stable plateau, cumulative concentration–relaxation curves were established for acetylcholine (–10 to –4.5 log M; Sigma-Aldrich, St Louis, MO, USA) and the H2S donor, NaHS (–11 to –2.5 log M; Sigma-Aldrich) (n = 8 for each treatment). IMA relaxation was expressed as the percent reversal of U-46619-induced precontraction. One concentration-relaxation curve was obtained for each IMA ring.
All drugs used in the present study were prepared in distilled water, except for indomethacin, glibenclamide and TRAM-34, which were dissolved in ethanol.
Procedures and measurements
Effect of CAT inhibition on endogenous H2S-induced vasorelaxation
To investigate the effect of CAT inhibitors on the acetylcholine response in human IMA, ring segments were divided into three groups and incubated with Krebs solution containing the NO synthase inhibitor, NG-nitro-L-arginine (L-NNA, 100 µmol/L; Sigma-Aldrich) plus the cyclooxygenase inhibitor, indomethacin (10 µmol/L) (control); L-NNA plus indomethacin plus the CAT inhibitor, aminooxyacetic acid (AOAA, 100µmol/L; Sigma-Aldrich); or L-NNA plus indomethacin plus a competitive CAT substrate, L-aspartate (1000 µmol/L; Sigma-Aldrich). After equilibration for 45 min in the organ chamber, ring segments were pre-contracted with –8 log M of U-46619. Acetylcholine (−10 to −4.5 log M)-induced relaxation curves were then established.
Acetylcholine-induced release of H2S in IMA
Levels of H2S in human IMA tissue were quantified using a methylene blue assay method, as previously described.20,21 Human IMA was pre-incubated for 4 h (as determined by pilot experiments) in Krebs solution (control), or in Krebs containing acetylcholine (1000 µmol/L). Tissues were then homogenized in ice-cold phosphate buffered saline (PBS; pH 8.0), and the homogenate was centrifugated at 5 200 × g for 10 min at 4°C. Next, 75 µl of the supernatant fluid was transferred to a test tube containing 450 µl of dH2O and mixed with 250 µl of 1% acetic acid zinc (Amresco [VWR Life Science], Radnor, PA, USA) at 37 °C for 10 min, before adding 250 µl of 10% trichloroacetic acid (Amresco). After centrifuging the mixture at 5 200 × g at 4°C for 10 min, 750 µl of the supernatant was incubated with 221 µl of 20 mmol/l N, N-dimethyl-p-phenylenediamine sulfate (Amresco) in 7.2 mol/l HCl (Sigma), immediately followed by the addition of 221 µl of 30 mmol/l FeCl3 (Amresco) in 1.2 mol/l HCl, to generate methylene blue. The absorbance of the resulting solution at 670 nm was measured after 20 min using a microplate reader. The H2S concentration was calculated against a linear standard curve of H2S (measured using NaHS standard solutions of 0, 18.75, 37.5, 75, 150 and 300 µmol/l), and the results were expressed as µmol of H2S per gram of vascular tissue (µmol/g tissue).20,21
Immunohistochemistry
Formaldehyde-fixed tissues were paraffin embedded and cut into 4-µm transverse sections onto Adhesive slides (Cat. No. SLI-20010501, MXB Biotechnologies, FuZhou, China) and then placed in an oven maintained at 60 °C for 1 h. The sections were microwaved for antigen retrieval then incubated with primary antibodies against cytosolic CAT (cCAT; rabbit-anti-aspartate aminotransferase antibody [ab170950], Abcam Cambridge, MA, USA; 1:50 dilution), mitochondrial CAT (mCAT; mouse anti-MCAT [sc-100477], Santa Cruz Biotechnology Dallas, TX, USA; 1:50 dilution) and 3-MPST (rabbit-anti-MPST antibody [HPA001240-100UL], Sigma-Aldrich; 1:250 dilution) overnight at 4 °C. The sections were rinsed three times in PBS followed by incubation with horseradish peroxidase-conjugated anti-mouse or anti-rabbit secondary antibody at 37 °C for 30 min. The immunoreactive signal was developed using a 3,3′-diaminobenzidine colour developing substrate for 5 min, then the sections were mounted and visualized under a light microscope (BX41, Olympus Corporation, Japan), as previously described. 22 Image processing and analyses were performed using CellSens Standard software (Olympus Corporation, Japan).
