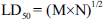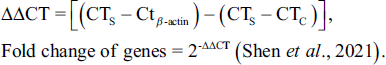Abstract
Objectives
This study investigated the effects of BP and doxorubicin (DOX) on 4T1 tumor cells in a mouse model.
Materials and Methods
After inducing breast tumors, 70 4T1-tumor-bearing BALB/c mice were divided into seven groups (n = 10/group). The groups were treated with DOX and BP for 35 days. On the 36th day, blood was taken from the heart, and serum was separated to measure levels of cytokines, estrogen, progesterone, testosterone, and nitric oxide. Antioxidant enzyme activities, as well as tissue ferric-reducing antioxidant power (FRAP) and malondialdehyde (MDA) levels, were evaluated. The expression of apoptotic genes and metastasis was measured using real-time polymerase chain reaction (PCR), while the expression of apoptotic proteins was evaluated using Western blotting. Finally, Ki-67 and p-53 were examined using immunohistochemistry to determine apoptosis.
Results
The study found that BP, with its synergistic effects with DOX, reduced the volume of tumors and increased the expression of apoptotic genes and proteins. In a dose-dependent manner, groups receiving BP and DOX with their synergistic effects reduced the level of estrogen and nitric oxide and also reduced the level of pro-inflammatory cytokines. BP along with DOX increased serum interferon-γ (IFN-γ) levels. Tumor tissue FRAP and thiobarbituric acid reactive substances (TBARS) levels increased in BP-treated groups. Ki-67 and Bcl-2 proliferation markers levels decreased, and p53 levels increased in 4T1-breast tumors.
Conclusion
The study concluded that BP with its synergistic effects along with DOX has the ability to suppress the growth of tumors and can also inhibit the oxidative damage of DOX.
Introduction
Breast cancer is the most common malignancy in women worldwide, with an annual registration of 1 million new cases and a prevalence of 18%. On average, one out of every eight women will be affected by breast cancer in their lifetime, with the highest incidence observed in the Asia-Pacific region (Sancho-Garnier & Colonna, 2019). The incidence of breast cancer is higher in women over 50 years old, and the mortality rate from breast tumors is increasing in developed countries. The causes of breast cancer can be attributed to lifestyle factors, age, genetics, pregnancy and menstruation age, previous benign breast tumors, and ionizing radiation (Łukasiewicz et al., 2021). Breast tumors contain heterogeneous cell populations that have different receptors, including estrogen receptors (ERs), progesterone receptors (PRs), and human epidermal growth factor receptor 2 (HER2). These receptors are also used as therapeutic targets. In hormone therapy, ERs and PRs (such as letrozole) are targeted, while, in treatment with monoclonal antibodies, drugs targeting the HER2 receptor (such as trastuzumab) are used (Yin et al., 2020). However, 40% of breast tumor cells lack these receptors and are known as triple-negative breast cancer cells (TNBCs). Because TNBCs do not have these receptors, they cannot be treated with hormone therapy or monoclonal antibody drugs that target these receptors. Therefore, chemotherapy is the only option for treating TNBCs. The 4T1 cell line is a member of the TNBC cell cluster, and the only way to treat them is through chemotherapy drugs (Johnson et al., 2021).
Chemotherapy drugs are commonly used to treat cancer, but they can also induce oxidative stress and activate inflammatory pathways in the body. Chemotherapy drugs damage not only the deoxyribonucleic acid (DNA) of rapidly dividing cancer cells but also the DNA of healthy cells, leading to the production of reactive oxygen species (ROS) and oxidative stress (Schirrmacher, 2019). Chronic oxidative stress can lead to the activation of signaling pathways that promote cell growth and survival, such as the nuclear factor-kappa B (NF-κB) pathway and the mitogen-activated protein kinase (MAPK) pathway. These pathways can activate genes involved in cell proliferation and survival, leading to the growth and spread of cancer cells (Liu et al., 2021). Chemotherapy drugs also induce the production of pro-inflammatory cytokines [tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-1 (IL-1)], which can contribute to the activation of inflammatory pathways and promote cancer cell proliferation and metastasis (Grova et al., 2021). Doxorubicin (DOX) is a chemotherapy drug commonly used in the treatment of breast cancer, as well as other types of cancer such as lymphoma and leukemia (Daneshforouz et al., 2021). DOX works by damaging the DNA of cancer cells, which prevents them from dividing and growing. However, it can also damage the DNA of healthy cells, leading to various side effects such as suppression of bone marrow, neurological toxicity, severe weight loss, suppression of ovarian function, toxicity, heart failure, secondary tumor formation, nausea, physical exhaustion, reduced lifespan, loss of cognitive function, severe gastrointestinal, liver, kidney toxicity, and glomerular destruction (van der Zanden et al., 2021). Therefore, reducing chemotherapy-induced oxidative stress and inflammation may help alleviate some of the side effects of chemotherapy and improve treatment outcomes. Antioxidant supplementation, such as with vitamins C and E, may help reduce chemotherapy-induced oxidative stress (Ilghami et al., 2020).
Bee pollen is a mixture of pollen, nectar, enzymes, honey, and beeswax that is collected by bees and stored in the hive. It is considered a superfood due to its high nutritional content, which includes vitamins, minerals, amino acids, and antioxidants. It has been shown to have anti-inflammatory and antioxidant properties, which may help protect against chronic diseases such as heart disease, diabetes, and cancer. Bee pollen has also been studied for its potential to improve athletic performance, reduce allergy symptoms, and support immune function (Thakur & Nanda, 2020). Bee pollen contains various bioactive compounds, including isoflavonoids (including genistein, daidzein, glycitein, biochanin A, formononetin, puerarin, coumestrol, and equol) and flavonoids (including quercetin, kaempferol, apigenin, luteolin, myricetin, hesperetin, naringenin, rutin, catechin, epicatechin, epigallocatechin, and proanthocyanidins), which have a similar structure to estrogen and can bind to ERs in the body (Mayda et al., 2020). Polyphenolic compounds (flavonoids and isoflavonoids) in bee pollen have potential anticancer effects through modulation of cancer-related pathways including apoptosis, cell cycle, PI3K/Akt/mTOR, NF-κB, and MAPK pathways (Zughaibi et al., 2021).
The aim of this study is to investigate the potential synergistic effects of bee pollen and DOX in inhibiting the proliferation and metastasis of breast tumors in 4T1 tumor-bearing BALB/c mice. The study utilizes a biochemical, immunohistochemical, and molecular approach to evaluate the efficacy of the combination therapy in inhibiting tumor growth and metastasis, as well as the underlying mechanisms involved.
Materials and Methods
Preparation of BP
To obtain the BP extract, 3000 grams of dried BP were ground into a powder using a soil grinder (Cat. No. H-4199.5F; Humboldt Company, USA). The resulting powder was mixed with a solution of hexane and methanol (30:70 v:v) and incubated in a dark room at 40°C for 72 h. After incubation, the mixture was filtered through a No. 42 paper filter (Cat. No. 1442-125; Millipore, USA) and concentrated using a rotary evaporator (model: 9230; Buchi Rotavapor, Switzerland). The final yield of BP extract was 20% (250 g), and it was stored at −4°C (Lawag et al., 2021).
