Abstract
Matrine possesses a broad spectrum of pharmacological activities as one of the primary components of Chinese herbal medicine Sophora flavescens extract, along with fewer side effects and low toxicity. Matrine possesses anti-inflammatory, anti-cancer, anti-virus, antibiosis, and anti-oxidative properties and protects the central nervous and cardiovascular systems, immunosuppression, intestinal flora balance regulation, and broad-spectrum insecticidal activities. We reviewed the recent progress in matrine research in terms of the pharmacological activities and associated mechanisms to provide a reference for the wide and safe application of matrine.
Introduction
Natural Chinese herbal extracts with a broad range of biological activities and low toxicity are abundant in Chinese herbal medicine. Sophora flavescens, also known as Sophora japonica or Sophora alopecuroides, is a plant belonging to the Leguminosae Sophora family. Matrine is a unique natural alkaloid extracted from S. flavescens with a tetracycline quinidine structure (Duan, 2007). It is inexpensive and has relatively few side effects. Matrine has promising anti-inflammatory, anti-cancer, anti-tumor, heart protection, diuresis detumescence, immunosuppression, and biological regulatory applications (Ji et al., 2018). We have summarized here matrine’s action mechanism behind the pharmacological activities based on the recent studies on the pharmacological effects so as to provide a reference for the clinical application of matrine.
Molecular Structure of Matrine
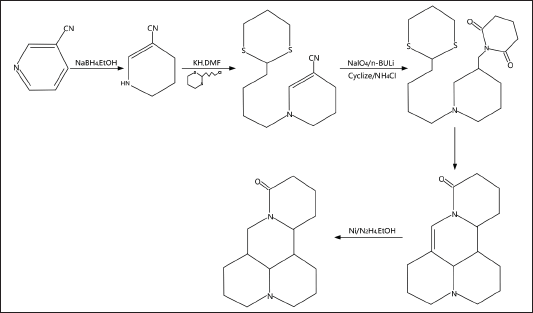
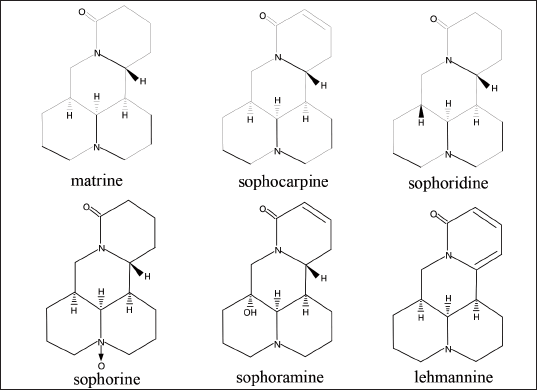
Matrine is an optically active isomer of lupanine, and it occurs in four different forms: α-matrine, β-matrine, γ-matrine, and δ-matrine. α-matrine is the most prevalent of the four forms. Matrine often exists as white needle-like crystals or white crystal-like powder (Fu, 2017). Matrine has a melting point of 76°C, and it is soluble in water, benzene, chloroform, methanol, and ethanol, as well as being slightly soluble in petroleum ether. Matrine has the molecular formula C15H24N2O and a relative molecular mass of 248.36. Figure 1 depicts the classical method of synthesizing matrine (Liu, 2013). Matrine belongs to the quinolizidine derivatives. Matrine’s molecular skeleton is a dense body formed by two quinolizidine rings thickened with each other, including 4 six-membered rings, and the nitrogen atom contains a pair of unshared electron pairs, which has a specific ability to attract protons, making it weakly alkaline. Furthermore, the pH of the matrine-saturated aqueous solution is 8.7, allowing it to react with acid to generate salt. The base weakens as the nitrogen atom is oxidized, and the pH of its saturated aqueous solution rises to 7.8. As a result, some oxidizing agents, such as hydrogen peroxide, can oxidize matrine to oxymatrine. Oxymatrine regenerates matrine by reacting with some weak reducing agents (Fleming et al., 1997). In addition, matrine alkaloids also include sophocarpine, sophoridine, sophorine, sophoramine, and lehmannine. The specific structure is shown in Figure 2.
Pharmacological Activity and Action Mechanism of Matrine
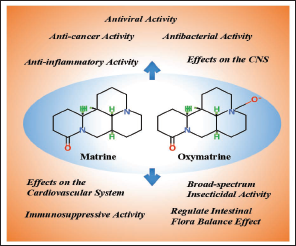
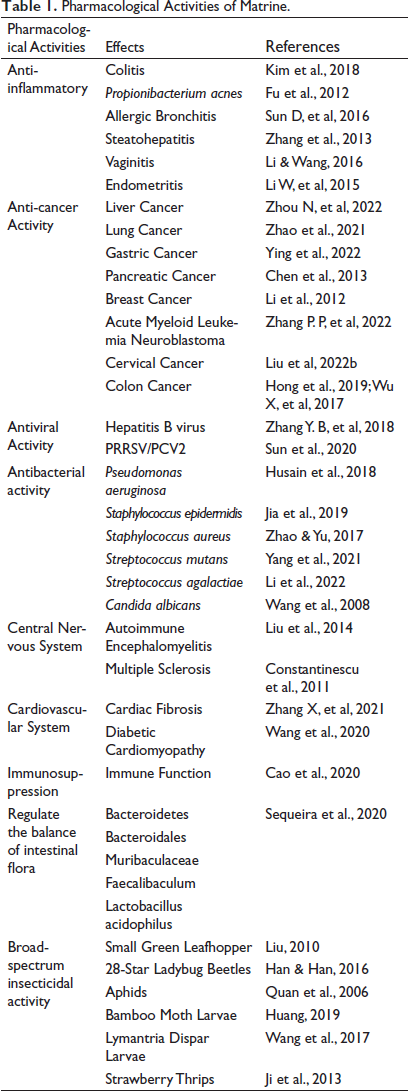
Matrine has important effects in anti-inflammatory, anti-cancer, anti-viral, anti-bacterial, antioxidant, central nervous system (CNS) regulation, cardiovascular system regulation, immunosuppression, regulation of intestinal flora balance, and broad-spectrum insecticidal activity without inducing additional toxic effects or side effects based on recent research (Figure 3). Matrine’s pharmacological effects have become a hot spot for both domestic and international research. As a result, Table 1 summarizes the primary pharmacological activities of matrine.