Effect of blocking KCa or KATP channels on acetylcholine-induced relaxation in the presence of L-NNA and indomethacin
To investigate the effect of potassium channel inhibitors in H2S-mediated relaxation, IMA ring segments were divided into four groups and incubated with Krebs solution containing L-NNA plus indomethacin (control); L-NNA plus indomethacin plus the KATP channel blocker glibenclamide (10 µmol/L); L-NNA plus indomethacin plus the large-conductance calcium-activated K+ [BKCa] channel blocker iberiotoxin (10−2 µmol/L; Sigma-Aldrich); or L-NNA plus indomethacin plus the intermediate-conductance Ca2+-activated K+ [IKCa] channel blocker TRAM-34 (1 µmol/L; Sigma-Aldrich) plus the small-conductance Ca2+-activated K+ [SKCa] channel blocker apamin (10−1 µmol/L; Sigma-Aldrich). Acetylcholine-induced relaxation curves were then established as mentioned above.
NaHS (exogenous H2S donor)-induced relaxation in IMA
To demonstrate the relaxation induced by exogenous H2S, the H2S donor NaHS was applied in cumulative concentration (–9 to –2.5 log M) to endothelium-denuded and endothelium-intact IMA ring segments with contraction induced by U-46619.
Effect of H2S on levels of endothelial NO synthase (eNOS), phosphorylated (p)-eNOS and PDE5A
Fresh IMA rings were incubated with Krebs solution (control), or Krebs containing 100 µmol/L NaHS, for 24 h at 37 °C. IMA segments from 15 patients were divided into 5 groups: (1) NaHS-treated (to measure total eNOS); (2) NaHS-treated (to measure p-eNOS); (3) without NaHS treatment (control for groups 1 and 2); (4) NaHS-treated (to measure PDE5A); and (5) without NaHS treatment (control for group 4). Each group was homogenized in lysis solution (KeyGEN Biotech, Nanjing, China). Supernatants were collected after centrifugation at 12 800 × g for 10 min at 4°C, and were used for measuring eNOS and p-eNOS by Western blotting, with GAPDH used as the internal control, as previously described 1 or for measuring PDE5A levels by ELISA, using a PDE Assay Kit (Cat No. 60300; Amsbio, Lake Forest, CA, USA) according to the manufacturer’s instructions.
Statistical analyses
All results are presented as mean ± SEM. Student’s t-test was used for comparisons between two sets of data and analysis of variance (ANOVA) was used for group comparisons, followed by post-hoc Bonferroni test to detect the individual differences. Statistical analyses were performed using SPSS software, version 20.0 (IBM, Armonk, NY, USA) and a P value <0.05 was considered statistically significant.
Results
Fresh IMA tissue was obtained from 47 patients who underwent CABG surgery, comprising 30 male and 17 female patients, with a mean age of 62.4 ± 10.7 years. All patients had received standard preoperative drug therapy including antiplatelet, antiangina, or antihypertensive drugs, as appropriate. It should be noted that CABG patients commonly have hypertension and diabetes, but specific clinical data for the patients who donated tissue for inclusion in the present study were not recorded.