Animal Care, 4T1-Breast Cancer Induction, Grouping, Treatment, Experimental Design, and Research Timeline
Cell Culture and 4T1-Breast Cancer Induction
The 4T1 cancer cells (Cell Bank of the Chinese Academy of Sciences, Shanghai, China) were cultured in RPMI-1640 (Gibco, Life Technologies, Inc., USA) supplemented with 10% fetal bovine serum (FBS) (Hyclone, USA) and 1% penicillin-streptomycin. The cells were maintained in a humidified incubator at 37°C with 5% CO2. The medium was changed every 2 days, and the cell surface was washed with phosphate-buffered saline (PBS) 2–3 times daily to remove serum, eliminate dead cells, and regulate cell osmolality. To detach the cells, 0.25% trypsin-ethylenediaminetetraacetic acid (EDTA) solution was added to each 25 cm2 flask and incubated at 37°C for 5 min. After 80%–90% of the cells detached, trypsin was neutralized by adding a medium containing 10% FBS, and the cells were centrifuged at 1,500 rpm for 5 min. The supernatant was discarded, and a fresh culture medium was added to the cell pellet. For the animal study, 100 adult female BALB/c mice (6–8 weeks old) were selected and injected with 1 × 106 cells along with 100–80 µL of culture medium into the back and flank region of each mouse. Ten days after injection, tumors were induced and palpable, and 50 4T1 tumor-bearing mice were selected for the study (Barnett et al., 2022).
Animal Care
The mice were housed in a controlled environment with a 12-h light/12-h darkness cycle, a temperature of 25 ± 2°C, and a humidity of 45 ± 5%. The mice were provided with standard laboratory pellets and had access to sanitary tap water. The mice were kept in accordance with international standard guidelines and protocols, as well as the ethics committee of the University of Melbourne. The pellets were formulated to contain all the necessary nutrients for proper nutrition, including 4.5 Mcal of energy, 21 grams of protein per 100 grams, 3 grams of total fat per 100 grams, 0.8 grams of calcium per 100 grams, 0.4 grams of phosphorus per 100 grams, and 6 grams of total fatty acids per 100 grams (including palmitic acid, oleic acid, linoleic acid, and arachidonic acid), following the manufacturer’s instructions (Barnett et al., 2022).
Animal Grouping, Experimental Design, and Research Timeline
Sixty female BALB/c mice were used in the study and divided into six groups (n = 10/group) as follows:
During the 35-day study, the mice were given BP daily at 9:00 a.m. and DOX at 3:00 p.m. on days 10, 20, and 30. The doses and administration schedule were determined based on previous studies and preliminary investigations to ensure the safety and efficacy of the treatments. The optimal therapeutic dose of BP was identified through a combination of methods, including a literature review, a preliminary investigation, and the LD50 technique. The aim of this approach was to identify a safe and effective dose of BP that could reduce tumor growth without causing toxicity or adverse effects in the mice (Shaldoum et al., 2021; Tian et al., 2021).
Weight of Mice, Volume of Tumors, and Assessment of Survival Rate
After tumor induction and palpation on the 10th day of the study, tumor volume was measured on the 10th, 17th, 24th, 31st, and 35th days using the formula V = 0.5 × d2 × D, where d represents the larger diameter and D represents the smaller diameter of the tumor. Mortality was recorded throughout the study to assess the survival of the mice, and a Kaplan-Meier curve was generated (Figure 1). These measurements and analyses were conducted to evaluate the effect of the treatments on tumor growth and mouse survival throughout the study (Tian et al., 2021).
BP Acute Toxicity Test (LD50)
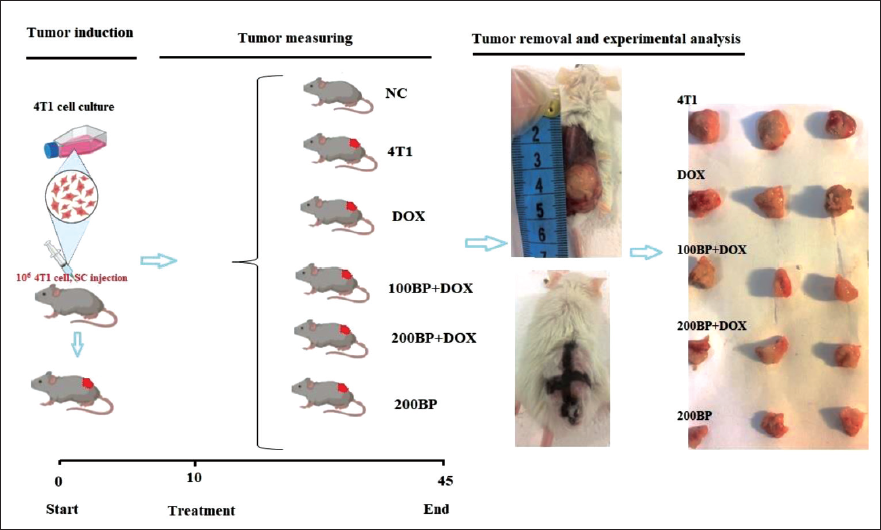
To determine the optimal treatment dose with minimal toxicity, Lork’s two-step technique was employed. First, nine female mice were divided into three groups (n = 3/group) and administered doses of 50, 500, and 5,000 mg/kg of BP via gavage. Three additional mice were then assigned to three separate groups and administered doses of 10, 100, and 1000 mg/kg of BP via gavage. Following a 24-h monitoring period, the LD50 of BP was calculated using a formula, and a safe and effective dose was selected for the study. This approach was used to identify a therapeutic dose of BP that could reduce tumor growth while minimizing toxicity and adverse effects in the mice.
In this method, M denotes the lowest dose at which the group exhibited toxic symptoms, such as weight loss, diarrhea, nausea, skin rashes, or mortality. N represents the highest dose at which no toxic symptoms or mortality were observed in the group (Cui et al., 2022).
Serum Levels of Estradiol (E2), Follicle-Stimulating (FSH), Luteinizing (LH), and Testosterone (T) Hormones
At the end of the study, the mice were euthanized using pre-anesthesia and anesthesia protocols involving 100 mg/kg xylazine 2% and 15 mg/kg ketamine 10%, respectively. Blood was obtained from the heart and centrifuged at 10,000 g for 15 min to isolate serum samples. The levels of E2 (catalog number: KA1907), FSH (catalog number: KA2330), LH (catalog number: NBP2-68054), and T (catalog number: NBP2-42044) hormones were measured in the serum using commercially available Novus ELISA kits based on a colorimetric assay, following the manufacturer’s recommendations and protocol (Ibrahim et al., 2021).
Serum Levels of IL-6, IL-8, Interferon-γ (IFN-γ), IL-1β, and TNF-α Levels
Commercial R&D ELISA kits (R&D Systems, Inc., USA) were used to measure the serum levels of IFN-γ (catalog number: DY585), IL-6 (catalog number: R6000B), IL-8 (catalog number: D8000C), IL-1β (catalog number: RLB00), and TNF-α (catalog number: RTA00) based on a colorimetric assay, following the manufacturer’s recommendations and protocol (Ibrahim et al., 2021).
Serum Nitric Oxide (NO) Levels
Serum total NO levels were assessed using a commercial kit from ZellBio (catalog number: ZX-44107-96; ZellBio GmbH, Germany). The manufacturer’s instructions were followed, wherein 50 µL of standard or serum sample was added to all wells of a 96-well plate. A zero standard of 50 µL of assay buffer was also added. Thereafter, 10 µL of nitric reductase solution was added to each well, followed by incubation at 25°C for 20 min. Subsequently, 25 µL of solution A and 25 µL of solution B were added to each well and incubated for another 20 min at 25°C. Finally, the absorbance of the final mixture was read at a wavelength of 540–570 nm using a UV-visible spectrophotometer (Model No. V-630 Bio; JASCO, USA) (Zahedi et al., 2022).