Pharmacological Activities of Matrine.
Anti-inflammatory Activity
Matrine, a Traditional Chinese Medicine active component, has significant anti-inflammatory effects in inflammatory diseases, such as colitis, allergic bronchitis, steatohepatitis, vaginitis, and endometritis.
Pro-inflammatory cytokines secreted by immune cells during inflammatory infection and pathological injury act as critical mediators and are primarily responsible for the induction of inflammation (Chen et al., 2018). IL-1β and TNF-α are the central pro-inflammatory cytokines secreted by macrophages and epithelial cells (Jiang et al., 2018a; Jiang et al., 2018b). The expression of IL-1β, TNF-α, and IL-6 was reportedly increased in the dextran sodium sulfate (DSS)-induced colonic inflammation model. Matrine was discovered to improve the intestinal barrier function and protect the intestinal barrier from DSS damage by significantly downregulating IL-1β, TNF-α, and IL-6 expression (Yao et al., 2021). Matrine could reduce histopathological lung damage, lung index, the content of IL-6, IL-10, and TNF-α inflammatory factors, and the viral load in mice lung tissues and increase the percentage of peripheral blood lymphocytes in an allergic bronchitis experimental model, thereby improving the mouse immunity (Liou et al., 2016). These findings suggest that the matrine can exert anti-inflammatory properties by inhibiting the expression of pro-inflammatory cytokines in a dose-dependent manner.
In an experimental model of liver steatosis transformed into non-alcoholic steatohepatitis in rats, matrine was reported to exert a protective effect against non-alcoholic steatohepatitis caused by a high-fructose diet. Matrine reduced lipid droplets, inflammatory cell infiltration, and intrahepatic necrosis foci in hepatocytes, as well as alanine aminotransferase and aspartate aminotransferase levels in blood and liver triglyceride. In addition, low to moderate dosages of matrine decreased lipid accumulation, oxidative stress, and cytosolic calcium levels (Ca2+) in hepatocyte lineages. Therefore, matrine can inhibit lipid peroxidation and improve the antioxidant capacity in rats with chronic alcoholic liver injury (Gao et al., 2018).
Currently, matrine has been reported to exert anti-inflammatory and antioxidant functions in a variety of experimental models. Matrine at high dosages has been demonstrated to be effective in treating bacterial vaginitis (Patricio et al., 2019; Yang et al., 2022). When Pseudomonas aeruginosa infects an organism, it causes vaginal environment disruption and increases the level of IL-1β expression in the serum, which in turn results in an inflammatory response (Willems et al., 2018). Matrine’s capacity to minimize intravaginal osteopathy, alleviate vaginal tissue inflammation, and restore sIgA and IgG antibody levels was primarily responsible for its defense against Pseudomonas-induced vaginitis. The vaginal epithelium secretes IgG, which is essential for Pseudomonas infection (Niu et al., 2016), and sIgA is one of the principal antibodies against Pseudomonas infection in vaginectomy (Wang et al., 2018).
Endometritis caused by Staphylococcus aureus (S. aureus) is one of the most prevalent illnesses of dairy cows’ sexual organs, resulting in high financial costs for the dairy sector. Endometritis is a common mucosal illness caused by several pathogenic microbial infections during mating or childbirth (Jiang K., et al.). As long-term antibiotic therapy for endometritis leads to the formation of drug-resistant strains (Jiang et al., 2017), establishing new treatment methods for endometritis in dairy cows is an intriguing research topic. Endometritis can be exacerbated by IL-1β and TNF-α expressions (Wu et al., 2018). Consequently, decreasing IL-1β and TNF-α production has become a strategic goal for treating endometritis. The experimental model of endometritis was induced by lysophosphatidic acid (LTA), and the Toll-like receptor 2 (TLR2) of LTA was demonstrated to activate the extracellular NF-κB signaling pathway, resulting in the overexpression of IL-1β and TNF-α. Matrine treatment hindered TLR2-mediated stimulation of the NF-κB signaling pathway and dramatically decreased the IL-1β and TNF-α expression levels. Therefore, matrine exerts crucial anti-inflammatory activities in endometritis induced by S. aureus LTA (Jiang et al., 2019).
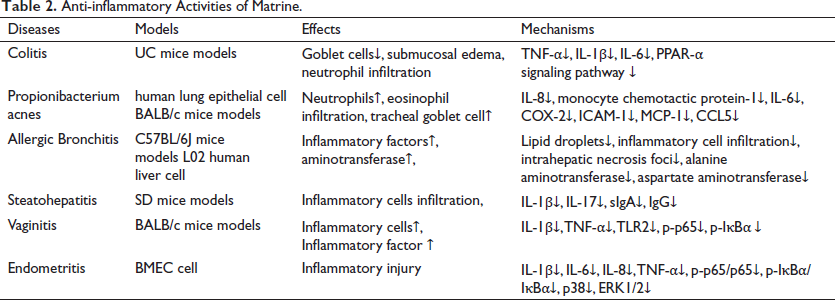
NF-κB is a typical inflammatory signaling pathway mediated by PI3K/AKT. Once the PI3K/AKT signaling pathway becomes active, NF-κB produces large amounts of inflammatory factors, thereby exacerbating an inflammatory response. NF-κB frequently forms inactive NF-κB/IκB complexes in natural biological settings by connecting to the inhibitory protein IκBα. The inflammatory stimuli induce the phosphorylation of NF-κB p65 with IκBα, resulting in the dissociation of IκBα from NF-κB, nuclear translocation of p65, and stimulation of NF-κB target genes for inflammation (Xiang et al., 2015). Matrine protects bovine mammary epithelial cells from S. aureus-induced inflammation by inhibiting the NF-κB signaling pathway activation, thereby reducing S. aureus-induced apoptosis (Zhang X. J, et al., 2020), downregulating pro-inflammatory cytokine expression, and inhibiting the phosphorylation of key proteins in the NF-κB and MAPK pathways during the pathogenesis of mastitis. In addition, matrine attenuates lipopolysaccharide (LPS)-induced inflammatory responses by downregulating IL-1 and IL-17 pro-inflammatory factor expression and inhibiting malondialdehyde (MDA) pro-inflammatory compound production (Sun et al., 2018; Wu et al., 2017). These findings suggest that matrine may be a beneficial anti-inflammatory drug for the treatment of inflammatory illnesses. The anti-inflammatory mechanism of matrine is summarized in Table 2.