Resting vessel parameters
The IMA ring segments were suspended at an equivalent transmural pressure of 70.4 ± 2.3 mmHg at 90% of the internal diameter at 100 mmHg after normalization. 19
Effect of CAT inhibition on endogenous H2S-induced relaxation
Acetylcholine induced almost full relaxation (92.37% ± 0.77%) in the endothelium-intact (E+) IMA rings whereas the relaxation was abolished in the endothelium-denuded (E-) rings (Figure 1A and B). In the endothelium-intact groups treated with CAT inhibitors, acetylcholine-induced relaxation was partially inhibited by L-NNA plus indomethacin (P <0.01, two-way ANOVA) and the inhibition (circa 30%) was similar to previously reported values. 19 Acetylcholine-mediated relaxation was also significantly inhibited in the presence of L-NNA plus indomethacin plus AOAA (P <0.01, two-way ANOVA) and L-NNA plus indomethacin plus L-aspartate (P <0.01, two-way ANOVA) compared with controls (L-NNA plus indomethacin; Figure 1A and B). Acetylcholine-induced Emax presented no significant differences between rings treated with L-NNA plus indomethacin plus AOAA (23.81% ± 0.84%) and rings treated with L-NNA plus indomethacin plus L-aspartate (24.29 ± 0.54%, P > 0.05, unpaired t-test). EC50 values in the L-NNA plus indomethacin plus AOAA group (–7.12 ± 0.01 log M) were higher than in the control group (–7.29 ± 0.04 log M; P <0.01, one-way ANOVA and Bonferroni test), but there were no statistically significant differences between L-NNA plus indomethacin plus L-aspartate treatment (–7.29 ± 0.03 log M) and the control group (L-NNA plus indomethacin; P >0.05, one-way ANOVA and Bonferroni test; Figure 1C).

(A) Effect of cysteine aminotransferase (CAT) inhibitors on endogenous H2S-induced relaxation. Acetylcholine (ACh) induced almost full relaxation in human endothelium-intact (E+) internal mammary artery (IMA) rings, and this relaxation was inhibited by NG-nitro-L-arginine (L-NNA) plus indomethacin (Indo) (**P <0.01, two-way ANOVA); ACh-induced relaxation in E+ rings was further inhibited by L-NNA plus Indo plus aminooxyacetic acid (AOAA; a general inhibitor of pyridoxal 5′-phosphate-dependent enzymes), and L-NNA plus Indo plus L-aspartate (a competitive CAT substrate). In contrast, there was no ACh-induced relaxation in endothelium-denuded (E-) IMA rings (n = 8; **P <0.01 versus E+; two-way ANOVA); (B) Original trace of Figure 1A; (C) –Log EC50 for each group; L-NNA plus Indo plus AOAA was significantly lower than L-NNA plus Indo (**P <0.01; n = 8; one-way ANOVA and Bonferroni test). Data presented as mean ± SEM.
H2S production in IMA
A linear standard curve of H2S was established to quantify H2S in the IMA tissues (R2 = 0.995; Figure 2A). The fresh human IMA tissue generated detectable amounts of H2S (Figure 2B) and the detected H2S was increased by acetylcholine (P <0.01 versus control, unpaired t-test), demonstrating that human IMA is capable of generating H2S when stimulated with acetylcholine.

(A) Standard curve of NaHS gradient solutions that was produced to quantify the concentration of H2S in fresh human internal mammary artery (IMA) tissue; (B) The concentrations of H2S in human IMA were increased by incubating with acetylcholine (ACh) for 4 h (**P <0.01 versus control; n = 5; unpaired t-test). Data presented as mean ± SEM.
Immunohistochemistry
Immunohistochemical staining showed that 3-MPST was located in both the endothelium and the smooth muscle (Figure 3 left), cCAT was located only in the endothelial cells (Figure 3 middle), and mCAT was located in both the endothelium and the smooth muscle (Figure 3 right).