Serum Activity of Superoxide Dismutase (SOD), Glutathione Peroxidase (GPx), and Catalase (CAT)
To determine the serum activity of GPx, SOD, and CAT, commercially available ELISA kits were used, including catalog number E-EL-R2491 from Elabscience (USA) for GPx, catalog number E-EL-R1424 from Elabscience (USA) for SOD, and catalog number ab100764 from Abcam (USA) for CAT. The measurement process followed the manufacturer’s instructions and protocol (Wankhar et al., 2020).
Tumor Total Thiol, Lipid Peroxidation Levels (TBARS), and Ferric-reducing Antioxidant Power (FRAP) Levels
To determine the overall antioxidant capacity of femur tissue using the FRAP method, the femur was dissected and a tissue homogenous mixture was prepared. Next, 200 µL of the homogenous tissue was combined with a FRAP solution that contained 5 mL of 2, 4, 6-tripyridyl-triazine (TPTZ), 50 mL of acetate buffer, and 3 mL of ferric chloride, and then incubated at 37°C for 10 min. The absorbance of the resulting mixture was subsequently measured using a Stat Fax ELISA reader (Stat Fax, USA) at a wavelength of 593 nm, and the total antioxidant capacity was calculated as µmol/mg protein.
The TBARS level measurement method was utilized to assess the degree of lipid peroxidation in femur tissue. To perform this method, a homogeneous femur tissue sample of 100 µL was combined with a TBARS solution, containing 50 µL of thiobarbituric acid, 2 µL of butylated hydroxytoluene, and 50 µL of phosphoric acid. The mixture was then incubated at 37°C for 30 min. After incubation, the final mixture was evaluated for absorbance using a Stat Fax ELISA reader (Stat Fax, USA) at a wavelength of 532 nm. The TBARS level was then determined as nmol/mg protein.
To quantify the total thiol level in femur tissue, a homogenized sample of 50 grams was combined with 250 µL of Tris-EDTA and incubated at 25°C for 10 min. Using a Stat Fax ELISA reader (Stat Fax, United States), the initial absorbance (α) of the mixture was recorded at a wavelength of 412 nm. Next, 20 µL of DTNB [5,5-dithio-bis-(2-nitrobenzoic acid)] was added to the mixture, which was then incubated for 20 min at 25°C. The final absorbance (β) was measured again at 412 nm. Additionally, the absorbance of the DTNB blank solution (µ) was recorded at 412 nm. The tissue thiol level was calculated using the following formula:
Total thiol concentration (µM) = (β – α – µ) × 1.07/(6.8) (Campestrini et al., 2019).
Real-Time Polymerase Chain Reaction (RT-PCR) Assay
Total RNA Extraction
The Bio Basic column animal total RNA purification kit (catalog number: EZ-10; Bio Basic Inc., Canada) was employed to extract total RNA from tumor tissue following the manufacturer’s guidelines. The extracted RNA was stored at −80°C (Kingston et al., 2021).
cDNA Synthesis and Quantitative Real-Time PCR (qPCR)
The Takara Bio RT Kit (RTK0104; Takara Bio., Japan) was used to synthesize cDNA templates from 1 µg of total RNA, following the manufacturer’s instructions. Real-time PCR was performed using 1000 ng of cDNA templates, 1 µL each of reverse and forward primers, and 8 µL of Takara Bio Mastermix. The expression levels of Bax, Bcl-2, p53, and caspase-3 genes were quantified using 42 temperature cycles in an Applied Biosystems thermocycler (Applied Biosystems, Quantstudio 1, USA), with each cycle including specific temperature stages. In the real-time PCR, there was a series of temperature cycles. These cycles consisted of a 5-minute holding stage at 50°C, a 15-s denaturation stage at 95°C, a 1-min annealing stage at 60°C, and a 5-min extension stage at 70°C. Following the temperature cycles, a melting curve was generated by gradually increasing the temperature from 60°C to 95°C at a rate of 1°C per second. CT values were recorded for the control, sample, and β-actin genes, and the 2−∆∆CT formula was used to calculate the fold change. The primers used in the real-time PCR were designed using Primer-3 software and their specificity was confirmed by blast search from the National Center for Biotechnology Information (NCBI) database. The internal reference gene β-actin was used, and the primer sequences are provided in Table 1.
Primer Sequences.

Western Blotting Assay
The Western blotting technique was used to assess the expression levels of mitochondrial apoptosis pathway proteins (Bax, Bcl-2, p53, and caspase-3). Initially, tumor tissue was washed with 200 µL of PBS, and 50 mg of the tissue was lysed with radio-immunoprecipitation assay buffer containing Tris-HCl (50 mmol/L), NaCl (150 mmol/L), and 0.1% NP-40 [w/v]. The lysate was centrifuged at −4°C for 10 min at 12,000g, and 50 µg of the resulting mixture was combined with loading buffer containing Tris-HCl (62.5 mmol/L), β-mercaptoethanol (1 µL), 10% glycerol (5 µL), and 0.5% sodium deoxycholate (5 µL) [w/v]. The mixture was then loaded onto a 10% sodium dodecyl sulfate (SDS)-polyacrylamide gel and transferred onto a polyvinylidene fluoride (PVDF) membrane (Cat. No. WBKLS0100; Millipore Bio Inc., UK). The PVDF membrane was incubated with GeneTex Bax (catalog number: GTX109683; 1:500), Bcl-2 (catalog number: GTX100064; 1:1000), p53 (catalog number: GTX102965; 1:500), and caspase-3 (catalog number: GTX86900; 1:500) antibodies (GeneTex Bio., USA) overnight at 4°C. Excess antibodies were removed by washing the membrane with Tris-buffered saline and Tween-20 (0.05%) [w/v]. horseradish peroxidase (HRP)-conjugated secondary antibody was added and incubated for 1 h at 37°C. The signals of the protein bands were recorded and evaluated using the Image J software and Bio-Rad software, as well as an enhanced chemiluminescence reagent (e-BLOT company, China) (Luo et al., 2020).
Immunohistochemistry (IHC) Assay
Ki-67, Bcl-2, and p53 positive cells were used as apoptotic-differentiation markers for tumor cells. The tumor tissues were washed with PBS and underwent routine tissue processing, and paraffin blocks were prepared from the tumor. Notably, 5-µm sections were placed on slides and incubated with primary Ki-67 (1:1000; catalog number: AF7649), Bcl-2 (1:1000; catalog number: MAB8272), and p53 (1:1000; catalog number: GAF1355) antibodies (R&D Systems, Inc., USA) overnight at 95°C and then for 1 h at 25°C. Tween-20 was used as the washing buffer, and bovine serum albumin (5%) was utilized to block residual antibodies. The slides were then treated with 3% hydrogen peroxide (H2O2) for 20 min at 25°C followed by 3,3′-diaminobenzidine (DAB). Hematoxylin was used to counterstain all slides. The slides were examined using an optical microscope (model No. BX61TRF; Olympus, Japan) connected to ImageJ software at 100× magnification. The percentage (%) of Ki-67, Bcl-2, and p53 positive cells/all cells was reported in 10 random fields of view in each sample (Missaoui et al., 2019).
Tumor Tissue Histopathology
An optical microscope (Model No. BX61TRF; Olympus, Japan) connected to ImageJ software was used to assess tumor cell apoptosis and histopathological findings at 100× magnification. Similar to the IHC assay, 5-µm sections were mounted on slides and subjected to Hematoxylin and Eosin (H&E) and Masson’s trichrome staining to evaluate tumor histopathology. H&E staining is a common staining technique used in histopathology to visualize cell nuclei, cytoplasm, and extracellular matrix, while Masson’s trichrome staining is used to visualize collagen fibers in tissues (McMullen et al., 2019).