Anti-inflammatory Activities of Matrine.
Anti-cancer Activity
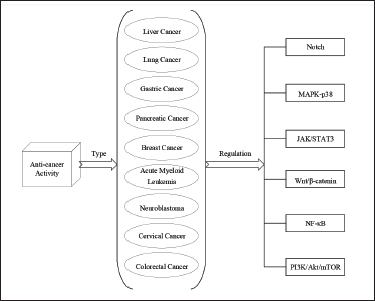
Past studies have shown that basic cellular processes are disrupted in cancer development and tumor progression (Pavlova et al., 2022). Cancer cells’ irregular development and metastasis are crucial cancer biological characteristics, and invasion is the most significant and defining phase in the complicated metastatic process. Matrine’s superior antitumor properties are highlighted by its ability to accelerate apoptosis, restrict tumor cell growth and proliferation, stimulate cell cycle arrest, hinder tumor progression and invasion, inhibit neovascularization, stimulate autophagy, reverse multidrug resistance, and inhibit differentiation. The anti-cancer activity of matrine is shown in Figure 4.
The Anti-cancer Activity of Matrine.
Liver Cancer
Increasing numbers of studies have demonstrated the efficacy of matrine in the treatment of liver fibrosis and anti-hepatocellular carcinoma (HCC) (Feng et al., 2016; Yin et al., 2018), which effectively decreases the growth of HCC cells. The primary modality of matrine in treating HCC inhibits HCC cell proliferation by regulating creatine levels and choline content. Creatine is among the power sources utilized by tumor cells during growth (Kazak & Cohen, 2020), and choline can enhance fatty acid usage to avoid the aberrant accumulation of liver fat. Furthermore, matrine can effectively boost intracellular choline levels and ameliorate lipid metabolism issues in HCC (Wang et al., 2022).
Matrine may inhibit tumor cell proliferation indirectly by regulating multiple signaling pathways. The Notch signaling pathway, for example, is critical in regulating the development of various organs, mainly by regulating cell proliferation, differentiation, and apoptosis (Dayekh & Mequanint, 2020; Luo et al., 2021; Zeng et al., 2021). Moreover, matrine has been linked to tumor stem cell formation, epithelial–mesenchymal transition (EMT), and tumor resistance to chemotherapeutic agents (Weber et al., 2019). Matrine alleviates diabetes-induced coronary atherosclerosis and liver injury by inhibiting the Notch signaling pathway activation (Liu et al., 2018). In addition, matrine could downregulate the ERK1/2 signaling pathway and reduce HCC cell growth, invasion, and migration. When extracellular signals are received, one of the MAPK sub-pathways, ERK1/2, binds to the corresponding protein receptors on the cell membrane. As a result, the phosphorylation of ERK1/2 pathway-associated proteins is increased. To complete the process of extracellular signal transmission to the intracellular or nucleus, phosphorylated proteins are activated by extracellular signals and chain reactions with intracellular protein kinases. This event regulates the biological behaviors of cell growth and development, division and differentiation, proliferation, and apoptosis (Yu et al., 2020).
Lung Cancer
Lung cancer accounts for approximately 21% of all cancers, with non-small cell lung cancer (NSCLC) comprising approximately 80%. Matrine is recognized as an effective treatment for NSCLC, either alone or in combination with chemotherapeutic drugs. According to research, matrine may diminish the transcriptional activity of lung cancer stem cells (CSC) by primarily upregulating Let-7b and blocking the activation of the Wnt signaling pathway (Li X, et al., 2020).
Bcl-2 is a crucial protein in the regulation of apoptosis, as it may maintain a cell’s normal state and prevent cell apoptosis through various signaling pathways (Czabotar et al., 2014). In contrast, Bax, a major Bcl-2 inhibitor, is a protein that accelerates apoptosis in tumor cells. The overexpression of Bcl-2 inhibits apoptosis and enhances cell survival, while Bax overexpression accelerates cell death (Lin & Tongyo, 2014). Matrine stimulates apoptosis in cancer cells by activating the p38/JNK signaling pathway, regulating Bcl-2, Bax, and Caspase-3 protein expressions, and reducing the protein proportion of Bcl-2/Bax. Matrine may inhibit the proliferation of lung and liver cancer cells through this mechanism (Zhang et al., 2009). The PI3K/AKT signaling pathway enhances cancer cell motility and invasion in several lung cancer disorders by regulating the increase in vascular endothelial growth factor (VEGF-A) expression. Nevertheless, matrine decreased the VEGF-A release as well as lung cancer cell proliferation and migration (Chen et al., 2009).
Gastric Cancer
Gastrointestinal (GI) cancers remain the second-most prevalent type of cancer. Consumption of food products and infections by Helicobacter pylori (H. pylori) are the primary risk factors for enteric and diffuse gastric cancers. Matrine has the potential to develop novel natural anti-gastric cancer drugs as it interferes with gastric cancer cell cycle changes and inhibits cancer cell proliferation (Liu Z. M, et al., 2020). Matrine can also regulate the phosphorylation of key signaling molecules in the NF-κB pathway, inhibit proliferation, and promote apoptosis. NF-κB is a pleiotropic transcriptional factor that regulates multiple biological processes, including inflammation, apoptosis, and tumorigenesis (Didonato et al., 2012). It can inhibit the expression of several proteins, including the NF-κB subunit, CIAP, XIAP, and p-ERK, all of which have significant effects on gastric cancer cell proliferation (Luo et al., 2012). Matrine also inhibits human prostate cancer cell proliferation, migration, and invasion by interfering with the activation of the NF-κB signaling pathway (Li et al., 2016).