Representative photomicrographs showing immunohistochemistry staining of 3-mercaptopyruvate sulfurtransferase (3-MPST) and cysteine aminotransferase (CAT) in human internal mammary artery tissue; 3-MPST was shown to be located in both the endothelium and smooth muscle (
Effect of KCa or KATP channel blockers on acetylcholine-induced relaxation in the presence of L-NNA plus indomethacin
The addition of L-NNA plus indomethacin inhibited acetylcholine-mediated relaxation. The presence of L-NNA plus indomethacin plus iberiotoxin, or L-NNA plus indomethacin plus glibenclamide, further inhibited the acetylcholine-induced relaxation versus L-NNA plus indomethacin (both P <0.01, two-way ANOVA). The greatest inhibition of acetylcholine-mediated vasorelaxation was observed in vessels treated with L-NNA plus indomethacin plus TRAM-34 plus apamin, and this inhibition was significantly higher versus vessels treated with L-NNA plus indomethacin plus iberiotoxin (P <0.01, two-way ANOVA). Emax inhibition of vessels treated with L-NNA plus indomethacin plus TRAM-34 plus apamin (12.42% ± 0.29%) was also significantly higher (P <0.01, unpaired t-test) than in vessels treated with L-NNA plus indomethacin plus iberiotoxin (23.12% ± 0.52%; Figure 4A). The EC50 values in the group treated with L-NNA plus indomethacin plus iberiotoxin (–7.56 ± 0.07 log M) were significantly lower than in the control group treated with L-NNA plus indomethacin (–7.29 ± 0.04 log M), and EC50 values in the groups treated with L-NNA plus indomethacin plus TRAM-34 plus apamin (–6.49 ± 0.02 log M) and L-NNA plus indomethacin plus glibenclamide (–6.09 ± 0.09 log M) were significantly higher versus the control group (all P <0.01; one-way ANOVA and Bonferroni test; Figure 4B).
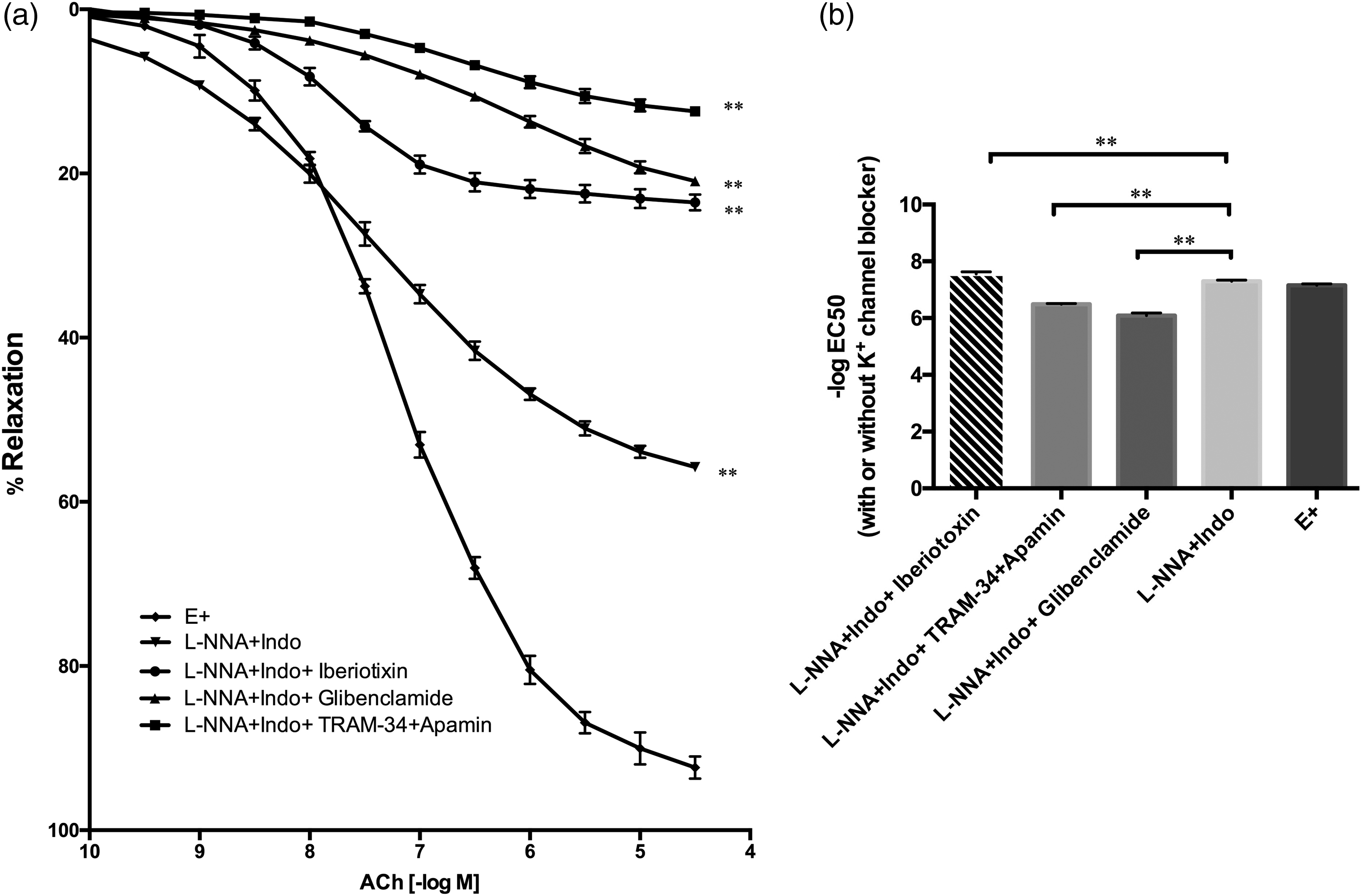
Effect of different K+ channel blockers on acetylcholine (ACh)-induced relaxation in human internal mammary artery rings. (A) ACh-induced relaxation was significantly reduced in the presence of NG-nitro-L-arginine (L-NNA) plus indomethacin (Indo). The K+ channel blockers iberiotoxin, glibenclamide and TRAM-34 plus apamin further inhibited the residual ACh-induced relaxation that was resistant to L-NNA plus