Statistical Analysis Approach
The quantitative results obtained from the different groups were compared using a one-way analysis of variance (ANOVA) and a Newman-Keuls post hoc test. Statistical significance was determined by a p-value less than 0.05. To assess the normality of the data, the Kolmogorov-Smirnov test was conducted, and data with p-values greater than 0.05 were considered to be normally distributed and homogeneous. The mean and standard deviation (SD) were used to express all results. The data were analyzed using IBM Inc.’s SPSS software (Ver. 16) and GraphPad Inc.’s Prism software (Ver. 9) was used to create graphs.
Results
LD50 of BP
Based on the evaluation of the LD50 of BP extract using Lork’s approach and monitoring of the groups for 24 h, it was found that the highest safe dose was 5,000 mg/kg and the lowest dose resulting in death was 1,000 mg/kg. Applying Lork’s formula, the LD50 was calculated to be 2.37 g/kg. This means that doses below 2,370 mg/kg are considered safe and can be administered in animal models.
Body Weight, Tumor Volume, and Survival Rate (%)
The weight of 4T1 tumor-bearing mice decreased during the study period compared with the NC group, and this decrease was significant (p < 0.05) in the results of the end of the study (day 45) of DOX, 4T1, and 100BP+DOX groups. While 200 BP and 200BP+ DOX showed a significant (p < 0.05) increase compared with the 4T1 and DOX groups (Figure 2a).
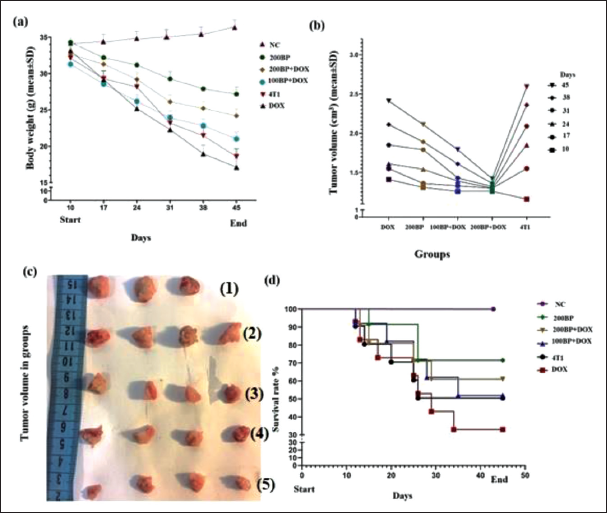
Throughout the study, the tumor volume in all mice bearing 4T1 tumors increased steadily on days 10, 17, 24, 31, 38, and 45. However, at the end of the study (day 45), the tumor volume in both the 200BP + DOX and 100BP + DOX groups exhibited a significant (p < 0.05) decrease compared with the 4T1 group. Furthermore, the tumor volume in the 200BP + DOX group at day 45 showed a significant (p < 0.05) decrease in comparison to the DOX group (Figure 2b and c).
The Kaplan-Meier diagram used to evaluate the survival of mice during the study period revealed that the highest number of deaths occurred in the 4T1, DOX, and 100BP + DOX groups. Additionally, the evaluation of results at the end of the study indicated that the survival rate (%) of mice in the 4T1, DOX, and 100BP + DOX groups was significantly (p < 0.05) lower compared with the NC group. On the contrary, the survival rate (%) of mice in the 200BP and 200BP + DOX groups were significantly (p < 0.05) higher than that of the DOX group (Figure 2d).
Serum Levels of E2, FSH, LH, and T Hormones
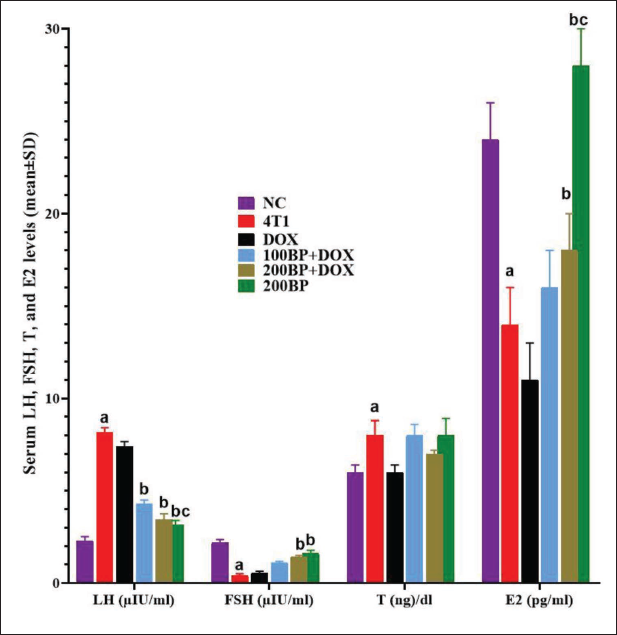
The levels of LH and T were significantly (p < 0.05) higher in 4T1 tumor-bearing mice compared with the NC group, whereas the levels of FSH and E2 were significantly (p < 0.05) lower. Treatment with BP in a dose-dependent manner, along with DOX (in 100BP + DOX and 200BP + DOX groups), resulted in a significant (p < 0.05) decrease in LH levels and a significant (p < 0.05) increase in FSH and E2 levels when compared with the 4T1 group. In the 200BP group, the level of LH decreased significantly (p < 0.05) compared with the 4T1 and DOX groups, while the level of FSH and E2 increased significantly (p < 0.05) (Figure 3).
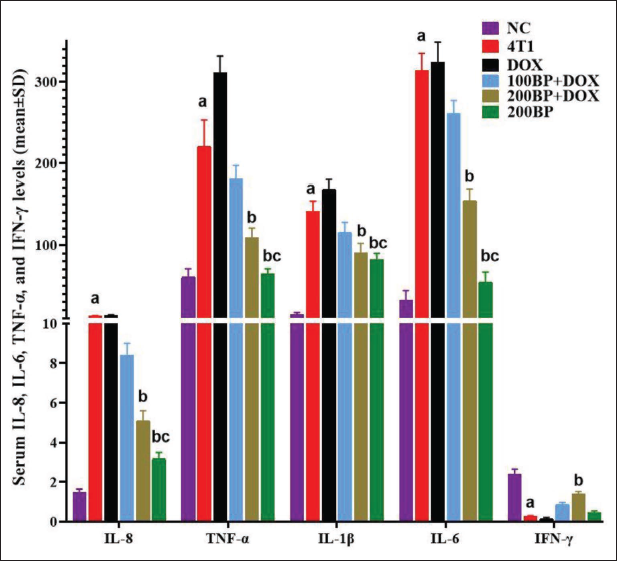
Serum Levels of IL-6, IL-8, IFN-γ, IL-1β, and TNF-α Levels
The results of the cytokine analysis showed that the levels of IL-6, IL-8, IL-1β, and TNF-α were significantly (p < 0.05) higher in 4T1 tumor-bearing mice compared with the NC group, whereas the level of IFN-γ was significantly (p < 0.05) lower. Treatment with DOX increased the levels of IL-6, IL-8, IL-1β, and TNF-α, as well as a decrease in the level of IFN-γ, although these changes were not significant (p > 0.05) compared with the 4T1 group. In contrast, treatment with BP in a dose-dependent manner (in 100BP + DOX and 200BP + DOX groups) significantly (p < 0.05) decreased the levels of IL-6, IL-8, IL-1β, and TNF-α compared with the 4T1 and DOX groups (Figure 4).