Pancreatic Cancer
Pancreatic cancer is among the most prevalent forms of cancer, with pancreatic ductal carcinoma (PDAC) being highly aggressive and often difficult to detect until cancer has spread beyond the pancreas. The surgical cure for PDAC is less efficient, and most patients with advanced pancreatic cancer are limited to these options, such as cytotoxic chemotherapy, targeted drugs, and radiotherapy. A deeper comprehension of adaptable metabolic alterations in cancer cells is required to successfully target cancer metabolism. Autophagy, whether basal or induced, is essential for the survival of certain malignancies. Thus, autophagy stimulates tumor growth (White et al., 2015). In contrast, normal cells rely less on autophagy-mediated metabolism than cancer cells. Matrine inhibits apoptosis by impairing lysosomal protease function and disrupting the autophagy-mediated mitochondrial metabolic process, thereby substantially decreasing the pancreatic cancer cell growth and proliferation, blocking the cell cycle, and inducing apoptosis. Matrine is currently considered a novel therapeutic agent for various cancer cell-dependent metabolisms (Cho et al., 2018).
Breast Cancer
As breast cancer is the most prevalent malignant tumor, most patients experience severe toxin accumulation and tolerance disorders after receiving chemotherapy with anthracyclines and paclitaxel drugs. The search for innovative natural chemicals with nontoxicity and excellent selectivity for the destruction of breast cancer cells is now a crucial aspect of cancer research. Matrine has been acknowledged to have considerable therapeutic effects in the practical management of breast cancer, as it decreases both primary and advanced breast cancer growth. Matrine inhibits breast carcinogenesis by reducing the AKT phosphorylation levels in breast cancer cells, upregulating the Bax protein expression, and downregulating the apoptotic factor Bcl-2 protein expression in MCF-7 cells (Li et al., 2015). Matrine has the potential to suppress the growth of breast cancer MCF-7 cells and trigger apoptosis in cancer cells, thereby reversing multidrug resistance in breast cancer cells (Zhou et al., 2018).
Matrine effectively inhibits breast cancer cell growth and migration by decreasing the expression of the Wnt/β-linked protein signaling pathway (Xiao et al., 2018). VEGF is an important target downstream of the Wnt / β-catenin signaling pathway. Matrine affects the mRNA expression levels of Bcl-2 / Bax / VEGF / p-AKT / NF-κB protein and gene. Matrine can also block the activation of MMP-9 / MMP-2 in AKT signaling, thereby reducing the proliferation and invasion of breast cancer cells. (Yu et al., 2009). As a result, matrine has comprehensive therapeutic and adjuvant therapy to treat breast cancer.
Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is caused by the aberrant proliferation and differentiation of immature myeloid cells in the bone marrow and peripheral blood. It is characterized mostly by an abnormal increase of immature leukocytes (Pelcovits & Niroula, 2020). Matrine, through multiple mechanisms, inhibits AML cell proliferation, alters the tumor cell cycle, and inhibits telomerase activity (Zhou et al., 2014).
Moreover, lncRNAs and miRNAs exert a role in the development and progression of several cancers (Li Y, et al., 2020). Most cancer cells are regulated at the transcriptional and post-transcriptional levels by lncRNAs and miRNAs (Tsagakis et al., 2020). LINC01116 acts as a pro-tumor development factor in lncRNA, and matrine exerts anti-cancer effects by reducing LINC01116 in AML cells. In addition, LINC01116 stimulates osteosarcoma cell growth and migration via the JAK/STAT3 pathway, which profoundly affects cell proliferation and apoptosis by modulating the activation of several downstream effector molecules (Johnson et al., 2018). Matrine inhibits the JAK/STAT3 pathway in AML cells by modulating LINC01116 and miR-592 expression. Matrine induces apoptosis in AML cells by drastically decreasing IL-1β, IL-6, and TNF-α expression and blocking the PI3K/AKT/mTOR pathway. Apoptosis in cancer cells is associated with a decrease in the expression of Bcl-2 protein—a crucial intracellular anti-apoptotic molecule (Hao et al., 2019). These findings imply that the matrine may decrease AML activity by inducing AML cell apoptosis, lowering AML cell proliferation, and reducing the expression of proinflammatory cytokines. Therefore, matrine was considered a drug candidate for treating AML.
Neuroblastoma
Neuroblastoma (NB) originates from the paravertebral sympathetic ganglia or adrenal medulla. There have been a few findings regarding how matrine affects NB cell migration and proliferation. Matrine’s anti-cancer activity against NB may be attributed to the induction of apoptosis in NB cells by inhibiting the PI3K/AKT/mTOR pathway and the reduction of NB cell growth. The PI3K/AKT/mTOR signaling pathway regulates physiological activities such as cell growth, cell cycle progression, and apoptosis. It has an indispensable influence on cancer development and anti-cancer drug resistance. The AKT/mTOR pathway participates in numerous physiological activities involving metabolic, cell growth, proliferating, differentiating, apoptosis, and autophagy (Cho, 2011).
Triptolide 3 (TRB3) is a pseudokinase that binds to the AKT activity and inhibits its activity. TRB3 acts as an inhibitor of AKT activation, and past studies indicate that TRB3 affects insulin signaling by interacting with AKT (Zhang et al., 2011). Matrine exhibited anti-tumor effects in the body without significant side effects by upregulating the TRB3 expression in NB cells and blocking the TRB3-mediated PI3K/AKT pathway to inhibit NB cell migration and proliferation (Shen et al., 2018). Matrine has an essential impact as an autophagy inducer in NB cell anti-tumor therapy and might be explored as an NB treatment.