Indo (**P <0.01 versus L-NNA plus Indo [control]; two-way ANOVA); (B) Regarding –logEC50 values, each experimental group had significantly higher EC50 values versus the control group (L-NNA plus Indo) except L-NNA plus Indo plus iberiotoxin, in which the EC50 was lower (**P <0.01 versus control; n = 8; one-way ANOVA and Bonferroni test). Data presented as mean ± SEM.
Effect of endothelium in exogenous H2S-induced relaxation
There were no statistically significant differences in NaHS-mediated relaxation between endothelium-denuded and endothelium-intact IMA (Figure 5), suggesting that exogenous H2S-induced relaxation in the present experiments was endothelium-independent.

The NaHS-mediated relaxation in human internal mammary artery (IMA) rings was not affected by denudation of the endothelium (P >0.05, endothelium-intact IMA versus endothelium-denuded IMA; n = 8; two-way ANOVA). Data presented as mean ± SEM.
Effect of H2S on eNOS, p-eNOS and PDE5A levels
Western blots showed that there was no difference in levels of eNOS protein between IMA rings exposed to NaHS (H2S donor; –4 log M) for 24 h at 37°C, and IMA rings incubated in Krebs only (controls; Figure 6A), however, NaHS was associated with significantly increased levels of eNOS phosphorylation at its activation site, ser1177 (increased p-eNOSser1177 levels) versus controls (P <0.01, unpaired t-test; Figure 6A).

Levels of endothelial nitric oxide synthase (eNOS), phosphorylated (p)-eNOS, and phosphodiesterase (PDE)5A in human internal mammary artery (IMA) rings. (A) Top panel shows representative Western blots of IMA protein extracts from IMA rings that had been incubated with Krebs alone (control; two left lanes) or Krebs with −4 log M NaHS (two right lanes) for 24 h. Results normalised to GAPDH (bottom figure) showed that exogenous H2S did not affect eNOS levels but increased p-eNOSser1177 levels in human IMA (**P <0.01 versus control; n = 5; unpaired t-test). (B) Results from enzyme-linked immunosorbent assay showing that stimulation of IMA with NaHS (−4 log M) significantly down-regulated PDE5A levels compared with the control (**P <0.01 versus control; n = 5; unpaired t-test). Data presented as mean ± SEM.