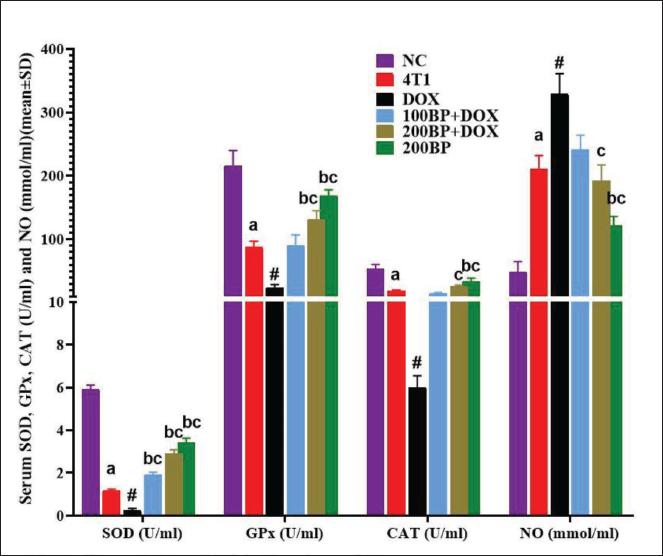
Serum Activity of SOD, GPx, and CAT as well as NO Levels
The activity level of SOD, GPx, and CAT in 4T1 tumor-bearing mice and DOX significantly (p < 0.05) decreased and the NO level increased significantly (p < 0.05) compared with the NC group, while BP caused a significant (p < 0.05) increase in the activity of SOD, GPx, and CAT in a dose-dependent manner and a significant (p < 0.05) decrease in NO compared with the 4T1 and DOX groups (Figure 5).
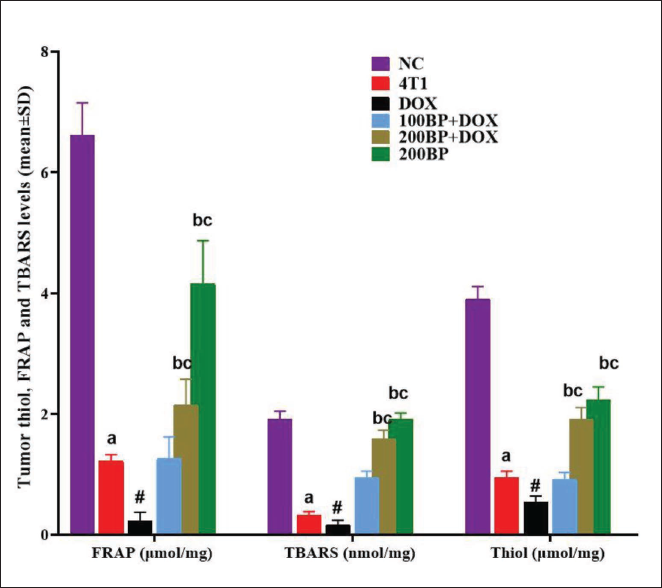
Tumor Total Thiol, TBARS, and FRAP Levels
The results related to oxidative stress parameters of tumor tissue showed that the amount of total thiol, TBARS, and FRAP in tumor group 4T1 and DOX significantly (p < 0.05) decreased compared with the NC group (non-tumor breast tissue). Even DOX significantly (p < 0.05) reduced all three parameters compared with the 4T1 group. While BP increased significantly (p < 0.05) in all three parameters with its antioxidant effects (Figure 6).
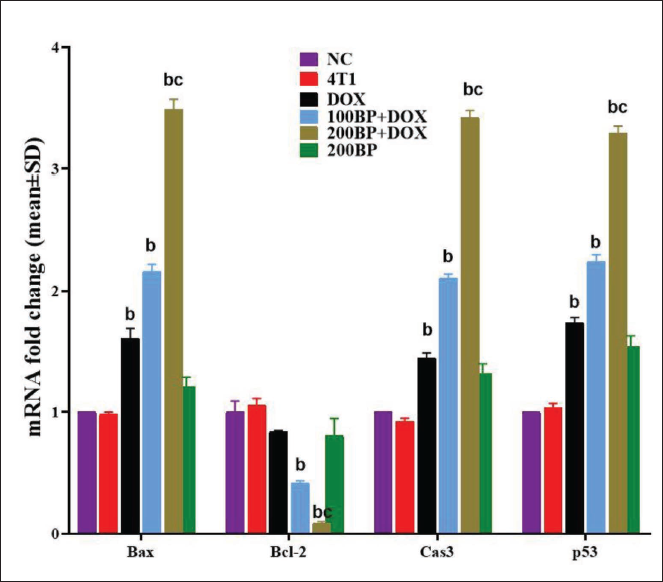
Bax, Bcl-2, Caspase-3, and p53 Genes Expression
Following 4T1 induction, the expression of apoptotic genes (Bax, caspase-3, and p53) decreased significantly (p < 0.05) compared with the non-tumor tissue of the NC group, while the expression of the anti-apoptotic gene Bcl-2 decreased significantly (p < 0.05). This was even though DOX was able to significantly (p < 0.05) increase the expression of apoptotic genes (Bax, caspase-3, and p53 genes) and significantly (p < 0.05) decrease the expression of the anti-apoptotic gene Bcl-2 compared with the 4T1 group. BP was able to significantly (p < 0.05) increase the expression of apoptotic genes (Bax, caspase-3, and p53 genes) and the expression of the anti-apoptotic gene Bcl-2 in a dose-dependent manner with synergistic effects in the groups co-treated with DOX (in 100BP + DOX and 200BP + DOX groups) compared with the 4T1 and DOX groups significantly reduced (p < 0.05) (Figure 7).
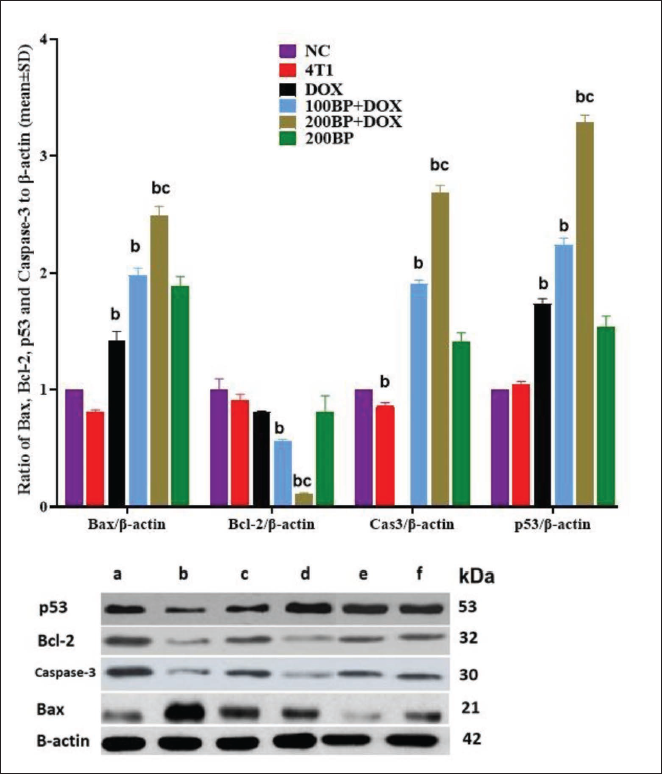
Bax, Bcl-2, Caspase-3, and p53 Proteins Expression
Similar to the results obtained for the expression of pro/anti-apoptotic genes, the expression of pro-apoptotic proteins (Bax, caspase-3, and p53) was significantly (p < 0.05) lower in the 4T1 group compared with non-tumor NC tissue, whereas the expression of anti-apoptotic protein Bcl-2 was significantly (p < 0.05) lower in the 4T1 group compared with the NC group. Treatment with DOX resulted in increased significantly (p < 0.05) expression of pro-apoptotic proteins (Bax, caspase-3, and p53) compared with the 4T1 group. Treatment with BP significantly (p < 0.05) increased the expression of pro-apoptotic proteins (Bax, caspase-3, and p53) in the group co-treated with DOX (in 100BP + DOX and 200BP + DOX groups) compared with the DOX and 4T1 groups (Figure 8).