Cervical Cancer
Matrine inhibits cervical cancer cell proliferation by blocking the AKT/mTOR signaling pathway to promote autophagy (Zhang et al., 2022), and it exerts anti-cancer activities by inhibiting the P38 mitogen-activated protein kinases (MAPKs) signaling pathway. MAPKs are indispensable components of intracellular signaling networks that regulate cell growth, diversification, cellular stress reaction, apoptosis, and tumor formation. The p38 signaling pathway is essential in cervical cancer invasion and progression (Wang et al., 2015). Matrine inhibits HeLa cervical cancer cell adherence and migration by downregulating P38 and reducing the activities of cAMP-dependent protein kinase (PKA) and vasodilator-stimulated phosphoprotein (VASP). Inhibition of the cell cycle inhibits the proliferation of cervical and ovarian cancer cells by downregulating the HIF-1α expression and blocking the MAPK/ERK and P38MAPK signaling pathways (Zhang et al., 2019; Zou et al., 2019). These findings demonstrate that matrine might be used to treat the progress and metastasis of cervical cancer. Matrine, in conjunction with autophagy inducers, may offer viable treatment options for cervical cancer.
Colorectal Cancer
Colorectal Cancer (CRC) is the world’s third-most prevalent cancer. It has become imperative to explore the molecular mechanisms of herbal medicine as an alternative therapy for treating and preventing CRC. Matrine is a novel approach to treating CRC, and its mode of action is to decrease CRC cell proliferation and invasion by downregulating p38 and microRNA-22 expression (Liu J, et al., 2020; Ren et al., 2014). Matrine, an ER membrane-associated protein with an N-terminal transmembrane structural domain, may inhibit the G1 phase of the cell cycle and cause apoptosis in CRC cells by downregulating the Erlin1 overexpression (Ren et al., 2022). High concentrations of matrine have a significant toxic effect on CRC cells. As a result, matrine, a conventional medicine, may act as a targeted drug for CRC.
Hypoxia-inducible factor-1 (HIF-1) is a heterodimeric protein that comprises two subunits HIF-1α and HIF-1β. The HIF-1α expression was upregulated in cancer cells, probably due to hypoxia-induced or activated oncogene expression (Masoud & Li, 2015). HIF-1α, a key transcription factor that restricts glucose uptake and produces the lactic acid effect (Warburg effect), increases the production and activation that enhances the suppression of the onset of the Warburg effect during cancer development. Therefore, targeting HIF-1α has emerged as an anti-cancer treatment strategy to counteract the Warburg effect.
Matrine’s anti-cancer mechanism against CRC is primarily manifested in its ability to substantially reduce the mRNA and protein expression levels of HIF-1α, inhibit HIF-1α regulation of GLUT1, HK2, and LDHA, the downstream targets of glucose metabolism, and finally decrease the uptake of glucose and lactate production, thereby preventing the growth of colorectal cancerous cells. The key enzymes in cancer cells’ glycolytic energy metabolism are GLUT1, HK2, and LDHA (Hong et al., 2019). Matrine might also inhibit the HIF-1α mRNA expression and protein synthesis by regulating the PI3K/AKT/mTOR pathway, thereby reversing the Warburg effect (Rius et al., 2008). With the HIF-1α-mediated Warburg effect, matrine demonstrates a remarkable therapeutic benefit on CRC in general.
Anti-viral Activity
Antiviral natural herbal compound screening is currently a prominent research area both at home and abroad. According to a recent study, numerous Chinese herbal medicine formulations or single components exhibited antiviral activities in fundamental or clinical application investigations. Matrine, the principal active component of the Chinese herbal remedy radix sophora flavescent, has a wide range of applications in the antiviral treatment of the hepatitis B virus (HBV). Matrine is an excellent clinical drug for enhancing autoimmunity and as adjuvant therapy for HBV chronic infection. It inhibits HBV DNA replication by decreasing the activity of the PKC protein kinase family, downregulating the MAPK-ATF2/CREB signaling pathway, and upregulating the expression of the inflammatory chemokine CXCL8, thereby acting as a potential therapeutic agent for HBV chronic infection (Zhou et al., 2022).
Furthermore, matrine possesses indispensable advantages in PRRSV/PCV2 blue ear disease (PRRSV) with porcine circovirus type 2 (PCV2) antiviral infections. Vaccination is the most effective approach to prevent PRRSV/PCV2 illness. Immunization is less effective in treating combined PRRSV/PCV2 infections due to the high mutation rate of PRRSV/PCV2 viruses and the development of complex mixed infection states. The vaccination does not directly eradicate PRRSV/PCV2 infection because of the limitations of the vaccine’s protection period against the disease (Feng et al., 2014). By using an experimental model of PRRSV/PCV2 viral co-infection in mice, the researchers determined that both PRRSV and PCV2 were transferred during infection and that mixed virus infection increased the spleen and thymus indices while also activating the body’s immune response. Moreover, matrine was reported to inhibit the replication of PCV2 in the liver of PRRSV/PCV2 co-infected mice. Furthermore, 40 mg/kg of matrine enhanced the phagocytosis of macrophages by modulating the immune function of mice, thereby reducing the virus-induced lesions (Sun et al., 2020).
It was reported that PCV2 infection increased the mRNA expression levels of NF-κB upstream regulators TLR2, TLR3, TLR4, and TLR9 in piglet spleen lymphocytes. PRRSV infection raises the mRNA expression of the NF-κB upstream regulators TLR3, TLR4, and TLR7 in tracheobronchial lymph glands and the brain. TLR3 and TLR4 mRNA expressions were enhanced after PRRSV/PCV2 co-infection. However, matrine could significantly reduce the expressions of TLR3 and TLR4 induced by PRRSV and PCV2 viruses (Sun et al., 2016). Therefore, it is believed that matrine might be a future antiviral agent against mixed PRRSV/PCV2 infections. The antiviral mechanism of matrine is summarized in Table 3.