The ELISA experiments showed that stimulation of IMA with NaHS (−4 log M) for 24 h at 37°C significantly down-regulated the PDE5A levels compared with control IMA tissue exposed to Krebs only (P <0.01, unpaired t-test; Figure 6B).
Discussion
The present study revealed that in human IMA conduit arteries (1) the endothelium releases H2S under both basal and acetylcholine-stimulated conditions; (2) the release of H2S involves 3-MPST coupled with CAT pathway; and (3) the H2S-induced relaxation involves stimulating the phosphorylation of eNOS and inhibiting PDE5A activity, as well as opening of potassium channels.
In mammals, H2S is enzymatically produced through pyridoxal 5′-phosphate (PLP)-dependent pathways. 20 CAT is a PLP-dependent enzyme and endogenous H2S is generated by 3-MPST coupled with CAT in rat thoracic aortic endothelial cells. 4 CAT is found in both cytosolic and mitochondrial forms in the rat liver. 23 H2S is reported to be a potent vasodilator of human pulmonary arteries 24 and mesenteric arteries. 13 Further, it has been previously suggested that cystathionine ɣ-lyase, and the H2S it generates, together play a physiological role in regulating the diameter of arteries in human IMA. 25
The present study demonstrated release of H2S from the human conduit artery under both basal and acetylcholine-stimulated conditions, by acetylcholine-induced relaxation and by direct measurement of H2S levels in IMA tissue. In addition, to the best of the authors’ knowledge, this is the first time that L-NNA and indomethacin-resistant relaxation induced by acetylcholine is shown to be further inhibited by both 3-MPST/CAT inhibitor AOAA (a general inhibitor of PLP-dependent enzymes) 20 and the competitive CAT substrate L-aspartate, 20 suggesting that the release of H2S from the human conduit artery involves the 3-MPST/CAT pathway. Direct immunohistochemistry evidence revealed that 3-MPST and mCAT are located in both endothelial and smooth muscle cells and cCAT is located in endothelial cells only. Although the H2S producing enzymes, MPST and CAT, were detected in both endothelium and smooth muscle cells, the acetylcholine-induced relaxation was abolished when the endothelium was removed. These results may be due to a number of factors: First, the present study showed that cCAT is located in the endothelial cells only and mCAT is located in both the endothelium and smooth muscle. The role of endothelium for endothelium-dependent relaxation is mainly due to endothelium-derived relaxing factors, such as NO, endothelium-dependent hyperpolarizing factor (EDHF), and H2S. Therefore, when the endothelium is removed, endothelium-dependent relaxation is actually abolished. This is true for NO, as has been repeatedly reported previously, although eNOS is present in the smooth muscle. Similarly, when the endothelium is removed, the H2S-mediated relaxation is also abolished. These observations suggest that endothelium-dependent relaxation related to endothelium-derived NO or endothelium-derived H2S is a dominant mechanism in acetylcholine-mediated relaxation. Secondly, the above facts may suggest that cCAT is the major enzyme, since it is located only in the endothelium. It appears that any mCAT-derived H2S, if any, would not be enough to induce significant smooth muscle CAT-mediated relaxation. Taken together, the authors believe that in the present study, L-NNA and indomethacin-resistant relaxation induced by acetylcholine is mediated by endothelium-derived H2S.
In the present study, the role of cGMP was explored in H2S-induced, endothelium-dependent relaxation in human IMA. p-eNOS was found to be increased and PDE5A was decreased in response to NaHS. There are few possibilities to explain this: (1) NaHS (H2S donor) may increase p-eNOS at its activation site, Ser1177 in endothelial cells, leading to NO production and induced cGMP expression, subsequently inhibiting PDE5A; (2) NaHS (H2S) directly inhibits PDE5A activity; (3) there may be a combination of the above two mechanisms; and (4) other possibilities may also exist, for example, NaHS (H2S) may stimulate other molecules in this pathway. Without measuring all the intermediate molecules (such as cGMP), it is difficult to determine which is the real mechanism. Regardless, the present experiments demonstrated that the H2S donor, NaHS, decreased purified PDE5A levels. As reported previously,1,26 PDE5A is involved with cGMP degradation and therefore inhibition of PDE5A enhances cGMP pathway-mediated relaxation.