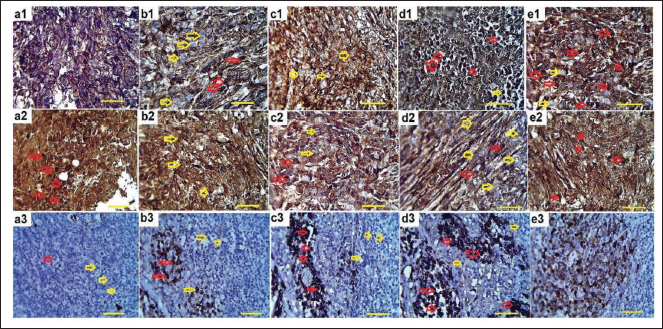
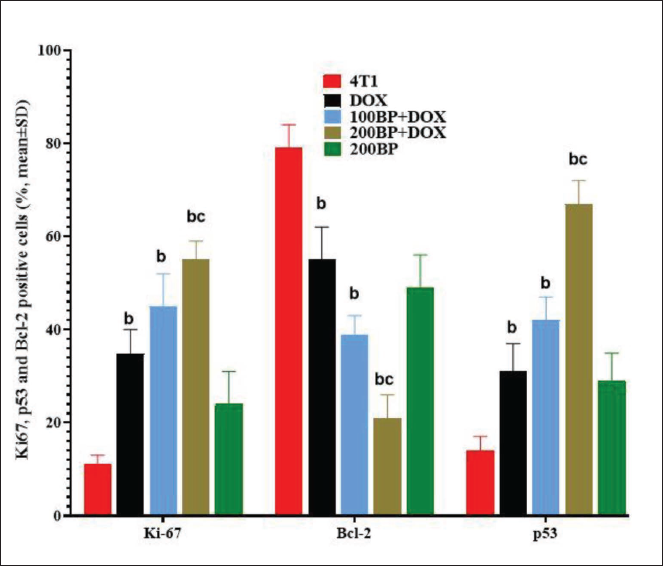
IHC Assay Results for Ki-67, Bcl-2, and p53 Expression
DOX treatment resulted in a significant (p < 0.05) increase in the percentage of p53 and Ki-67 cells, as well as a significant (p < 0.05) decrease in the percentage of Bcl-2 cells, compared with the 4T1 group. Treatment with BP in combination with DOX (in 100BP + DOX and 200BP + DOX groups) also resulted in a significant (p < 0.05) increase in the percentage of p53 and Ki-67 cells, as well as a significant (p < 0.05) decrease in the percentage of Bcl-2 cells, compared with the DOX and 4T1 groups (Figures 9 and 10).
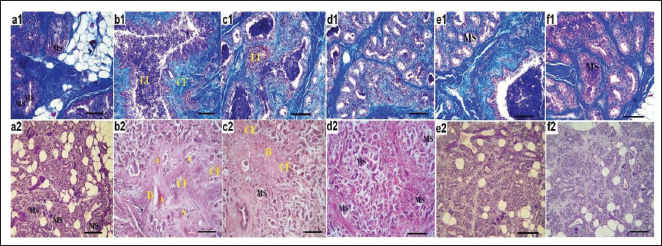
Tumor Tissue Histopathology
Histopathological evaluations, using Masson’s trichrome and H&E staining, revealed that, following the induction of breast tumors with 4T1 cell injections, the density of these cells in the tissue parenchyma of the injected area increased. The mammary glands atrophied, and lymphocyte infiltration was observed. The synthesis of collagen fibers increased, and the lumens of the glands were filled with 4T1 cells, leading to the degeneration of the mammary parenchyma. DOX treatment was able to reduce the density of 4T1 cells and promote the organization of mammary sinuses. Meanwhile, in the BP and DOX co-treatment groups (100BP+DOX and 200BP+DOX), the normal architecture of the mammary sinuses was visible, the mammary glands and their ducts were well-organized, and the density of 4T1 cells was significantly reduced. There was no lymphocytic infiltration in the main parenchyma of the breast, and the tissue was not degenerative or atrophic (Figure 11).
Discussion
The results of the study suggest that BP extract, in combination with DOX, may have a potential role in inducing apoptosis of 4T1 tumor cells. The increase in the expression of pro-apoptotic proteins such as Bax, p53, and caspase-3 observed in the BP and DOX co-treatment groups indicates a potential mechanism for inhibiting breast cancer growth. Additionally, the reduction in free radicals and pro-inflammatory cytokines observed in the BP-treated groups may contribute to the anti-tumor effects of the extract. Importantly, the protective effects of BP extract on normal cells and the main parenchyma from oxidative and inflammatory damage suggest that it may have a potential role in reducing the side effects of chemotherapy.
Endogenous estrogens, like estradiol, are known to promote the growth and proliferation of breast cancer cells, including those of triple-negative breast cancer (TNBC) because breast cancer cells have ERs. When estrogen binds to the ER, it activates a signaling pathway that stimulates cell growth and proliferation (Starek-Świechowicz et al., 2021). Phytoestrogens, such as flavones and isoflavones found in soybeans and other natural products, have both estrogenic and anti-estrogenic effects. These compounds can bind to ERs and act like estrogen, but they can also compete with naturally occurring estrogens for binding to the ER and act as anti-estrogens. This dual action has sparked interest in their potential as agents for breast cancer prevention and treatment (Garcia-Oliveira, 2021). Isoflavones, in particular, can competitively bind to ERs by blocking estrogen-binding sites, which are known to promote tumor growth in breast cancer, including TNBC. By inhibiting the activation of genes that respond to estrogen (17-β-estradiol), isoflavones can suppress tumor growth (Zhang et al., 2020). Additionally, isoflavones and other flavonoids can inhibit aromatase enzymes involved in the production of estradiol (E2) and estrone (E1), as well as suppress the conversion of endogenous estrogens by the enzyme 17-β-hydroxysteroid dehydrogenase (17-β-HSD). Studies conducted on animal models and breast cancer patients have shown that high levels of estradiol in the blood are often associated with breast tumors and can even be considered an indicator of tumor growth and treatment prognosis (Montano et al., 2022). Isoflavones have been shown to inhibit the growth of breast cancer cells, including TNBC cells, and counteract the effects of endogenous estrogens. A study on cynomolgus monkeys conducted by Wood et al. (2006) demonstrated that isoflavones can decrease the levels of estradiol in the blood and reduce the expression of Ki-67 in breast tissue. Ki-67 is an important marker for monitoring the progression of breast tumors and predicting treatment outcomes (Wood et al., 2006). In this study, we found that BP, specifically its polyphenolic compounds like isoflavonoids, can regulate the levels of E2 hormone and other hormones influenced by the hypothalamic-pituitary-gonad (HPG) axis. These findings suggest that BP extract may have potential as a complementary therapy for breast cancer treatment, particularly in the case of TNBC.