Antiviral Activities of Matrine.

Antibacterial Activity
In a rising number of investigations, small molecules and components of Chinese herbal medicine have been identified as potential biofilm formation inhibitors in the Quorum-sensing (QS) system of drug-resistant bacteria. For example, Coptidis Rhizome and Psoralen (seed) extract inhibited the QS system of P. aeruginosa (Husain et al., 2018). Matrine demonstrated superior antibacterial activity against Staphylococcus epidermidis, inhibiting biofilm formation by reducing AI-2 activity, and targeting the QS system (Pourahmad Jaktaji & Mohammadi, 2018). Thus, matrine prevents S. epidermidis biofilm-forming infections (Jia et al., 2019). Research has revealed that matrine inhibited Streptococcus pyogenes biofilm formation in a concentration-dependent manner.
Streptococcus mutans is the main pathogenic bacteria causing dental caries. Its pathogenic mechanism is primarily to establish a biofilm on the surface of teeth to create an anaerobic and acidic environment, thereby accelerating the formation of dental caries. The mature biofilms are highly viable. It is difficult to remove the biofilm after it has developed and adhered to the surface of an object. The effect of matrine was examined on an S. mutans biofilm model using a 24-hour mature S. mutans biofilm model. The observations suggested that matrine could inhibit the growth of S. mutans, antagonize its organic acid production, disperse the pre-formed biofilm, and reduce its cellular metabolic activity (Yang et al., 2021). As a result, matrine was believed to have the potential for development as a novel anti-caries drug.
Staphylococcus aureus is a significant cause of mastitis in dairy cows that acts mainly by invading host cells to trigger an inflammatory response in the cow’s mammary gland, which in turn initiates a series of programmed cell deaths such as apoptosis and necrosis (Zhang et al., 2018). The minimum inhibitory concentrations of matrine against Escherichia coli and S. aureus were determined to be 12.5 and 25 mg/mL using the paper diffusion method and the Oxford cup method, respectively (Feng et al., 2018). Matrine attenuates S. aureus-induced bovine mammary epithelial cell (BMEC) damage by inhibiting S. aureus alpha-hemolysin secretion and downregulating the antimicrobial peptide gene expression in BMECs (Zhang X. J, et al., 2020). Therefore, matrine is a prospective medication for preventing and treating mastitis caused by S. aureus.
Matrine is potentially a possible treatment option for acne. All synthetic compounds included in matrine have demonstrated better activities against Propionibacterium acnes and Streptococcus albicans. In a previous study, the mechanism of action involved the introduction of hydrophilic groups that slightly enhanced the matrine’s anti-bacterial activity. As a result, the matrine’s antibacterial activity was more potent than that of the positive control drugs doxycycline hydrochloride and fluconazole compounds (Alkhawaja et al., 2020).
Matrine has also shown antifungal activity in vitro experiments, specifically by inhibiting the formation of the fungal biofilm. In clinical infections, for example, Saccharomyces albicans is the primary pathogenic fungus. Matrine’s antibacterial activity mechanism against S. albicans is mainly a strong inhibitory effect on biofilm formation. Matrine inhibits the growth of Aspergillus fumigatus, Trichoderma mentagrophytes, and Cryptococcus neoformans. Anaphylactic responses are caused by A. fumigatus in immunocompetent organism groups. T. mentagrophytes cause superficial infections such as capitis, tinea corporis, tinea manus, and pedis, as well as deep infections such as psoriasis and granuloma. C. neoformans is a well-known species of seriously infective fungi, and matrine has demonstrated an excellent inhibitory effect on infections caused by these bacteria (Zhang J, et al., 2020). These findings imply that matrine may be utilized as an antifungal medication enhancer.
Effects on the CNS
Autoimmune encephalomyelitis (EAE) is a chronic inflammatory demyelinating disease of the CNS. Matrine improves EAE neurological function, reduces CNS inflammatory infiltration and demyelination, inhibits inflammatory cytokine or chemokine production, and promotes the secretion of IL-4 and IL-10 (Liu et al., 2014). IL-10 is an essential anti-inflammatory cytokine that inhibits T-cell proliferation and Th1/Th17 cell differentiation by reducing the antigen-presenting ability of cells. The primary mechanism by which matrine exerts its inhibitory effect in neuropathic pain and autoimmune diseases is the high expression of IL-10 anti-inflammatory factor (Chu et al., 2020). Increased IL-10 anti-inflammatory factor expression within the CNS EAE is essential for its recovery, and IL-10 could significantly enhance neural regeneration by transfecting neural stem cells (Yang et al., 2009). Furthermore, multiple sclerosis (MS) is an inflammatory demyelinating disease of the CNS that causes axonal and neuronal degeneration. Myelin-specific T-cells were reported to be activated and enter the CNS across the blood–brain barrier in an experimental animal model for MS and EAE, which promoted the apoptosis of oligodendrocytes (Ols). Therefore, when matrine was administered, it not only suppressed CNS inflammation but also protected Ols by regulating the PI3K/AKT and Wnt3a/β-catenin/TCF7L2 signaling pathways, thereby promoting Ols maturation and repairing myelin (Liu S. Q, et al., 2017; Ma et al., 2022).
Effects on the Cardiovascular System
Oxidative stress is a crucial pathological process that contributes to cerebral ischemia injury. Matrine can further reduce focal ischemia-reperfusion injury in mice by enhancing antioxidant activity and reducing apoptosis. Matrine can significantly enhance the serum MDA levels and promote the body’s free-radical scavenging balance, inhibiting the aggregation of free radicals and lipid peroxides in focal cerebral ischemic injury by enhancing the activities of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-PX), and total antioxidant capacity (T-AOC) (Zhao et al., 2015), ultimately exerting a protective function for the brain.