To investigate the role of potassium channels in H2S-induced relaxation, the effect of various KCa and KATP channel inhibitors were tested. First, the role of IKCa and SKCa channels was explored in the endothelial cells. The inhibitory role of apamin (inhibitor of SKCa) plus TRAM-34 (inhibitor of IKCa) was demonstrated in the residual component of acetylcholine-induced vasorelaxation that was resistant to L-NNA plus indomethacin. Further, to assess whether acetylcholine-induced relaxation involves activation of the BKCa and KATP channels in the vascular smooth muscle, the effects of the BKCa channel blocker iberiotoxin and KATP channel blocker glibenclamide on the acetylcholine-induced responses were tested. Relaxation was found to be significantly blocked, suggesting a role for BKCa and KATP in acetylcholine-induced relaxation in the smooth muscle of human IMA. In fact, H2S opens BKCa, and induces [K+]i efflux 27 that inhibits the capacitative [Ca2+]i influx through inhibition of L-type voltage-gated Ca2+ channels. 28 In the present study, inhibition of the BKCa channel by iberiotoxin reduced the H2S-mediated relaxation, suggesting that H2S-induced relaxation is related to opening of the BKCa channel. The present study therefore provides evidence that H2S released from the endothelium may induce vasorelaxation via opening of vascular smooth muscle KCa channels in human IMA.
In previous studies, the present authors have demonstrated that acetylcholine-induced, L-NNA and indomethacin-resistant relaxation in human IMA is mediated by EDHF 29 and KCa channels are involved in the EDHF-mediated relaxation29,30 and hyperpolarization.31,32 Further, the authors hypothesized that H2S is a candidate of EDHF in rat cerebral vessels.31,32 The present study supports this notion in human arteries and the findings are consistent with observations in small mesenteric arteries, or aortae in male animal models.33,34
The possible role of KATP channels in H2S-mediated relaxation was also assessed. KATP channel opening is known to mediate many of the biological activities of H2S. 35 Sarcolemmal KATP channels are involved in intracellular [Ca2+]i handling and their opening results in resistance to apoptosis during myocardial ischaemia/reperfusion injury. 36 Interestingly, administration of glibenclamide in vivo reduced the infarct-limiting effects of NaHS, 12 indicating that sarcolemmal KATP channel activation participated in the cardioprotective signalling of H2S. 12 Further, potent vasodilatory activity by H2S both in vitro and in vivo is assumed to be realized by opening of vascular smooth muscle KATP channels. 37 The present study also demonstrated that KATP channel is involved in H2S-induced relaxation in human IMA.
In conclusion, the results of the present study demonstrated that in the human IMA conduit artery, the endothelium releases H2S under both basal and acetylcholine-stimulated conditions and the release of H2S involves 3-MPST coupled with CAT pathway. Further, H2S-induced relaxation involves stimulating the phosphorylation of eNOS and inhibiting PDE5A activity. In addition, potassium channels are involved in H2S-induced vasorelaxation in this human artery, involving two-fold actions: opening of the endothelial IKCa and SKCa channels and opening of the smooth muscle BKCa channels; both producing hyperpolarization and relaxation. These findings may provide new therapeutic targets for the treatment of vasospasm in coronary artery bypass graft vessels, and may provide a pharmacological basis for the development of new vasodilator drugs.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was fully supported by grants from the National Natural Science Foundation of China (81870288, 81870227), the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2018TX31002), Binhai New Area Health Bureau (2016BWKY007, 2016BWKZ003), and China-Serbia Exchange Program (2-13, 4-12).