ROS, are highly reactive radicals that are formed during oxygen metabolism and play a crucial role in regulating various cellular processes, including proliferation, migration, tissue repair, and angiogenesis. They can mediate multiple signal transduction pathways and have been implicated in tumor development, survival, and metastasis (Hecht et al., 2016). Certain chemotherapy drugs, such as DOX, can increase ROS production, further enhancing the tumorigenic potential and metastasis of cancer cells. Both ROS and estrogen can activate and stimulate the synthesis of activator protein 1 (AP-1), an essential regulator of tumor cell growth and proliferation. Additionally, ROS can facilitate tumor cell metastasis by diminishing the attachment of tumor cells to the basal lamina of the basement membrane or by increasing the activity or expression of proteins that govern cell motility and attachment, such as E-cadherin and collagen IV (Alsherbiny et al., 2021). In a study conducted by Monte et al. (2007), intravenous injection of hydrogen peroxide (H2O2) was observed to activate angiogenesis by generating ROS, thus intensifying lung metastasis in a mouse model of breast cancer. Moreover, during the early stages of tumorigenesis, ROS-induced oxygen deprivation can prompt signal pathways that foster tumor cell proliferation and angiogenesis (Ahlina et al., 2020). Arfin et al. (2021) demonstrated that ROS-induced Nox1 could heighten the expression of vascular endothelial growth factor (VEGF) receptors and enhance matrix metalloproteinases (MMPs) activity, crucial indicators of angiogenesis and metastasis, ultimately promoting rapid tumor proliferation (Arfin et al., 2021). ATP-binding cassette (ABC) transporters, such as P-gP and multidrug resistance-1 (MDR-1), are present in tumor cells and can contribute to cancer cell resistance by eliminating ROS and other metabolites produced by chemotherapy compounds, thereby reducing their effectiveness. Additionally, studies have revealed that cancer cells have developed systems to combat ROS, leading to a notable upregulation of genes encoding superoxide dismutase, catalase, and glutathione peroxidase enzymes (Goebel et al., 2021). Numerous studies have demonstrated the potent antioxidant properties of BP or bitter melon extract. For instance, Pratheeshkumar et al. (2012) conducted a study on quercetin, a major flavonoid in BP, and found that it can regulate the expression of VEGFR-2 through the AKT/mTOR/P70S6K signaling pathway, inhibiting angiogenesis in an angiogenesis model in the chorioallantoic membrane (CAM) and human prostate cancer cell line (PC-3). Another study conducted on an animal model of human breast cancer cell line transplantation (MCF-7) in nude mice demonstrated that quercetin can decrease VEGFR-2 expression by targeting the calcineurin/NFAT pathway, ultimately reducing angiogenesis in tumor tissue (Zhao et al., 2023). In terms of antioxidant properties, a study by Shaldoum et al. (2021) explored the protective effects of BP against DOX-induced damage to the immune system, bone marrow, and spleen in rats. The study found that the plant extract increased the activity of enzymes involved in reducing ROS, including CAT, SOD, and GPx. Additionally, the extract decreased the serum level of NO and lowered the tissue level of malondialdehyde (MDA), suggesting reduced oxidative stress (Shaldoum et al., 2021). Similarly, in another study by Shahba and Mansour (2022) on the impact of BP extract on the reproductive performance of male v-line rabbits, it was noted that the extract increased the activity of CAT and GPx, leading to improved reproductive performance in the rabbits (Shahba & Mansour, 2022). In this study, the BP extract was found to enhance the activity of endogenous antioxidant enzymes, subsequently increasing FARP, total thiol, and TBARS, while simultaneously decreasing the level of NO.
Inflammatory pathways are known to play a central role in the proliferation, differentiation, and metastasis of tumor cells. One such inflammatory cytokine is IL-6, which is produced and secreted by various types of cells, including tumor cells. IL-6 is found at high levels in serum and tumor tissues in most tumors, especially breast tumors, and plays a role in the proliferation and differentiation of tumor cells (Hartman et al., 2013). Studies have shown that a high serum level of IL-6 in 4T1 breast cancer animal models weakens the efficacy of DOX treatment (Li et al., 2020). Increased levels of IL-6 are directly related to the growth and metastasis of tumors, and patients with high circulating levels of IL-6 are generally associated with a poor prognosis and shorter survival, while lower levels of IL-6 are associated with a better response to treatment. The IL-6/JAK/STAT3 pathway regulates the expression of several genes involved in the proliferation, survival, and metastasis of breast cancer cells. IL-6-induced JAK/STAT-3 and NF-κB signaling facilitates nuclear translocation of STAT-3 and NF-κB. Activation of these signaling pathways leads to the expression of anti-apoptotic genes such as Bcl-2, Bcl-xL, Mcl-1, survivin, and so on (Hartman et al., 2013; Manore et al., 2022). The role of IL-8 in the invasion, progression, and metastasis of tumors has also been well-established. High levels of IL-8 in TNBCs are associated with the aggressive behavior of these cells. IL-8 affects the expression of cell adhesion molecules such as E-cadherin and fibronectin on human breast cancer cells. It increases the expression of β3 integrin and enhances the invasive potential of breast cancer cells through the activation of the PI3K/Akt pathway and subsequently NF-κB. NF-κB, in turn, plays an important role in inducing IL-8 gene transcription through several signaling mediators such as LPS, TNF-α, and oxidative stress (Deng et al., 2020). The IL-8/PI3K/Akt/NF-κB/β3 integrin axis is considered one of the most important therapeutic targets for inhibiting the growth of metastatic breast tumors. As mentioned earlier, ROS stimulates and activates the Erk signaling pathway through the induction of IL-8 cytokine secretion, which subsequently stimulates the migration of TNBCs. There is also an inverse relationship between IL-8 levels and ER expression (Shao et al., 2015). IL-12 and IFN-γ have important roles in regulating the expression of p53 and p27, activating macrophages, stimulating the differentiation of (CD4+) T cells into the Th1 subclass, increasing the apoptosis of tumor cells, and increasing the sensitivity of tumors to recognition and destruction by the immune system (Jorgovanovic et al., 2020). Even a study in mice on breast cancer has shown that inhibiting the IFN-γ signaling pathway facilitates the initiation of tumorigenesis in tumor cells (Todorović-Raković et al., 2022). Studies on the effect of isoflavones on the levels of these two cytokines have shown that these compounds can increase the secretion of IL-12 and IFN-γ from immune cells. Studies have shown that isoflavones can increase the production of IFN-γ from cytotoxic T and TH1 cells in mice, as well as affect the production of IL-12 and IFN-γ by NK cells. Additionally, there are significant connections between IL-12 and IFN-γ and ERs (ERα and ERβ) even in breast cancer cell lines lacking ERs (Mace et al., 2019). Autocrine IFN-γ increases the mRNA expression of ER ERβ, thus increasing the sensitivity of MCF-7 breast cancer cells to tamoxifen. Estrogen analogs such as tamoxifen and phytoestrogens have been shown to regulate the ERα-induced signaling pathway and IFN-γ production in ER (ER+) cell lines, including MCF-7 and BT-474, as well as ER-deficient (ER-) cells, including SK-BR-3 and MDA-MB-231 (Niu et al., 2015). A recent study by Shaldoum et al. (2021) investigated the protective effects of BP against DOX-induced damage to the immune system, bone marrow, and spleen in rats. The study found that BP, with its anti-inflammatory effects, reduced pro-inflammatory cytokines (IL-6 and IL-1β) and increased anti-inflammatory cytokine (IL-10) levels (Shaldoum et al., 2021). This study, consistent with previous research, demonstrates that BP can suppress inflammatory pathways, leading to a decrease in pro-inflammatory cytokines and an increase in anti-inflammatory cytokines. These effects are likely due to the presence of polyphenolic compounds, especially flavonoids.