Matrine is also used to treat arrhythmias and cardiac fibrosis, as well as to improve cardiac function, where cardiac fibrosis is a common feature in the development of various cardiovascular diseases. Matrine is a targeted drug for treating pathological cardiac fibrosis and cardiac insufficiency in mice. It inhibits cardiac fibrosis in mice by reducing p38 phosphorylation and decreasing the α-SMA protein levels and mRNA expression levels of Col1 and Col3 in cardiac fibroblasts. Matrine can also reduce the incidence of cardiac fibrosis in diabetic cardiomyopathy (DCM) by modulating the ATF6-signaling pathway (Liu Z, et al., 2017), and matrine may show when rat hearts are subjected to advanced glycosylation end products by modulating Ryanodine receptor two activity (Wang J. T, et al., 2019). Matrine administration not only protected the mouse hearts from adriamycin-induced cardiac insufficiency but also alleviated oxidative stress and inhibited mouse cardiac fibroblast proliferation, migration, and differentiation in vitro (Hu et al., 2019).
DCM in rats is frequently accompanied by increased apoptosis and reactive oxygen species (ROS) generation in the heart tissues. Excessive ROS production activates both the TLR4/MYD-88 and the Caspase-8/Caspase-3 signaling pathways. Matrine enhances cardiac functions in DCM rats by lowering the non-fasting glucose levels, inhibiting TLR4/MYD-88/Caspase-8/Caspase-3 signaling, and preventing cardiomyocyte apoptosis and ROS generation (Liu et al., 2015). When analyzing the effects of matrine and its derivative oxymatrine on left ventricular contractility in rats, it was found that both herbs increased left ventricular contractility and improved cardiac mechanical functions. However, matrine’s dose was significantly lower than that of oxymatrine. The effect of matrine on myocardial contractility was found to be more substantial than that of oxymatrine (Dai et al., 2022). In addition, matrine enhanced the viability of cardiac myoblasts after an inflammatory response induced by LPS. These results prove that matrine is vital in cardioprotection (Liu et al., 2021).
Immunosuppressive Activity
In recent years, the development of some immunologically active substances in Chinese herbal medicine has become a hot research topic. Chinese herbal medicines can improve the immunity of an organism, and hence the rational development and utilization of Chinese herbal medicine have multiple economic and social values (Su et al., 2016). Matrine might be used to treat autoimmune diseases in clinical practice as an immunosuppressant, and its immune regulatory role is more complex (Wang, R. H. 2019). Matrine has been proven in studies to regulate the mice’s immune system bidirectionally. It can significantly reduce the immune function of normal T-lymphocytes while improving the immunological function of T-lymphocytes in immunocompromised individuals. T lymphocytes’ functional status can broadly reflect the resistant level of body cells. Matrine may therefore dramatically increase the conversion rate of mouse thymic cells as an immune enhancer and improve mice’s cellular immune function by producing an immunodeficiency mouse model (Wynn & Vannella, 2016).
Matrine directly inhibited the serum lysozyme activity, antibody-forming cell production, and serum hemolysin production, possibly through enhanced host antitumor immune response (Liu et al., 2010). The immunological response of immune effector cytotoxic T cells (CTLs) is the body’s primary defense mechanism against tumors, and its effective activation could significantly improve tumor prognosis. When the mice were administered matrine aqueous decoction, the researchers observed changes in the systemic immune function. In mice, matrine was discovered to block the immunoreactive actions of T-cells, B-cells, and abdominal macrophages. Matrine has been demonstrated to significantly increase T-cell subsets (i.e., CD3, CD4, CD8, and CD4/CD8) and serum immunoglobulin (i.e., IgG, IgA, and IgM) levels in peripheral blood, improving the immune function and therapeutic efficacy (Zhang & Shen, 2019).
Regulate Intestinal Flora Balance Effect
The interaction between the intestinal microbiota and herbal compounds is well-known to be complex. Herbal blends can promote, inhibit, eliminate, or rebuild the intestinal microbiota directly or indirectly. Chinese herbs and herbal compounds can act as prebiotics, thereby promoting the growth of intestinal microbiota and changing the pH, transit duration, and productivity of microfiber in the GI environment, which affects the composition of the intestinal gut microbiota indirectly (Li Y, et al., 2018). Matrine involves structural changes in the intestinal flora of mice, which may enhance the abundance of some dominant flora such as Bacteroidetes, Bacteroidales, Muribaculaceae, Faecalibaculum, and Lactobacillus acidophilus. Among them, Bacteroidetes prevented the colonization of pathogenic bacteria in the gut and increased the host’s resilience to viruses (Sequeira et al., 2020; Stefan et al., 2020). Bacteroidales protected the intestinal barrier, which served as the basis of their defensive position (Kuhn et al., 2018). Faecalibaculum exerts antitumor effects in a CRC mouse model (Zagato et al., 2020). The matrine may boost the ability of the dominant L. acidophilus in the mice’s intestinal flora to colonize various intestinal segments and subsequently exert pharmacological effects such as antiviral, anti-pneumonia, antioxidant, and anti-tumor activities (Cao et al., 2022). In this previous study, researchers evaluated the effects of matrine and amoxicillin on the intestinal microbial community of mice. Amoxicillin administration not only altered the number and diversity of the intestinal microbial community but also increased the proportion of pathogenic bacteria relative to probiotic bacteria.
In contrast, matrine significantly enhanced the abundance of several gut probiotics, such as Clostridium rumen, Lachnospiraceae, and Ruminococcaceae. Among them, C. rumen produces short-chain fatty acids, which are crucial in maintaining intestinal epithelial cells’ function and morphology as well as in regulating the intestinal flora’s balance (Tan et al., 2014). The core intestinal bacterium Lachnospiraceae is a potential probiotic that produces short-chain fatty acids and is negatively associated with intestinal inflammation (Zhao et al., 2017). Ruminococcaceae resides inside the colon and cecum and degrades cellulose and starch, as well as actively regulates the balance of the intestinal environment (Kong et al., 2016). Therefore, matrine possibly enhances the anti-inflammatory capabilities of the digestive tract by boosting the number of probiotic bacteria, which is beneficial for preserving the intestinal microbiota’s stability and gut health (Wu et al., 2021). Conversely, gut microbiota can modulate the metabolic activity of the Chinese herbal medicine matrine (Feng et al., 2019), and matrine has been shown to significantly reduce the expression of PPAR-α—a critical immunological and inflammatory response regulator—by indirectly regulating the gut microbiota (Tan et al., 2021).