As a tumor suppressor protein, p53 plays a vital role in regulating cell death by increasing the expression of a pro-apoptotic gene called Bax, while decreasing the expression of anti-apoptotic genes like Bcl-2. This leads to a higher ratio of Bax/Bcl-2, which in turn triggers the activation of caspase-3, increases the permeability of the mitochondria membrane, and induces the release of cytochrome c, ultimately resulting in cell apoptosis (Katiyar et al., 2005). In an in vitro study on breast cancer cell lines MCF-7 and MDA-MB231, Pilco-Ferreto and Calaf (2016) discovered that DOX, a chemotherapy drug, reduces the expression of pro-apoptotic genes, including p53, Bax, caspase-8, and caspase-3. Moreover, DOX increases apoptosis in these cells by suppressing the expression of the NF-κB gene (Pilco-Ferreto & Calaf, 2016). Polyphenolic compounds, particularly flavonoids, have a dual impact as they help maintain normal cells and promote apoptosis in tumor cells. These compounds can suppress pro-apoptotic factors and increase anti-apoptotic factors in healthy cells under stress. However, in cells with a high growth rate like tumor cells, they can indirectly and directly influence ROS and modify the behavior of these apoptotic factors (Soobrattee et al., 2006). Flavonoids and isoflavones found in BP (presumably a plant or supplement) can hinder the progression of the cell cycle in the G1 or G2/M stages. They elevate the expression of pro-apoptotic genes, inhibit ROS production, alter the activity of enzymes involved in the growth and proliferation of tumor cells, and suppress mediators such as VEGF and FGF, leading to anti-tumor effects (Sánchez-Carranza et al., 2017). A study by Su et al. (2003) demonstrated that tablets containing isoflavones such as genistein, biochanin-A, and daidzein increased the expression of p53, Bax, and caspase-3 while reducing the expression of Bcl-2. Consequently, these tablets effectively inhibited the growth of five human hepatoma cell lines in a dose-dependent manner (Su et al., 2003). Moon et al. (2008) conducted an in vivo study using a human breast cancer cell line (MCF-7) implanted in nude mice. They found that tablets containing isoflavones such as genistein and biochanin-A effectively suppressed tumor growth by upregulating the expression of p53, Bax, and Caspase-3 while downregulating the expression of Bcl-2. The inhibitory effect was observed in a dose-dependent manner (Moon et al., 2008). In a recent study by Shaldoum et al. (2021), it was demonstrated that BP (specifically not specified) can safeguard bone marrow and spleen cells from apoptotic damage caused by DOX chemotherapy. This protective effect was achieved by reducing the expression of pro-apoptotic genes within the mitochondrial cascade, particularly the Bax/caspase-3/p53 pathway (Shaldoum et al., 2021). In this study, BP increased the expression of 53 and caspase-3 (effector caspase) in tumor cells by strengthening the mitochondrial apoptotic pathway by increasing the Bax/Bcl-2 ratio.
Ki-67 is a reliable prognostic indicator for the efficacy of chemotherapy in breast cancer treatment. The WHO index categorizes breast cancer into four stages based on the percentage of Ki-67 positive cells: stage 3 for advanced breast cancer with over 44% Ki-67, stage 2 for cases with 25%–44% Ki-67, stage 1 for cases with 16%–24% Ki-67, and subclinical breast cancer for cases with less than 15% Ki-67. High levels of Ki-67 expression, which reflects cell proliferation, are associated with a poorer prognosis in breast cancer patients (Davey et al., 2021). Another significant clinical measure for determining the stage and prognosis of breast cancer is the p53 index. This index is based on the percentage of positive p53 staining samples, where more than 50% indicates a good prognosis or appropriate treatment, while less than 22% suggests a poor prognosis or inadequate treatment (Bacinschi et al., 2020). Patients with TNBC tumors have a greater sensitivity to chemotherapy using anthracyclines like DOX and exhibit a higher rate of favorable pathological response compared with other breast tumors, as indicated by the immunohistochemical marker Ki-67. In a clinical study by Messina et al. (2022) on breast tumor patients, the use of isoflavones was shown to decrease Ki-67 expression to less than 15 in these patients’ biopsies (Messina et al., 2022). Similarly, a study by Jaskulski (2019) involving postmenopausal women with breast tumors revealed an inverse correlation between genistein dosage and Ki-67 expression. As the dose of genistein increased, the expression of this marker decreased to less than 20%, suggesting a positive prognosis for the disease (Jaskulsk, 2019). Furthermore, this study demonstrated that BP (presumably referring to a treatment or substance) increases the activation of the mitochondrial apoptotic pathway (Bax/Bcl-2/p53 and caspase-3). This activation leads to the modulation of apoptotic markers, including Ki-67, p53, and Bcl-2 (%), in tumor cells. These findings affirm the role of BP in promoting apoptosis in tumor cells.
Conclusion
According to the findings of this study, the use of BP extract as a dietary supplement alongside DOX may be beneficial, particularly for TNBC patients. This extract was found to halt the growth of breast tumors by triggering the production of cytokines that inhibit tumor growth (IFN-γ) while suppressing the production of cytokines that promote breast tumor growth (IL-6, IL-1β, and IL-8). Additionally, it reduced the production of ROS stimulated by DOX metabolites and inhibited estradiol synthesis, resulting in increased apoptosis in 4T1 cells. The co-treatment of 400 mg/kg BP and DOX was found to be the most effective dose in inhibiting tumor growth, although all doses in the study showed antitumor effects. These findings suggest that chemotherapy in combination with BP extract may have potential as an anticancer treatment for TNBC, but further research is necessary to determine its clinical effects in humans, particularly in those undergoing DOX treatment.
Abbreviations
TNBC: Triple negative breast cancer; BP: Bee pollen; DOX: Doxorubicin; ELISA: Enzyme-linked immunosorbent assay; FRAP: Ferric-reducing antioxidant power; TBARS: Thiobarbituric acid reactive substances; IFN-γ: Interferon-γ; ER: Estrogen receptors; PR: Progesterone receptors; HER2: Human epidermal growth factor receptor; ROS: Reactive oxygen species; DNA: Deoxyribonucleic acid; NF-κB: Nuclear factor-kappa B; MAPK: Mitogen-activated protein kinase; NK: Natural killer; TNF-α: Tumor necrosis factor-α; IL-6: Interlekine-6; NSAIDs: Non-steroidal anti-inflammatory; EDTA: Drugs ethylenediaminetetraacetic acid; FBS: Fetal bovine serum; PBS: Phosphate-buffered saline; DW: Distilled water; IP: Intraperitoneal; E2: Estradiol; FSH: Follicle-stimulating; LH: Luteinizing; T: Testosterone; NO: Nitric oxide; SOD: Superoxide dismutase; GPx: Glutathione peroxidase; CAT: Catalase; TPTZ: 2,4,6-tripyridyl-riazine; DTNB: 5,5-dithio-bis-2-nitrobenzoic acid; PCR: Real-time polymerase chain reaction; real-time qPCR: Quantitative real-time polymerase chain reaction; NCBI: National center for biotechnology information; PVDF: Polyvinylidene fluoride; SDS: Sodium dodecyl sulfate polyacrylamide; HRP: Horseradish peroxidase; ANOVA: One-way analysis of variance HPG: Hypothalamic-pituitary-gonad; AP-1: Activator protein 1; ABC: ATP-binding cassette; MDR-1: Multidrug resistance-1; VEGF: Vascular endothelial growth factor; MMPs: Matrix metalloproteinases; MDA: Malondialdehyde.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
This research adhered to ethical guidelines for the use of animal subjects. Animal welfare considerations were taken into account, minimizing pain, distress, and suffering. Ethical approval was obtained from the University of Melbourne for animal research.