Broad-Spectrum Insecticidal Activity
Matrine, a commonly used plant-derived pesticide, possesses insecticidal, bacteriostatic, and acaricidal activities (Zanardi et al., 2015). Its mechanism paralyzes neural activity by directly acting on the central and peripheral nervous systems (Liu et al., 2008). Matrine’s biological activity improves when it is modified by introducing active groups or synthesizing matrine derivatives (Huang & Xu, 2016). The introduction of dimethyl carbonate condensation ester to matrine at the C14 position, reduction to produce alcohols, and decarboxylation by lithium aluminum hydride yielded matrine derivatives with significantly higher antibacterial activity against Bacopa mosaic virus and plants compared with matrine (Ni et al., 2017). In addition, matrine is easily acidolysis, particularly when 1,3,4-thiadiazole, 4-methyl benzyl, and 2-chlorobenzyl reactive groups are present. Their acidic derivatives include insecticidal solids and have acaricidal activities (Lv et al., 2018; Zhang et al., 2018). After modification with coumarin and piperazine moieties, matrine acidic products can treat cancer and tumors (Li Z, et al., 2018; Wu et al., 2016).
Several studies have demonstrated that the complexes formed by matrine and other substances have efficient and prominent insecticidal properties. The combination of matrine and neem, for example, was found to be >86% effective against leafhoppers. The mixture of matrine with mineral oil showed a 76% insect-control effect against small green leafhoppers. Therefore, combining matrine with neem or mineral oil has a strong control effect against small green leafhoppers. Matrine significantly reduced the power of 28-star ladybug beetles. The lethality of 0.04 mg/mL matrine extract to 28-star ladybug beetles was approximately 90%.
Moreover, the compound prepared with a specific concentration of matrine and zymosan showed a good control effect on aphids. After 48 hours, the corrected mortality rate of aphids was as high as 93.5%. The emulsified oil complex synthesized by matrine and nicotine destroyed bamboo moth larvae, with a lethality rate of approximately 90%. The insecticidal activity of three plant secondary metabolites—matrine, oxymatrine, and kaempferol—on the gypsy moth, was investigated to reveal that all three substances had significant insecticidal effects on gypsy moth larvae. Of these, matrine had the most substantial insecticidal effect, with an LC50 value of 0.420 mg/mL. In addition, the spinosa and matrine showed high toxicity to strawberry thrips. The LC50 value was 0.0556 mg/mL, and 3% matrine suspension showed an excellent rapid and sustained effect on strawberry thrips.
Conclusion
Matrine is an alkaloid derived from the Chinese traditional plant S. flavescens with anti-bacterial, anti-viral, antioxidant, anti-inflammatory, anti-tumor, immunomodulatory, and cardioprotective effects. Matrine, a biotin compound, exhibited pharmacological properties against various diseases. Therefore, it is typically employed in clinical treatment as an immunosuppressive medicine for viral infections, autoimmune illnesses, and malignancies; however, it can also serve as an efficient anti-inflammatory or anti-cancer agent in the treatment of a variety of inflammatory conditions. Matrine has better antibacterial activity and can hence be a potential agent in preventing and treating S. aureus mastitis. In addition, matrine regulates the structural state of the intestinal microflora while increasing the number of the dominant probiotic bacteria. These pharmacological effects suggest that matrine has broader application prospects in future clinical studies. Although matrine exhibited a variety of biological activities, its low bioavailability and high doses restrict its therapeutic applicability. Therefore, further studies are warranted to yield the best derivatives with high bioactivity and common side effects.
Footnotes
Abbreviations
S. flavescens: Sophora flavescens; P. aeruginosa: Pseudomonas aeruginosa; S. aureus: Staphylococcus aureus; HCC: Hepatocellular carcinoma; DSS: Dextran sodium sulfate; CNS: Central nervous system; LTA: Lysophosphatidic acid; TLR2:Toll-like receptor 2; LPS: Lipopolysaccharide; MDA: Malondialdehyde; EMT: Epithelial–mesenchymal transition; NSCLC: Non-small cell lung cancer; CSC: Cancer stem cells; VEGF: Vascular endothelial growth factor; GI: Gastrointestinal; PDAC-: Pancreatic ductal carcinoma; AML: Acute myeloid leukemia; NB: Neuroblastoma; TRB3: Triptolide 3, MAPKs: Mitogen-activated protein kinases; VASP: Vasodilator-stimulated phosphoprotein; CRC: Colorectal cancer; HBV: Hepatitis B virus; PCV2: Porcine circovirus Type 2; QS: Quorum–sensing; S. epidermidis: Staphylococcus epidermidis; S. mutans: Streptococcus mutans; E. coli: Escherichia coli; BMEC: Bovine mammary epithelial cell, P. acnes: Propionibacterium acnes, S. albicans: Saccharomyces albicans, A. fumigatus: Aspergillus fumigatus; T. mentagrophytes: Trichoderma mentagrophytes; C. neoformans: Cryptococcus neoformans; EAE: Encephalomyelitis; MS: Multiple sclerosis; Ols: Oligodendrocytes; SOD: Superoxide dismutase, GSH-PX: Glutathione peroxidase; CAT: Catalase; T-AOC: Total antioxidant capacity; DCM: Diabetic cardiomyopathy; L. acidophilus: Lactobacillus acidophilus; C. rumen: Clostridium rumen.
Acknowledgments
Thanks to all the teachers and students who have contributed to this paper.
Authors’ Contributions
XC: Conceptualization, methodology, and writing – original draft. PL and YM: Visualization and investigation. GW: Writing – review and editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
This work was funded by the National Natural Science Foundation of China (Grant No. 32160852).
